Abstract
La–Ni–S2O82–/ZrO2–Al2O3 catalysts were successfully prepared by two different methods of sulfate impregnation, and the physico-chemical properties of the catalysts were characterized by X-ray diffraction, Brunauer–Emmett–Teller analysis, Fourier transform infrared spectroscopy, pyridine adsorption–infrared spectroscopy, and X-ray photoelectron spectroscopy techniques. Catalytic activities were evaluated in a fixed-bed flow reactor using n-pentane isomerization as the probe reaction. Compared with catalyst La–Ni–S2O82–/ZrO2–Al2O3-I, prepared by the traditional impregnation method, the catalyst La–Ni–S2O82–/ZrO2–Al2O3-W, prepared by the incipient-wetness impregnation method, possessed higher pore volume, pore size, sulfur content, and stronger Brønsted acid sites. The catalytic activity for La–Ni–S2O82–/ZrO2–Al2O3-W was maintained at around 56% within 3000 min with an isopentane selectivity of 88% which showed much greater stability than that of La–Ni–S2O82–/ZrO2–Al2O3-I. This can be attributed to the fact that (1) the large pore size and pore volume of La–Ni–S2O82–/ZrO2–Al2O3-W can largely suppress carbon deposition and (2) the more numerous and stronger Brønsted acid sites for La–Ni–S2O82–/ZrO2–Al2O3-W guaranteed to provide enough acid sites for isomerization during the reaction process.
Introduction
To improve the quality of gasoline and meet the increasingly stringent environmental protection regulations, there has been a sharp rise in the demand for processes that can produce clean fuels with non-aromatic hydrocarbons and high octane numbers. The isomerization of alkanes, such as n-pentane, is one of the best procedures to achieve high quality clean gasoline. Therefore, developing high-activity catalysts for alkane isomerization is very important for the production of clean fuels. 1 The bifunctional catalysts of platinum on chlorinated alumina, which have a strong catalytic activity below 473 K, are the state-of-the-art catalysts for isomerizing heterogeneous light naphtha streams which are predominantly C5/C6 alkanes. Nevertheless, a strictly dry and oxygen-free feed as well as a continuous addition of small amounts of organic chlorides are required to maintain the catalyst activity in this process. In an alternative system, metal oxide-based catalysts are applied for the isomerization of light naphtha at temperatures between 453 and 483 K. 2 As environmentally benign catalysts, inorganic solid acids have gained much attention in recent years due to recognized advantages of heterogeneous catalysts, like simplified product isolation, mild reaction conditions, high selectivities, ease in recovery and reuse of the catalysts, and reduction in the generation of wasteful by-products. 3 Among various acidic solids, sulfated zirconia is a promising isomerization catalyst due to its super-acidity, thermal stability, and excellent activity for skeletal isomerization of short alkanes at low temperatures. 4 It is well known that the catalytic properties of sulfated zirconia are highly influenced by the preparation method. 5 The zirconia and sulfate ions in the synthesis system may play a key role in stabilizing the mesoporous structure and enhancing the acidic properties of the catalyst. 6
In our previous work, 7 La–Ni–S2O82–/ZrO2–Al2O3 (La–Ni–SZA) was prepared and the effects of La and Ni on the structure and isomerization performance of the catalyst were investigated. The La–Ni–SZA catalyst showed an excellent catalytic activity (isopentane) yield of 66.5% at low temperature, which can be attributed to the synergistic interaction of La and Ni. In this study, ZrO2–Al2O3 (ZA) was sulfated by two different methods, and the obtained sulfated ZA catalyst precursors (SZA) were used to prepare La–Ni–SZA catalysts, to understand the effect of sulfating method on the performance of the La–Ni–SZA catalysts obtained.
Experiment
Catalyst preparation
A co-precipitated support was prepared from a mixed solution of ZrOCl2·8H2O and Al(NO3)3·9H2O using NH4OH solution as a precipitating agent. The resulting precipitates were aged, filtered off, washed with distilled water, and then dried at 110°C for 12 h to obtain the ZA support. Subsequently, the ZA was sulfated by two different methods: (1) impregnating the ZA with 0.75 mol L−1 (NH4)2S2O8 solution (15 mL g−1) under continuous stirring at room temperature for 6 h, filtering off, and then drying overnight at 110°C to obtain the catalyst precursor (denoted as SZA-I) and (2) incipient-wetness impregnation of the ZA with aqueous 0.75 mol L−1 (NH4)2S2O8 and then dried overnight at 110°C to obtain the catalyst precursor (denoted as SZA-W). SZA-I and SZA-W were impregnated with aqueous La nitrate and aqueous Ni nitrate in turns as described in our previous work. 7 The resulting precursors were dried and then calcined at 650°C for 3 h. The obtained La–Ni–SZA catalysts with 1 wt% of Ni and 1 wt% of La were denoted as La–Ni–SZA-I and La–Ni–SZA-W for preparation from SZA-I and SZA-W, respectively.
Catalyst characterization
X-ray diffraction (XRD) was carried out on a D/max-2200PC-X-ray diffractometer (Rigaku Corporation, Tokyo, Japan). The typical physico-chemical properties of supports and catalysts were analyzed by the Brunauer–Emmett–Teller (BET) method using Quantachrome micromeritic adsorption desorption equipment model NOVA2000e. Fourier Transform Infrared (FTIR) spectroscopy measurements were carried out with a Spectrum GX FTIR spectrometer. The surface sulfur and Ni contents were analyzed by X-ray photoelectron spectroscopy (XPS) using an ESCALAB MKII spectrometer.
