Abstract
Two new molecules based on 2-(2-alkoxy-1-naphthyl)-2,3-dihydro-1h-perimidine are synthesized. The binding properties are investigated by fluorescence spectroscopy showing that one of the products (

Introduction
Perimidines are tricyclic heterocycles containing a dihydropyrimidine ring
Recently, perimidine derivatives have drawn more extensive examinations because of their diverse range of biological activity.5–8 Because of the continuing interest in this field, investigations into new perimidine derivatives and their biological activities is of great importance. However, the biological activity of perimidine derivatives is mostly focused on applications in the biomedical field, while little attention has been paid to their application in the recognition field.9,10
The dihydropyrimidine ring present in the structure of perimidine compounds can provide metal ion recognition sites. Based on this, we have introduced additional oxygen atoms and nitrogen atoms to improve the recognition ability of such compounds towards metal ions.11,12 We now report the synthesis of perimidine-containing ethyl 2-((1-(2,3-dihydro-1h-perimidin-2-yl)naphthalen-2-yl)oxy)acetate (
Results and discussion
The synthesis of the new perimidine derivatives

Synthetic route to

The crystal structure of compound

(a) View of
Crystals of
The binding properties of molecules
Changes in the fluorescence properties of

(a) Fluorescence emission changes of
The sensitivity of the fluorescence emission response of

(a) Fluorescence emission spectra (excitation at 365 nm) of
A proposed mechanism for the fluorescence enhancement of
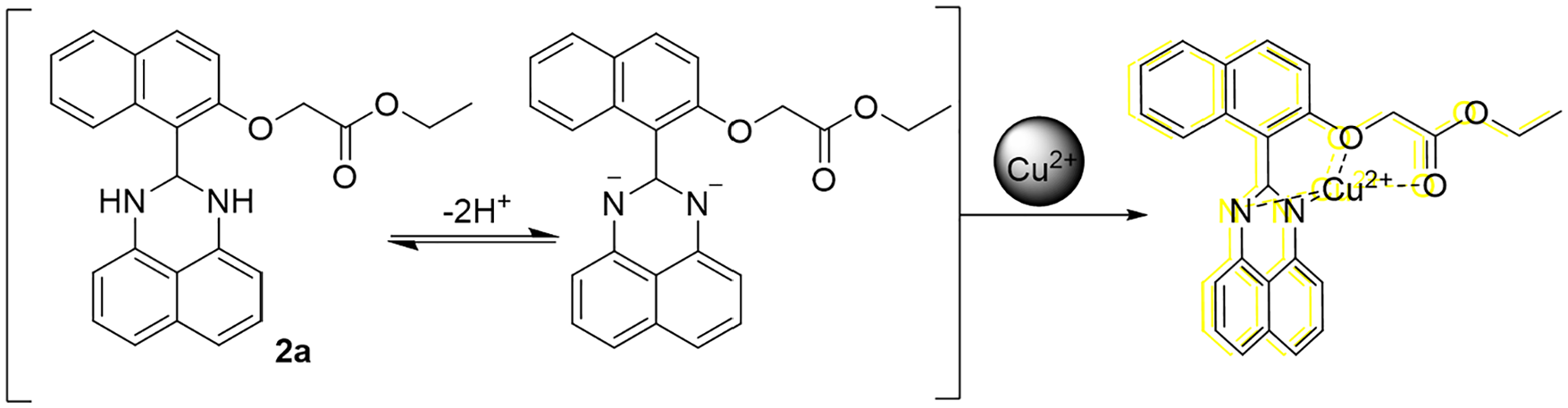
Recognition mechanism.
To understand more clearly the structure of the Cu2+–

Job plot of a 1:1 complex of

1
H NMR (300 MHz) spectra of
According to the Job plot and nuclear magnetic resonance (NMR) spectroscopy, the chemosensor
Conclusion
In conclusion, two new perimidine products had been designed and synthesized. The structures of
Experimental
General
All reagents were obtained from commercial sources and were of AR (Analytical Reagent) grade. Melting points were determined with a XT4A micromelting point apparatus and are uncorrected. The 1H NMR spectra were recorded on a Mercury Plus-400 spectrometer with tetramethylsilane (TMS) as the internal reference and CDCl3 as the solvent. MS was performed with Finnigan Trace MS instrument using the electron ionization (EI) method. Elemental analyses were carried out on a Vario EL III instrument.
1-(2,3-dihydro-1h-perimidin-2-yl)naphthalen-2-ol (1 ):
To a solution of naphthalene-1,8-diamine (632 mg, 4 mmol) in ethanol (30 mL) was added 2-hydroxy-1-naphthaldehyde (688 mg, 4 mmol), and the reaction mixture was heated at 70 °C for 4 h. Subsequently, it was cooled to room temperature. The resultant orange solution was filtered and a yellow precipitate was obtained. The precipitate was purified by recrystallization from ethanol/ethyl acetate (2:1) to give 749 mg of compound
Ethyl 2-((1-(2,3-dihydro-1h-perimidin-2-yl)naphthalen -2-yl)oxy)acetate (2a) :
Ethanol (30 mL) was added to a flask containing compound
2-(2-(pyridin-3-ylmethoxy)naphthalen-1-yl)-2,3-dihydro-1h-perimidine (2b) :
Ethanol (30 mL) was added to a flask containing compound
X-ray diffraction study of 2a
A white crystal of
The Cambridge Crystallographic Data Centre (CCDC) deposition number is 1957498 for compound
Supplemental Material
checkcif – Supplemental material for A new perimidine-based fluorescent turn-on chemosensor for selective detection of Cu2+ ions
Supplemental material, checkcif for A new perimidine-based fluorescent turn-on chemosensor for selective detection of Cu2+ ions by Yanning Ge, Dehua Zhang, Xiaoyan Zhang, Yang Liu, Longfei Du and Yingying Wang in Journal of Chemical Research
Supplemental Material
mo_190408A_0m_3 – Supplemental material for A new perimidine-based fluorescent turn-on chemosensor for selective detection of Cu2+ ions
Supplemental material, mo_190408A_0m_3 for A new perimidine-based fluorescent turn-on chemosensor for selective detection of Cu2+ ions by Yanning Ge, Dehua Zhang, Xiaoyan Zhang, Yang Liu, Longfei Du and Yingying Wang in Journal of Chemical Research
Supplemental Material
Support_Information – Supplemental material for A new perimidine-based fluorescent turn-on chemosensor for selective detection of Cu2+ ions
Supplemental material, Support_Information for A new perimidine-based fluorescent turn-on chemosensor for selective detection of Cu2+ ions by Yanning Ge, Dehua Zhang, Xiaoyan Zhang, Yang Liu, Longfei Du and Yingying Wang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work supported by the Hubei Key Laboratory of Pollutant Analysis & Reuse Technology (Grant No. PA190203) and the Hubei Provincial Department of Education (Grant No. B2019133).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
