Abstract
Atomic polarizability is an essential theoretical construct to define and correlate many physicochemical properties. It exhibits periodicity and has a relationship with other periodic descriptors. Although a number of scales are available to compute atomic polarizability, the final scale is yet to be designed. In this venture, we have invoked a new empirical approach to compute the atomic polarizability of 103 elements of the periodic table, considering the conjoint action of other periodic descriptors, namely effective nuclear charge (Zeff) and absolute radii (r). The proposed approach is
Keywords

Introduction
It is considered that the hydrogen atom is an advantageous system to be used for an introduction to classical or quantum methods. Calculation of the polarizability is used to determine the change of the energy level of the hydrogen atom caused by an electric field, and is known as the Stark effect. 1 The exact calculation of atomic polarizability has potential applications in quantum information processing and in trapping schemes. 2 Bonin and Kadar-Kallen 3 have reported the relationship between polarizability and other physical parameters.
Atomic polarizability is a size-dependent parameter which depends on the ease of deforming the valence electron clouds of a chemical species. 4 Polarizability offers numerous applications regarding bond energy and chemical reactions. 5 Nagle 6 studied how atomic polarizability can be used in conjunction with the number of s and p valence electrons. A semi-empirical method has been reported by Miller 7 to compute the components of molecular polarizability along with the dipole tensor. Politzer et al. 8 have also proposed a relationship between molecular polarizability (α), volume (V), ionization energy (I), and mean local ionization energy. Furthermore, Ghosh and Islam 9 have also computed global atomic hardness taking into account the relationship between Electronegativity and Hardness Polarizability can be expressed with good accuracy by invoking relationship to volume and an appropriate set of radii. 10 Chattaraj and Maiti 11 have already reported that atomic polarizability and hardness of atoms are two periodic properties and their periodicity has been established by the maximum hardness principle (MHP)12–17 and the minimum polarizability principle (MPP).18,19 According to MPP and MHP, we can say that an atom with a closed shell structure is the most stable and the least polarizable among all the atoms in each period, and in general, polarizability decreases along a period and increases along a group, while the hardness follows the reverse trend. Thus, it reveals that the global hardness and polarizability of elements are the periodic function of the atomic number. Ghanty et al. have reported the relationship between the size, the polarizability, and the hardness of different chemical species. This relationship supports the use of these periodic properties for qualitative characterization of the hard–soft acid–base concept.20–22
In this analysis, we have proposed a new equation to compute atomic polarizability invoking the relationship between atomic radii and effective nuclear charge.
Importance of absolute radii to compute the atomic polarizability
During a chemical reaction, the bond distance changes with a change of covalent radii and molecules become distorted. So, the use of covalent radii places a hindrance on the determination of the exact values of other periodic descriptors. Ghosh and Islam 9 have already established that the electronegativity of the atom cannot be an in situ property The validity of the electronegativity equalization principle strongly supports this concept. Also, there is a distinct disadvantage of in situ properties that it is not transferable. Since atomic polarizability is a similar theoretical construct like electronegativity, we may assume that atomic polarizability cannot be an in situ property. The use of covalent radii has certain disadvantages over the accurate calculation of periodic descriptors. Over a long period, scientists have utilized reported covalent radii to compute radial-dependent periodic descriptors. 23 However, there has been much debate about whether we should use covalent radii or not for computing theoretical descriptors. 9 The higher values of covalent radii24–27 compared to absolute radii for some atoms demonstrate this anomalous relation. Also, it is a well-known fact that the covalent radii of atom can be transferred from one compound to other. So the use of absolute radii in place of covalent radii will be more useful to compute an absolute property of atoms. Ghosh et al. 25 computed absolute radii in terms of orbital exponent considering Slater’s rule. Chakraborty et al. 26 also proposed an approach for computing absolute radii of the 103 elements of the periodic table considering conjoint action of two periodic properties, namely ionization energy and effective nuclear charge. We have followed this verdict in our computation and proposed an approach invoking the absolute radius of an atom.