Catalytic reaction
The isomerization of n-pentane was performed in a fixed-bed flow reactor (inner diameter 10 mm, length 600 mm). A quantity of catalyst (3.0 g, 16–32 mesh) was loaded into the isothermal region of the reactor tube for all reaction tests. Prior to the reaction, the catalyst was activated in a hydrogen stream at 300°C for 3 h, and then cooled down to the reaction temperature. A dose of n-pentane was passed over the activated catalyst under the following reaction conditions: reaction pressure of 2.0 MPa, molar H2/n-pentane ratio of 4:1, and weight hourly space velocity (WHSV) of 1.0 h−1. The products were analyzed by an online FL9790 gas chromatograph (Shimadzu GC-14C) equipped with a flame ionization detector.
Results and discussion
XRD
The XRD patterns of SZA-I, La–Ni–SZA-I, SZA-W, and La–Ni–SZA-W catalysts are shown in Figure 1. Evidently, the diffraction peaks of the tetragonal ZrO2 phase at 2θ = 30.3°, 35.1°, 50.4°, and 60.3° were detected in both samples. For SZA-I and La–Ni–SZA-I, no peaks assigned to the monoclinic ZrO2 phase could be observed, 8 while for SZA-W and La–Ni–SZA-W, peaks of the monoclinic ZrO2 phase at 2θ = 24.2°, 28.1°, and 31.3° could be seen clearly. This result indicates that sulfated ZA obtained by the incipient-wetness impregnation method is more likely to cause the transformation of tetragonal ZrO2 phase to the monoclinic ZrO2 phase. Mercera et al. 9 have proposed that the ZrO2 phase is affected by crystallite size. Crystallites smaller than the critical size result in tetragonal zirconia. However, transformation from tetragonal to monoclinic crystallites occurs as crystallite size becomes larger than the critical size. They suggested that the critical crystallite size after calcination at 650°C is within the range 90–123 Å. The mean particle diameters (Dc) based on the tetragonal crystal of ZrO2 calculated from Scherrer’s equation for the four catalysts are shown in Table 1. As shown in Table 1, the crystallite sizes of SZA-W and La–Ni-SZA-W are 9.1 and 9.4 nm, respectively, which are slightly larger than the critical crystallite size. This is the possible reason for the presence of the monoclinic ZrO2 phase in this catalyst. The crystallite sizes of SZA-I and La–Ni–SZA-I are 7.5 and 7.7 nm, respectively, which are much smaller than those corresponding to SZA-W and La–Ni–SZA-W. The smaller size of tetragonal ZrO2 crystals would lead to higher isomerization activity, and this will be further discussed in the “Catalytic activity and stability” section.
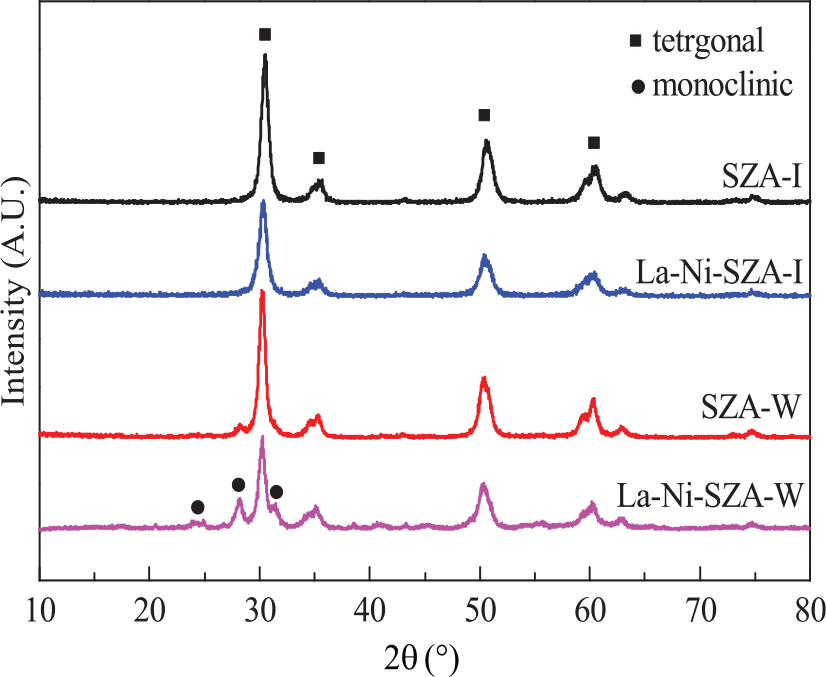
XRD patterns of La–Ni–SZA-I and La–Ni–SZA-W catalysts.
Textural properties of La–Ni–SZA-I and La–Ni–SZA-W catalysts.
