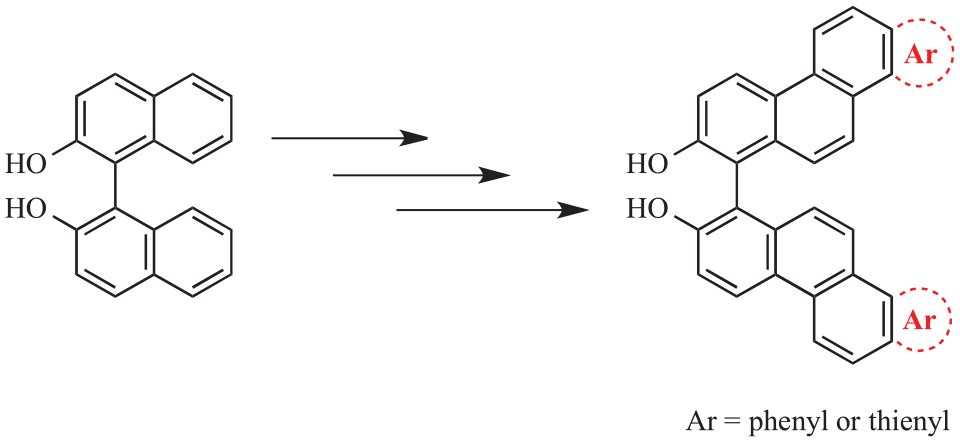
Experimental
All chemicals were commercially available as analytical or chemical grade. Solvents were purified via standard methods before use. All reactions sensitive to air or water were conducted under an Ar or N2 atmosphere. Reactions were monitored by thin-layer chromatography (TLC). Silica gel (Anhui Liangchen GF254) for column chromatography was 200–300 mesh. 1H nuclear magnetic resonance (NMR) and 13C NMR spectra were measured on a Bruker DRx500 NMR spectrometer (
1
H NMR: 500 MHz, 13C NMR: 125 MHz). Low-resolution mass spectra were recorded on an Agilent 5973N or a Waters GCT Premier spectrometer (mass spectrometry electron ionization (MS EI)). High-resolution mass spectra were obtained using a Bruker MicroTof II mass spectrometer (high-resolution mass spectrometry electrospray ionization (HRMS ESI)). Melting points were measured on an X-4 micrographic melting point apparatus.
Starting materials
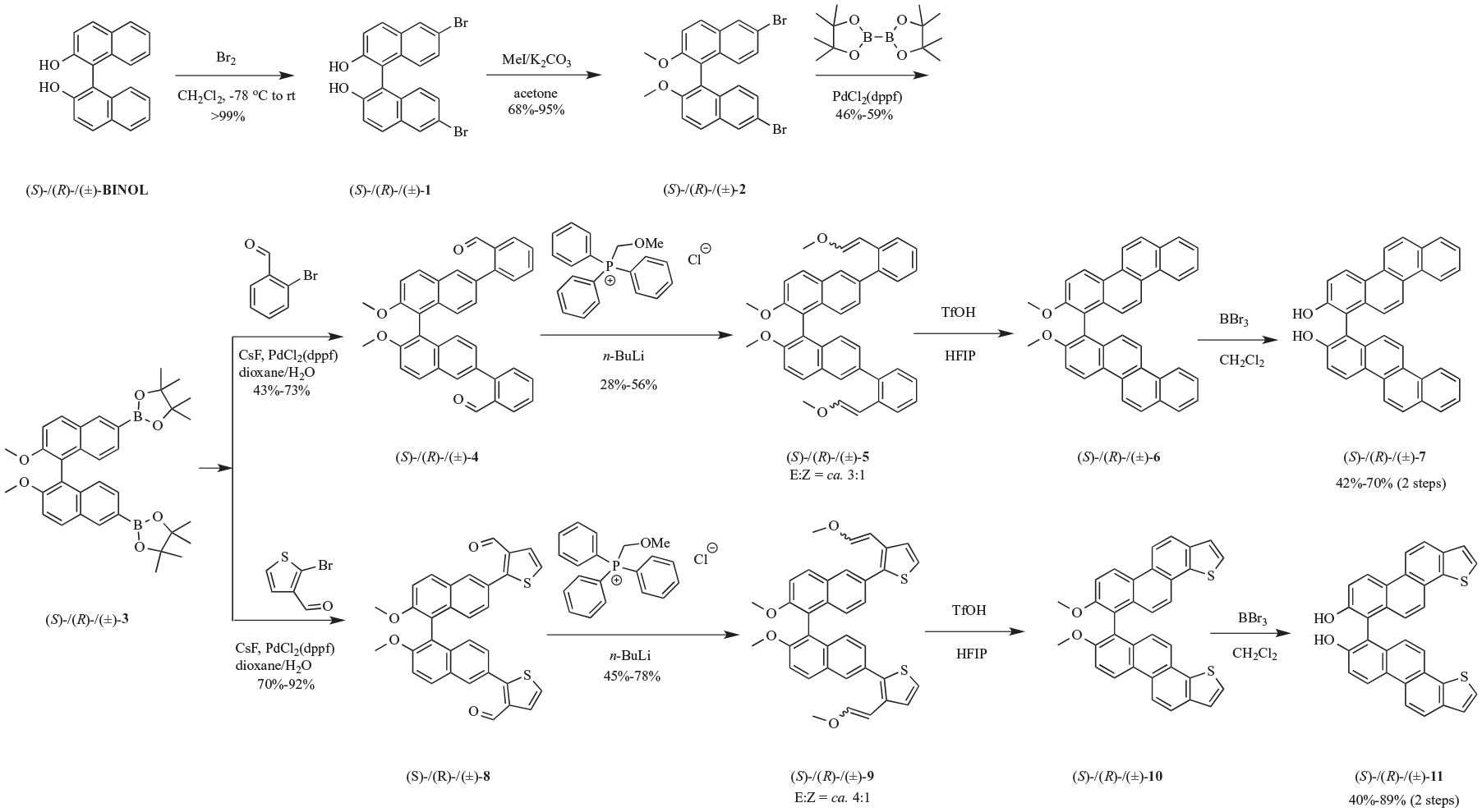
