Abstract
The main motivation of this work is to deposit two different metals (gold and silver) on titanium oxide nanoparticles surface in a one-step simple and fast physical process by applying a nitrogen plasma as the main source of nitrogen atoms to obtain nanostructured N-TiO2 −
Introduction
Titanium oxide (TiO2) has been widely used to eliminate organic contaminants mainly in aqueous media; it is also used in air purification through the process of photocatalytic oxidation which occurs on the surface of this semiconductor. However, one of the drawbacks of using TiO2 is the band gap value, which is found in the range of 3.0–3.2 eV, corresponding to ultraviolet radiation, thereby limiting the use of solar light in photocatalytic reactions. For this reason, therefore, and with the purpose of improving photocatalytic efficiency, several research groups have shown that the monometallic impregnation on the surface of TiO2 of metals such as copper (Cu), palladium (Pd), silver (Ag), and gold (Au) enhance the photogenerated electrons transfer, which causes an increase in the useful life of the corresponding electron-hole pairs.
Au has been extensively studied since the 1980s when M Haruta reported that this metal is catalytically active when it is dispersed as small particles on transition metal oxides matrix (cobalt oxide, iron oxide, and nickel oxide). 1 Since then numerous studies have been conducted related to TiO2/Au synergy, improving several catalysis reactions such as CO oxidation, water gas shift reaction (WGSR), H2O2 synthesis, and degradation of a diversity of contaminants. 2 In addition, other works can be found in the literature dealing with the impregnation of Ag on TiO2 to obtain TiO2/Ag 3,4 , in particular, those related to the bactericidal properties of this metal. Therefore, Au and Ag are transition metals that have been widely studied and were the first metals to be impregnated by chemical methods by means of their precursor salts and their subsequent reduction on TiO2.
Other research groups have also conducted studies on bimetallic impregnation of TiO2 by chemical methods; these investigations have reported promising results to be applied on the field of photocatalysis, in comparison with the monometallic impregnation, and are paving the way for new and promising research in the field of catalysis. Regarding this topic, it is suitable to mention the study carried out by Anna Zielińska-Jurek et al. on bimetallic impregnation of platinum (Pt) and Pd onto TiO2 to obtain Pt/Pd-TiO2; this research group demonstrated that the nanoparticles (NPs) Pt/Pd-TiO2 are highly active under visible light for the degradation of phenol and that this nanocomposite presents a more effective rate of phenol degradation in comparison with the monometallic photocatalysts Pt/TiO2 and Pd/TiO2. 5 Good synergy of Au and Ag on TiO2 has also been reported; Alberto Sandoval et al. obtained Au-Ag/TiO2 based on the process of sequential deposit precipitation (two-step impregnation) of Ag and Au and also reported a significantly greater activity of the Au-Ag/TiO2 in the oxidation of CO in comparison with the monometallic Au/TiO2. 6 The studies show that it is possible to improve the photocatalytic response or induce new reactions by depositing two metals on TiO2. However, as with the monometallic impregnation, the processes used to achieve bimetallic impregnation are chemical processes, such as deposit precipitation.
The plasma process has been applied successfully in monometallic impregnation separately, either Au or Cu on TiO2 to obtain TiO2/Au and TiO2/Cu, respectively. 7,8 Similarly, the process of plasma treatment has been successful in inducing phase changes on amorphous TiO2. 9 In this work, we reported positive results in bimetallic impregnation by applying nitrogen plasma technique, and this new proposal could contribute to improvement of novel processes in the search of new nanocomposite.
Materials and methodology
TiO2 Degussa P25 (Evonik Ind. AG, Germany), a mixture of 80% anatase and 20% rutile, was used. Au and Ag metal foils were employed, with 99.99% purity (Sigma-Aldrich, Toluca, México) and a thickness of 0.1 mm. Nitrogen gas flow was applied with 99.99% of purity (Praxair, Mérida, México).
The methodology of producing the nanocomposite with monometallic impregnation used in this work is similar to that proposed by Trejo-Tzab et al. 7,8 to the process followed is as follows: creation of bimetallic impregnation (Au and Ag) and doping of titania Degussa P25 powder; to achieve this goal, foils of this materials must cover half–half surface of the bottom electrode, and on top of the Au-Ag surface, the titania in its powder form is placed, covering 75–85% of the foil surface; the sample titania size is around 200 mg (see Figure 1(b)).
