Abstract
This work reports density functional theory and time-dependent density functional theory calculations of the optimized geometries, electronic structures and optical properties of molecular dyes
Keywords

Introduction
Dye-sensitized solar cells (DSSCs) have received considerable research attention due to the importance to replace nonrenewable and limited energy sources and reduce environmental pollution.1–5 The attraction toward DSSCs is due to their low cost and easy fabrication, unlike other solar cells.6,7 The energy conversion efficiency of the currently used technologies such as silicon solar cells has become high and stable; however, the resulting efficiency does not transfer to reduced costs of the materials and fabrication techniques.8,9 This limits the future application of these technologies. DSSCs are environmentally friendly and cost-effective and might have high power conversion efficiency.9–13 However, more studies are needed to make this practical. DSSCs operate through incorporation of components such as dye sensitizers, mesoporous semiconductors (arguably a metal oxide like TiO2), electrolytes, counter electrodes, and photoelectrodes.14,15
Dye sensitizers play an essential role in attaining a high power conversion efficiency of DSSCs 16 and have received more research attention compared to other components as they are involved in light harvesting, electron conduction, and regeneration process. 9 Dyes used in DSSCs are categorized as metal-free organic and metal complexes.17,18 The former are further classified as natural and synthetic, 19 both being conjugated molecules. This is because conjugated molecules possesses particular properties which make them precise materials for solar cells,20–22 optoelectronic devices,23–25 and transistors. 26 Furthermore, these molecules have benefits of not being amorphous and can be synthesized easily for practical use.27–29 Metal-free organic dyes have been reported to achieve a power conversion efficiency of 17.3%, 30 while metal complexes have been reported to achieve only 13%. 31 Ongoing experimental and theoretical research has reported studies on metal-free organic dyes alternatively to metal-based examples due to the expense and toxicity associated with metal-based dyes. Theoretical studies have gained more interest as they analyze valuable information for use in experimental studies. 32 The information gained from theoretical studies contributes to the rationalization of the properties of known materials and helps to estimate those of unknown compounds33–36 as the basis for any future experimental work.
Many studies have been reported on natural dyes11,37–40 due to their easy availability, cost-effectiveness, and nontoxicity, even though their power conversion efficiency is still low.41–44 Alizarin (1,2-dihydroxy-9,10-anthraquinone) is a previously reported natural dye and one of the first natural dyes to be duplicated synthetically.
45
Alizarin is a prominent red dye most used previously in dyeing textile fabrics. It is naturally derived from the roots of madder plants and was first reported as a synthetic dye in 1869.
45
Apart from other applications, alizarin is confirmed to be applicable in the DSSC technology reaching up to 3.57% conversion efficiency.
46
Despite being one of the oldest dyes, studies on derivatives of alizarin are rarely reported, if any. This paper reports theoretical studies on the substitution of alizarin using 2-hexylthiophene as the donor group based on structures
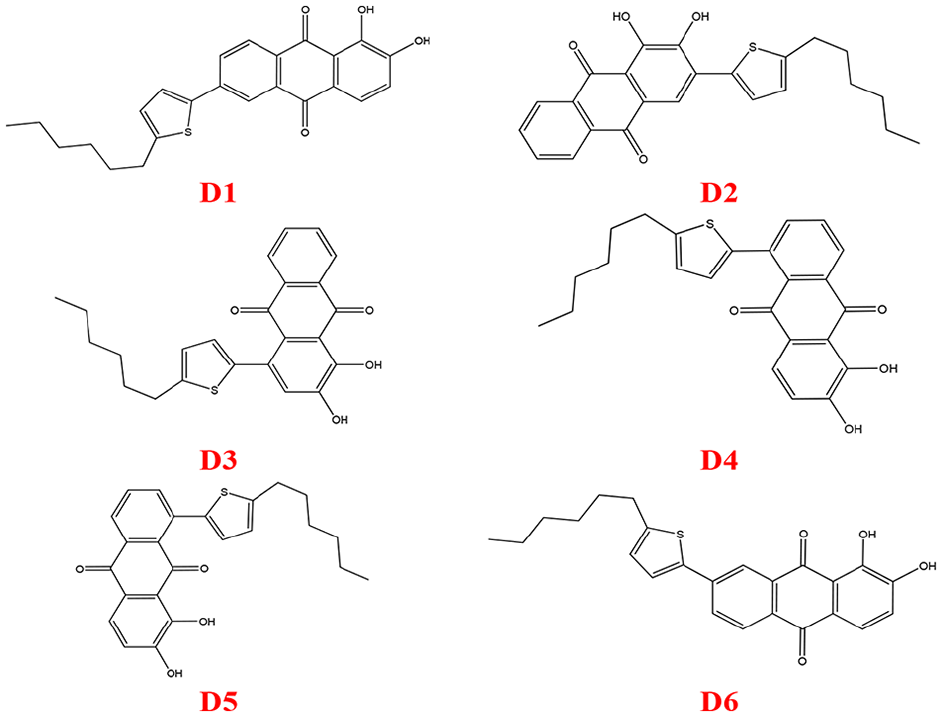
Structures of the dyes considered for the theoretical studies.
Results and discussion
Geometries optimization
Geometry optimization for