(S)-/(R)-/(±)-6,6′-dibromo-[1,1′-binaphthalene]-2,2′-diol [(S)-/(R)-/(±)-1], 6,6′-dibromo-2,2′-dimethoxy-1,1′-binaphthalene [(S)-/(R)-/(±)-2], and 2,2′-(2,2′-dimethoxy-[1,1′-binaphthalene]-6,6′-diyl)bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolane) [(S)-/(R)-/(±)-3] were prepared according to previous work (see Supplemental Material for details).15–17
(S)-/(R)-/(±)-2,2′-(2,2′-dimethoxy-[1,1′-binaphthalene]-6,6′-diyl)dibenzaldehyde [(S)-
4/(R)-
4/(±)-
4]
A 250-mL three-neck bottom flask was charged with (S)-3/(R)-3/(±)-3 (4 g, 7.06 mmol, 1.0 equiv.), 2-bromobenzaldehyde (2.9 g, 15.52 mmol, 2.2 equiv.), CsF (6.45 g, 42.44 mmol, 6.0 equiv.), and dioxane/H2O (180 mL, 2:1). The flask was filled with argon and evacuated. This procedure was repeated three times. Then Pd(dppf)Cl2 (258.5 mg, 0.35 mmol, 0.05 equiv.) was added and the solution was heated under argon at 90 °C for 8–10 h. After cooling to room temperature (rt), the mixture was extracted with EtOAc (150 mL). The organic layer was washed with brine (3 × 50 mL) and dried over anhydrous MgSO4. After removing the solvent, the crude residue was purified by silica gel chromatography (hexane/EtOAc, 15:1) to furnish the product as a white solid.
