Abstract
A CuI coordination polymer based on the
Introduction
To date, adsorption is a widely used technique for the separation and removal of pollutants from wastewaters.1–3 Carbon nanotubes (CNTs) are emerging as potential adsorbents because of their well-defined cylindrical hollow structure, large surface area, high aspect ratios, and easily modifiable surfaces. 4 In the last decade, the use of CNTs for the removal of organic and inorganic pollutants has been investigated, and performances comparable to those of commercial adsorbents have been obtained.5–8 Considering the relatively high cost of single-walled CNTs (SWCNTs), multi-walled CNTs (MWCNT) adsorbents may be more promising for practical use.
In addition, many techniques have been developed to fabricate MWCNTs including arc discharge, 9 carbon dioxide reduction, 10 and chemical vapor deposition (CVD). 11 For the CVD method, Fe, Co, and Ni have been most reported.12–14 Some assisted techniques, such as high-intensity electron irradiation, 15 direct current plasma enhancement, 16 electrostatic-control, 17 and magnetism-assisted methods have been utilized. 12 However, most of these methods suffer from the disadvantage of introducing external templates and additives or exhibit poor uniformity.
Coordination polymers (CPs) with various architectures have been employed as sacrificial precursors for synthesizing highly dispersed metal/metallic compounds/carbon materials or composites with high specific surface areas and thermal stability by the calcination–thermolysis method.18–21 Herein, using Cu-CP as a catalyst precursor, a simple and controllable method is described for the synthesis of MWCNTs with uniform diameters. CuCl2·2H2O and
Results and discussion
Single-crystal X-ray diffraction analysis revealed that complex

(a) The coordination environment of the Cu(II) ions in
The IR spectrum of complex
Next, we use the title CP as a combined catalyst precursor for preparing CNTs by the CVD method. The morphology of the MWCNTs was characterized using scanning electron microscopy (SEM) and Transmission electron microscopy (TEM), and typical images are shown in Figure 2(a) and (b). The diameter distribution measured from the TEM images is given in Figure S4, which shows a narrow peak centered at about 60 nm. The Raman spectrum of the MWCNTs is shown in Figure 2(c), where two independent D and G peaks are observed, and the G/D intensity ratio is 0.94, indicating the high quality of the MWCNTs. 25 To investigate the main origin of the defects of functionalities and/or the presence of other carbonaceous impurities in our sample, we characterized the MWCNTs using TGA and IR analysis. The TGA curve and IR spectrum are given in Figures 2(d) and S5, respectively. It can be seen that the MWCNTs start to oxidize at 432°C, and rapid oxidation occurs at 707°C. The catalyst residue is about 7 wt% after oxidation, suggesting that a part of the catalyst residue exists in the sample. The IR spectrum (Figure S5) shows many peaks, indicating that the MWCNTs contain rich functional groups. The strong signals around 3425 cm−1 and 1400 cm−1 in the spectrum are attributed to the O–H stretching vibrations of the phenolic groups and the bending vibrations of the O–H groups, respectively. In addition, the stretching band for C=O at 1635 cm−1 and the stretching vibrations of C–O around 1138 cm−1 are also observed. 26 The OH and COOH moieties of the MWCNTs may provide additional coordination and/or adsorption sites, which may coordinate with multiple metal centers and/or adsorb organic molecules to afford MWCNTs with enhanced adsorption properties.