Optimized geometries of the considered molecular dyes.
The total energy (ET) values of the optimized molecules
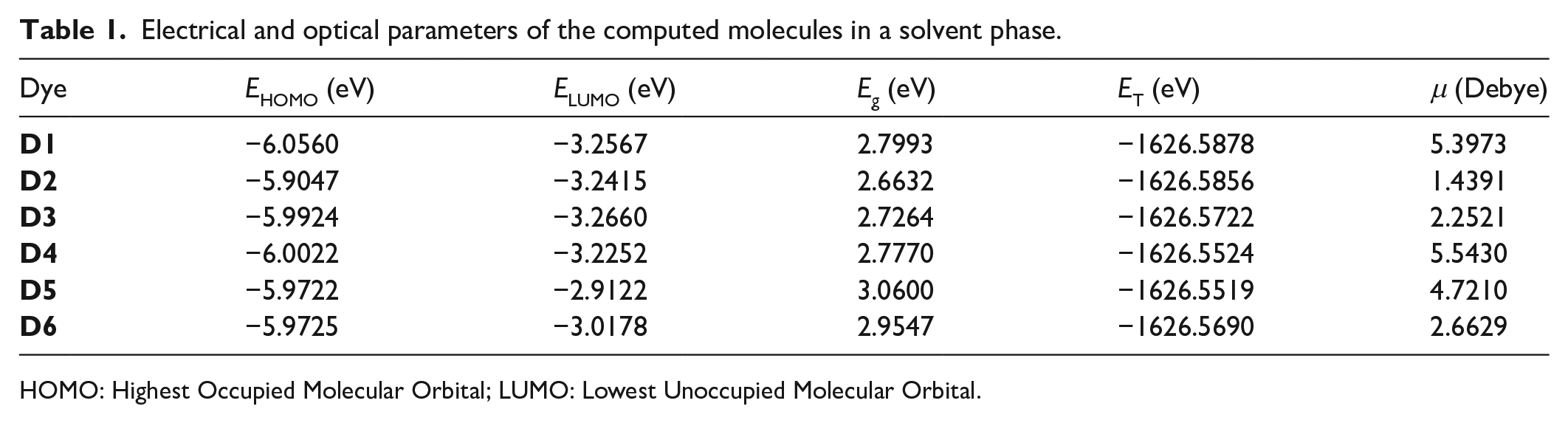
Electrical and optical parameters of the computed molecules in a solvent phase.
HOMO: Highest Occupied Molecular Orbital; LUMO: Lowest Unoccupied Molecular Orbital.
Intramolecular charge transfer
Charge transfer from a donor to an acceptor is one of the important features of metal-free organic dyes as it is used to indicate conduction and regeneration in the cell.
48
It is generally referred to as intramolecular charge transfer (ICT). The investigation of ICT in molecular dyes is possible through the study of HOMO and LUMO energies, which is scientifically referred to as the frontier molecular orbital (FMO) contribution.49,50 The study of these molecular orbital energies is accurately done through the energy gap created by the two, as the energy gap directly affects the photocurrent of the dye molecule. The