(S)-
4: White solid (1.85 g, 50%); Rf = 0.47 (PE/EtOAc, 3:1); m.p. 134.6–136.8 °C; 1H NMR (500 MHz, CDCl3): δ = 10.10 (s, 2H), 8.09–8.06 (m, 4H), 7.89 (d, J = 1.7 Hz, 2H), 7.68 (td, J = 7.5, 1.4 Hz, 2H), 7.61–7.50 (m, 6H), 7.33 (dd, J = 8.7, 1.8 Hz, 2H), 7.27 (d, J = 9.1 Hz, 2H), 3.87 (s, 6H); 13C NMR (125 MHz, CDCl3): δ = 192.77, 155.64, 146.07, 133.90, 133.54, 133.38, 132.76, 130.98, 129.96, 129.76, 128.71, 128.38, 127.61, 127.59, 125.47, 119.10, 114.87, 56.87; MS (EI): m/z [M]+ calcd for C36H26O4: 522.18; found: 522.
(R)-
4: White solid (1.59 g, 43%); Rf = 0.47 (PE/EtOAc, 3:1); m.p. 138.1–142.8 °C; 1H NMR (500 MHz, CDCl3): δ = 10.10 (s, 2H), 8.09–8.05 (m, 4H), 7.89 (d, J = 1.7 Hz, 2H), 7.67 (td, J = 7.5, 1.5 Hz, 2H), 7.60–7.50 (m, 6H), 7.33 (dd, J = 8.7, 1.9 Hz, 2H), 7.27 (d, J = 8.8 Hz, 2H), 3.87 (s, 6H); 13C NMR (125 MHz, CDCl3): δ = 192.77, 155.64, 146.07, 133.89, 133.54, 133.37, 132.75, 130.98, 129.96, 129.76, 128.71, 128.38, 127.61, 127.59, 125.46, 119.09, 114.87, 56.86.
(±)-
4: White solid (2.68 g, 73%); Rf = 0.47 (PE/EtOAc, 3:1); m.p. 233.3–236.4 °C; 1H NMR (500 MHz, CDCl3): δ = 10.10 (s, 2H), 8.10–8.05 (m, 4H), 7.89 (d, J = 1.9 Hz, 2H), 7.68 (td, J = 7.5, 1.5 Hz, 2H), 7.61–7.50 (m, 6H), 7.33 (dd, J = 8.7, 1.8 Hz, 2H), 7.27 (d, J = 8.9 Hz, 2H), 3.87 (s, 6H); 13C NMR (125 MHz, CDCl3): δ = 192.76, 155.65, 146.07, 133.90, 133.54, 133.38, 132.76, 130.98, 129.96, 129.76, 128.71, 128.38, 127.60, 127.60, 125.47, 119.10, 114.87, 56.87.
(S)-/(R)-/(±)-2,2′-dimethoxy-[6,6′-bis(2-methoxyethenyl)phenyl]-1,1′-binaphthalene [(S)-
5/(R)-
5/(±)-
5]
A mixture of Ph3P(CH2OMe)·Cl (3.61 g, 10.52 mmol, 5.0 equiv.) and n-BuLi (674.14 mg, 10.52 mmol, 5.0 equiv.) was dissolved in anhydrous tetrahydrofuran (THF; 15 mL) at 0 °C. After stirring at 0 °C for 30 min, a THF (10 mL) solution of (S)-4/(R)-4/(±)-4 (1.10 g, 2.10 mmol, 1.0 equiv.) was added. The mixture was stirred at rt for 2 h and then quenched with water. After removing spare THF, the water layer was extracted with EtOAc (3 × 20 mL). The combined extracts were washed with brine and dried over Na2SO4. After removing the solvent, the crude residue was purified by silica gel column chromatography (hexane/EtOAc 15:1) to give a white solid.