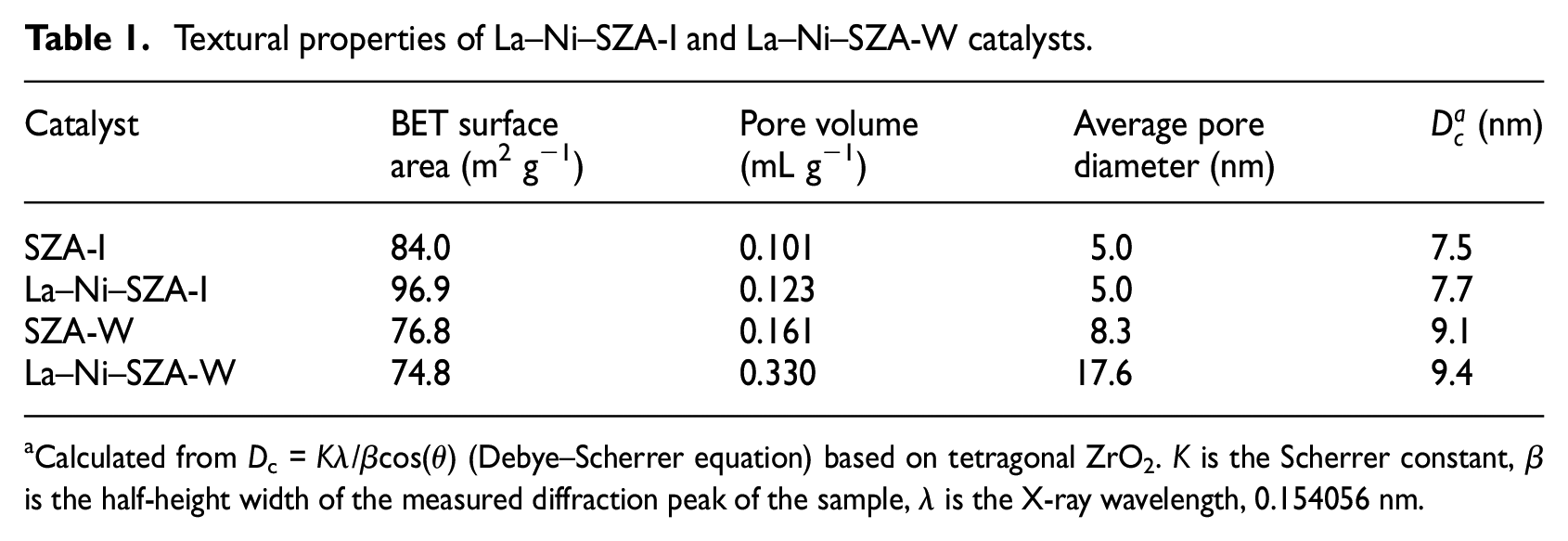
Calculated from Dc = Kλ/βcos(θ) (Debye–Scherrer equation) based on tetragonal ZrO2. K is the Scherrer constant, β is the half-height width of the measured diffraction peak of the sample, λ is the X-ray wavelength, 0.154056 nm.
BET
The surface areas obtained by N2 adsorption and the BET method are shown in Table 1. The BET surface area of SZA-I was 84.0 m2 g−1. Compared with the SZA-I catalyst, the BET surface area of the SZA-W catalyst showed a slight decrease from 84.0 to 76.8 m2 g−1. This showed that the incipient-wetness impregnation method, which is beneficial to loading more sulfur species on the catalyst surface would give a low surface area. It should be noted that the SZA-W catalyst showed a much larger pore size than that of SZA-I, showing that the two SZAs, as prepared with different methods, possessed totally different pore structures even though the compositions were essentially the same. The BET surface area, pore volume, and pore diameter of the La–Ni–SZA-I catalyst were 96.9 m2 g−1, 0.123 mL g−1, and 5.0 nm, respectively. Compared with the La–Ni–SZA-I catalyst, the BET surface area of the La–Ni–SZA-W catalyst showed a dramatic decrease from 96.9 to 74.8 m2 g−1. However, the pore volume (0.330 mL g−1) and pore diameter (17.6 nm) of the La–Ni–SZA-W catalyst were both significantly (2.68 and 3.52 times) larger than those of the La–Ni–SZA-I catalyst. The results revealed that the pore structures of the La–Ni–SZA-W catalyst were also entirely different from those of the La-Ni-SZA-I catalyst because of the different sulfating methods.
Figure 2 shows the pore-size distributions of La–Ni–SZA-I and La–Ni–SZA-W catalysts. The unimodal Barrett–Joyner Halenda (BJH) pore-size distribution curves for the La–Ni–SZA-I and La–Ni–SZA-W catalysts were observed. It can be seen that the peak of the La–Ni–SZA-W catalyst was obviously shifted to the right compared with that of the La–Ni–SZA-I catalyst, and the La-Ni-SZA-W catalyst with high sulfur content (see Table 3) has a larger pore size. This phenomenon can be explained as follows: during the sulfating process, the sulfate reacts with Zr to form Zr(SO4)2, which possesses small pores. Aside from being a structural promoter, the sulfate group is also a textural promoter for ZrO2; sintering of ZrO2 crystallite can be suppressed by sulfate groups. 10 Therefore, the more active catalyst with high sulfur content has a larger pore size.
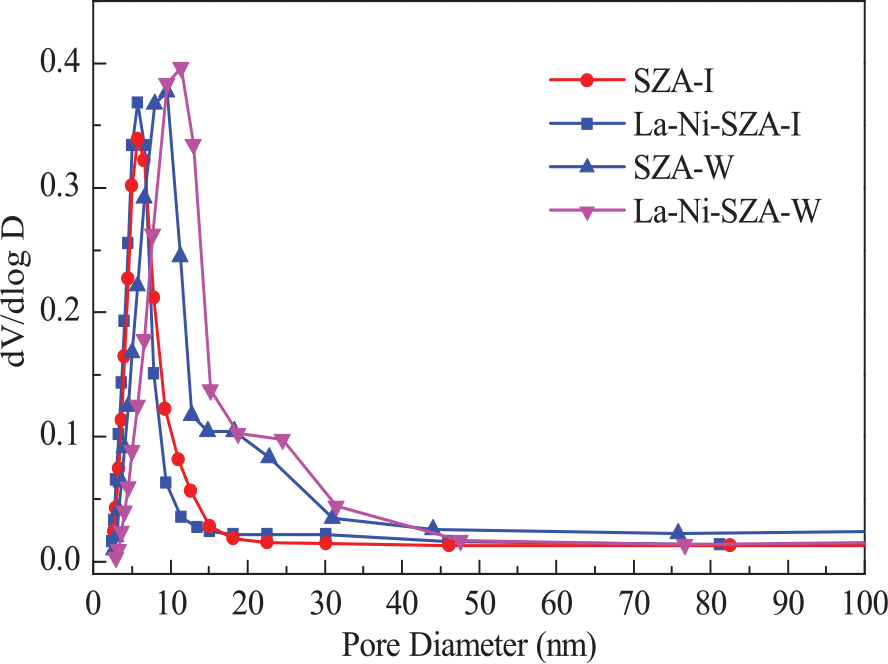
BJH pore-size distributions of La–Ni–SZA-I and La–Ni–SZA-W catalysts.
