Abstract
The adsorption of toluene and 1,3,5-trimethylbenzene and the catalytic transformation of 1,3,5-trimethylbenzene are applied as probing approaches to characterize the pore system of hierarchical EU-1 zeolites prepared using organofunctionalized fumed silica as the silicon source. The adsorption and diffusion of toluene and 1,3,5-trimethylbenzene are significantly improved in the hierarchical EU-1 zeolites compared with the conventional microporous EU-1 zeolite. The adsorption kinetics of toluene and 1,3,5-trimethylbenzene suggested that introducing mesopores significantly increases the rate of adsorption and improved the diffusion of large molecules. In the catalytic transformation of 1,3,5-trimethylbenzene, the conversion of 1,3,5-trimethylbenzene on the hierarchical EU-1 zeolites is doubled compared with the conventional microporous EU-1 zeolite, due to the improved diffusion of bulky molecules and enhanced accessibility of active sites in the hierarchical EU-1 structure. Although isomerization is the main reaction, differences are observed in the product ratios of isomerization to disproportionation between the hierarchical EU-1 zeolites and the microporous counterpart with different times on stream. The transformation of 1,3,5-trimethylbenzene over the hierarchical EU-1 zeolites has a higher isomerization to disproportionation ratio than that over the microporous EU-1 zeolite; this is due to the increased mesoporosity.
Keywords

Introduction
EU-1 zeolite has a one-dimensional pore system of 10-membered ring (10-MR) channels (0.54 × 0.41 nm) running along the [100] direction and connected to side pockets by a 12-membered ring (12-MR) (cross section of 0.68 × 0.58 nm; 0.8 nm depth) in the [001] direction. The unique pore system with narrow channels and wide pockets in EUO-type zeolite allows it to demonstrate very interesting and special catalytic performance in different reactions involving hydrocarbon transformation.1–6 However, the medium-size one-dimensional channel EU-1 zeolite has the drawbacks of a slow diffusion rate and low accessibility of the active sites within the channels and side pockets for large organic molecules. It is found that the narrow channel of EU-1 zeolite is easily blocked by the deposition of coke molecules. 1 In order to solve this problem, mesopores were introduced into the EU-1 zeolite making a hierarchical pore structure, which can improve the diffusion of large reactants and product molecules and can make more active sites accessible to bulky molecules.2–7
There are two main strategies for making hierarchical zeolites, namely, top-down approaches (post-synthesis treatment, for example, desilication or dealumination) and bottom-up approaches (e.g. templating methods). 8 The post-synthesis treatment often changes the chemical composition of the zeolite, which reduces its thermal and hydrothermal stability, and to loss of a significant amount of Brønsted acid sites. 4 In the templating methods, soft templates such as cationic polymers and multivalent cationic surfactants have been applied to synthesize hierarchical EU-1 zeolite.3,7 We have reported another route using organofunctionalized fumed silica as the silicon source in the conventional hydrothermal system to synthesize hierarchical EU-1 zeolite. 9
Characterization of the texture of the hierarchical zeolites is an important but challenging task. Gas adsorption of N2 at 77 K, Ar at 87 K, and CO2 at 273 K has become the standard method to characterize the pore structure of a porous material, with particular reference to the assessment of surface area and pore size distribution. 10 However, these small gaseous molecules are not enough to monitor the real applications. Several organic vapors with large molecular size are applied as probe molecules to study the pore structure of porous materials, especially for the pores with molecular sieve effects.11,12 Xylenes and 1,3,5-trimethylbenzene (TMB) have been applied as probes to study the adsorption and diffusion in mesoporous zeolites in our previous studies.13,14 Moreover, the use of catalytic reactions with probe molecules is also a versatile tool to characterize the pore structure of a porous catalysts. 15 The activity and product selectivity are usually analyzed to evaluate the pore structure based on the shape selectivity effects on the sizes of reactant molecules, transition states, intermediates, and product molecules. TMB is such a probe molecule and has three isomers with kinetic molecular size in the order of 1,3,5-TMB > 1,2,3-TMB > 1,2,4-TMB. The transformation of TMBs can be catalyzed by zeolites, among which isomerization between the TMB isomers, disproportionation of TMBs to xylenes and tetramethylbenzenes (TeMBs), and dealkylation to xylenes, toluene, benzene, and lower olefins have been recognized as the main reaction pathways in this complex reaction system. 16 Medium pore zeolites including EU-1 impose significant steric constraints for large TMB molecules. Diffusion of TMBs into the one-dimensional channel system of EU-1 is rather slow, and the conversion of TMB over EU-1 zeolites is usually significantly slow. It was shown that the transformation of 1,3,5-TMB molecules occurs mainly on the “external” surface of the zeolites with medium pore size. 17
Hierarchical EU-1 zeolites with extra mesopores could potentially improve the diffusion of large molecules within the structure. Herein, we used the adsorption of toluene and 1,3,5-TMB as probe molecules and the catalytic transformation of 1,3,5-TMB to characterize the pore structure of hierarchical EU-1 zeolites synthesized from organofunctionalized fumed silica.