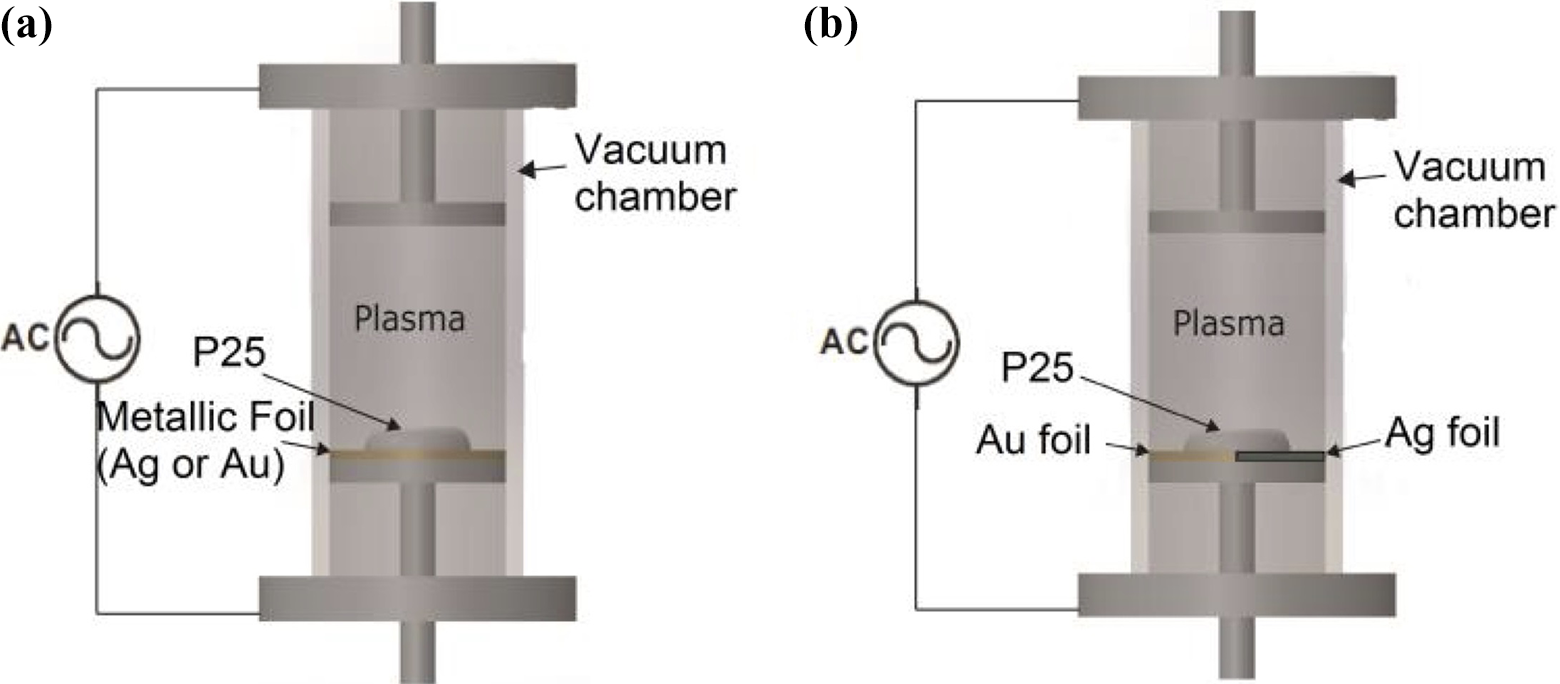
Reactor’s glow discharge configuration: (a) monometallic impregnation and (b) bimetallic impregnation.
A constant flow of nitrogen at a working pressure in the order of 10−2 torr was introduced into the reactor; on reaching the working pressure, discharge between the electrodes was applied, thereby generating the nitrogen plasma inside the reactor with a power of 200 watts. The effective treatment time was 120 min, in a “batch” process of 60 min, in which the sample is mechanically homogenized every 60 min in an agate mortar to ensure homogeneous metal particles distribution on the sample and to avoid metal agglomeration over extended periods of time plasma treatment. Even more, in bimetallic impregnation, there is a possibility that half the sample is mostly impregnated by only one metal, for such reasons, it is important to homogenize the sample to ensure that both metals have an homogeneous distribution over the powder sample. At the end of this procedure, the samples of N-TiO2 −
Characterization
Plasma characterization and optical properties
The nitrogen plasma emission throughout the entire period of the plasma treatment was observed using optical emission spectroscopy (OES). This optical characterization technique did not produce any perturbation in the plasma formation process, thus information of the optical characteristics are reliable. The intensity of the light emitted at particular wavelengths from an excited state is proportional to the density of species in that excited state and the excitation collisions and the subsequent radiative decays. Therefore, measurement of intensities provides a qualitative indicator of species concentration involved in the optical emission. These studies were conducted using an ultraviolet–visible (UV-Vis) Starline spectrophotometer (AvaSpec-3648, Avantes, Louisville, CO, USA).
