Abstract
A new triazine ligand salt, 2,2′,2″-(1,3,5-triazine-2,4,6-triyl)tris(hydrazin-1-ium) chloride [THT·(HCl)3], and the one-dimensional coordination polymer of cadmium, [Cd2(µ-THT)(µ-Cl)(µ-I)I2]·2(H2O), are prepared and characterized by elemental analysis, Fourier-transform infrared spectroscopy, 1H NMR spectroscopy, and single-crystal X-ray diffraction (for the cadmium polymer). X-ray analysis revealed that the polymeric backbone is extended alternatively by di-iodo and di-chloro bridges; this type of bridge is not observed previously between any metal atoms. There are two types of cadmium atoms in the polymer: cadmium of the polymeric chain and terminal cadmiums. The geometry around the first (CdN2Cl2I2) is octahedral, while the latter (CdN3I2) has an incline to square-pyramidal geometry. The triazine ligand of this structure also bridges two cadmium atoms and acts as an N3 donor toward the terminal cadmium atoms and as an N2 donor toward the cadmium atoms of the chain. In the crystal network of the cadmium polymer, the hydrogen bonds of N–H···X (X: O, N, I) form different hydrogen bond motifs, including

Introduction
1,3,5-Triazine (s-triazine) has been widely used in organic reactions1–6 and offers access to a multitude of useful molecules7,8 due to its specific structure and electronic properties. This scaffold has provided the basis for the design of biologically active molecules with diverse therapeutic profiles,9–13 herbicides,14,15 and polymer photostabilizers. 16 Some trisubstituted-1,3,5-triazines are also used as liposomes. 17
Cyanuric chloride is an essential organic intermediate in which three chlorines can be replaced by –NH2, –OH, –SH, or –NHR, step by step, with high yields.18,19 Cyanuric chloride derivatives have been studied for decades, especially the amino derivatives. It is generally accepted that the first chlorine of cyanuric chloride can be easily substituted by an NH2 group at 0–5
The last decade has witnessed an upsurge of research efforts toward the development of coordination polymers (CPs). Due to their novel and innovative methodologies with diversified and tunable structural frameworks, significant interest has been created in coordination chemistry, focusing mainly on applications such as catalysis, 21 drug delivery, 22 gas storage, 23 luminescence, 24 separation, 25 adsorption, 26 photo-physical studies, antimicrobial properties, materials chemistry, 27 and pollutant sequestration. 28
To extend the chemistry of CPs containing 1,3,5-triazines, in this work, the preparation and characterization (elemental analysis, Fourier-transform infrared spectroscopy (FTIR), and 1H NMR spectroscopy) of a new triazine chloride salt, 2,2′,2″-(1,3,5-triazine-2,4,6-triyl)tris(hydrazin-1-ium) chloride [THT·(HCl)3] (Figure 1), and a one-dimensional (1D) cadmium(II) CP, [Cd2(µ-THT)(µ-Cl)(µ-I)I2]·2(H2O) (