Results and discussion
Characterization of the EU-1 zeolites
The EU-1 zeolites prepared using organofunctionalized fumed silica as the silicon source (EU-1-1, 2) showed the typical characteristic peaks of an EUO framework topology structure, with similar crystallinity as compared to the conventional microporous EU-1 zeolite synthesized with unfunctionalized fumed silica (EU-1-0), as shown in the X-ray diffraction (XRD) patterns in Figure 1(a). The involvement of organic functional groups did not influence the crystallization significantly. However, the N2-adsorption/desorption results shown in Figure 1(b) confirmed the introduction of mesopores into the EU-1 zeolites using organofunctionalized fumed silica as the silicon source. As shown in the N2 adsorption/desorption isotherms, the isotherm of EU-1-0 has a small hysteresis loop in the high relative pressure region, due to the nature of the small crystalline assembled particle as shown in Supplemental Figure S1. The adsorption increased gradually as the ratio of organic functional groups increased, which is caused by the introduction of mesopores. The pore structure parameters derived from the isotherms are summarized in Table 1. The Brunauer-Emmet-Teller (BET) surface area and pore volume of the EU-1 zeolites increased with an increase in the functionalization, which is related to the increase of mesoporosity. The micropore volume remained similar among the EU-1 zeolites, while the pore volume due to the mesopores increased from EU-1-0 to EU-1-2. The mesoporous EU-1 zeolites have similar composition compared with the conventional EU-1. The framework Si/Al ratios of EU-1(0) and EU-1(2) are 12.6 and 12.9, from the 29Si magic angle spinning nuclear magnetic resonance (MAS-NMR) results.

(a) XRD patterns and (b) nitrogen adsorption/desorption isotherms at 77 K for the EU-1 zeolites.
Pore structure parameters of the EU-1 zeolites.

SBET is the surface area determined by the BET model.
Vtot is the total pore volume.
Vmic is the micropore volume.
Vmeso is the mesopore volume.
Toluene adsorption
Toluene was used as a probe molecule to characterize the pore structure of the EU-1 zeolites. The toluene adsorption isotherms of the EU-1 zeolites in the pressure range of 0–30 mbar at three different temperatures are presented in Figure 2. The conventional microporous EU-1 zeolite showed the isotherm close to a type I isotherm (Figure 2(a)), which has a sharp increase of adsorption in the low-pressure region corresponding to micropore filling, followed by a plateau at higher pressures. The isotherms of the hierarchical EU-1 zeolites shown in Figures 2(b) and (c) are a combination of both type I and type IV isotherms, which show adsorption at low pressure by micropores, but also gradually increased adsorption at higher pressures by mesopores. At the same temperature, the toluene adsorption by EU-1-2 is higher than those of EU-1-0 and EU-1-1, which agrees well with the trend of the amount of mesopores. It is therefore apparent and obvious that the introduction of mesopores enhanced the adsorption of toluene by the EU-1 zeolites.

Toluene adsorption isotherms of the EU-1 zeolites (EU-1-0 (a), EU-1-1 (b), EU-1-2 (c)) at different temperatures.