Studies of UV-Vis diffuse reflectance spectroscopy were done to determine band gap changes of the samples after the plasma treatments, by means of a spectrophotometer (AvaSpec-3648), coupled to an integration sphere (AvaSphere-50-REFL), deuterium–halogen UV-Vis light source (AvaLight DH-S-BAL) and the WS-2 (Avantes) reference tile as white diffuse polytetrafluoroethylene (PTFE)-based material.
Particle size, elemental analysis, and surface composition
High-resolution transmission electron microscopy (HRTEM) investigations were carried out by a JEOL (Japan; model JEM 2100) transmission electron microscope operating at 200 kV. Shape and size of the particle were determined by field-emission scanning electron microscopy (FESEM) and by the technique of elemental analysis (energy-dispersive X-ray analysis (EDX)), employing a field-emission scanning electron microscope (JEOL model 7600F) applying an acceleration voltage of 15 kV.
The studies of the chemical states and surface composition were conducted with an X-ray photoelectron spectroscopy with a source of aluminum
Analysis of the crystalline structure was conducted using an X-ray diffractometer (Bruker D8 Advance), which provides X-rays with a wavelength of 1.5418 Å at 30 mA. The diffraction experiments were carried out in a 2
Results and discussion
Nitrogen plasma spectrum from OES is observed in Figure 2, and the peaks with higher intensities at 391.1 nm and 337.0 nm belong to excitation emissions due to radiative decays that correspond to N2 + molecular ion and molecular N2, respectively. The magnitude of the emission intensity lines is proportional to the concentration of the aforementioned nitrogen species and the amount of electrons that have energy greater than the excitation threshold from ground state. Figure 2 also shows that N2 + peak intensity is higher than N2 peak emission because N2 + emission is more sensitive to high-energy electrons than N2. The emission spectra are consistent with other studies conducted by different research groups. Qayyum et al. and Abdel-Fattah et al. reported emission peaks that are similar to those reported in our studies. 10,11