Method of computation
Molecular polarizability is a vital descriptor to explain intermolecular interactions.28–32 Polarizability has been established as an essential factor in determining the chemical reactivity. 33 In this analysis, we have tried to propose a theoretical model to compute atomic polarizability invoking other periodic properties.
It is well established that polarizability is considered equal to r 3 , where r is the radius of the conducting sphere. 34 The generality of linear volume with polarizability has been assessed quantum mechanically in the case of atoms. We can write the following equations as suggested by Hati and Datta 35
where K is a constant.
For atoms, the value of K was determined by Dmitrieva and Plindov 36 through atomic oscillation theory, as 0.585, whereas Hati and Datta 35 used the value of K as 0.792.
Equation 1 reveals a direct relationship between atomic polarizability and the size of atoms. It supports the fact that atomic polarizability is periodic in nature and can be computed in terms of other periodic descriptors.
Electronegativity, an important periodic descriptor, has already been defined in terms of an energy model as 37
Ghosh and Islam
9
have also proved that another periodic descriptor, hardness, is numerically equal to electronegativity, that is,
So we can assume the following relationship
Szarek and Grochala 38 suggested a simple relationship between atomic radius, atomic hardness, and electronic polarizability as
From equation (4), it can be concluded that there is a relationship between atomic polarizability with effective nuclear charge and atomic radii.
We propose that atomic hardness is directly proportional to the effective nuclear charge
From the relationship between atomic hardness, effective nuclear charge, and absolute radii, we can propose the following
Now we have proposed that atomic polarizability is not equal to the parameter
The proposed equation is
where “a” and “b” are regression coefficients and “e” represents electronic charge.
In this report, we have tried to maintain the proper dimension of evaluated polarizability as an energy unit. It is already reported that the exact value of hardness is calculated from the absolute radii, and not from the covalent radii. In formula (6), we have used the absolute radii from the literature. 38 In addition to the resolution of the fundamental problems relating to the correct dimension of the measured quantity and the proper size descriptors of atoms, many researchers39–41 have calculated the screening constants and effective nuclear charge theoretically. We have used the most reliable set of effective nuclear charge (Zeff) from the reported data, 9 and have considered the charge in an atomic unit and the distance in Å to compute the polarizability in the energy model.
Equalization of polarizability
The equalization of the periodic property is an essential theoretical concept and comes into play during molecule formulation from atoms. Sanderson42,43 first popularized the concept of equalization regarding electronegativity. According to his research, when two or more atoms that are initially different in electronegativity combine chemically, their electronegativity becomes equalized in the molecule. The equalization of electronegativity occurs through the adjustment of the polarities of the bonds, which is pictured as resulting in a partial charge on each atom. Electron loss causes an increase and electron gain causes a decrease in electronegativity. When an electrophile interacts with a nucleophile, the electrophilicity of the former is reduced (via electronic charge transfer and other related processes, from the nucleophile to the electrophile) and that of the latter is increased until they become equalized (roughly the geometric mean or arithmetic mean).44,45 It is expected that as a periodic descriptor, polarizability is also equalized like electronegativity. The equality between local and global polarizability values reaffirm this fact. Beg et al. 46 proposed an approach to compute molecular polarizability invoking the polarizability equalization principle (PEP)
In this report, we have computed the molecular polarizability in terms of our evaluated atomic polarizability on the basis of PEP.
Result and discussion
Using our proposed approach, we have computed the atomic polarizability of the first 103 elements of the periodic table, with the data presented in Table 1. All elemental values demonstrate a similar periodic trend as expected from the periodic table.25,47 From Table 1, it is evident that the value of the computed polarizability of the nitrogen atom is lower than oxygen. This proves the half-filled shell characteristics of nitrogen. Due to the half-filled shell structure, nitrogen is less polarized under perturbation than oxygen. The computed polarizability values show a minimum for alkali metals and a maximum for noble gas atoms.
Periodic table with computed atomic polarizabilities of the first 103 elements.