The temperature-programmed desorption (TPD) curves in Figure 3 show the desorption of toluene from the EU-1 zeolites with increasing the temperature. The derivative curve (solid line) of the desorption curve (dashed line) shows the desorption speed of toluene molecules from the EU-1 zeolites as a function of temperature. The desorption speed increased with the rising of temperature and slowed down after reaching a peak at around 80°C. The derivative curves have mainly one peak at around 80°C with a small peak at around 280°C, which indicates that the EU-1 zeolite has two adsorption sites with one being the major adsorption site. It is worth noting that the desorption of toluene occurs more easily for the hierarchical zeolites (EU-1-1, 2) than for the conventional microporous zeolite (EU-1-0). The peak at around 80°C shifted to a lower temperature when the mesoporosity increased, indicating the weaker interaction between the adsorbate and adsorbent.

Toluene TPD curves for the EU-1 zeolites.
The adsorption kinetics of toluene on the EU-1 zeolites were measured at 308 K at a pressure range of 0.5–1 mbar to obtain the adsorption rate constant (k), and under a constant pressure of 1 mbar to determine the effective diffusion time constant (Deff/R2). As shown in Figure 4(a) and (b), the hierarchical EU-1 zeolites adsorb toluene much faster than the conventional microporous EU-1 zeolite. The adsorption speed increased with increasing mesoporosity. The kinetic curves obtained in the pressure range of 0.5–1 mbar were fitted to the pseudo first-order kinetic model (equation (1)) to give the adsorption rate constants (k), which are presented in Table 2. The equation fitted well with the kinetic curve, as is supported by the high values of R2. The adsorption rate constant (k) reflects the adsorption rate of an adsorbate being adsorbed onto an adsorbent. A high adsorption rate constant (k) indicates a high adsorption rate. In the adsorption of toluene, the hierarchical EU-1 zeolites with mesopores have higher values of adsorption rate constants (k) compared with conventional microporous EU-1 zeolite, and the k values increased with the increasing of mesoporosity following the order of EU-1-0 < EU-1-1 < EU-1-2.

The toluene adsorption kinetics on the EU-1 zeolites at 308 K, under pressure of 0.5-1 mbar (a) and 1mbar (b).
The adsorption rate constants (k values) and effective diffusion time constants (Deff/R2) of toluene adsorption on the EU-1 zeolites.

The effective diffusion time constants (Deff/R2) were extracted from the kinetic curves obtained under a constant pressure of 1 mbar following equation (3) and are presented in Table 2. The high values of R2 showed good fitness. The effective diffusion time constant (Deff/R2) is an indicator of the diffusion rate of an adsorbate being adsorbed into an adsorbent, which excludes the influence of the morphology of the zeolite particle by involving the radius (R). A high effective diffusion time constant (Deff/R2) refers to a high diffusion rate. It can be seen that the introduction of mesoporosity dramatically improved the diffusion of toluene into the adsorption site of EU-1 zeolites, which is indicated by the higher Deff/R2 value of hierarchical EU-1 zeolites than that of the conventional microporous EU-1 zeolite, following the same order as the adsorption rate constants (k): EU-1-0 < EU-1-1 < EU-1-2.
1,3,5-TMB adsorption and catalytic transformation
The 1,3,5-TMB adsorption isotherms of the EU-1 zeolites were measured at three different temperatures (308, 323, and 338 K) and are presented in Figure 5. It can be seen in Figure 5(a) that EU-1-0 has a very low adsorption of 1,3,5-TMB, which is attributed to the fact that 1,3,5-TMB molecules with a molecular size of 0.87 nm cannot be adsorbed into the one-dimensional channel of EU-1 zeolite (pore opening is 0.58 × 0.41 nm). The adsorption of 1,3,5-TMB can only happen on the external surface of the microporous EU-1-0. However, with the increase of mesoporosity in the hierarchical EU-1 zeolites, the adsorption of 1,3,5-TMB increased gradually (Figures 5(b) and (c)). It is obvious that the introduction of mesopores offers more adsorption sites for larger molecules in the hierarchical EU-1 zeolites.