Figure 3 illustrates the adsorption–desorption isotherms of N2 on SZA-I, La–Ni–SZA-I, SZA–W, and La–Ni–SZA-W catalysts. The nitrogen adsorption–desorption isotherms for all the samples can be classified as type IV isotherms. 11 The appearance of a H2 hysteresis loop revealed that all the samples have a major mesoporous structure. 12 The SZA-I and La-Ni-SZA-I catalysts showed a sudden increase in absorption for P/P0 = 0.5–0.6, which indicates that the mesopores were distributed fairly uniform. In addition, the increasing tendencies of these two samples are basically the same, indicating the similarity of the pore sizes. The amounts of adsorption of nitrogen for the SZA-W and La–Ni–SZA-W catalysts were larger than those for the SZA-I and La–Ni–SZA-I catalysts, which indicated that the SZA-W and La–Ni–SZA-W catalysts had larger pore volumes. The change of pore structure could lead to the improvement of catalyst stability.
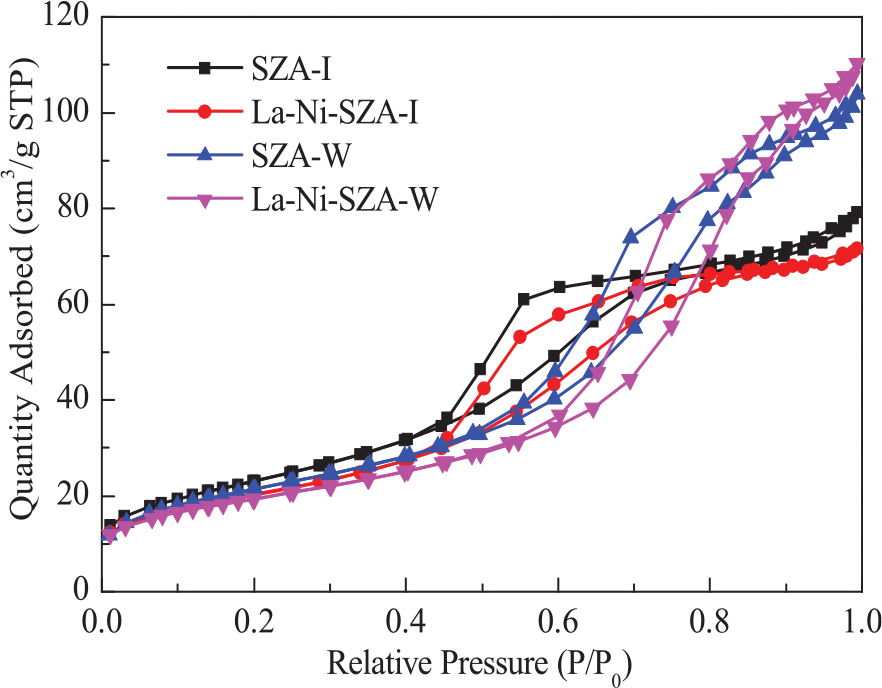
Adsorption–desorption isotherms of SZA-I, La–Ni–SZA-I, SZA-W, and La–Ni–SZA-W catalysts.
FTIR
Figure 4 shows the FTIR spectra of the solid super acid catalysts SZA-I, La–Ni–SZA-I, SZA-W, and La–Ni–SZA-W. A strong and broad band in the 3600–3000 cm−1 region can be assigned to physisorbed and coordinated water. Also, the broad band at 1634 cm−1 can be assigned to the bending mode (∠HOH) of coordinated water. 13 The bands at 1064 and 1128 cm−1 can be assigned to the symmetric O–S–O stretching mode of bidentate persulfate ions coordinated to the metal ion. The band at 1264 cm−1 corresponds to the antisymmetric O=S=O stretching peak of persulfate ions under the condition of dehydration. The characteristic vibrational peaks of S–O and S=O bonds were observed in all samples, which confirms the presence of the solid superacid structures. The intensities and extent of splitting of these three peaks reveal the dispersion of S2O82– on the metal oxide surface and the amount of acid sites on the surface of the catalyst. As compared with the SZA-I and La–Ni–SZA-I catalysts, the intensities and extents of splitting these three peaks of the corresponding SZA-W and La–Ni–SZA-W catalysts were stronger, indicating that the samples obtained by the incipient-wetness impregnation method are more likely to yield a better dispersion of S2O82– and more and stronger acid sites on the surface of these catalysts. To confirm this, analyses of the acid type, acid strength, and acid content of the samples were performed.
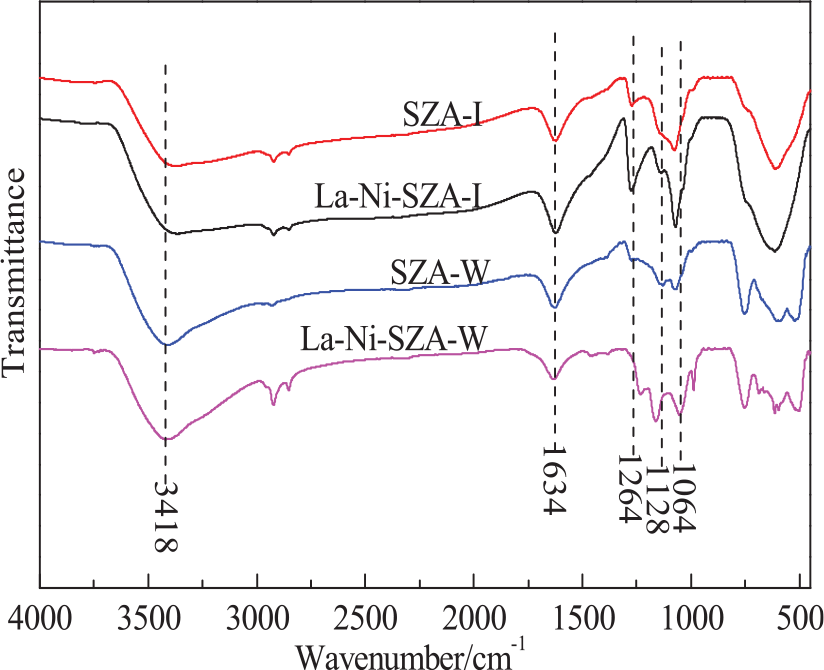
FTIR spectra of SZA-I, La–Ni–SZA-I, SZA-W, and La–Ni–SZA-W catalysts.
