Abstract
Horseradish peroxidase–H2O2-mediated oxidative cross-coupling reactions of oxyresveratrol 2-methyl ether and brominated isorhapontigenin or brominated resveratrol efficiently produce two 8-5’-coupled dihydrobenzofuran-type heterodimers. The LiAlH4-catalysed reductive debrominations of these cross-coupled dimeric intermediates provided the first synthesis of two unnatural oxyresveratrol-related heterodimers.

Introduction
Natural polyphenolic oligostilbenes possess complex structures and have diverse biological properties as potential therapeutic agents in human health. Most of them are the homooligomers of stilbene units including resveratrol (

Representative stilbenes and several natural heterooligomers.
The key to synthesis of oxyresveratrol-related heterodimers
Results and discussion
The oxidative coupling reactions of monomer
As shown in Scheme 1, when monomer

HRP–H2O2-promoted cross-coupling products of stilbenes
The dihydrobenzofuran skeleton of heterodimer
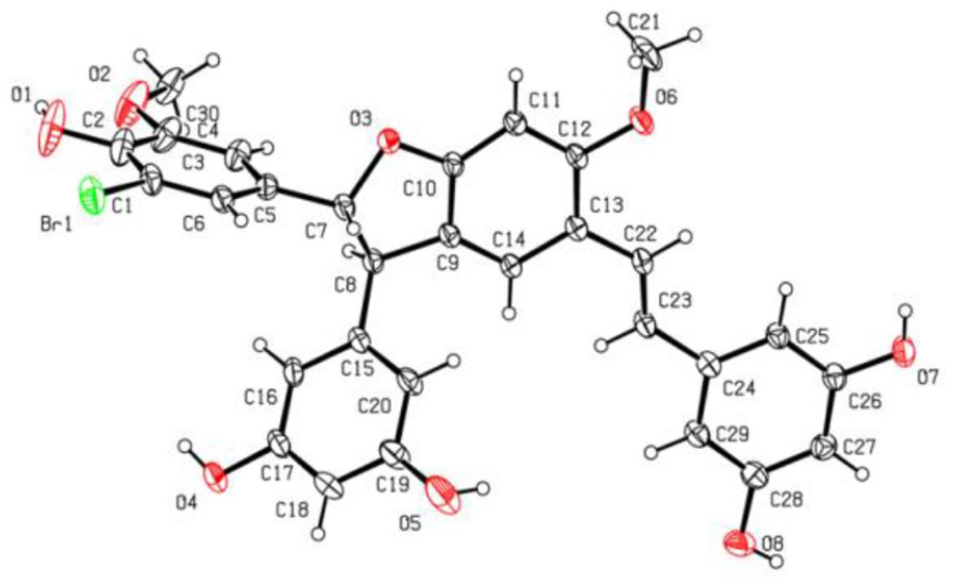
X-ray crystal structure of the heterodimer
Based on the above result, we further studied the oxidative cross-coupling reaction of
We next attempted to remove the bromine protecting groups and methyl groups from the heterodimeric intermediates
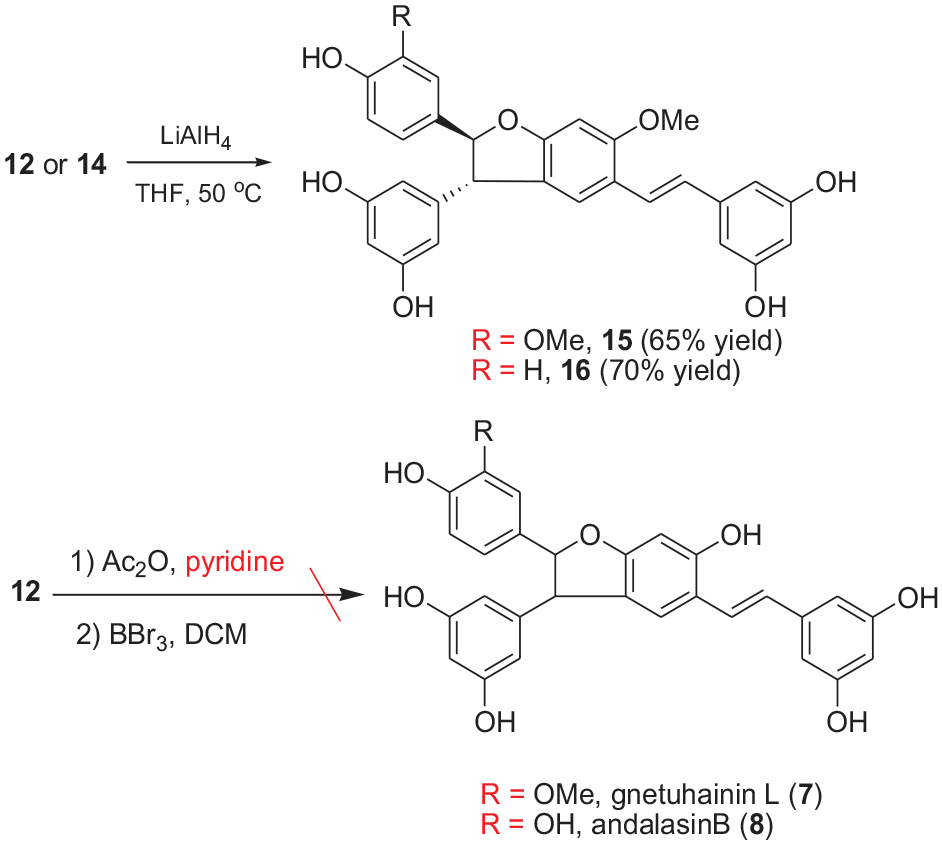
Reductive debromination products of heterodimers
Conclusion
In conclusion, we have investigated the biomimetic cross-coupling reactions of two different stilbene monomers under HRP–H2O2–acetone–H2O oxidative conditions. The key to the regioselective synthesis of heterodimers
Experimental
General methods