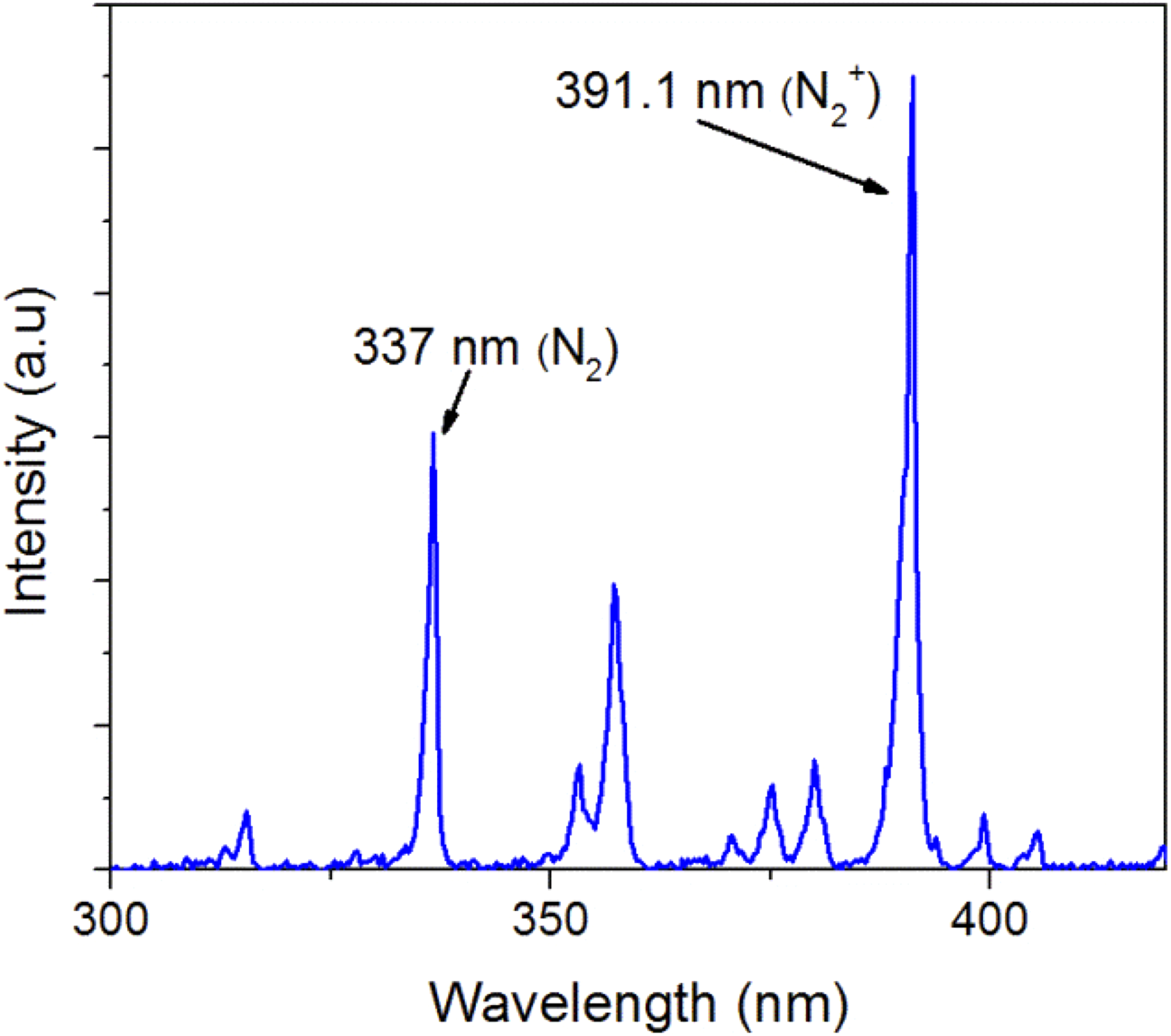
Optical emission spectrum of nitrogen plasma.
The samples obtained at 60 and 120 min of treatment, applying 200 watts of plasma power on the Au-Ag foil, present a gray–violet coloration, in comparison with the TiO2 P25 which presents a characteristic intense white. After 120 min, the intensity of the violet color of the samples increases, in comparison with the samples treated for 60 min. (Figure 3).

Monometalllic and bimetallic samples obtained after the plasma treatments at (a, c, and e) 60 min and (b, d, and f) 120 min.
Morphological studies using FESEM equipped with EDX was used to establish the influence of the plasma related to the metallic impregnation process after the 60-min and 120-min treatments. Figures 4 and 5 present the FESEM micrographs and EDX analysis corresponding to the bimetallic sample N-TiO2/Ag/Au with different time treatment with nitrogen plasma. The EDX analysis for both treatments proves the existence of Au and Ag in the samples. Moreover, the FESEM images demonstrate that the power employed, and the treatment time required to obtain the bimetallic samples do not generate significant changes in the geometry of the NPs; both TiO2/Au/Ag samples reveal a uniform distribution of particle size and both metals can be distributed uniformly on the surface of TiO2 using the method of impregnation by plasma.

FESEM micrograph of TiO2/Ag/Au after 60 min of nitrogen plasma treatment: (a) ×200,000, (b) ×100,000, and (c) EDX analysis. FESEM: field-emission scanning electron microscopy; TiO2: titanium oxide; Au: gold; Ag: silver; EDX: energy-dispersive X-ray spectroscopy.

FESEM micrograph of TiO2/Ag/Au after 120 min of nitrogen plasma treatment: (a) ×200,000, (b) ×100,000, and (c) EDX analysis. FESEM: field-emission scanning electron microscopy; TiO2: titanium oxide; Au: gold; Ag: silver; EDX: energy-dispersive X-ray spectroscopy.
