Abstract
A facile and effective route to synthesize graphene quantum dots for cell imaging and as a deoxidizer by using glucan as a precursor is developed. AuNPs are successfully synthesized by mixing of graphene quantum dots and Au(III) salts without any additional reductants. The reducing driving force of these graphene quantum dots is much weaker than that of strong reducing agents such as NaBH4. The sizes of the as-synthesized AuNPs are much larger, with an average size of 15 nm. Notably, this size range is specifically useful and optimal for the application of AuNPs in biomedical applications. In addition, the as-synthesized graphene quantum dots are also successfully applied in cell imaging.
Graphene quantum dots, synthesized from glucan as a green precursor, are applied to deoxidizer and cell imaging.

Introduction
Graphene quantum dots (GQDs), as the latest addition to the family of nanocarbon materials, are gaining wide attention due to their exceptional chemicophysical properties.1–6 GQDs have an atomically thin graphitic plane (<2 nm thick), a large specific surface area, and external functional oxygen-containing groups. 7 These functional groups can provide abundant active sites for carrying or loading of drug molecules and have compatibility with polymers.8–10 Hence, GQDs have significant potential applications in biological imaging and displays, chemical/photo/electro-catalysis, flexible devices, energy conversion and storage, sensing, and theranostics.11–13 At present, the synthesis of GQDs has been divided into two categories: the top-down method and the bottom-up method. 14 The former is a classical and extensively used method, in which GQDs are synthesized from carbon materials (including graphene, fullerenes, and carbon nanotubes) by cutting them via chemical or physical methods, such as oxidative cleavage, electrochemical oxidation, and laser ablation. 15 For the latter, GQDs are prepared through controllable synthesis or carbonization from suitable organic molecules or polymers. However, there are still some disadvantages to be overcome in these methods.16–21 For examples, in oxidative cleavage, the most used classical method results in potential environmental requirement of pollution due to a large excess of a strong acid, such as sulfuric acid and/or nitric acid. In addition, the reported precursors (e.g. graphene, graphene oxide, graphene nanosheets, and graphite oxide) are prepared commonly through the Hummers methods, which increase both the complexity and the costs. For the electrochemical oxidation, both the pretreatment of raw materials and the purification of the GQD products are time-consuming. The low yield also makes it difficult to realize the mass production of GQDs. Controllable synthesis can be used to prepare GQDs with precise structures through the organic chemical method, but the process is complicated with many chemical steps, leading to low yields. Therefore, eco-friendly methods suitable for mass production have recently received high attention. 22
In fact, our research group has a long-term interest in the synthesis of GQDs and their applications in catalysis, cell imaging, and in antidotes.23–28 Earlier this year, our research group reported the first synthesis of GQDs from the commercial polymer glucan and water, via a green and efficient one-pot hydrothermal method. The obtained GQDs were characterized by transmission electron microscopy (TEM), high-resolution transmission electron microscopy (HRTEM), X-ray photoelectron spectroscopy (XPS), atomic force microscopy (AFM), X-ray diffraction (XRD), Fourier-transform infrared (FTIR) spectroscopy, photoluminescent (PL) spectra, and the Raman spectroscopy. The GQDs have been utilized to stabilize 11 transition metal nanoparticles. Among them, GQD-stabilized RhNP and PtNP are the most efficient catalysts for H2 evolution from the hydrolysis of tetrahydroxydiboron, in which both atoms of H2 evolution are provided by H2O.23–28 As a part of our current studies on the development of new routes for GQD synthesis and novel applications, herein, we report a facile synthesis of GQDs from glucan and their first applications as a deoxidizer and in cell imaging (Scheme 1). In this study, we used our previously reported GQDs (Figure 1) with an average size of 2.74 nm, in which the GQDs were synthesized from the commercial polymer glucan and water.29,30 The GQDs have been successfully used to reduce Au(III) salts for forming AuNP without any additional reductants. Finally, the as-synthesized GQDs have also been successfully applied in cell imaging, due to their favorable hydrophilicity, excellent biocompatibility, highly PL properties, and low cytotoxicity.