(a) SEM image, (b) TEM image, (c) Raman spectrum, and (d) TGA curve of the MWCNTs.
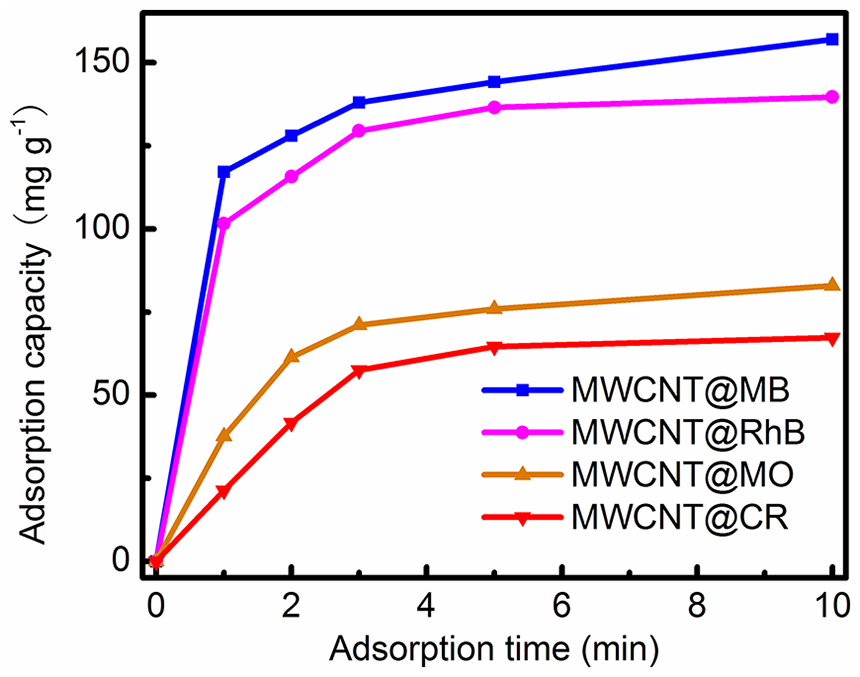
Nowadays, dye contaminants in water have become a potential risk to both human health and ecological systems.27–29 At present, a number of methods such as photocatalytic degradation, sonochemical degradation, Fenton-based oxidation, and ozonation have been developed to remove dyes from wastewater.30–33 However, the cost of the above techniques is high, mainly due to the fact that they cannot be reused. It is therefore important to find a low-cost adsorbent with reversible adsorption capability. The low cost of our MWCNTs motivated us to investigate their dye adsorption capability. Four dyes, methylene blue (MB), rhodamine B (RhB), methylene orange (MO), and congo red (CR), were used to investigate the adsorption performance of our MWCNTs. In a typical experiment, MWCNT powders (~5 mg) were put into an aqueous dye solution (100 mL, 10 mg L−1), followed by stirring at room temperature. At time intervals of 1 min, the dye concentration was measured by using a UV-Vis spectrophotometer at the maximum absorbance of each dye (664 nm, 553 nm, 465 nm, and 490 nm for MB, RhB, MO, and CR, respectively) (Figure 3). The dye adsorption amount
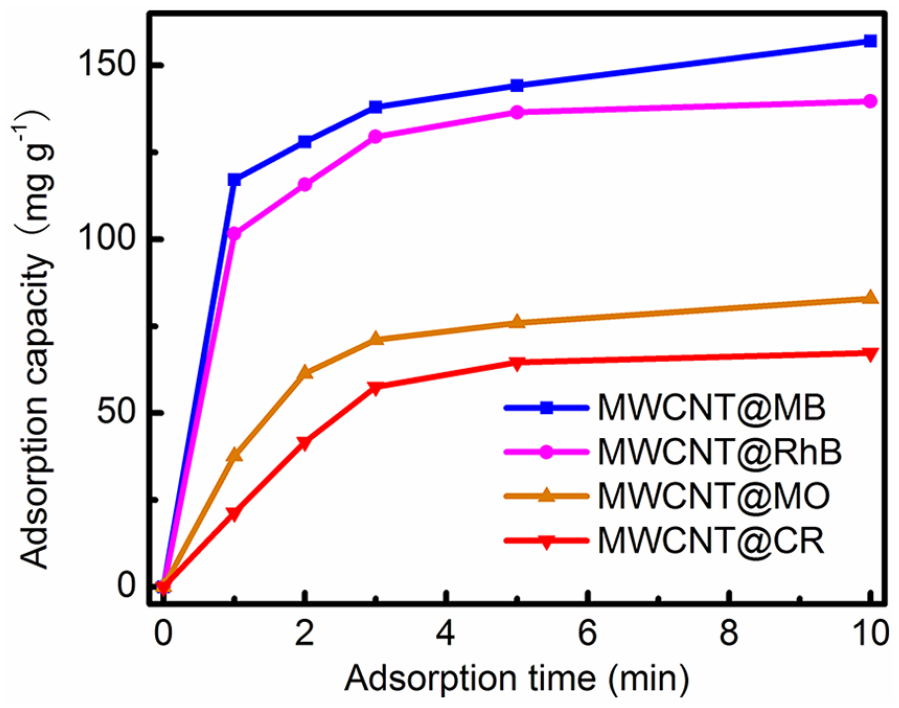
The adsorption capacities of MB/RhB/MO/CR at different times in the dark.

UV-Vis spectra of MB (a), RhB (b), MO (c) and CR (d) solutions after different adsorption times with the MWCNTs.
Adsorption performance comparison of the title MWCNTs with other materials.

The BET surface area of the MWCNTs is 40.11 m2 g−1 (Figure 5), demonstrating that their high performance toward dyes might not be due to their porosity. However, the pore size of the MWCNTs is ca. 1.97–11.08 nm, being larger than the molecule sizes of MB, RhB, MO, and CR, which indicates that the molecule size might contribute to the high uptake amount of MB, RhB, MO, and CR. The presence of OH and COOH in the MWCNTs and methyl groups in MB and RhB might contribute to the formation of abundant hydrogen-bonding interactions, which could favor MB and RhB adsorption. Furthermore, the greater number of π–π stacking interactions between the aromatic rings of MB and RhB and the benzene ring of the MWCNTs might contribute to their excellent adsorption performance toward MB and RhB.