(S)-
5: White solid (340 mg, 28%); Rf = 0.49 (PE/EtOAc, 3:1); m.p. 89.3–92.5 °C; 1H NMR (500 MHz, CDCl3): δ = 8.03 (d, J = 9.0 Hz, 2H), 7.89 (s, 2H), 7.52 (dd, J = 9.0, 1.6 Hz, 2H), 7.44 (d, J = 7.4 Hz, 2H), 7.37 (dt, J = 7.4, 1.8 Hz, 2H), 7.34–7.30 (m, 4H), 7.27–7.20 (m, 4H), 6.95 (dd, J = 12.9, 1.6 Hz, 2H), 5.87 (d, J = 12.8 Hz, 2H), 3.85 (s, 6H), 3.52 (s, 6H); 13C NMR (125 MHz, CDCl3): δ = 155.13, 148.91, 139.87, 136.53, 134.29, 132.89, 130.47, 129.63, 129.10, 128.68, 128.37, 127.38, 125.98, 125.04, 124.90, 119.39, 114.43, 104.68, 56.94, 56.54; MS (EI): m/z [M]+ calcd for C40H34O4: 578.25; found: 578.
(R)-
5: White solid (683 mg, 56%); Rf = 0.49 (PE/EtOAc, 3:1); 1H NMR (500 MHz, CDCl3): δ = 8.03 (d, J = 9.0 Hz, 2H), 7.90 (s, 2H), 7.53 (d, J = 9.0 Hz, 2H), 7.44 (dd, J = 7.6, 1.5 Hz, 2H), 7.37 (dd, J = 7.3, 1.8 Hz, 2H), 7.34–7.29 (m, 4H), 7.27–7.21 (m, 4H), 6.95 (d, J = 12.9 Hz, 2H), 5.87 (d, J = 12.9 Hz, 2H), 3.85 (s, 6H), 3.52 (s, 6H); 13C NMR (125 MHz, CDCl3): δ = 155.13, 148.91, 139.87, 136.53, 134.28, 132.89, 130.47, 129.63, 129.10, 128.68, 128.37, 127.38, 125.98, 125.03, 124.90, 119.39, 114.43, 104.68, 56.94, 56.54.
(±)-
5: White solid (426 mg, 35%); Rf = 0.49 (PE/EtOAc, 3:1); m.p. 118.2–121.4 °C.
(S)-/(R)-/(±)-2,2′-dimethoxy-1,1′-bichrysene [(S)-
6/(R)-
6/(±)-
6]
Trifluoromethane sulfonic acid (31 μL, 0.35 mmol, 0.6 equiv.) was added to a hexafluoroisopropanol (HFIP; 30 mL) solution of the crude mixture of (S)-5/(R)-5/(±)-5 (340 mg, 0.59 mmol, 1.0 equiv.) at rt.
18
After stirring at rt for 2 h, the reaction was quenched with saturated NaHCO3 aqueous solution (pH 9) and then filtered and dried to give the crude product of (S)-6/(R)-6/(±)-6 as a pink solid.
(S)-/(R)-/(±)-[1,1′-bichrysene]-2,2′-diol [(S)-
7/(R)-
7/(±)-
7]
The crude product of (S)-6/(R)-6/(±)-6 (0.59 mmol, 1.0 equiv.) was dissolved in CH2Cl2, and BBr3 (2.3 mL, 23.6 mmol, 40.0 equiv.) was slowly added at 0 °C. The mixture was stirred at 0 °C for 1 h and then at rt for 4 h, poured into the water, and then filtered and dried to give a brown solid.
(S)-
7: Brown solid (146 mg, 51%); Rf = 0.46 (PE/EtOAc, 1:1); m.p. > 300 °C; 1H NMR (500 MHz, DMSO-d6): δ = 9.56 (s, 2H), 8.93 (dd, J = 28.0, 9.3 Hz, 4H), 8.73 (d, J = 8.3 Hz, 2H), 8.57 (d, J = 9.6 Hz, 2H), 8.09 (dd, J = 18.8, 8.5 Hz, 4H), 7.62 (ddd, J = 35.6, 18.4, 8.3 Hz, 6H), 7.27 (d, J = 9.4 Hz, 2H); 13C NMR (125 MHz, DMSO-d6): δ = 154.57, 133.49, 131.59, 130.58, 128.89, 128.84, 127.73, 127.24, 126.45, 125.91, 125.10, 124.91, 124.53, 123.37, 121.96, 118.63, 117.79, 99.98; MS (EI): m/z [M]+ calcd for C36H22O2: 486.16; found: 486.