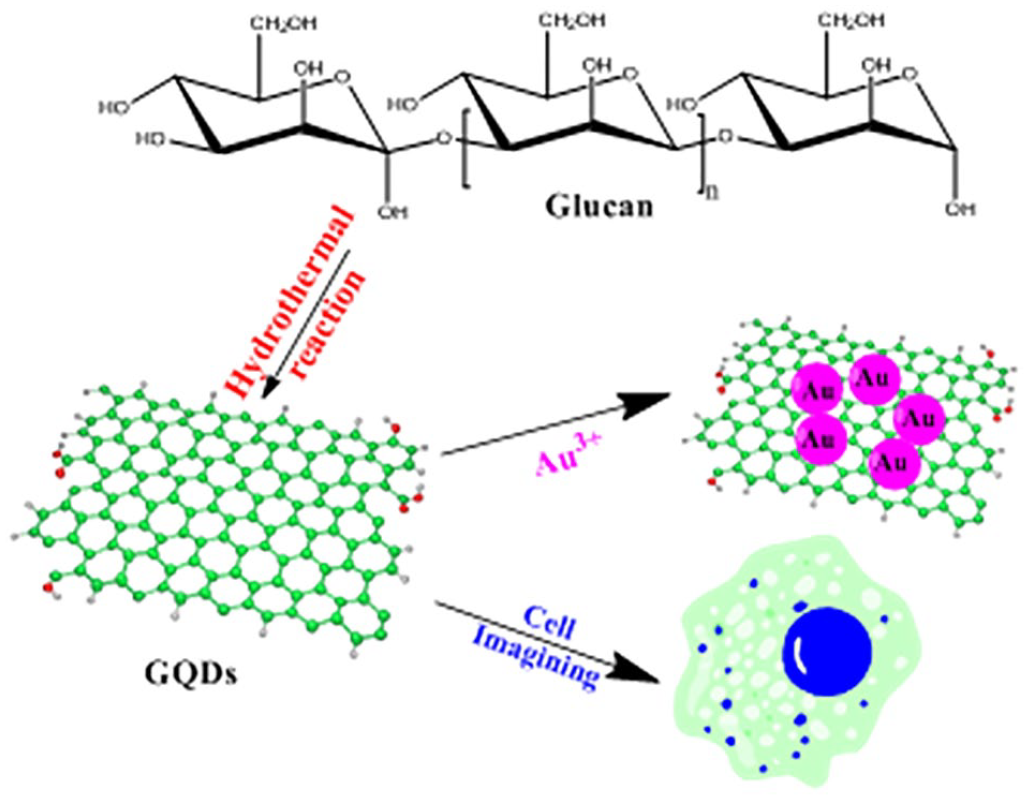
The synthesis of the GQDs and its applications.

TEM image of the GQDs.
Results and discussion
The deoxidization of GQDs
To test the deoxidization properties of the GQDs, they have been successfully used to reduce Au(III) salts to AuNP without any additional reductants. HAuCl4·3H2O (2.5 × 10−4 mmol) was added dropwise to the GQDs solution at 0 °C. The color of the solution changed immediately from yellowish-brown to wine red on reduction of the Au(III) to Au(0) and AuNP formation (Figure 2). These AuNPs are only stabilized by the oxidized GQDs and chloride anions generated by reduction of [AuCl4]−. Indeed, GQDs are a successful reductant of Au(III) salts to Au(0). Because of a surface plasmon band at 528 nm, the characteristic peaks of AuNPs appear in the UV-Vis spectrum (Figure 3). TEM and HRTEM have also been measured to confirm the morphology and size of the AuNPs. The reducing driving force of these GQDs is much weaker than that of strong reducing agents such as NaBH4. The sizes of the as-synthesized AuNPs are much larger (Figure 4(a) and S4), with an average size of 15 nm. Notably, this size range is specifically useful and optimal for the application of AuNPs in biomedicine and in cell imaging.31–33 The HRTEM of one representative particle exhibits Au (200) lattice fringe distances of 0.207 nm (Figure 4(b)).34–49 XPS has been used to further study the Au atomic configuration. The XPS spectra show two different peaks at 83.66 and 87.35 eV, assigned to Au 4f7/2 and 4f5/2, respectively. The binding energies of 83.66 and 87.35 eV are attributed to the zerovalent surface of the Au atoms (Figure 5), confirming that the Au3+ ions have been successfully reduced to Au(0) by the GQDs. For comparison, the commercial polymer glucan (instead of the GQDs) was investigated under the same conditions. The result showed that the color of the glucan solution remained unchanged (Figure 2) and the characteristic peak of AuNP did not appear in the UV-Vis spectrum (Figure 3). This result indicates that glucan does not reduce gold(III) salts to Au(0).