results of the HOMO/LUMO energy (eV), Eg (eV), ET (eV), and dipole moment (μ) in Debye are presented in Table 1. For conjugated molecules with electron delocalization, HOMOs always show typical aromatic features.51,52 For the dye molecules, HOMOs are concentrated at electron-rich points of the molecules with LUMOs at electron-deficient points.48,53 Thus, HOMO represents the bonding character and LUMO represents the antibonding character of molecular dyes.54,55 The HOMO/LUMO diagrams are presented in Figure 3, while the HOMO/LUMO energies are shown in Figure 4. The strength of the electron-donating group is directly proportional to the HOMO energy level;56,57 a dye molecule with a strong electron-donating group gives a high HOMO energy than examples with weakly donating groups. Considering all the studied dyes, the HOMO energy levels are in the following order:
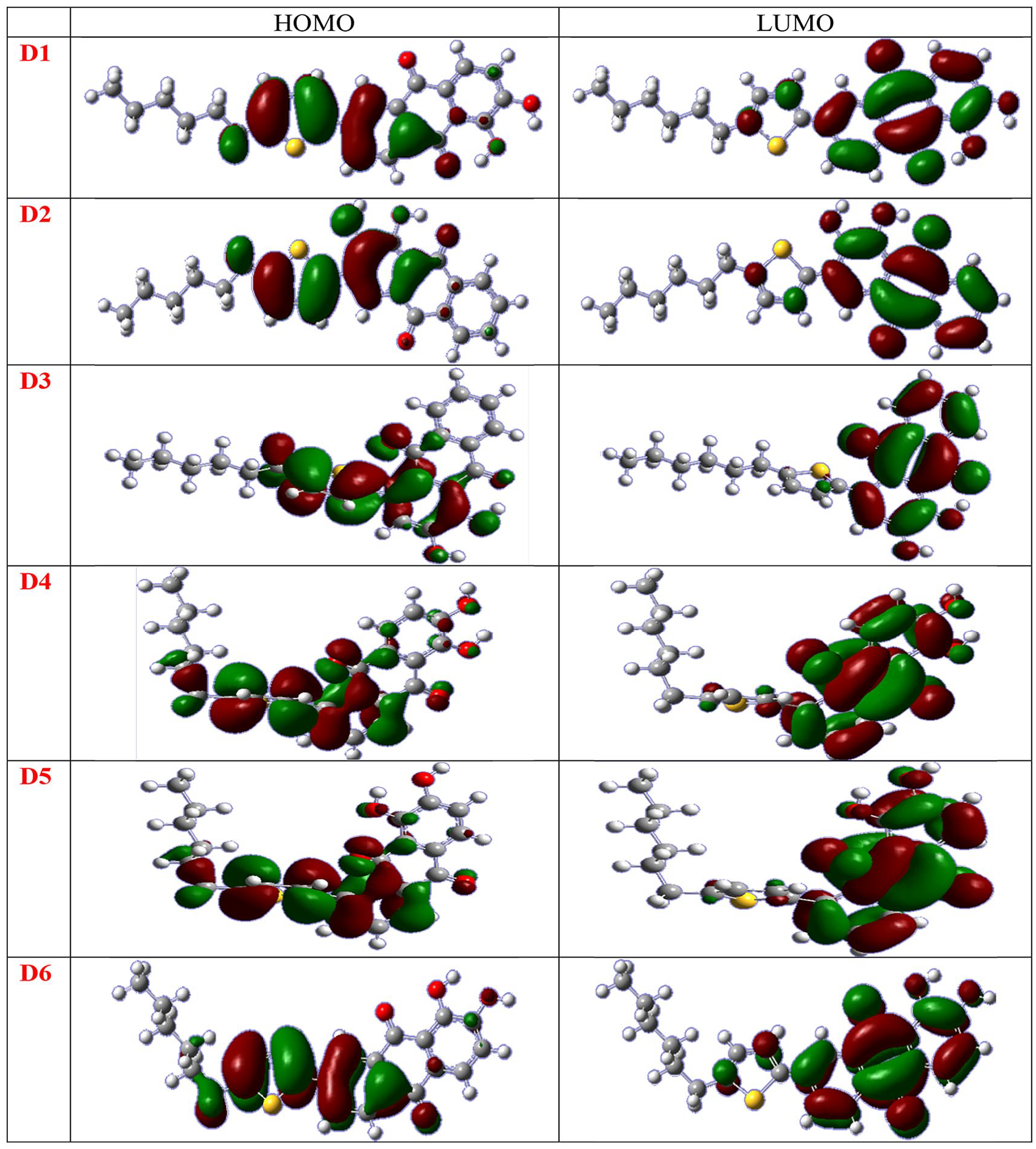
HOMO and LUMO diagrams of the computed dyes.