(R)-
7: Brown solid (200 mg, 70%); Rf = 0.46 (PE/EtOAc, 1:1); m.p. > 300 °C; 1H NMR (500 MHz, DMSO-d6): δ = 9.56 (s, 2H), 8.96 (d, J = 9.2 Hz, 2H), 8.90 (d, J = 9.2 Hz, 2H), 8.73 (d, J = 8.3 Hz, 2H), 8.57 (d, J = 9.4 Hz, 2H), 8.11 (d, J = 9.1 Hz, 2H), 8.07 (dd, J = 7.8, 1.6 Hz, 2H), 7.70–7.59 (m, 4H), 7.57 (d, J = 9.1 Hz, 2H), 7.26 (d, J = 9.4 Hz, 2H); 13C NMR (125 MHz, DMSO-d6): δ = 154.56, 133.49, 131.59, 130.58, 128.89, 128.85, 127.74, 127.24, 126.46, 125.90, 125.09, 124.92, 124.53, 123.37, 121.97, 121.93, 118.63, 117.79; MS (EI): m/z [M]+ calcd for C36H22O2: 486.16; found: 486.
(±)-
7: Brown solid (120 mg, 42%); Rf = 0.46 (PE/EtOAc, 1:1); m.p. > 300 °C; 1H NMR (500 MHz, DMSO-d6): δ = 9.56 (s, 2H), 8.96 (d, J = 9.2 Hz, 2H), 8.90 (d, J = 9.2 Hz, 2H), 8.72 (d, J = 8.2 Hz, 2H), 8.56 (d, J = 9.5 Hz, 2H), 8.11 (d, J = 9.2 Hz, 2H), 8.07 (dd, J = 7.9, 1.6 Hz, 2H), 7.69–7.60 (m, 4H), 7.57 (d, J = 9.1 Hz, 2H), 7.26 (d, J = 9.3 Hz, 2H); 13C NMR (125 MHz, DMSO-d6): δ = 154.57, 133.49, 131.58, 130.58, 128.89, 128.84, 127.72, 127.24, 126.45, 125.90, 125.10, 124.91, 124.53, 123.36, 121.95, 121.94, 118.63, 117.78; MS (EI): m/z [M]+ calcd for C36H22O2: 486.16; found: 486.
(S)-/(R)-/(±)-2,2′-(2,2′-dimethoxy-[1,1′-binaphthalene]-6,6′-diyl)bis(thiophene-3-carbaldehyde) [(S)-
8/(R)-
8/(±)-
8]
A 250-mL three-neck round-bottom flask was charged with (S)-3/(R)-3/(±)-3 (1.40 g, 2.47 mmol, 1.0 equiv.), 2-bromo-3-thiophenecarboxaldehyde (1.04 g, 5.44 mmol, 2.2 equiv.), CsF (2.25 g, 14.83 mmol, 6.0 equiv.), and dioxane/H2O (120 mL, 2:1). The flask was filled with argon and evacuated. This procedure was repeated three times. Then Pd(dppf)Cl2 (90.45 mg, 123.61 μmol, 0.05 equiv.) was added, and the solution was heated under argon at 90 °C for 8–10 h. After cooling to rt, the mixture was extracted with EtOAc. The organic layer was washed with brine (3 × 50 mL) and dried over anhydrous MgSO4. After removing the solvent, the crude residue was purified by silica gel chromatography (hexane/EtOAc, 10:1) to furnish the product as a white solid.