A comparison of the deoxidization of GQDs and glucan.
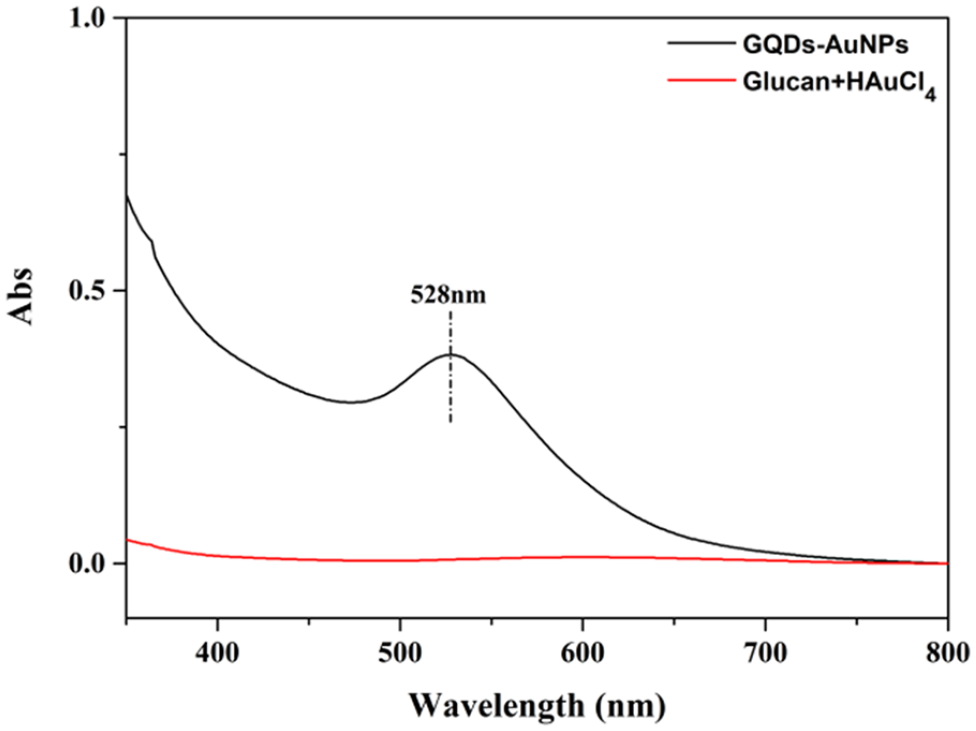
UV-Vis spectrum of GQDs-AuNPs and glucan with HAuCl4.

(a) TEM and (b) HRTEM images of AuNP.