It is also apparent from Table 1 that the magnitude of the polarizability of mercury and gold are exceptionally very low, indicating that these atoms would hold electrons very tightly and are less polarizable. The above observations support the intrinsic inertness of mercury and gold similar to with the relativistic effects of such atoms.48,49 Our computed data successfully explain the half-filled and full shells of the structures of d-block and p-block elements.
Lanthanide contraction in terms of atomic polarizability is also established through our computed data. The same trend is presented in Figure 1, which correlates the computed polarizability values with ionization energies. 50

Comparison between computed molecular polarizability and ionization energy (IE).
In order to apply computed atomic polarizability to the real field, we have adopted the PEP and computed molecular polarizability (mean polarizability) for some compounds. The computed molecular polarizability has been compared vis-à-vis with available experimental data 51 in Figures 2–10.

Comparison between computed molecular polarizability and experimental values of alkanes.

Comparison between computed molecular polarizability and experimental values for alkanols.

Comparison between computed molecular polarizability and experimental values for alkenes.

Comparison between the computed molecular polarizability and experimental values of carbonyl compounds.

Comparison between computed molecular polarizability and the experimental values for alkyl cyanides.
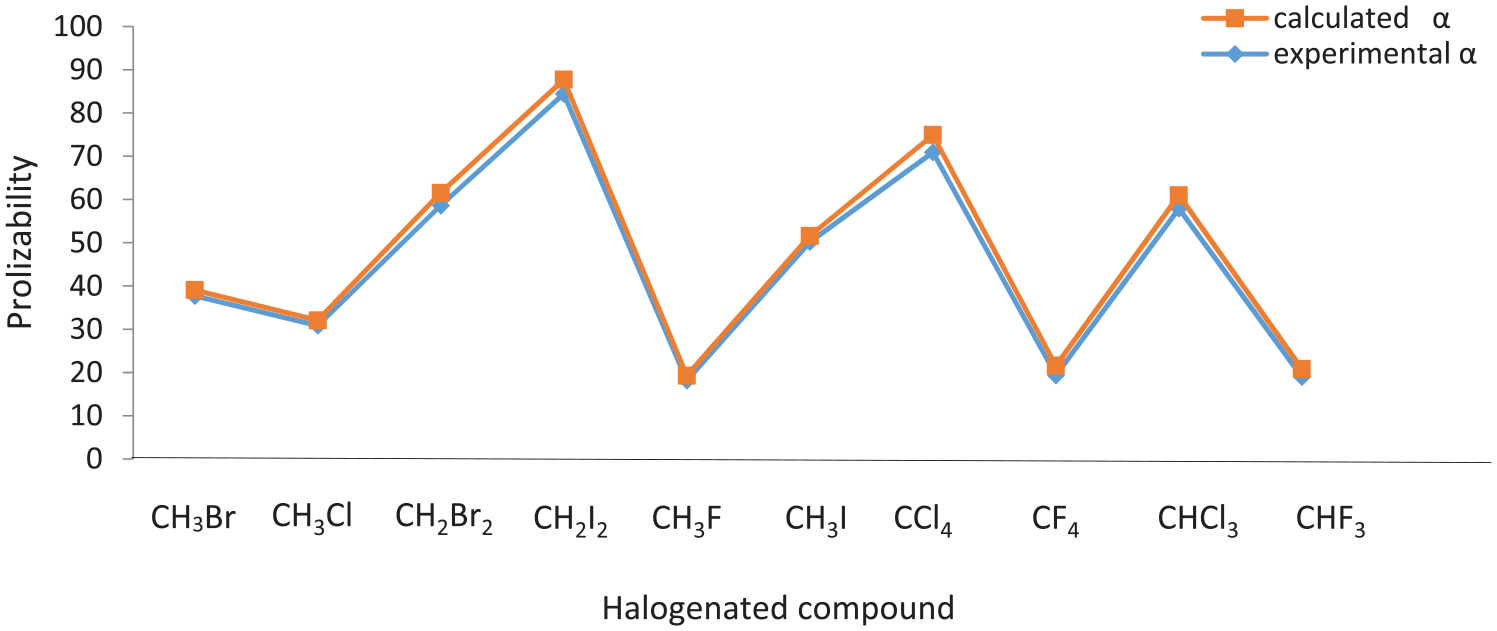
Comparison between computed molecular polarizability and the experimental values for halogenated compounds.

Comparison between computed molecular polarizability and the experimental values for sulfur compounds.

Comparison between computed molecular polarizability and the values for various molecules.