Structural determinations of the isolated compounds were based on 1H, 13C NMR, NOESY, 1H-1H COSY and 1H-1H NOSEY spectra, and HRMS analysis. All NMR spectra were recorded on Varian Mercury 500 or 600 MHz instruments in the indicated solvent. HRMS spectra were measured on an Autostec-3090 mass spectrometer. All solvents were freshly purified and dried by standard techniques prior to use. Purification of products was performed by column chromatography on silica gel (200–300 mesh), purchased from Qingdao Marine Chemical Co. (Qingdao, China). The monomer
5-[5-[(1E)-2-(3,5-dihydroxyphenyl)ethenyl]-2-(3-methoxy-4-hydroxyphenyl)-6-methoxy-2,3-dihydrobenzofuranyl]-1,3-benzenediol (12 )
The precursor
5-[5-[(1E)-2-(3,5-dihydroxyphenyl)ethenyl]-2-(3,5-dibromo-4-hydroxyphenyl)-6-methoxy-2,3-dihydrobenzofuranyl]-1,3-benzenediol (14 )
The precursor
5-[5-[(1E)-2-(3,5-dihydroxyphenyl)ethenyl]-2-(3-methoxy-4-hydroxyphenyl)-6-methoxy-2,3-dihydrobenzofuranyl]-1,3-benzenediol (15 )
LiAlH4 (4.0 mL, 2.5 M THF solution, 10.0 mmol) was slowly added to the solution of dimer
5-[5-[(1E)-2-(3,5-dihydroxyphenyl)ethenyl]-2-(4-hydroxyphenyl)6-methoxy-2,3-dihydrobenzofuranyl]-1,3-benzenediol (16 )
LiAlH4 (4.0 mL, 2.5 M THF solution, 10.0 mmol) was slowly added to the solution of dimer
Supplemental Material
sj-doc-1-chl-10.1177_17475198211068803 – Supplemental material for Enzyme-promoted oxidative cross-coupling for the synthesis of oxyresveratrol-related heterodimers
Supplemental material, sj-doc-1-chl-10.1177_17475198211068803 for Enzyme-promoted oxidative cross-coupling for the synthesis of oxyresveratrol-related heterodimers by Yuefei Wang, Li Han, Zongjun Tang, Haiping Liu and Wenling Li in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research work was financially supported by the National Natural Science Foundation of China (no. 21462024).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
