Abstract
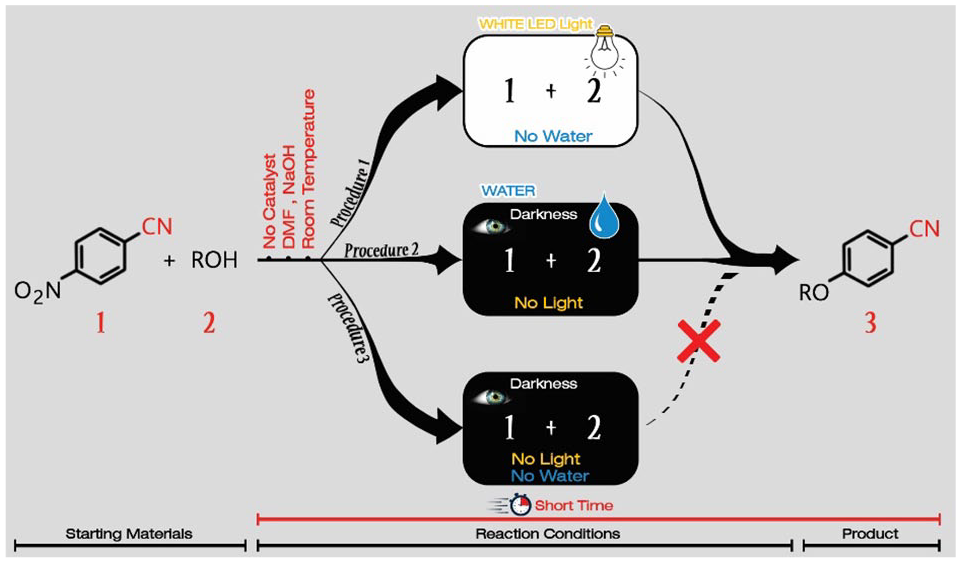
In this study, we have developed a catalyst-free procedure for denitrative etherification of electron-deficient nitroarenes. In this method, the reaction failed in the dark but was enabled by white light-emitting diode light (6 W) in the presence of NaOH/dimethylformamide at room temperature with short reaction times. Interestingly, the reaction in the dark is completed almost immediately once a small quantity of water is added to the reaction mixture. Ultraviolet irradiation (λ = 254 nm) was not effective for this reaction to proceed.
Keywords
Herein, we wish to report an effective procedure for denitrative photoetherification of 4-nitrobenzonitrile under catalyst-free conditions.
Introduction
In recent years, nitroarenes have been used in cross-coupling reactions as alternatives to aryl halides. 1 Using these compounds as the starting materials, we have developed three efficient approaches for the synthesis of aromatic sulfides under copper catalysis. 2 In addition to this report, conditions have been developed by our group and others for construction of C–O,3–8 C–N, 9 C–C,10,11 and C–S12,13 bonds in the presence of transition-metal catalysts. Apart from these metal-catalyzed reactions, some metal-free procedures are also available in the literature.14,15 One of the earliest observations for a denitrative etherification reaction under metal-free conditions was during studies of the reaction of N-substituted nitrophthalimides with sodium methoxide. When methyl 3-nitrophthalimidoacetate was treated with sodium methoxide in refluxing methanol, a displacement of the nitro group with methoxide occurred. 16 In addition, Beck has reported the synthesis of benzo[b]thiophene derivatives under metal-free conditions. The first step of this process was suggested to involve a denitrative C–S bond formation reaction.17–19 Displacement of activated nitro groups with various nucleophiles for construction of C–O, C–S, and C–C has also been reported in hexamethylphosphoramide (HMPA) at room temperature. 20 In continuation of our studies of denitrative functionalization reactions, we noticed that the metal-free reactions do not progress in the dark showing the involvement of a photon-catalyzed process in these reactions. In the above mentioned reports, such an effect was not considered for these reactions.