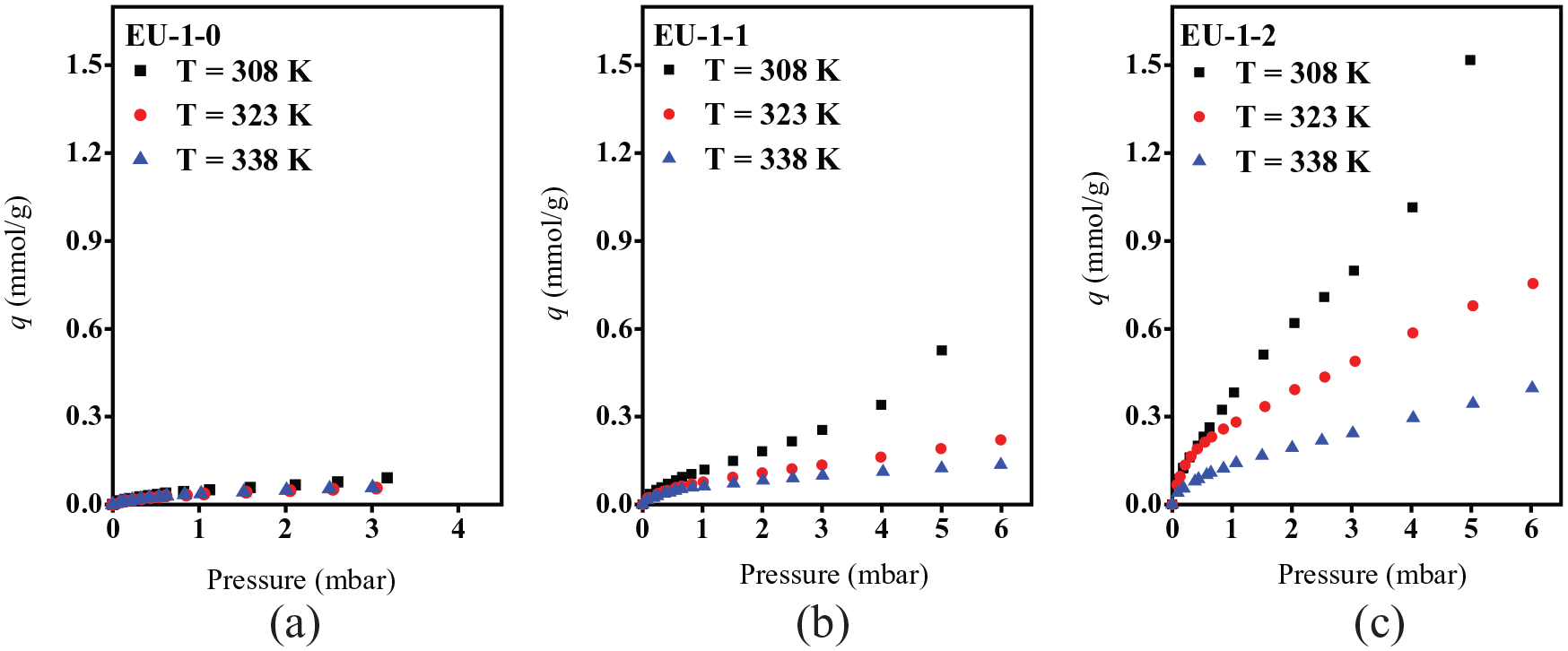
1,3,5-TMB adsorption isotherms of the EU-1 zeolites (EU-1-0 (a), EU-1-1 (b), EU-1-2 (c)) at different temperatures.
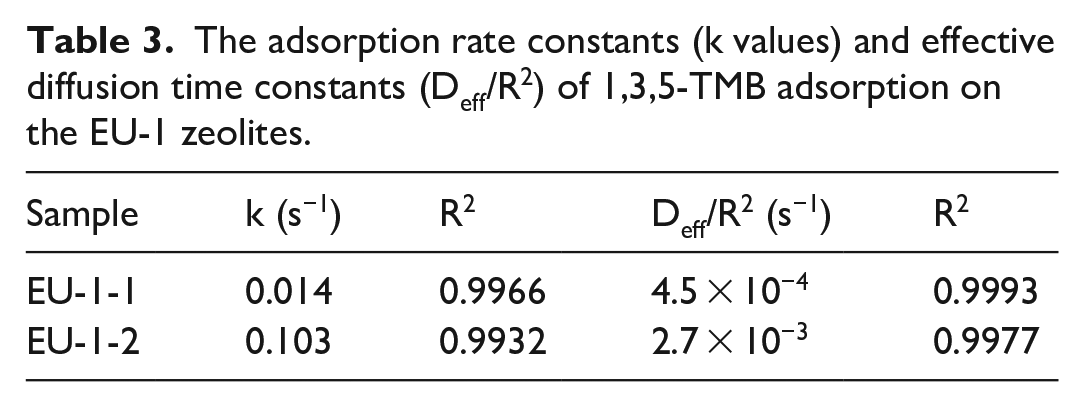
The adsorption kinetic curves of 1,3,5-TMB on the hierarchical EU-1 zeolites are shown in Figure 6(a) and (b). The conventional microporous EU-1 has very limited adsorption of 1,3,5-TMB, which is not presented. 1,3,5-TMB has a larger molecular size than toluene and cannot enter the 10-MR pores of EU-1 zeolite. However, the adsorption speed of 1,3,5-TMB increased when the mesoporosity of the hierarchical EU-1 zeolites increased from EU-1-1 to EU-1-2. As shown in Table 3, the adsorption rate constant (k) of EU-1-2 is larger than that of EU-1-1, which is attributed to the fact that the increased mesoporosity allows much faster adsorption into the adsorption site within the EU-1 zeolites. The effective diffusion time constant (Deff/R2) of EU-1-2 increased compared with that of EU-1-1, confirming the dramatically improved diffusion of 1,3,5-TMB into the adsorption site of the hierarchical EU-1 zeolites due to the increased mesoporosity.