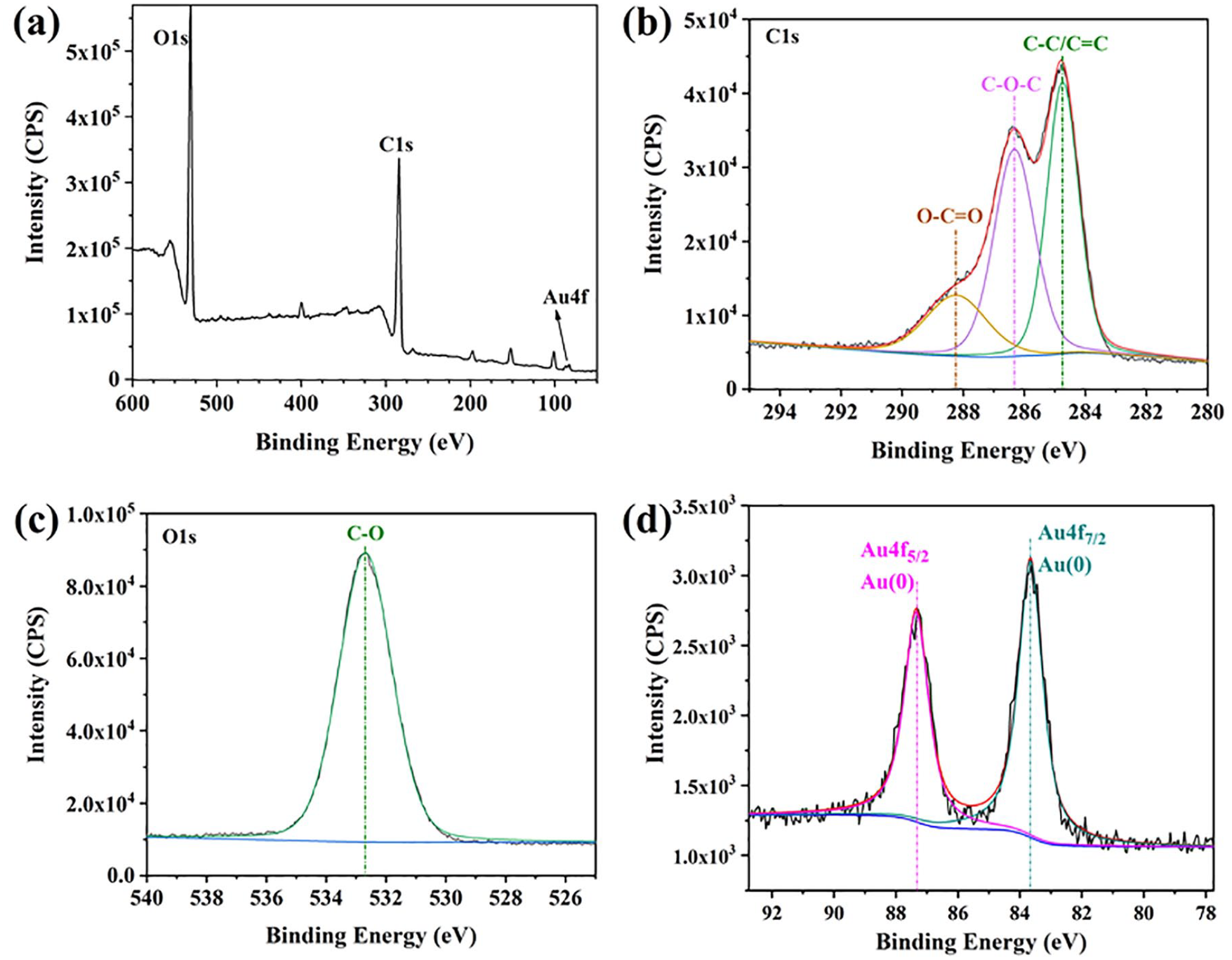
(a) XPS survey spectra; (b) C 1s XPS spectra; (c) O 1s XPS spectra, and (d) Au 4f XPS spectra of GQDs-AuNPs.