(S)-
8: White solid (1.22 g, 92%); Rf = 0.7 (PE/EtOAc, 1:1); m.p. 137.3–140.1 °C; 1H NMR (500 MHz, DMSO-d6): δ = 9.90 (s, 2H), 8.27 (d, J = 8.7 Hz, 4H), 7.74 (d, J = 9.1 Hz, 2H), 7.69 (dd, J = 5.4, 0.8 Hz, 2H), 7.54–7.48 (m, 4H), 7.07 (d, J = 8.8 Hz, 2H), 3.79 (s, 6H); 13C NMR (125 MHz, DMSO-d6): δ = 185.99, 156.20, 155.74, 136.96, 133.74, 130.97, 130.19, 128.87, 128.34, 127.14, 126.94, 126.54, 125.70, 118.32, 115.55, 56.71; MS (EI): m/z [M]+ calcd for C32H22O4S2: 534.10; found: 534.
(R)-
8: White solid (1.20 g, 89%); Rf = 0.7 (PE/EtOAc, 1:1); m.p. 131.7–134.2 °C; 1H NMR (500 MHz, DMSO-d6): δ = 9.90 (s, 2H), 8.27 (d, J = 9.5 Hz, 4H), 7.74 (d, J = 9.1 Hz, 2H), 7.69 (d, J = 5.3 Hz, 2H), 7.56–7.48 (m, 4H), 7.07 (d, J = 8.8 Hz, 2H), 3.79 (s, 6H); 13C NMR (125 MHz, DMSO-d6): δ = 185.99, 156.20, 155.74, 136.96, 133.74, 130.97, 130.19, 128.87, 128.34, 127.14, 126.94, 126.54, 125.70, 118.32, 115.55, 56.71.
(±)-
8: White solid (0.92 g, 70%); Rf = 0.7 (PE/EtOAc, 1:1); m.p. 138.2–141.3 °C; 1H NMR (500 MHz, DMSO-d6): δ = 9.90 (s, 2H), 8.27 (d, J = 9.3 Hz, 4H), 7.74 (d, J = 9.2 Hz, 2H), 7.69 (d, J = 5.3 Hz, 2H), 7.55–7.47 (m, 4H), 7.07 (d, J = 8.8 Hz, 2H), 3.79 (s, 6H); 13C NMR (125 MHz, DMSO-d6): δ = 186.00, 156.19, 155.75, 136.94, 133.73, 130.97, 130.18, 128.86, 128.32, 127.15, 126.93, 126.53, 125.69, 118.30, 115.55, 56.72.
(S)-/(R)-/(±)-2,2′-dimethoxy-[6,6′-bis(2-methoxyethenyl)thiophene]-1,1′-binaphthalene [(S)-
9
/(R)-
9
/(±)-
9
]
A mixture of Ph3P(CH2OMe)·Cl (1.64 g, 4.78 mmol, 5.0 equiv.) and n-BuLi (306.43 mg, 4.78 mmol, 5.0 equiv.) was dissolved in anhydrous THF (10 mL) at 0 °C. After stirring at 0 °C for 30 min, a THF (5 mL) solution of (S)-4/(R)-4/(±)-4 (500 mg, 0.96 mmol, 1.0 equiv.) was added. The mixture was stirred at rt for 2 h and then quenched with water. After removing THF, the water layer was extracted with EtOAc (3 × 10 mL). The combined extracts were washed with brine and dried over Na2SO4. After removing the solvent, the crude product was purified by silica gel column chromatography (hexane/EtOAc 15:1) to give a white solid.
(S)-
9: White solid (246 mg, 45%); Rf = 0.46 (PE/EtOAc, 3:1); 1H NMR (500 MHz, CDCl3): δ = 8.03 (d, J = 9.0 Hz, 2H), 7.99 (s, 2H), 7.52 (d, J = 9.0 Hz, 2H), 7.41 (dd, J = 8.8, 1.9 Hz, 2H), 7.25 (d, J = 5.4 Hz, 2H), 7.20 (d, J = 8.7 Hz, 2H), 7.12 (d, J = 5.3 Hz, 2H), 7.05 (d, J = 12.9 Hz, 2H), 6.02 (d, J = 13.0 Hz, 2H), 3.84 (s, 6H), 3.62 (s, 6H); 13C NMR (125 MHz, CDCl3): δ = 155.30, 149.76, 136.33, 133.03, 132.48, 129.76, 129.70, 129.16, 128.13, 127.96, 125.56, 125.41, 124.33, 119.28, 114.59, 99.96, 56.90, 56.63.