HOMO and LUMO energies of the computed dyes
As observed in Figure 3, the HOMOs in all the considered molecules are located more in the conjugated unsubstituted ring of the alizarin and thiophene side, thereby showing stronger electron-donating behavior. The LUMOs are located more on the two substituted rings of the alizarin molecules with the carbonyl and hydroxy groups, which indicates that the anchoring group for the dyes is C=O. This could lead to a strong electronic coupling with the semiconductor (TiO2) surface and improve the electron injection efficiency. The higher electron injection power will subsequently enrich the Jsc value of the dyes. As shown in Figure 4, the LUMO energy levels of the studied dyes are higher than those of the TiO2 conduction band edge, which is −4.0 eV.
58
Thus, injection of electrons from the excited dyes to the semiconductor, TiO2, is certain. In addition, the corresponding HOMO energies are lower than that of the electrolyte,
The dipole moments of the dyes are presented in Table 1. The dipole moment indicates the symmetry in the distribution of the electronic charge of the dye molecule. It should be noted that a larger dipole moment is attributed to the asymmetric behavior in the distribution of the electronic charge. This behavior makes a molecule more reactive and sensitive to the external electric field.48,53 From the results in Table 1, the dipole moment of dye
Photovoltaic properties
The parameters to explain the photovoltaic properties of the computed dyes are presented in Table 2. This includes the oxidation potential energy of the dye in the ground state (Edye), the electronic injection free energy (ΔGinject), the light-harvesting efficiency (LHE), the oxidation potential energy of the dye in the excited state (Edye*), and the open-circuit voltage (Voc). The other more important property, short-circuit current (Jsc), is not discussed in this paper as it can only be determined experimentally, instead it is analyzed through LHE and ΔGinject. The ability of the dye to absorb photons from the sun and inject photoexcited electrons from the LUMO to the conduction band is explained through these vital parameters.
Photovoltaic properties of the computed molecules in a solvent phase.
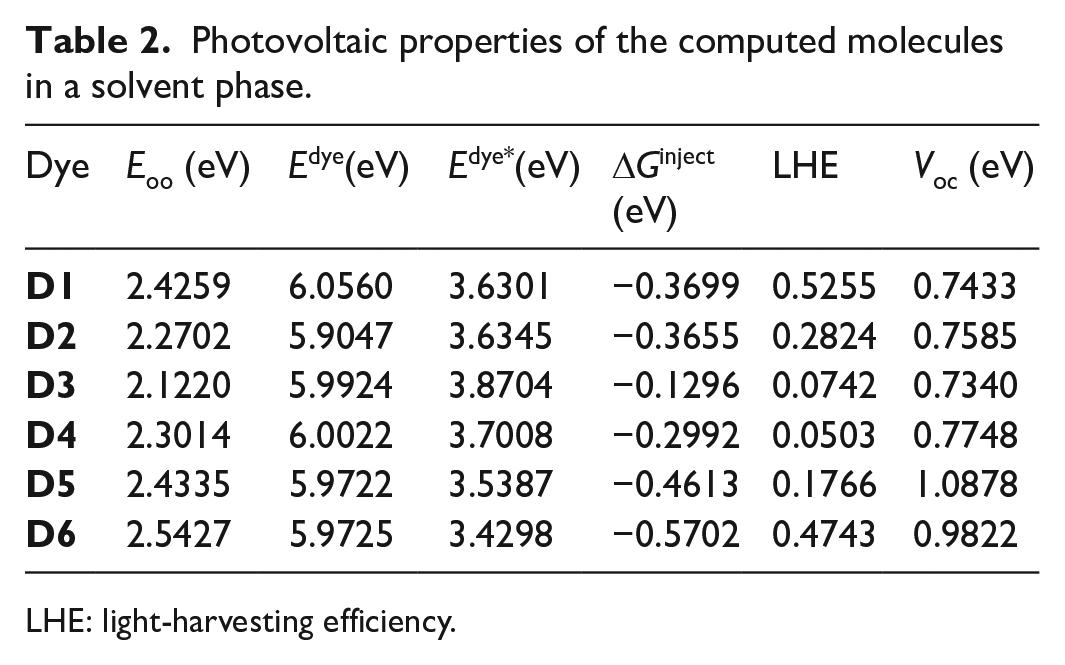
LHE: light-harvesting efficiency.
Considering the results from Table 2, all the computed dyes have negative values of ΔGinject: −0.3699, −0.3655, −0.1296, −0.2992, −0.4613, and −0.5702 eV for
Another parameter which influences the power conversion efficiency (η) of the dye is the open-circuit voltage (Voc). In molecular dyes, a higher value of Voc demonstrates higher electron injection power. From Table 2, the values of Voc for all the computed dyes show insignificant differences in the range of 0.3538 eV. The highest values are observed in
Absorption properties
To analyze the absorption behavior of the dyes, the oscillator strength (f), the vertical excited singlet state and the transition energy level are considered in the gas phase and in acetonitrile solvent (Table 3). In the same media, significant variation is observed between the molecular dyes for the vertical excited energy. In contrast, under the same excitation level, the dyes show insignificant differences between the solvent and gas media. Representatively, the first vertical excited energy decreases in the following order
Absorption properties of the considered dye derivatives in the gas phase and acetonitrile.