Pyridine adsorption-infrared spectroscopy
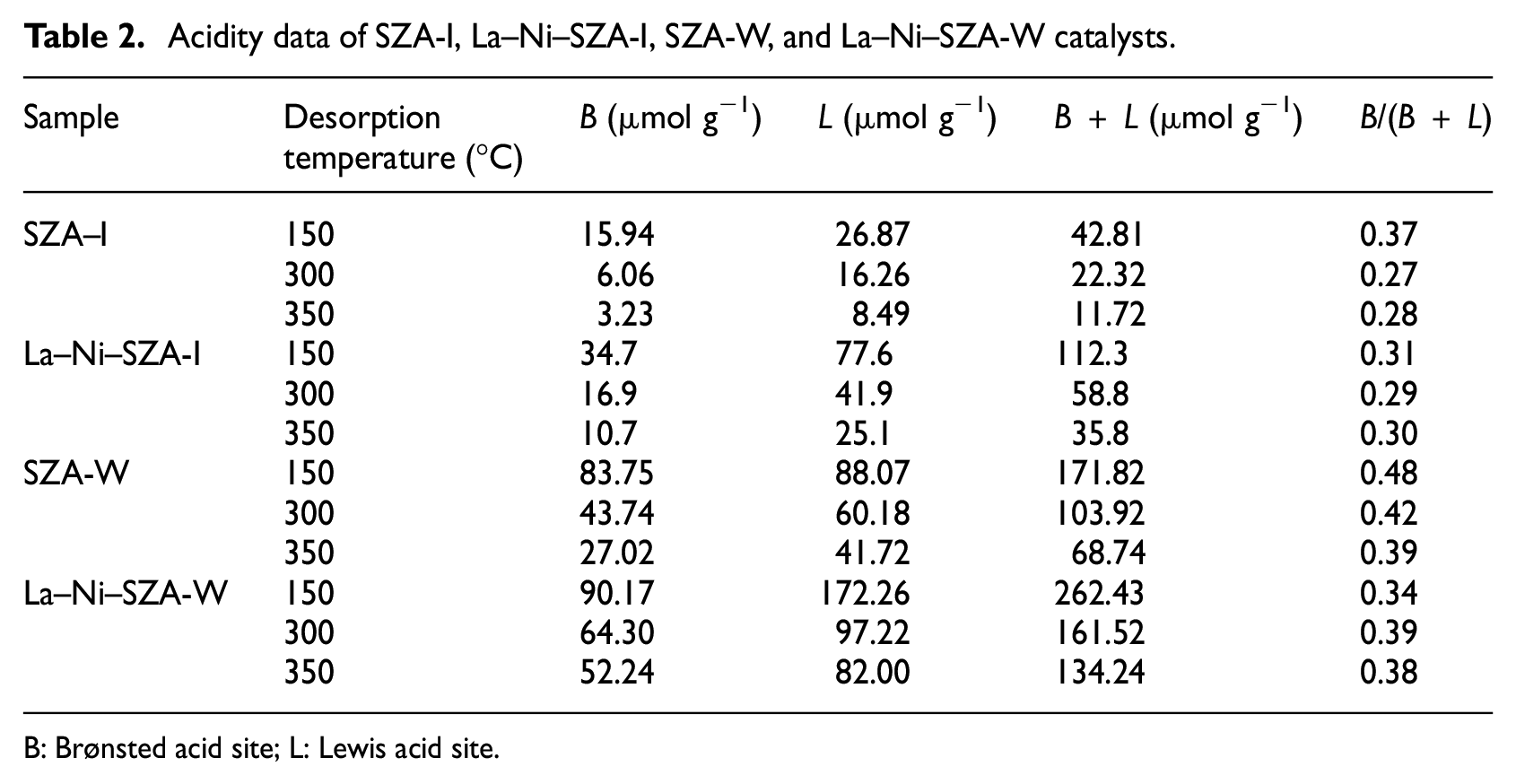
Pyridine adsorption-infrared (Py-IR) spectroscopy is a very effective method for the characterization of catalyst acid type, acid strength, and acid content. 2 Table 2 lists the acidity distribution for catalysts calculated from the IR spectra of pyridine desorption. Data from all samples revealed that a much larger number of Lewis acid (L) sites than Brønsted acid (B) sites were formed. 6 As compared with the SZA-I and La–Ni–SZA-I catalysts, the amounts of B and L sites on the corresponding SZA-W and La–Ni–SZA-W catalysts were much higher, especially for the stronger B sites, which leads to a substantial increase in the B/(B + L) value. This indicates that the samples obtained by the incipient-wetness impregnation method are beneficial to obtaining more and stronger acid sites, especially for the stronger B sites. It was reported that the Brønsted acidity, especially strong Brønsted acidity, plays an important role in isomerization and enhances the catalysts’ stability in isomerization. 14 Therefore, one can deduce that the La–Ni–SZA-W catalyst is more likely to show a better stability than the La–Ni–SZA-I catalyst.