Results and discussion
We started our studies on the reaction of 4-nitrobenzonitrile with ethanol under white light-emitting diode (LED; 6 W) irradiation at room temperature (Table 1). The reaction failed in the presence of either 1,8-diazabicyclo(5.4.0)undec-7-ene DBU, nPr3N, KF, or K2CO3 as the base in dimethylformamide (DMF) after 24 h (entries 1–4). A low yield of the product was obtained in the presence of K3PO4 as the base (entry 5). Interestingly, by switching the base to KOH, the yield of the desired product increased drastically to 80% within 20 min (entry 6). A further increase in the yield up to 97% was observed when using NaOH as the base with a 10-min reaction time, and we selected these as the optimized conditions (entry 7). Dimethyl sulfoxide (DMSO) was also found to be an effective solvent for this reaction (entry 8). However, polyethylene glycol (PEG), EtOH, CH3CN, and water were not suitable solvents (entries 9–12). To our surprise, when we added CuI and 1,10-phenanthroline to the standard conditions and raised the temperature up to 100 °C, the reaction failed even after a 24-h reaction time (entry 13). In addition, when the optimized conditions (entry 7) were carried out in the absence of LED light, no product was obtained after 24-h reaction time (entry 14). This observation shows the importance of the white LED irradiation in this reaction. When we increased the temperature to 100 °C, the reaction gave the desired product in 80% yield in the dark within 12 h (entry 15). We were surprised to observe that the reaction in the dark using 96% EtOH instead of absolute EtOH proceeded very quickly, giving a yield comparable to the optimized conditions (entry 16). To clarify the effect of water, we conducted the standard reaction using absolute ethanol and we added 2 mmol of water to the reaction. Again, a 97% of the desired product was isolated from the reaction mixture within 10 min (entry 17). The reaction failed under ultraviolet (UV) irradiation (λ = 254 nm) after a 30-min reaction time (entry 18). In the next experiment, we mixed all the