1,3,5-TMB adsorption kinetics on the EU-1 zeolites at 308 K, under pressure of 0.5-1 mbar (a) and 1mbar (b).
The adsorption rate constants (k values) and effective diffusion time constants (Deff/R2) of 1,3,5-TMB adsorption on the EU-1 zeolites.
The catalytic transformation of 1,3,5-TMB was applied as a probing reaction for the hierarchical EU-1 zeolites, which is a complex reaction including mainly isomerization and disproportionation.16,18–20 Moreover, the products of these reactions can react with each other and with the original reactants as well. 16 The conversion of bulky molecules tends to be more diffusion controlled, because the majority of the active sites of the zeolite are within its pores. Since the kinetic diameter of the 1,3,5-TMB molecule is larger than the pore openings of EU-1 zeolite with a one-dimensional pore system, the reaction of 1,3,5-TMB is presumed to occur only on the external surface of the 10-MR EU-1 zeolite, 21 which is supported by the low conversion of 1,3,5-TMB over the conventional microporous EU-1 zeolite as shown in Figure 7(a). The active sites within the micropore system of EU-1 zeolite are not accessible to the 1,3,5-TMB molecules. However, the hierarchical EU-1 zeolites with more mesopores show higher conversions (roughly doubled) compared to the conventional microporous EU-1 zeolite. It is well known that the transport and diffusion of reactants and products influences the outcome of the reaction.22,23 As has been confirmed by the adsorption performance, the introduction of mesopores dramatically improved the adsorption and diffusion of 1,3,5-TMB molecules within the hierarchical EU-1 zeolites. The improved activity of hierarchical EU-1 zeolites in the transformation of 1,3,5-TMB is largely contributed to the enhanced diffusion of the reactant molecules and the improved accessibility of active sites by mesopores. The conversion of 1,3,5-TMB over all the studied EU-1 zeolites dropped with longer time on stream in the continuous reaction, which is caused by the deactivation of the active sites located in the pore openings close to the surface.
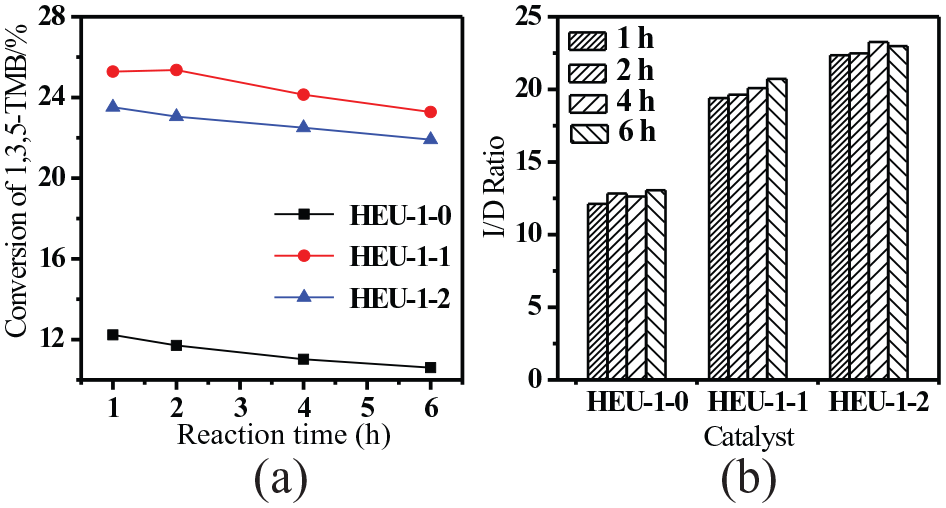
(a) Conversion of 1,3,5-TMB over the EU-1 zeolites and (b) the ratio of isomerization to disproportionation (I/D ratio) at 673 K.