Acidity data of SZA-I, La–Ni–SZA-I, SZA-W, and La–Ni–SZA-W catalysts.
B: Brønsted acid site; L: Lewis acid site.
XPS
XPS characterization was carried out to examine the chemical state changes of sulfur species of the catalysts (Figure 5). The La–Ni–SZA-I and La–Ni–SZA-W catalysts showed peaks belonging to S6+ species at binding energies of 168.9 and 169.9 eV, respectively. 15 The acidic strength of catalysts can be estimated from the binding energy of S6+ species. The La–Ni–SZA-W catalyst possessed a higher binding energy than that of the La–Ni–SZA-I catalyst, which indicates the interaction between sulfates and ZA substrate or metal becomes stronger. The La–Ni–SZA-W catalyst, with a higher binding energy of S6+ species has the higher acid strength, which is consistent with the results of the Py-IR analysis.
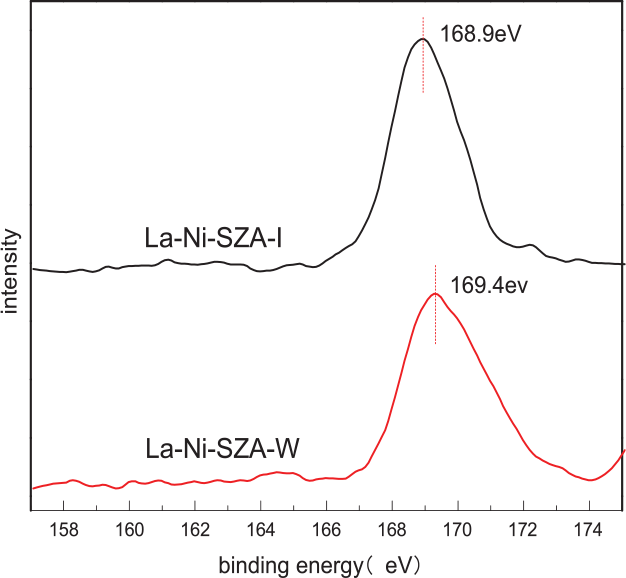
Comparison of S 2p XPS profiles of La–Ni–SZA-I and La–Ni–SZA-W catalysts.
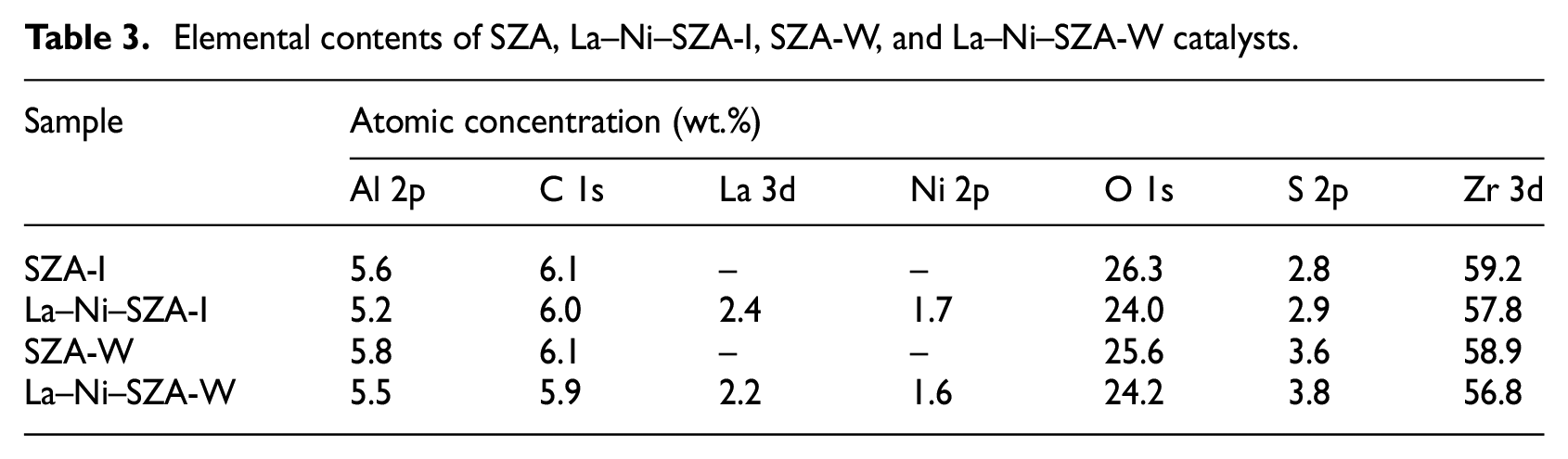
Based on XPS measurements, the surface elemental contents of SZA-I, La–Ni–SZA-I, SZA-W, and La–Ni–SZA-W catalysts are summarized in Table 3. 3 The carbon detected by XPS was adventitious carbon, which is typically introduced from carbon compounds existing in the environment under testing conditions. It can be seen that the SZA-W and La–Ni–SZA-W catalysts, which were sulfated by the incipient-wetness impregnation method, showed much higher sulfur contents on the surface than the corresponding SZA-I and La–Ni–SZA-I catalysts, which were sulfated by the normal impregnation method. The more stabilized sulfur on the surface of the SZA-W and La–Ni–SZA-W catalysts indicated more acid sites on these catalysts, which is consistent with results of FTIR and Py-IR analyses. However, compared with the La–Ni–SZA-I catalyst, the La and Ni contents of the La–Ni–SZA-W catalyst decreased slightly. This can be explained by the increase of sulfur element content which would lead to more active acid sites on the surface of the catalyst and therefore reduce the active metal sites on the catalyst surface.
Elemental contents of SZA, La–Ni–SZA-I, SZA-W, and La–Ni–SZA-W catalysts.