Application of GQDs in cell imaging
Recently, PL cell–imaging techniques have emerged as powerful and non-invasive analytical tools for visualizing biological species by virtue of their excellent temporal resolution, fast response times, simple operation, superb sensitivity, in situ workability, and good reproducibility.50–56 As a major branch of PL materials, GQDs are nowadays undergoing significant development, and possess exceptional advantages such as favorable hydrophilicity, excellent biocompatibility, highly PL properties, and low cytotoxicity. 57 As reported in our previous work, when the excitation wavelength is changed from 320 to 480 nm, the corresponding emission peak of GQDs red shifts from 444 to 537 nm in the PL spectra.29,30 This suggests that the PL processes are mainly influenced by surface defects of the GQDs. 58 The above optical results inspired us to apply the GQDs in biological systems. T24 cells have been cultured with a GQD solution in order to explore in vitro cell imaging. As shown in Figure 6(b), all the T24 cells have been stained with the photoluminescence from the GQDs, and show blue emission with the imaging EX channel at 420 nm, revealing a more evident morphology than that of the T24 cells in bright field (Figure 6(a)). This confirms that GQDs are successful for bioimaging and bio-labeling.

(a) Bright field image and (b) fluorescence image of T24 cells incubated with GQDs. Images were acquired using a confocal fluorescence microscope at 60× magnification and EX channel of 420 nm. Scale bar: 25 μm.
Conclusion
The facile synthesis of GQDs from glucan and their first applications as a deoxidizer and for cell imaging have been developed. In this study, GQDs had been synthesized from the commercial polymer glucan and water, according to our previous work.29,30 The AuNPs have been successfully synthesized by mixing of GQDs and Au(III) salts without any additional reductants. Finally, the as-synthesized GQDs have been successfully applied in cell imaging due to their favorable hydrophilicity, excellent biocompatibility, highly PL properties, and low cytotoxicity. Our work has the potential to promote new methods for the synthesis of efficient AuNPs and facilitate the exploration of bioprobe materials.
Experimental section
The preparation of the GQDs
50 mg of glucan was first dispersed in 50 mL deionized water and stirred at r.t. for 15 min. After dissolution, the solution was immediately poured into a 100-mL Teflon-lined stainless autoclave, which was then heated in an oven at 180 °C for 8 h. Then, the autoclave was taken out to be cooled freely. The final brown product was transferred into centrifugal tubes, and centrifuged at 10,000 r/min for 30 min to separate out the precipitate. The yellowish-brown liquid obtained was the solution of GQDs: 0.54 g/L.
The reduction of Au(III) salts to Au(0) by GQDs
The GQDs (1 mL, 0.54 g/L) and deionized water (3 mL) were added to Schlenk flask and the solution was stirred for 10 min at 0 °C. Next, a colorless solution of HAuCl4·3H2O (2.5 × 10−4 mmol in 1 mL of water) was added dropwise, and the color of the solution changed immediately from yellowish-brown to wine red, indicating the reduction of Au(III) to Au(0) and AuNP formation. The AuNPs were kept in aqueous solution for characterization and used as catalysts. A plasmon band was observed in the UV-Vis spectrum of the AuNPs (528 nm).
Cell imaging
T24 cells (1 × 105/well) were seeded into 35 mm cell culture dishes (Corning, USA) containing 1 mL of supplemented RPMI-1640 culture medium with 10% fetal bovine serum (FBS) and incubated for 24 h in 5% CO2 at 37 °C. Next, 1 mL of medium containing GQDs (0.054 mg/mL) was added and further incubated. Finally, the cells were observed under a laser confocal fluorescence microscope (Olympus FV1200, Japan).
Supplemental Material
sj-pdf-1-chl-10.1177_1747519820973934 – Supplemental material for Facile synthesis of graphene quantum dots from glucan and their application as a deoxidizer and in cell imaging
Supplemental material, sj-pdf-1-chl-10.1177_1747519820973934 for Facile synthesis of graphene quantum dots from glucan and their application as a deoxidizer and in cell imaging by Jialu Shen, Weifeng Chen and Xiang Liu in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The financial support from the National Natural Science Foundation of China (21805166), the 111 Project of China (D20015), the Engineering Research Center of Eco-environment in Three Gorges Reservoir Region, Ministry of Education, China Three Gorges University (KF2019-05), the outstanding young- and middle-aged science and technology innovation teams, Ministry of Education, Hubei province, China (T2020004), and the China Three Gorges University is gratefully acknowledged.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
