Abstract
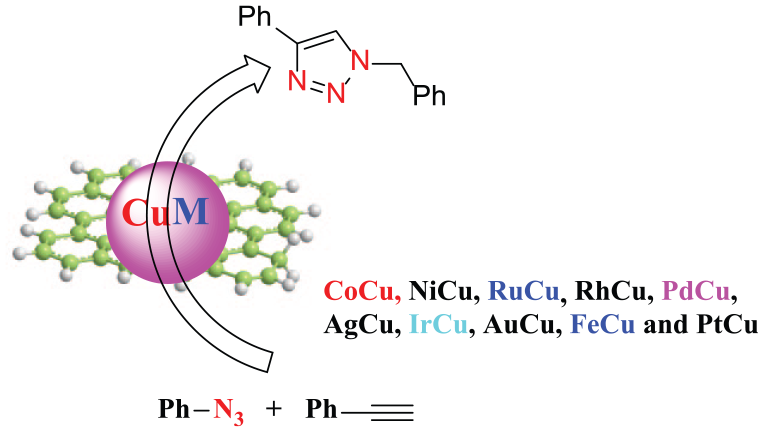
Compared to monometallic nanoparticles, bimetallic nanoparticles have attracted wide attention due to their physical properties, excellent catalytic activity, high regioselectivity, selectivity, and stability. Here, we have first synthesized 10 different kinds of graphene quantum dot–stabilized Cu-based bimetallic nanoparticles (including CoCu, NiCu, RuCu, RhCu, PdCu, AgCu, IrCu, AuCu, FeCu, and PtCu) and compared their catalytic activities in a CuAAC click reaction. Among them, RhCu provides the highest yield of the desired product in the click reaction (77%). The catalytic activity of these MCu in the click reaction is in the order: RhCu > PdCu > AuCu > CoCu > PtCu > AgCu > NiCu > CuNP > RuCu > FeCu > IrCu.
Here, we have first synthesized 10 different graphene quantum dot (GQD)-stabilized Cu-based bimetallic nanoparticles and compared their catalytic activities in a CuAAC click reaction. Among them, RhCu provided the highest yield of the desired product in the click reaction.
Introduction
Triazoles, as a type of significantly useful N-heterocycles,1–5 have been widely applied in fluorescent materials, drug discovery, chemosensing, biochemistry, dendrimers, agricultural products, materials science, corrosion-retarding agents, dyes, and polymer chemistry,6–8 due to the physically and chemically inertness against reduction, oxidation and hydrolysis under basic or acidic conditions. 9 Plenty of methods have been developed for the synthesis of 1,2,3-triazoles. Among them, the copper-catalyzed [3+2] cycloaddition of azides and alkynes (CuAAC), independently discovered by the groups of Sharpless and Meldal,10–12 has attracted significant interest due to the mild environmentally friendly conditions and high regioselectivity.13–19 Typically, CuAAC is accomplished using stoichiometric quantities of copper(I) salts, Cu(II) salts/Na ascorbate or Cu(0)/Cu(II).20,21 Recently, Cu nanoparticles (CuNP) have been increasingly explored as efficient catalysts for the CuAAC click reaction. 22 In addition, the CuAAC mechanism, involving a dicopper intermediate, was described by Fokin in 2013.23–25
Compared to monometallic nanoparticles, bimetallic nanoparticle (BNP) catalysts exhibit excellent chemical conversion due to their synergistic effect.26–29 In continuation of our interest in the synthesis of transition metal nanoparticles and their applications in catalysis,30–36 in this study, we have first synthesized 10 different graphene quantum dot (GQD)-stabilized Cu-based BNPs and compared their catalytic activities in the CuAAC click reaction. Among them, GQDs (average 3 nm) were synthesized from commercial starch and water, which have been previously described and employed as detoxicants and for cell imaging.37–39 The full characterization of GQD by UV-Vis spectroscopy, X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), transmission electron microscopy (TEM) and atomic force microscopy (AFM) had been reported. The construction of the Cu-based BNPs, including CoCu, NiCu, RuCu, RhCu, PdCu, AgCu, IrCu, AuCu, FeCu, and PtCu, was performed by here direct coordination of Cu2+ and the corresponding transition metal ions (Mn+) in the solution of the GQD, followed by reduction with sodium borohydride in neat water.
Results and discussion
The synthesis of Cu-based BNPs
The synthesis of the Cu-based BNPs (CoCu, NiCu, RuCu, RhCu, PdCu, AgCu, IrCu, AuCu, FeCu, and PtCu) was conducted by using 1.25 × 10−4 mmol of CuSO4·5H2O and 1.25 × 10−4mmol of Mn+ dissolved in the GQDs solution, followed by the addition of 2.5 × 10−3 mmol of sodium borohydride at 0 °C, resulting in the formation of Cu-based BNPs (Scheme 1). The Cu2+ and Mn+ ions are first coordinated by hydroxyl and carboxylic groups of the GQD and are then reduced by NaBH4. The as-synthesized MCu BNPs are stabilized by these hydroxyl and carboxylic groups. However, the characteristic surface plasmon band of the transition metal nanoparticles did not appear in the UV-Vis spectra of all the CuM-BNPs due to the interference of CuNP. As a comparative experiment, mono CuNP was also synthesized by using 2.5 × 10−4 mmol of CuSO4·5H2O under the same conditions. All the Cu-based BNPs (CoCu, NiCu, RuCu, RhCu, PdCu, AgCu, IrCu, AuCu, FeCu, and PtCu) and CuNP were characterized by transmission electron microscopy (TEM) to confirm their morphologies and sizes (Figures 1 and Supplemental Figures S5–S15). We found that the sizes of CuNP (1.61 nm) and RhCu (1.45 nm) were smaller than 2 nm, and AgCu (2.20 nm), PdCu (2.26 nm), PtCu (2.42 nm), FeCu (2.48 nm) and RuCu (2.35 nm) are still smaller than 3 nm. In contrast, AuCu and IrCu are 3.80 and 3.87 nm, respectively. While NiCu (10.66 nm) and CoCu (9.37 nm) were much bigger (Figure 2).