starting materials in a flask and exposed it to sunshine. When we carried out the reaction under these conditions, the product was obtained in a 80% yield within a 60-min reaction time (entry 19). Hence, the best conditions for the reaction of 4-nitrobenzonitrile with absolute ethanol were using NaOH as the base, DMF as the solvent under white LED irradiation at room temperature (entry 7).
Optimization of the reaction conditions. a
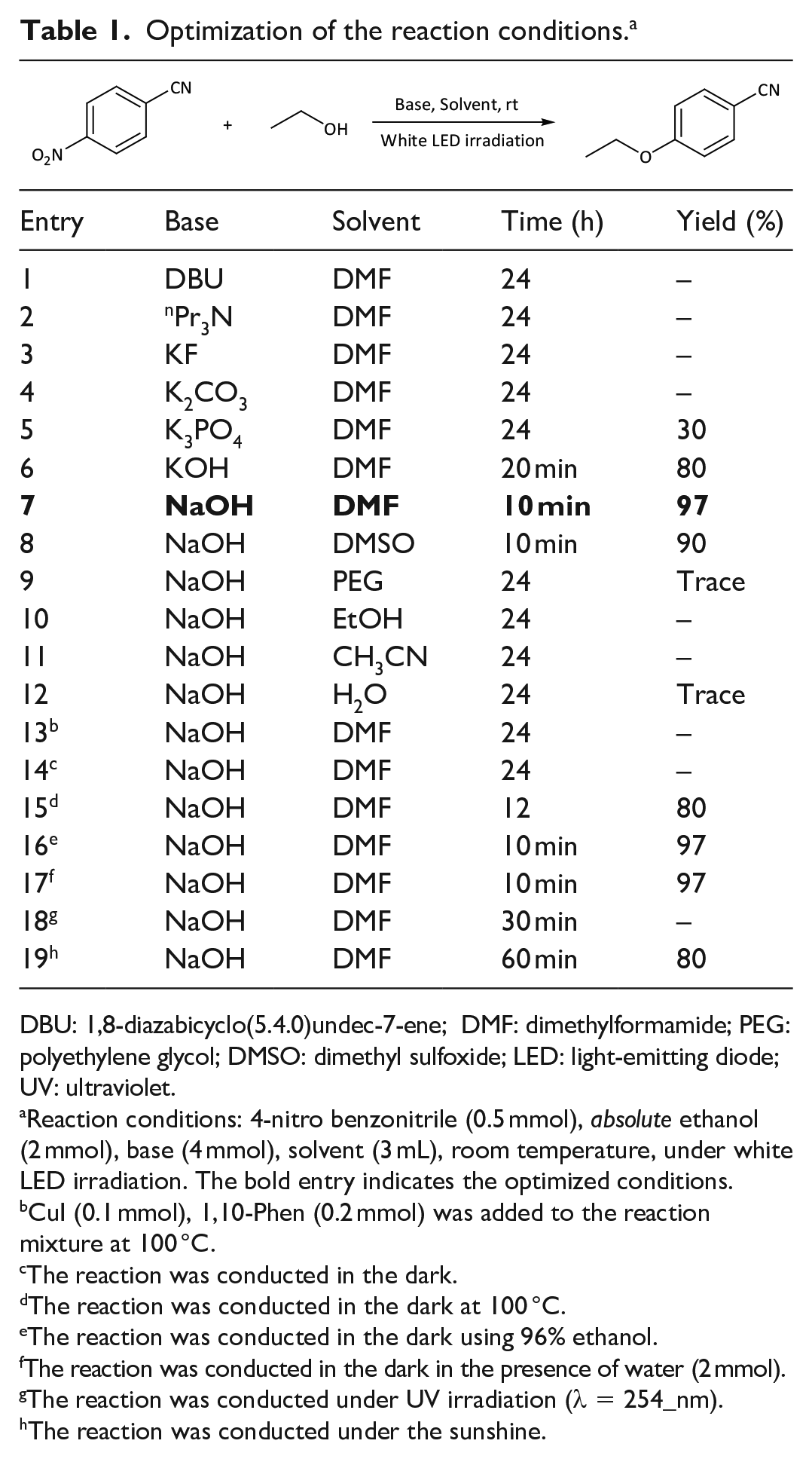
DBU: 1,8-diazabicyclo(5.4.0)undec-7-ene; DMF: dimethylformamide; PEG: polyethylene glycol; DMSO: dimethyl sulfoxide; LED: light-emitting diode; UV: ultraviolet.
Reaction conditions: 4-nitro benzonitrile (0.5 mmol), absolute ethanol (2 mmol), base (4 mmol), solvent (3 mL), room temperature, under white LED irradiation. The bold entry indicates the optimized conditions.
CuI (0.1 mmol), 1,10-Phen (0.2 mmol) was added to the reaction mixture at 100 °C.
The reaction was conducted in the dark.
The reaction was conducted in the dark at 100 °C.
The reaction was conducted in the dark using 96% ethanol.
The reaction was conducted in the dark in the presence of water (2 mmol).
The reaction was conducted under UV irradiation (λ = 254_nm).
The reaction was conducted under the sunshine.
Having optimized conditions in hand, we used different alcohols for denitrative etherification reactions (Scheme 1). Primary (

The reaction scope for denitrative functionalization of nitroarenes. Reaction conditions: nitrobenzene derivative (0.5 mmol), nucleophile (2 mmol), NaOH (4 mmol), DMF (3 mL), room temperature, under white LED irradiation; b Si(OEt)4 was used as the ethoxide source.
To obtain some information about the reaction pathway, we determined the UV-Vis spectrum of 4-nitrobenzonitrile, a potential chromophore for the reaction (Figure S1). No significant peak was observed in the visible region of the spectrum. This suggests that the utilized LED might cover some area in the near UV region. Alternatively, the visible LED light can be absorbed by the intermediates produced during the course of the reaction.