HRTEM was utilized to reveal the microstructure of the samples with metallic impregnation. It is important to mention that Ag and Au NPs have similar crystal characteristics. Figure 6 shows the typical images that were obtained through the HRTEM analyses of the samples; Figure 6(a) and (b), corresponding to the sample TiO2/Ag, shows some spherical particles dispersed on the surface of TiO2 with a diameter in a range between 5 nm and 10 nm. Moreover, the interplanar spacing allows the identification of the average values of lattice fringes of

HRTEM analysis for monometallic and bimetallic samples: (a and b) TiO2/Ag, (c and d) TiO2/Au, and (e and f) TiO2/Au/Ag and EDX mapping. HRTEM: high-resolution transmission electron microscopy; TiO2: titanium oxide; Au: gold; Ag: silver; EDX: energy-dispersive X-ray spectroscopy.
Taking the HRTEM images for the samples with monometallic impregnation of Ag and Au NPs as reference, it is observed that Au particles onto TiO2 show darker shades compared to the Ag ones, and HRTEM images shows a greater contrast for Au NPs. The difference between both metals is due to the procedure that was carried out to identify Au and Ag particles in the samples with bimetallic impregnation, in which it is presumed that the darkest belongs to Au. This procedure, by contrast identification, was carried out because the crystal lattice parameters of Au and Ag are very similar, and it was impossible to identify them independently in the samples with bimetallic impregnation. 15 The results were confirmed when measuring the interplanar spacing of the lattice fringes corresponding to the plane (111) of the Ag and Au with average values of 0.234 and 0.236 nm, respectively. The values measured for both metals are consistent with the interplanar distance of the plane (111) of these metals with fcc structures. 16 These results were confirmed through EDX mapping provind the presence of Au and Ag dispersed on the powder sample surface (Figure 6(e) and (f)). Once the particles of Au and Ag have been identified, the results of HRTEM reveal metallic NPs of both metals dispersed on the surface of TiO2/Ag/Au samples with spherical shape and an average particle size of approximately 10 nm. It reveals not only particles of both metals dispersed independently but also the presence of metallic particles that have been formed by the union of both metals without forming an alloy as has been observed by other research groups when applying other techniques in comparison with the plasma methodology. It is postulated that the low power and short time used in the process of bimetallic impregnation are the key factors that do not allow the formation of metal NP alloys of Au-Ag on the TiO2 surface.
X-Ray diffraction (XRD) pattern analysis (Figure 7) shows that the crystallinity of TiO2 P25 does not change after the plasma treatments. One can observe that the intensity of the main peaks of anatase (101) and rutile (110), as well as their relative ratio, remain constant and do not present significant changes due to the effects of nitrogen plasma. The ratios of the phases in the mixture are 70% for anatase and 30% of rutile, with a 26 ± 3 nm size of the crystallite. The XRD patterns corresponding to the TiO2/Au monometallic sample obtained after 120 min of treatment (Figure 7(b)) show three different diffraction peaks in the positions 2

Diffractograms of the monometallic and bimetallic samples obtained in (a, c, and e) 60-min and (b, d, and f) 120-min treatment times with nitrogen plasma (A: anatase, R: rutile, Au: gold, Ag: silver).
By means of the diffraction patterns, the presence of Ag is not as obvious as the one presented by Au. However, the deconvolution of the reflection at 2
Weight percent of N, Au, and Ag on the sample after 120-min plasma treatment (XPS analysis).

XPS: X-ray photo spectroscopy; N: nitrogen; TiO2: titanium oxide; Au: gold; Ag: silver.