As shown in Table 4, the products of the transformation of 1,3,5-TMB are mainly those derived from isomerization, disproportionation, transalkylation, and dealkylation reactions.16,18,23 In the isomerization reaction, 1,3,5-TMB was isomerized into 1,2,4-TMB and 1,2,3-TMB, following the potential monomolecular mechanism. 16 In the disproportionation reaction, one 1,3,5-TMB molecule reacts with another by transfer of one methyl group leading to one xylene and one TeMB molecule, followed by a bimolecular mechanism.23,24 The product distribution in Table 4 shows that 1,2,4-TMB and 1,2,3-TMB are the major products, among which 1,2,4-TMB was formed with a much higher rate than 1,2,3-TMB, since the diameter of 1,2,4-TMB is smaller than that of 1,2,3-TMB and the rate of formation is kinetically controlled by the pore system. It is obvious that isomerization is the main reaction in the catalytic transformation of 1,3,5-TMB over the EU-1 zeolites, which is quite different from the reaction over USY zeolite. 18 The isomerization of 1,3,5-TMB into 1,2,4-TMB and 1,2,3-TMB accounts for more than 90% of the product selectivity, which agrees well with the findings of Röger et al., 21 who reported that isomerization reached a selectivity of about 90% in the 1,2,4-TMB transformation over HZSM-5 zeolite and that the 1,2-methyl-shift was shown to take place on the external surface of the zeolite crystal. It has also been demonstrated by Se-Ho et al. 17 that the isomerization of 1,2,4-TMB occurs mostly on the external surface of the 10-MR zeolite, while disproportionation happened in the microporous channel system. From the product distribution, the ratios of isomerization to disproportionation (I/D ratio) from different times on stream were calculated and are presented in Figure 7(b). Although the isomerization is the main reaction in the transformation of 1,3,5-TMB, the I/D ratio showed some differences among the various EU-1 zeolites under different conditions. The I/D ratios over hierarchical EU-1 are higher than those obtained from the conventional microporous EU-1 zeolite, which indicates that more isomerization occurred over the hierarchical EU-1 zeolites due to the improved diffusion of reactants and enhanced accessibility of the active sites in the mesopores. The conversions of 1,3,5-TMB are similar over EU-1-1 and EU-1-2, and the I/D ratio could be reasonably compared between the two hierarchical EU-1 zeolites. It can be seen in Figure 7(b) that the I/D ratio over EU-1-2 is higher than that over EU-1-1, which is attributed to that the diffusion of reactants was enhanced and isomerization can happen more easily with a higher mesoporosity.
The distribution (mol%) of aromatic products obtained from the EU-1 zeolites after a time of 6 h on stream at 673 K.

TMB: trimethylbenzene.
Conclusion
The pore structure of hierarchical EU-1 zeolites, prepared using organofunctionalized fumed silica as the silicon source, is characterized using the adsorption of toluene and 1,3,5-TMB as probe molecules, and the transformation of 1,3,5-TMB as the probe reaction. The hierarchical EU-1 zeolites showed significantly improved adsorption and diffusion of bulky molecules compared with the conventional microporous EU-1 zeolite. The toluene and 1,3,5-TMB adsorption capacity of hierarchical EU-1 zeolites increased with increasing mesoporosity. The toluene TPD results showed that the desorption of toluene occurs more easily for the hierarchical zeolites than for the conventional microporous zeolite. In addition, the adsorption kinetics of toluene and 1,3,5-TMB showed that the increased mesoporosity in the hierarchical EU-1 zeolites resulted in higher adsorption rates and faster diffusion for large molecules.