The synthesis of the Cu-based bimetallic nanoparticles.

Photographs of the 11 different copper-based bimetallic nanoparticles.

TEM images of (a) RhCu, (b) PdCu, (c) AuCu, (d) CoCu, (e) PtCu, (f) AgCu, (g) NiCu, (h) CuNP, (i) RuCu, (j) FeCu, and (k) IrCu.
All the Cu-based BNPs were also characterized by XPS to confirm the valence state of the surface metal atoms. The XPS of RhCu showed that Rh 3d possesses only one pair of peaks for the 3d3/2 and 3d5/2 states at 311.88 and 306.49 eV, which are assigned to Rh0 (Figure 3(a)). 40 While Cu 2p exhibits two pairs of peaks for the 2p3/2 and 2p1/2 doublets, (at 933.48 and 953.23 eV) assigned to Cu0, whereas peaks at 934.67 and 956.40 eV correspond to Cu2+ (see Supplemental Figure S16). Similarly, the XPS of AgCu, FeCu, and IrCu only shows binding energies of 368.08, 704.35, and 63.00 eV for the 3d5/2 state of Ag (Figure 3(f)), the 2p3/2 state of Fe (Figure 3(j)) and the 4f7/2 state of Ir (Figure 3(k)), respectively, indicating the presence of only metallic Ag0, Fe0, and Ir0, whereas the Cu 2p states of AgCu (Supplemental Figure S21), FeCu (Supplemental Figure S24), and IrCu (Supplemental Figure S25) exhibit two pairs of peaks for the 2p3/2 and 2p1/2 doublets, indicating the coexistence of Cu0 and Cu2+. Moreover, the XPS of PdCu (Figure 3(b)), AuCu (Figure 3(c)), CoCu (Figure 3(d)), PtCu (Figure 3(e)), NiCu (Figure 3(g)) and RuCu (Figure 3(i)) show that Pd0, Au0, Co0, Pt0, Ni0, and Ru0 that have been partly oxidized to Pd2+, Au3+, Co2+, Pt2+ and Pt4+, Ni2+ and Ru3+ by air, respectively. Whereas the Cu 2p state of AuCu (Supplemental Figure S18) and PtCu (Supplemental Figure S20) show only one pair of peaks for 2p3/2 and 2p1/2, which are assigned to Cu0, respectively. Other Cu 2p state of PdCu (Supplemental Figure S17), CoCu (Supplemental Figure S19), NiCu (Supplemental Figure S22) and RuCu (Supplemental Figure S23) show two pairs of peaks for the 2p3/2 and 2p1/2 doublets, indicating the coexistence of Cu0 and Cu2+. In addition, the XPS of CuNP shows Cu 2p with two pairs of peaks for the 2p3/2 and 2p1/2 doublets, the 2p3/2 and 2p1/2 peaks at 932.32 and 952.25 eV are assigned to Cu0, respectively, whereas the peaks at 933.52 and 954.65 eV correspond to the 2p3/2 and 2p1/2 states of Cu2+, respectively (Figure 3(h)). It is clear that CuNP has been partly oxidized to Cu2+. We also summarized these in results Table 1.
Click reactions catalyzed by the CuM-BNPs in water. a

Reaction conditions: phenylacetylene (0.2 mmol), benzyl azide (0.3 mmol), H2O (5 mL), 30 °C, 24 h.
Core size (TEM).
Valence states by XPS.
Isolated yield.