XPS analysis
XPS was used to analyze the composition and surface chemical states in monometallic and bimetallic samples (N-TiO2/Au, N-TiO2/Ag, N-TiO2/Au/Ag) after 120 min of plasma treatment. Figure 8(a) and (c) shows Au 4f binding energy values at 83.2–83.6 eV (Au 4f 7/2) and 87.0–87.4 eV (Au 4f 5/2) on N-TiO2 −

XPS spectra for monometallic and bimetallic sample after 120-min plasma treatment for (a) Au on N-TiO2 −
On the other hand, the XPS spectrum from 395 eV to 405 eV (Figure 9) shows the nitrogen binding energies values for monometallic and bimetallic samples. The signal observed at 399.6, 399.8, and 400.0 eV corresponds to nitrogen spectrum N 1 s, which is attributed to the nitrogen when it occupies an interstitial site with Ti–O–N bond in TiO2 doped with nitrogen, reported by Viswanathan and Krishanmurthy, which indicates that nitrogen was successfully incorporated. 22 Also, Valentin et al. propose that both substitutional and interstitial doping generate optical properties modifications in titania, and these are associated with the improvement of the catalytic processes. 23
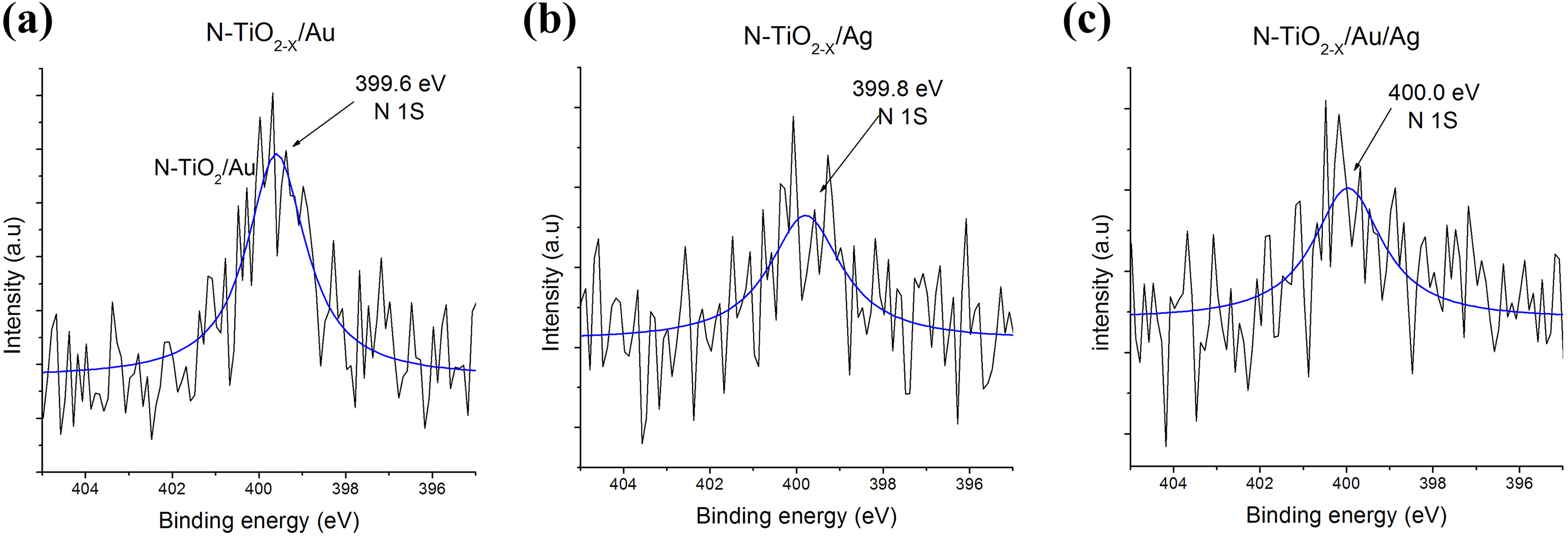
XPS spectra N1s nitrogen in monometallic and bimetallic sample after 120-min plasma treatment for (a) N-TiO2 −
The metal concentrations in the mono- and bimetallic samples with load noble metal particles were examined after 120 min of plasma treatment. By means of the high-resolution XPS spectra for Au, Ag, and N of all samples, their atomic percentage was determined. However, Table 1 summarizes the results in weight percentage. Au concentration on N-TiO2 −
The change of color observed on these materials (Figure 3) after plasma treatment is characteristic of the TiO2 samples with impregnation of noble metal NPs due to surface plasmon resonance (SPR). It has been reported by other research groups for similar samples, for example, the ones obtained using tetrachloroauric (III) acid (HAuCl4) and silver nitrate (AgNO3) used as the starting material on Ag/Au-TiO2 preparation in water-in-oil microemulsion system.