Because of the enhanced diffusion of the bulky reactant molecules and improved accessibility of active sites within the EU-1 zeolite pore system, the conversion was doubled in the transformation of 1,3,5-TMB over the hierarchical EU-1 zeolites compared with the conventional microporous EU-1 zeolite. The product I/D ratios in the reaction catalyzed by the hierarchical EU-1 zeolites were higher than those over the microporous EU-1 zeolite, which is ascribed to the better diffusion and improved accessibility of active sites in the mesopores of the hierarchical EU-1 zeolites.
It can be concluded that the significantly improved adsorption and diffusion of toluene and 1,3,5-TMB and the catalytic performance in the transformation of 1,3,5-TMB confirmed the improvement of mesoporosity in the hierarchical EU-1 zeolites using organofunctionalized fumed silica as the silicon source, which leads to more active sites in the one-dimensional pore system with 10-MR channels accessible to large reactant molecules. Such hierarchical EU-1 zeolites could further enhance the catalytic performance in reactions involving other bulky molecules.
Experiment
Preparation of hierarchical EU-1 zeolite
Hierarchical EU-1 zeolites were prepared using organofunctionalized fumed silica as the silicon source, following the protocol we developed previously. 9 The fumed silica was functionalized with TPOAC (dimethyloctadecyl[3-(trimethoxysilyl)propyl]ammonium chloride) according to the molar ratio of SiO2/x organosilane/6 CH3OH/60 H2O (x = 0.072, 0.182). In a typical process, 20 g of fumed silica (Degussa, Aerosil 200) was dissolved in 400 mL of distilled water. The mixture was stirred (100 r/min) under reflux at 373 K for 1 h, before 20 mL of TPOAC (here x is 0.182) was added, and the mixture was continuously refluxed for another 8 h at 373 K. After being washed thoroughly with ethanol and dried at 393 K for 12 h, organofunctionalized fumed silica with different ratios was obtained. Thermogravimetric (TG) analyses indicated the percentage of the organic moiety of the organofunctionalized fumed silica as 17.5% and 30.5%, which corresponds to x = 0, 0.072, and 0.182, respectively.
The synthesis of hierarchical EU-1 zeolites was started from a solution prepared by mixing distilled H2O, NaOH, NaAlO2, HMBr2 (hexamethonium bromide), NaF, NH4NO3, seed (0.5 wt% of the total solution) and organofunctionalized fumed silica, according to the molar composition of 60 SiO2/Al2O3/5.5 Na2O/700 H2O/2 NH4NO3/2 NaF/6 HMBr2. After being stirred at room temperature for 4 h, the resulting gel was transferred into an autoclave and heated at 373 for 18 h, followed by crystallization at 413 K for 12 days. The resulting product was washed with distilled H2O until the pH was neutral, and then dried at 373 K for 12 h, followed by calcination under an air atmosphere at 873 K for 5 h. The hierarchical EU-1 zeolites were coded as EU-1-n (n = 1, 2, following the increase of organic functionalization). A conventional microporous EU-1 was synthesized following the same process but with fumed silica without functionalization and was named EU-1-0.
To make the catalysts for 1,3,5-TMB transformation, the EU-1 zeolites were subjected to ion-exchange three times using 1 M NH4NO3 with a liquid-to-solid ratio of 100 mL/g at 333 K for 2 h. After being washed with distilled H2O, dried at 373 K for 12 h and calcinated at 823 K for 5 h, the catalysts were obtained and named as HEU-1-n (n = 0, 1, 2).
Characterization
The powder XRD patterns of the as-synthesized EU-1 zeolites in the range of 5°–35° were recorded on a Shimadzu XRD-6000 diffractometer with CuKα radiation, using a step size of 0.02° and a counting time of 10 s. Quantachrome QUADRASORB SI equipment was used to obtain the N2 adsorption–desorption isotherms (77 K). Before each measurement, the sample was degassed at 613 K under vacuum for 5 h. The total surface area, SBET, was obtained from the BET equation; the micropore volume (Vmic) was calculated from the t-plot method; the total pore volume (Vtot) was measured from the N2 adsorption at a relative pressure p/p0 of 0.99. The mesopore volume (Vmeso) was calculated from the difference between Vtot and Vmic. 29Si solid-state MAS-NMR spectroscopy analysis was performed on a Bruker Avance III 500-MHz spectrometer operating at resonance frequencies of 99.36 MHz.