XPS spectra of (a) Rh 3d over RhCu, (b) Pd 3d over PdCu, (c) Au 4f over AuCu, (d) Co 2p over CoCu, (e) Pt 4f over PtCu, (f) Ag 3d over AgCu, (g) Ni 2p over NiCu, (h) Cu 2p over CuNP, (i) Ru 3p over RuCu, (j) Fe 2p over FeCu, and (k) Ir 4f over IrCu.
Compared catalytic performance of MCu in click reaction
These 10 different MCu BNPs and CuNP have been tested for in the CuAAC click reaction of benzyl azide and phenylacetylene (Table 1). The CuAAC click reactions were carried out with PhCH2N3 (0.3 mmol) and PhC≡CH (0.2 mmol) in H2O (5 mL) at 30 °C in the presence of the MCu (0.01 mol% Cu) for 24 hours. Firstly, we found that CuNP provided a 31% yield of the desired product, 1-benzyl-4-phenyl-1H-1,2,3-triazole (Table 1, entry 8). The results showed that Ru-, Fe-, and Ir-alloyed MCus only yielded 25%, 17%, and 16%, respectively (entries 9–11), while Rh-, Pd-, Au-, Co-, Pt-, Ag-, and Ni-alloyed MCus improved the click reaction, yielding from 32–77% of the desired product (entries 1–7). Among them, RhCu provided the highest yield of the desired product (77%). The catalytic activity of these MCus in click reactions is in the order: RhCu > PdCu > AuCu > CoCu > PtCu > AgCu > NiCu > CuNP > RuCu > FeCu > IrCu.
Next, different acetylenes and benzyl azides were studied in the click reaction (Scheme 2). The alkynes and azides were suited well for this click reaction with RhCu as the catalyst.

CuAAC reactions between various azides and alkynes.
Conclusion
A green and facile method for the synthesis of 10 different GQD stabilized Cu-based BNPs (including CoCu, NiCu, RuCu, RhCu, PdCu, AgCu, IrCu, AuCu, FeCu, and PtCu) has been developed. The construction of the MCu BNPs was performed by direct coordination of Cu (II) and the corresponding transition metal ions into the GQD solution, followed by reduction with sodium borohydride in neat water. XPS showed that Cu is more easily oxidized by air than Rh, Ag, Fe, and Ir, whereas Rh, Ag, Fe, and Ir remained in the zero-valent state in RhCu, AgCu, FeCu, and IrCu, respectively. In contrast, Au and Pt were more easily oxidized by air than Cu, whereas Cu remained in the zero-valent state in the AuCu and PtCu. Interestingly, the both two metals in PdCu, CoCu, NiCu, and RuCu were partly oxidized by air. These 10 MCu BNPs and CuNP were tested in the CuAAC click reaction of benzyl azide and phenylacetylene. The catalytic activity of these MCu in click reaction follows the order of RhCu > PdCu > AuCu > CoCu > PtCu > AgCu > NiCu > CuNP > RuCu > FeCu > IrCu.
Reaction conditions: alkyne (0.2 mmol), azide (0.3 mmol), RhCu (0.01 mol%), and H2O (5 mL) at 30 °C for 24 h.
Experimental section
The general procedure of the click reaction
All reactions were performed on a 0.20 mmol scale of triazole. The phenylacetylene (0.2 mmol), benzyl azide (0.3 mmol), RhCu (0.01% mmol) and 5 mL H2O were taken into a round bottom flask equipped with stirrer. The resulting mixture was stirred for 24 h at 30 °C. After cooling to room temperature, extracted with ethyl acetate (3 × 10 mL). The combined organic phases were washed with brine (2 × 5 mL), dried over anhydrous MgSO4 and concentrated under reduced pressure. The residue was subjected to flash column chromatography with hexanes/EtOAc (5:1) as eluent to obtain the desired 1-benzyl-4-phenyl-1H-1,2,3-triazole as a light yellow solid (77% yield). 1H NMR (CDCl3, 400 MHz) δ: 7.81–7.79 (d, 2H), 7.66 (s, 1H), 7.40–7.38 (m, 5H), 7.38–7.26 (m, 3H), 5.59 (s, 2H). Note that the 1H NMR spectra of triazoles are shown in Supplemental Figures S1–S4 in ESI.
Supplemental Material
Supporting_information_2.19 – Supplemental material for A comparison of the catalytic efficiency of copper-based bimetallic nanoparticles in the click reactions
Supplemental material, Supporting_information_2.19 for A comparison of the catalytic efficiency of copper-based bimetallic nanoparticles in the click reactions by Ning Li and Xiang Liu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support from the National Natural Science Foundation of China (No. 21805166) and the 111 Project (No. D20015) and sponsorship by the Research Fund for Excellent Dissertation of China Three Gorges University is gratefully acknowledged.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