20
Diffuse reflectance absorbance (K/S) of TiO2 P25 and the samples loaded with Au/Ag, Ag, and Au for the TiO2 −
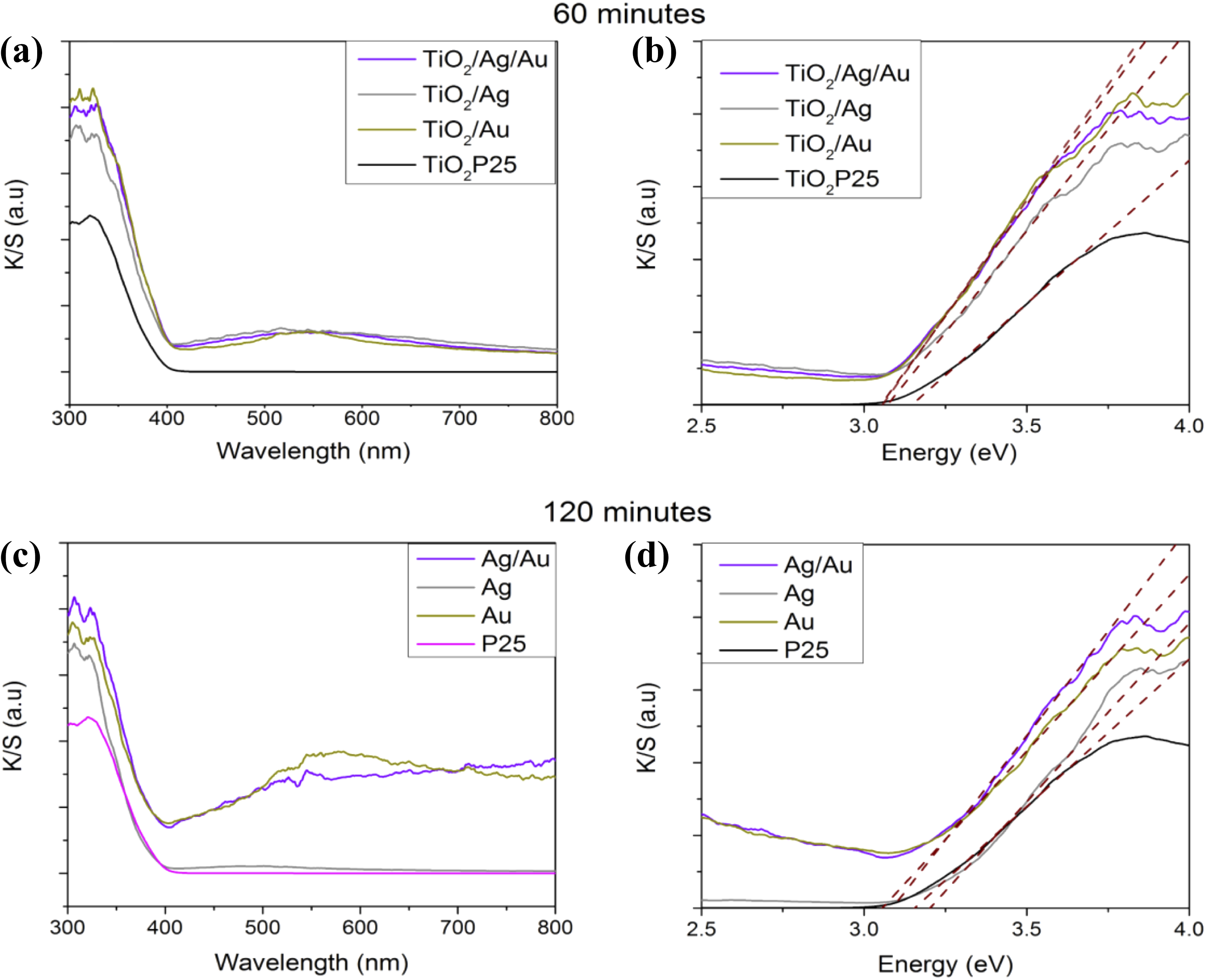
Absorption spectrum and band gap analyses of the monometallic and bimetallic samples at different treatment times with nitrogen plasma (a and b) 60 min and (c and d) 120 min.
The SPR peak of Ag NP appeared at 576 nm; however, Au-NPs give a plasmonic signal located at 580 nm due to SPR of Au-NPs. The plasmon resonance of Au and Ag loaded on TiO2/Au/Ag around 450–700 nm is overlapped, making it difficult to observe the individual SPR response of both metals. The SPR peak of Au/Ag-NPs over TiO2 P25 surface increased even more (approximately 410–750 nm) with loads of 1.7 wt% of Au and 1.3 wt% of Ag, probably due to the particles formed by the junction between Au and Ag particles confirmed by the HRTEM analyses.
From the obtained data, the band gap has been calculated by means of a linear adjustment of the section corresponding to the K/S versus energy of each sample, as presented in Figure 10(b) and (d); the procedure has been described in previous works, 7,8 and the results are presented in Table 2.