Adsorption of toluene and 1,3,5-TMB
The adsorption isotherms and kinetic curves with toluene and 1,3,5-TMB as the adsorbates using the EU-1 zeolites were measured on an intelligent gravimetric analyzer (IGA-002; Hiden, Warrington, UK). The apparatus has a very sensitive microbalance with an accuracy of ±0.1 μg to record the weight changes of the adsorbents under the control of a computer.
Before the adsorption measurements, the EU-1 zeolites (50 mg) were treated at 673 K under a vacuum of 10−7 mbar for 5 h to remove the impurities and moisture until the sample weight became constant. After being cooled to the set temperature (308–338 K in this study), the vapor of the adsorbate was gradually introduced into the sample chamber until the set pressure was reached. The adsorption isotherms were obtained by recording the mass changes at the set pressures (0–30 mbar for toluene and 0–6 mbar for 1,3,5-TMB) when the adsorption reached equilibrium.
The TPD curves of toluene on the EU-1 zeolites was obtained from the same equipment by recording the weight changes of the saturated samples at 298 K under a vacuum of 10−7 mbar, while the temperature increased from 298 to 673 K.
The kinetic curves at 308 K were obtained by recording the mass change with time, in the pressure range of 0.5–1 mbar, and at a constant pressure of 1 mbar, respectively. The kinetic curves obtained at the pressure range of 0.5–1 mbar were fitted to the pseudo first-order kinetic model (equation (1)) 25

where k is the pseudo first-order rate constant, t is the adsorption time, and q∞ and qt are the adsorption capacity of the adsorbent at equilibrium and at time t, respectively.
Assuming the morphology of the sorbent particle is spherical, the kinetic curves obtained at a constant pressure of 1 mbar were fitted by the following equation (2) to calculate the diffusion coefficient (D) 26
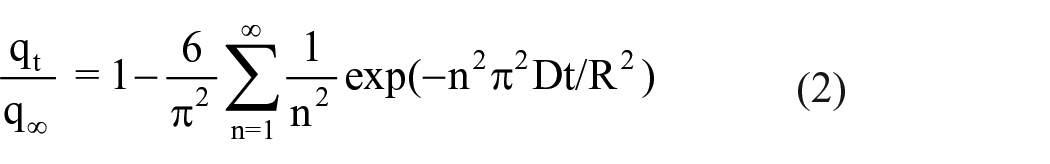
The simplified version of equation (2) is

where q∞ is the adsorption capacity of the adsorbent at equilibrium, qt is the adsorption at time t, R is the radius of the assumed adsorbent sphere, t is the adsorption time, and D refers to the diffusion coefficient. Considering the zeolite crystal is not spherical, the effective diffusion time constant Deff/R2 was applied to eliminate the effect of morphology from the zeolite crystal in this study.
Transformation of 1,3,5-TMB
Transformations of 1,3,5-TMB were carried out at 673 K under atmospheric pressure in a continuous flow fixed bed reactor. Prior to the reaction, the catalyst was pelletized, crushed, sieved into particles with a size of 200–400 µm, and loaded into the reactor, followed by in situ heat treatment at 723 K under a nitrogen atmosphere for 4 h. The weight hourly space velocity (WHSV) of 1,3,5-TMB was kept at 0.26 h−1. The product effluents were analyzed online using an Agilent 7890B gas chromatograph equipped with a flame ionization detector (FID) and a HP-AL/S capillary column. The products were studied according to different aspects: the I/D ratio is calculated by I/D = (1,2,4-TMB + 1,2,3-TMB, mole)/(xylenes + TeMBs, mole).
Supplemental Material
Supporting_information_4 – Supplemental material for Probing the pore structure of hierarchical EU-1 zeolites by adsorption of large molecules and through catalytic reaction
Supplemental material, Supporting_information_4 for Probing the pore structure of hierarchical EU-1 zeolites by adsorption of large molecules and through catalytic reaction by Zaibin Guo, Wenming Hao, Jinghong Ma and Ruifeng Li in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Applied Basic Research Program of Shanxi Province (no. 201801D221127).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