(R)-9: White solid (305 mg, 55%); Rf = 0.46 (PE/EtOAc, 3:1); m.p. 138.9–144.3 °C.
(±)-
9: White solid (430 mg, 78%); Rf = 0.46 (PE/EtOAc, 3:1); m.p. 112.7–115.1 °C.
(S)-/(R)-/(±)-7,7′-dimethoxy-6,6′-biphenanthro[1,2-b]thiophene [(S)-
10/(R)-
10/(±)-
10]
Trifluoromethane sulfonic acid (14.52 μL, 0.16 mmol, 0.6 equiv.) was added to an HFIP (20 mL) solution of the crude mixture of (S)-9/(R)-9/(±)-9 (162 mg, 0.27 mmol, 1.0 equiv.) at rt. After stirring at rt for 2 h, the reaction was quenched with saturated NaHCO3 aqueous solution (pH 9) and then filtered and dried to give the crude product of (S)-10/(R)-10/(±)-10 as a gray solid.
(S)-/(R)-/(±)-[6,6′-biphenanthro[1,2-b]thiophene]-7,7′-diol [(S)-
11/(R)-
11/(±)-
11]
The crude product (S)-10/(R)-10/(±)-10 (0.27 mmol, 1.0 equiv.) was dissolved in CH2Cl2, and BBr3 (1.06 mL, 11.01 mmol, 40.0 equiv.) was slowly added at 0 °C. The mixture was stirred at 0 °C for 1 h and then at rt for an additional 4 h, poured into the water, and then filtered and dried to give a brown solid.
(S)-
11: Brown solid (121 mg, 89%); Rf = 0.4 (PE/EtOAc, 1:1); m.p. 165.6–168.2 °C; 1H NMR (500 MHz, DMSO-d6): δ = 8.79 (d, J = 9.1 Hz, 2H), 8.75 (d, J = 8.9 Hz, 2H), 8.33 (s, 2H), 8.07 (d, J = 8.9 Hz, 2H), 7.77 (d, J = 5.3 Hz, 2H), 7.69 (d, J = 9.2 Hz, 2H), 7.62 (d, J = 5.3 Hz, 2H), 7.42 (d, J = 9.0 Hz, 2H), 7.16 (d, J = 9.2 Hz, 2H); 13C NMR (125 MHz, DMSO-d6): δ = 154.49, 138.09, 137.46, 132.99, 127.61, 126.57, 125.73, 125.56, 125.07, 124.79, 124.54, 123.13, 122.92, 120.29, 118.61, 118.51; HRMS (ESI): m/z [M + Na]+ calcd for C32H18NaO2S2: 521.0648; found: 521.0640.
(R)-
11: Brown solid (54 mg, 40%); Rf = 0.4 (PE/EtOAc, 1:1); m.p. 164.2–166.8 °C; 1H NMR (500 MHz, DMSO-d6): δ = 9.54 (s, 2H), 8.89 (d, J = 9.5 Hz, 2H), 8.80 (d, J = 9.4 Hz, 2H), 8.12 (d, J = 8.9 Hz, 2H), 7.85–7.80 (m, 4H), 7.64 (d, J = 5.3 Hz, 2H), 7.53 (d, J = 9.1 Hz, 2H), 7.18 (d, J = 9.2 Hz, 2H); 13C NMR (125 MHz, DMSO): δ = 154.48, 138.09, 137.46, 132.99, 127.60, 126.58, 125.73, 125.57, 125.07, 124.80, 124.54, 123.13, 122.93, 120.29, 118.60, 118.50; MS (EI): m/z [M]+ calcd for C32H18O2S2: 498.07; found: 498.
(±)-
11: Brown solid (92 mg, 68%); Rf = 0.4 (PE/EtOAc, 1:1); m.p. 164.6–167.2 °C; MS (EI): m/z [M]+ calcd for C32H18O2S2: 498.07; found: 498.