Nitrogen adsorption and desorption isotherms of the title MWCNT. The inset shows the pore size distribution.
We further investigated the desorption performance of the MWCNTs by dipping them (after 10 min of MB, RhB, MO, or CR adsorption) into ethanol solution. The adsorbed (Ad) MB, RhB, MO, and CR gradually desorbed (De) and changed the color of the ethanol to blue, pink, yellow, and orange, respectively. The amount of MB, RhB, MO, and CR in ethanol reached about 141.1, 133.9, 78.8, and 62.7 mg g−1 after 30 min (Figure 6), which suggests that about 96% adsorbed MB, 95.9% adsorbed RhB, 95.1% adsorbed MO, and 93.2% adsorbed CR can be desorbed. The residual unreleased dyes can be ascribed to chemical adsorption. Therefore, considering this high reversible adsorption ability and the low cost of our MWCNTs, they show significant potential for use as an adsorbent to remove dyes efficiently.

Photographs showing the color change of the dyes after adsorption (Ad) and desorption (De).
An adsorption isotherm expresses the relationship between the amount of adsorbate adsorbed per unit weight of adsorbent (

where

where
The experimental results showed that the adsorption data could be fitted with both the Langmuir and Freundlich isotherms. The adsorption constants evaluated from the isotherms for MWCNTs are listed in Table 2. The Freundlich model was found to be a better fit for dye adsorption by MWCNTs, which suggests the adsorbent surface was heterogeneous in nature. The
Langmuir and Freundlich isotherm constants and their correlation coefficients.

During an adsorption process, it is necessary to investigate the adsorption kinetics. Figure 3 shows the data for the adsorption of the dye solutions by MWCNTs at different time intervals. About 73.5% adsorption occurred within 10 min for MB, suggesting that the MWCNTs had fast adsorption kinetics toward dye solutions. The kinetic data were analyzed using pseudo-second-order kinetics,45,48,49 which is based on the assumption that chemisorption is the rate-determining step, and can be expressed as

where

Kinetic adsorption data plots for MWCNT@MB: the transformed rate plot
Rate constants and correlation coefficients of the pseudo-second-order kinetic model.

Conclusion
In summary, high-quality MWCNTs were successfully synthesized over a CuI-CP as a catalyst precursor
Experimental
Materials and measurements
All reagents were obtained from commercial sources and were used without further purification. The 3-dpyb ligand was prepared according to the literature method. 50
FTIR spectra were recorded on a Varian 640-IR spectrometer (KBr pellets). The elemental analyses (C, H and N) were carried out on a PerkinElmer 240°C elemental analyzer. Powder X-ray diffraction (PXRD) data were collected with an Ultima IV instrument with a D/teX Ultra diffractometer at 40 kV and 40 mA with Cu–Kα (
Preparation of the title complex [Cu(3-dpyb)0.5Cl] (1 )
CuCl2·2H2O (0.034 g, 0.20 mmol), 3-dpyb (0.030 g, 0.10 mmol), and NaOH (0.016 g, 0.40 mmol) were dissolved in water (12 mL) at room temperature, then sealed in a 23 mL Telfon reactor and heated at 120°C for 4 days. After slow cooling to room temperature, the obtained red block crystals were filtered and washed with distilled water. Yield: 15% based on Cu. Anal. calcd for C8H9ClCuN2O: C, 38.72; H, 3.66; N, 11.29. Found: C, 38.75; H, 3.61; N, 11.26.
Preparation of the title MWCNT
The above synthesized complex (80 mg) powder was placed in a quartz boat inside a quartz tube (diameter 80 mm, length 1100 mm) located in a horizontal electrical furnace. The furnace temperature was increased to 800°C under an argon flow (300 mL min−1, 99.999% purity) over 40 min, followed by the reduction of the catalyst under a hydrogen atmosphere (200 mL min−1, 99.999% purity) for 60 min, and the CuI-CP was calcined to CuO nanoclusters. The reactor was then heated to the desired temperature under an argon flow. The MWCNT synthesis was performed under a gas mixture of C2H4 (10 mL min−1), Ar (300 mL min−1), and H2 (200 mL min−1) for 30 min. In subsequent heating under a H2 atmosphere, the CuO nanoclusters were reduced to Cu and aggregated into Cu nanoparticles. Following completion of the experiment, the system was cooled to room temperature under Ar to give the MWCNTs.
X-ray crystallography
X-ray diffraction data for complex
Crystal data and structure refinements for complex

Supplemental Material
Revised_Supporting_information – Supplemental material for From coordination polymer to carbon nanotube: Preparation, characterization and rapid adsorption capacity toward organic pollutants
Supplemental material, Revised_Supporting_information for From coordination polymer to carbon nanotube: Preparation, characterization and rapid adsorption capacity toward organic pollutants by Hong-Yan Lin, Yi-Fei Wang, Yuan Tian, Guo-Cheng Liu and Jian Luan in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The support of the National Natural Science Foundation of China (No. 21501013, 21671025, 21971024) and the Key Laboratory of Polyoxometalate Science of the Ministry of Education are gratefully acknowledged.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