Comparison between computed molecular polarizability and the experimental values for diatomic molecules.
Since alkanes are non-polar compounds and have only carbon and hydrogens, they require more heat to combust. They respond poorly to other polar and ionic species. The same trend is observed in Figure 2. Among the hydrocarbons, methane shows the least polarizability and reactivity. For the alcohol series, due to inclusion of a polar oxygen atom, the magnitude of polarizability of alcohols increases compared to alkanes containing equivalent numbers of carbon atoms. A comparative analysis of the polarizability of alcohols is presented in Figure 3.
From Figure 4, it is established that nitrobenzene has the highest polarizability as it has both nitrogen and oxygen atoms.
In Figure 5, formaldehyde exhibits the lowest value of polarizability due to the absence of a nitrogen atom.
A similar trend of polarizability with experimental values for cyanomethane compounds is observed in Figure 6. Cyanomethane has the lowest polarizability in comparison to other compounds.
The close agreement between our computed polarizabilities and experimental results are observed for some halo and sulfur compounds in Figures 7 and 8, respectively.
The impact of the half-filled shell structure of nitrogen on the polarizability is distinct in Figure 9.
Due to the lower electronegativity of mononuclear diatomic molecules, H2 shows the least polarizability. This trend is visible in Figure 10.
In Figure 11, the computed polarizabilities of some molecules such as hydrogen, carbon, nitrogen, oxygen, fluorine, sulfur, chlorine, bromine, and iodine are compared with the ab initio (mean) linear polarizabilities proposed by van Duijnen and Swart. 51 A close agreement between the two data is distinct from the Figure 11.
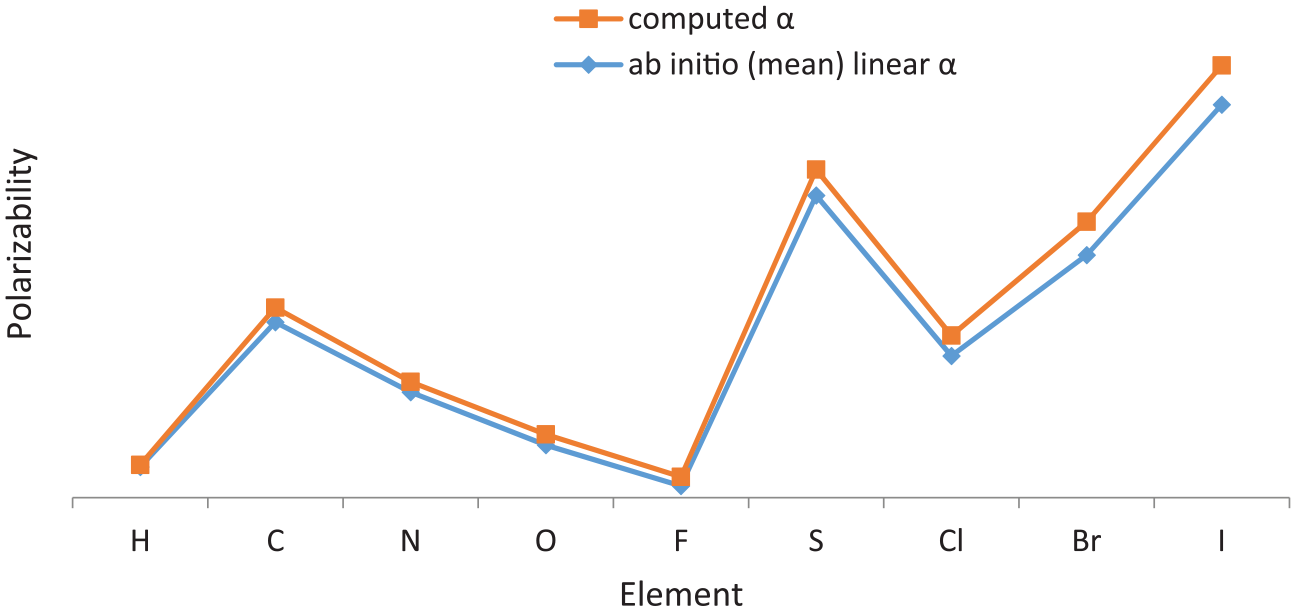
Comparison between computed molecular polarizability and the experimental values for some elements.
Conclusion
Based on the potential energy concept, we have established a new scale of atomic polarizability invoking two other periodic descriptors, namely effective nuclear charge and absolute atomic radius. Our proposed approach establishes the dependence of chemical reactivity on polarizability. The computed atomic polarizability distinctly exhibits all periodic properties. The relativistic effect is also pronounced in our computations. Comparative analysis with available data, considered as a benchmark, reveals the efficacy of our proposed equation. Furthermore, we have computed molecular polarizability in terms of our computed atomic polarizability through the PEP. Excellent correlation with experimental data establishes our model.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.