HOMO: Highest Occupied Molecular Orbital; LUMO: Lowest Unoccupied Molecular Orbital.
Another parameter presented in Table 3 is the oscillator strength. The relationship between oscillator strength and wavelength is shown in Figures 5 and 6. The oscillator strength of the dyes trends randomly for the reflected excitations. Mostly, the values of the oscillator strengths are seen to be higher in the solvent phase, as represented in Figure 5, than in the gas phase, as shown in Figure 6. From the results in Figures 5 and 6,

Theoretical UV–Vis spectra for the computed molecules in the solvent phase.
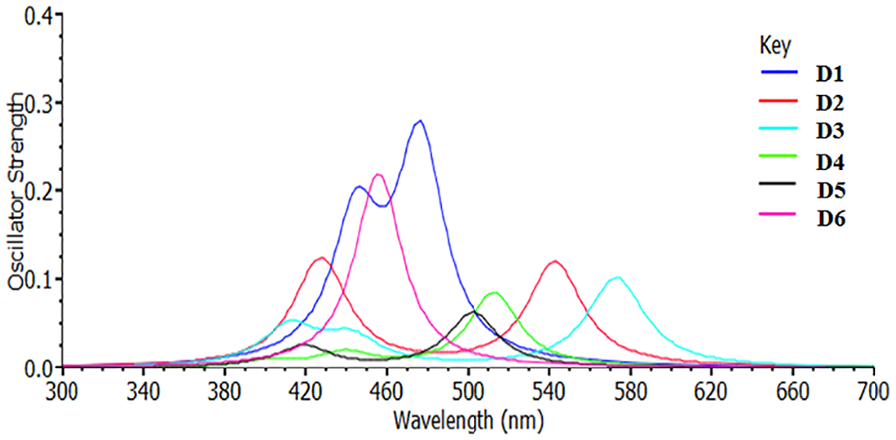
Theoretical UV–Vis spectra for the computed molecules in the gas phase.
Methodology
Theoretical details
To analyze the applicability of the computed dyes in DSSCs, different parameters were calculated using density functional theory (DFT) and time-dependent density functional theory (TD-DFT) methods. One of the crucial parameters in DSSCs is the power conversion efficiency (η), which is obtained using the formula below 60

where FF is the fill factor, Jsc is the short-circuit current density, Voc is the open-circuit voltage, and Pinc is the incident solar power on the cell. Jsc and FF values are experimental parameters, and Jsc is calculated by the following relation 48

where

where f is the oscillator strength of the dye associated with the maximum wavelength (λmax). It may be noted that LHE and f, which are presented in equation (3), are directly related. The increase in oscillator strength influences the increase in LHE and vice versa. The other parameter in the calculation of Jsc, as shown in equation (2), is

where Edye* denotes the oxidation potential energy of the dye in the excited state and ECB denotes the reduction potential of the conduction band of the semiconductor. In this study, we considered TiO2 as a semiconductor, following on from previous works7,62,63 where this semiconductor gave the best performance. Moreover, the Edye* value from equation (4) is calculated using equation (5)7,63,64

where Edye is the ground-state oxidation potential of the dye, which in this work is approximated to be −EHOMO, and E00 is the electronic vertical transition energy corresponding to λmax.
The analysis of electron conduction from dye to semiconductor is related to Voc and the energy of the LUMO (ELUMO) as in the following relation 64

It should be noted that in this work the reduction potential (ECB) for the considered TiO2 semiconductor is −4.0 eV. 58
Computational details
Presented in this work are molecular dyes obtained by substitution of 2-hexylthiophene on the alizarin molecule (see Figure 1). All calculations were performed with the Gaussian 09W package 65 using DFT and TD-DFT methods. The methods used Becke’s three-parameter functional and the Lee–Yang–Parr functional (B3LYP)66–68 coupled with the 6-31G (d,p) basis set. 69 The UV–Vis spectra of the computed dyes were obtained by performing single-point TD-DFT calculations through the use of optimized ground-state geometries. The TD-DFT method was also used to calculate the electronic absorption spectra based on allowed excitations and oscillator strength together with excited-state properties, which allowed derivation of the HOMO and LUMO energy values. The polarizable continuum model (PCM) 70 was used in the calculation performed in the solvent phase; the solvent used is acetonitrile.
Conclusion
This paper has presented a discussion on the sensitizer donor effects of 2-hexylthiophene-substituted alizarin photosensitizers for use in DSCCs using DFT and TD-DFT. Different parameters were considered, including geometric, photovoltaic, absorptional, and charge transfer ability aiming at finding potential sensitizers for use in solar cells. Generally, all the considered dyes showed better performance with insignificant variation, and especially the results of dye
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this paper.