Although the mechanism of the reaction is not clear to us yet, but based on our experiments and related reports in literature, we proposed a mechanism as shown in Scheme 2. In the first step, ethanol is converted to ethoxide. The presence of water can help NaOH and sodium ethoxide to dissolve in the DMF solution. Then, ethoxide attacks to 4-nitrobenzonitrile to form intermediate
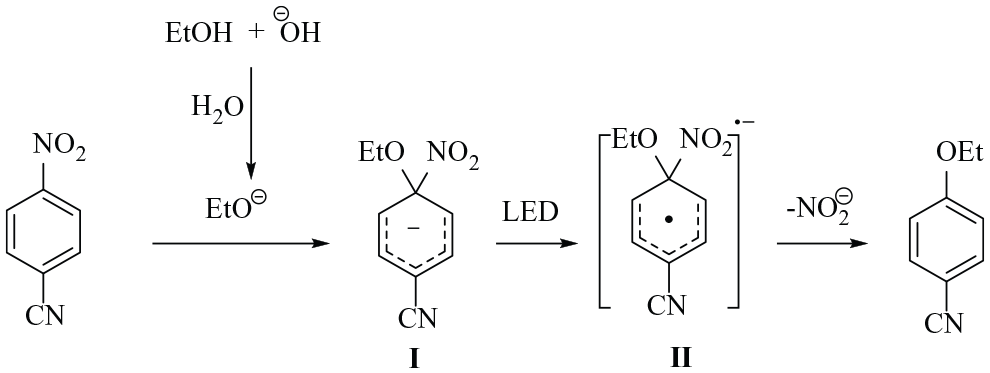
The reaction pathway for denitrative etherification of 4-nitrobenzonitrile.
Conclusion
In summary, we report a novel procedure for the synthesis of ethers using activated nitroarenes as the coupling partners. In this approach, a transition metal-free denitrative etherification of 4-nitrobenzonitrile or ethyl 4-nitrobenzoate is reported in the presence of NaOH under white LED irradiation at room temperature.
Experimental
General
All chemicals were purchased from Merck and Aldrich chemical companies and used as received without further purification. All experiments were run under white LED light and air atmosphere unless stated otherwise. Nuclear magnetic resonance (NMR) data were determined in the Bruker instrument (300 MHz) using CDCl3 as the solvent.
Typical procedure for the denitrative etherification of 4-nitrobenzonitrile with ethanol
A mixture of 4-nitrobenzonitrile (0.5 mmol), NaOH (4 mmol), and absolute ethanol (2 mmol) were mixed in DMF (3 mL) under white LED irradiation in air atmosphere at 25 °C. The progress of reaction was monitored by thin layer chromatography (TLC). After the completion of reaction, the product was extracted with ethylacetate/water. The solvent was evaporated leaving the crude product, which was purified by column chromatography over silica gel using n-hexane/ethylacetate (8:2) as the eluent.
4-Ethoxybenzonitrile (
4-Isopentyloxybenzonitrile (
4-Isopropoxybenzonitrile (
4-sec-butoxybenzonitrile (
4-Cyclohexyloxybenzonitrile (
4-(Prop-2-ynyloxy)benzonitrile (
4-Phenoxybenzonitrile (
Ethyl 4-ethoxybenzoate (
Supplemental Material
Supporting_Information_2 – Supplemental material for Light-emitting diode light–enabled denitrative etherification of 4-nitrobenzonitrile under catalyst-free conditions at room temperature
Supplemental material, Supporting_Information_2 for Light-emitting diode light–enabled denitrative etherification of 4-nitrobenzonitrile under catalyst-free conditions at room temperature by Roghaye Soltani, Fatemeh Abdollahi and Arash Ghaderi in Journal of Chemical Research
Footnotes
Acknowledgements
The authors are thankful to the Iran National Science Foundation and the University of Hormozgan research council. We are also grateful to Abdolvahed Pouryousef for his assistance in this project. Valuable discussion with Professor Hossein Eshghi regarding the mechanism of the reaction is highly appreciated.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study received financial support from the Iran National Science Foundation (INSF; Grant number: 95825781) and University of Hormozgan research council.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