Band gap values for the bimetallic samples.
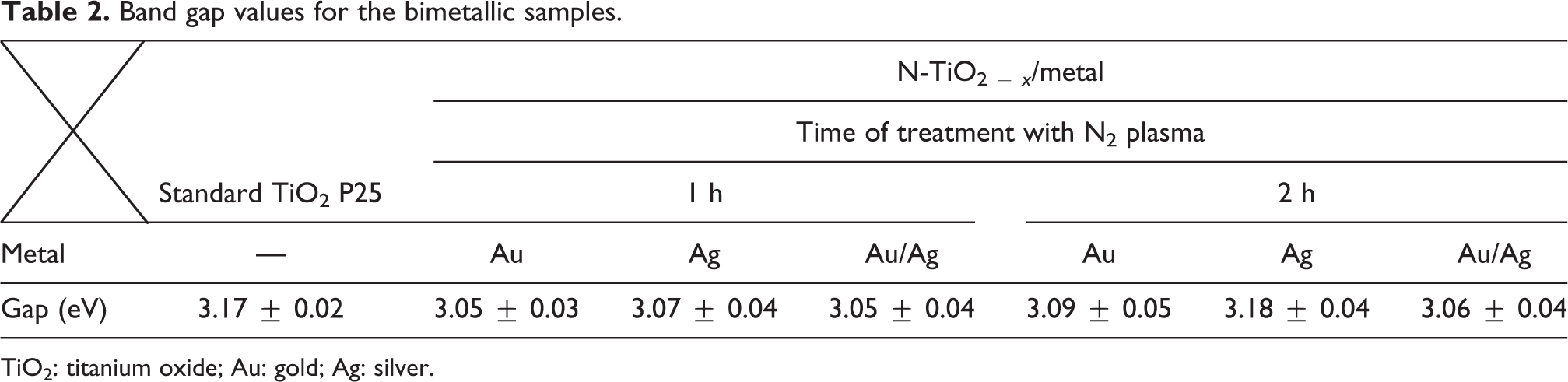
TiO2: titanium oxide; Au: gold; Ag: silver.
Au/Ag, Ag, and Au were loaded on the TiO2 surface with the purpose of increasing the light energy absorption related to the visible range of the electromagnetic spectrum and to act as electron traps to further improve the charge separation on the TiO2 surface. Similarly, the addition of metal particles succeeded in displacing the TiO2 band. This is possible because of the SPR observed for the metallic elements such as Ag and Au, owing to the induction in the visible region absorption, caused by means of the collective excitation of the free electrons. The incoming electric field of the light wave will cause an oscillating behavior in the conduction of the band electrons, and thus, when the light frequency is in resonance with the movement of the electrons, a strong absorption is produced, resulting in SPR.
One can observe a displacement in the band gap for bimetallic samples with respect to P25, from 3.17 eV to a minimum value of 3.05 eV. The change of band gap values when the bimetallic samples increases in the treatment time, from 60 min to 120 min, is minimum. However, the band gap values for both samples are lower compared to that reported for P25, 3.05 and 3.06 eV, respectively. Within the limits of the error, it can be confirmed that a longer time of plasma treatment has minimal effects on the band gap shift.
Conclusions
One-step nitrogen plasma process for bimetallic impregnation is a new methodology to impregnate two metals in only one plasma treatment without changing the crystalline structure of TiO2 (P25). Applying this technique successfully produces N-TiO2 −
Footnotes
Acknowledgements
The authors would like to extend their deepest appreciation to CONACYT (Consejo Nacional de Ciencia y Tecnologia) for financial support of this research project under SEP-CONACYT no. 221418, FOMIX-Yucatan 2008-108160, and CONACYT LAB-2009-01 no. 123913, 188345, 204822, 292692, 294643, 299083. HRTEM analyses were carried out in the Research Center in Applied Science and Technology of Tabasco, UJAT-DAIA (project 225962, CONACYT-INFRA 2014). The authors acknowledge Dora A Huerta, D Aguilar and W Cauich of LANNBIO (CINVESTAV, Mérida) for technical assitance in SEM, XRD, and XPS; the authors also thank S García López and L Díaz Flores for their support with TEM-SAED (Centro de Investigación en Ciencia y Tecnología Aplicada de Tabasco).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