Catalytic activity and stability
The La–Ni–SZA-I and La–Ni–SZA-W catalysts were tested for the isomerization of n-pentane at a pressure of 2.0 MPa, a molar H2/n-pentane ratio of 4:1, and a WHSV of 1.0 h−1.
Figure 6 shows that the yield and selectivity for isopentane in the isomerization process vary with temperature. For both the samples, the yield of isopentane first increased and then decreased with increase of temperature. At lower reaction temperatures, both samples exhibited excellent selectivity. However, the selectivity began to decline as the temperature rose. The highest isopentane yields for the La–Ni–SZA-I and La–Ni–SZA-W catalysts were obtained at the optimum reaction temperatures of 220°C and 280°C, respectively. In addition, as compared with the La–Ni–SZA-I catalyst (yield 65%, selectivity 96%), the optimum yield for the La–Ni–SZA-W catalyst (yield 56%, selectivity 88%) was low owing to its low selectivity at higher temperature. The low temperature isomerization activity of the La–Ni–SZA-W catalyst prepared by incipient-wetness impregnation was low as compared with the La–Ni–SZA-I catalyst prepared by normal impregnation. This is possibly due to the smaller crystal size of tetragonal ZrO2, lower surface area, and metal site content compared with the La–Ni–SZA-W catalyst.
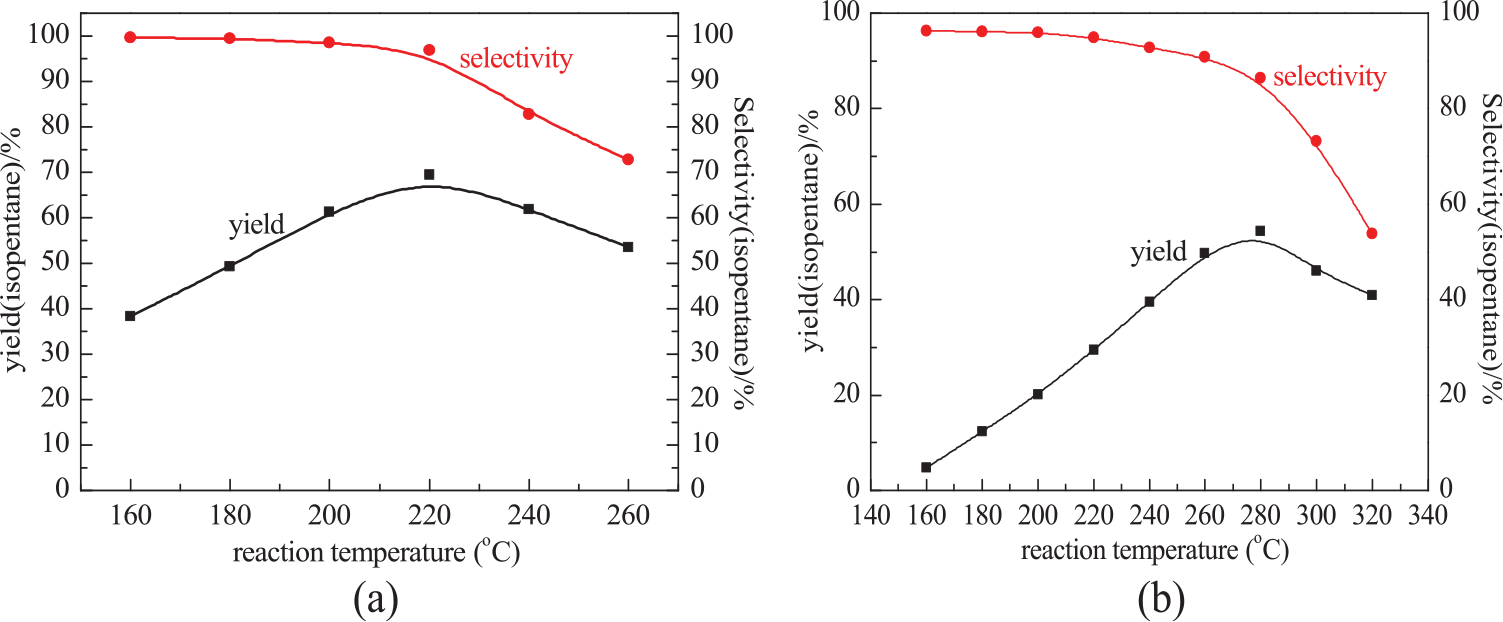
Catalytic activity and selectivity of (a) La–Ni–SZA-I and (b) La–Ni–SZA-W catalysts.
The stability and selectivity of the catalytic isomerization at optimum temperature were tested. Figure 7(a) shows that the yield of isopentane for the La–Ni–SZA-I catalyst remained above 65% with an isopentane selectivity of 96% within 1000 min. After 1000 min, the yield of isopentane decreased more rapidly and reached 20% at a reaction time of 3000 min. Figure 7(b) shows that the catalytic activity for the La–Ni–SZA-W catalyst was maintained at around 56% within 3000 min with an isopentane selectivity of 88%. This indicated that the incipient-wetness impregnation sulfating method was beneficial to obtaining a catalyst with great stability, although the initial activity was low. The improved stability of the catalyst can be explained due to the (1) large suppression of carbon deposition. The pore size and volume of the La–Ni–SZA-W catalyst were 17.6 nm and 0.330 cm3 g−1, respectively (Table 1). The large pore size and pore volume of the La–Ni–SZA-W catalyst promoted the diffusion of the reactant, and thus reduced the residence time of hydrocarbon compounds on the catalyst surface. Thus, the deposition of carbon may be largely suppressed. Therefore, the La–Ni–SZA-W catalyst sulfated by incipient-wetness impregnation effectively resisted deactivation, since carbon deposition is one of the main reasons for deactivation of catalysts of this kind; and (2) it is known that the acidity of catalysts plays an important role in isomerization. The acid sites can promote the formation of carbonium ions from light hydrocarbons. The greater number of and stronger B sites for the La–Ni–SZA-W catalyst (Figure 4 and Table 2) were guaranteed to provide enough acid sites for isomerization during the reaction process.
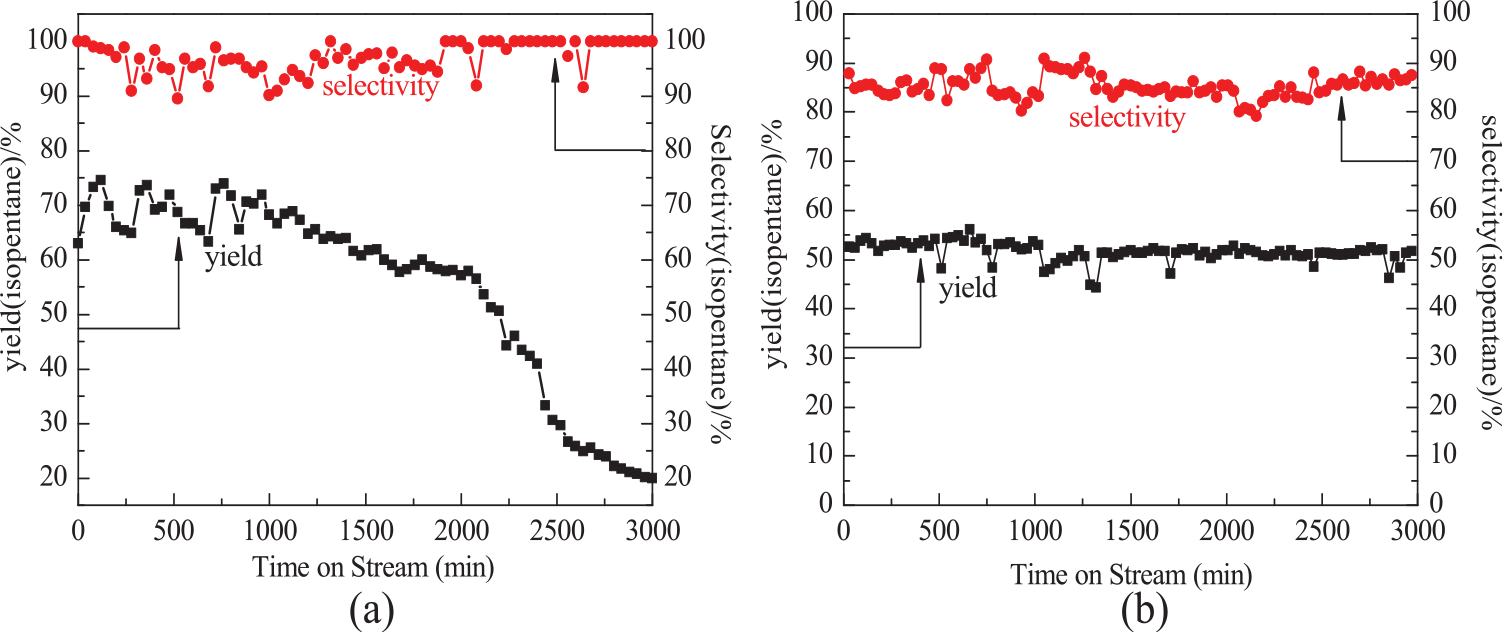
Stability and selectivity of (a) La–Ni–SZA-I and (b) La–Ni–SZA-W catalysts.
Conclusion
La–Ni–SZA catalysts were successfully prepared by two different sulfating methods. The results showed that the pore structures of the La–Ni–SZA-W catalyst were entirely different from those of the La–Ni–SZA-I catalyst because of the different sulfating methods. The surface area of the La–Ni–SZA-W catalyst, prepared by incipient-wetness impregnation was lower than that of the La–Ni–SZA-I catalyst. However, the pore volume (0.330 cm3 g−1) and pore diameter (17.6 nm) of the La–Ni–SZA-W catalyst were both significantly (2.68 and 3.52 times) larger than those of the La–Ni–SZA-I catalyst. As compared with the La–Ni–SZA-I catalyst, the amounts of B and L sites of the La–Ni–SZA-W catalyst were both much higher, especially for the stronger B sites. This indicates that the samples obtained by incipient-wetness impregnation were beneficial in obtaining more and stronger acid sites, especially for the stronger B sites. In addition, the XPS analysis also revealed that the SZA-W and La–Ni–SZA-W catalysts, sulfated by incipient-wetness impregnation, showed much higher sulfur contents on the surface than the corresponding SZA-I and La–Ni–SZA-I catalysts, which were sulfated by the normal impregnation method. The more stabilized sulfur on the surface of the SZA-W and La–Ni–SZA-W catalysts indicated more acid sites on these catalysts.
The catalytic activity for the La–Ni–SZA-W catalyst was maintained at around 56% within 3000 min with an isopentane selectivity of 88% which showed much greater stability than that of the La–Ni–SZA-I catalyst. The improved stability of the La–Ni–SZA-W catalyst can be explained by the fact that (1) carbon deposition was largely suppressed. The large pore size and pore volume of the La–Ni–SZA-W catalyst promoted the diffusion of the reactant, and thereby reduced the residence time of hydrocarbon compounds on the catalyst surface. Thus, the deposition of carbon may have been largely suppressed. Therefore, the La–Ni–SZA-W catalyst sulfated by incipient-wetness impregnation effectively resisted deactivation, since carbon deposition is one of the main reasons for deactivation of catalysts of this kind and (2) the more numerous and stronger B sites for the La–Ni–SZA-W catalyst (Figure 4 and Table 2) were guaranteed to provide enough acid sites for isomerization during the reaction process.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