Structure of 2,2′,2″-(1,3,5-triazine-2,4,6-triyl)tris(hydrazin-1-ium) chloride (THT·(HCl)3).
Results and discussion
The reaction between THT·(HCl)3 and CdI2 in a branched tube provided the 1D CP of
Spectroscopic characterization
In the FTIR spectrum of THT·(HCl)3, the presence of ν (N–H) stretching vibrations confirms the successful condensation of hydrazine with 2,4,6-trichloro-1,3,5-triazine. These vibrations shifted by −24 and +67 cm−1, respectively, after complexation to cadmium(II) iodide. In the FTIR spectra of ligand and
The presence of a water molecule in
In the 1H NMR spectrum of the ligand salt, all the protons of the protonated hydrazine arms were observed as a broad peak with a maximum at 4.02 ppm. This peak is shifted to lower magnetic field after the complexation process and is split into two singlets. The protonated salt ligand loses three protons after binding to the cadmium(II) ion, which leads to an increase in the electron density on this ligand, and thus, all protons are shifted to lower magnetic field in
Crystal structure of [Cd2(µ-THT)(µ-Cl)(µ-I)I2]·2(H2O)
X-ray structural analysis of
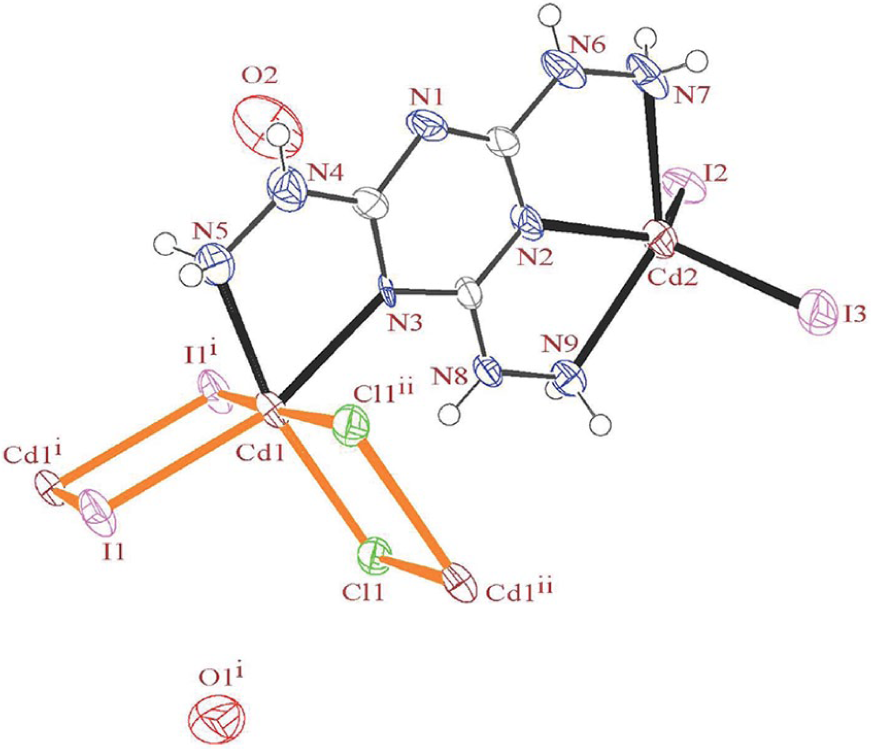
The ORTEP-3 diagram of the molecular structure of
A search of the CSD for all structures containing di-chloro or di-iodo bridges revealed the results shown in Figure 3. This comparison shows that the bond lengths and angles of Cd2(μ-X)2 (X: Cl, I) units are comparable with those of CSD analogues, except for one of the Cd–Cl bonds which is 0.232 Å longer than the CSD average (Figure 3). The Cd–Ibridge bond length among the CSD examples containing a Cd2(μ-X)2 moiety is 0.331 Å longer than the Cd–Clbridge example. The Cd–Cl–Cd bond angle is larger than the Cl–Cd–Cl angle while replacing the chloride ions (Figure 3(b)) with iodide (Figure 3(c)) leads to the order of the angles changing to Cd–I–Cd < I–Cd–I owing to the larger size of the iodide ion. In another comparison, deviation of the atoms from a plane through the atoms of the Cd2(μ-X)2 (X: Cl, I) units was calculated. The CSD average for all deviations was calculated to be 0.043 and 0.015 Å, which means that the two units are planar and have a rectangular shape. No deviation has been observed for similar units of

Coordinated bond lengths and average angles for (a) Complex
In this structure, there are two cadmium(II) ions with different environments, including Cd(Namine)2(Ntz)(Iter)2 and Cd(Namine)(Ntz)(µ-I)2(µ-Cl)2. The Cd1 atom with a coordination number of six has an octahedral geometry. A pentacoordinate geometry in Cd2 may adopt either a square-pyramidal or a trigonal bipyramidal structure. To determine the geometry for such complexes, the formula of Addison et al. 34 was applied. The angular structural parameter (τ) value for Cd2 was calculated to be 0.20, indicating an incline to square-pyramidal geometry.
The THT ligand acting as a bridging pentadentate (Namine)3(Ntz)2-donor forms three five-membered chelate rings. A mean plane through the chelate rings revealed that these rings are almost planar (maximum deviation of atom from plane: 0.042 Å (C3), 0.023 Å (N2), and 0.076 Å (N4)). The dihedral angles between the mean planes through the triazine unit and each chelate rings are 4.23°, 4.01°, and 1.90°, confirming that these rings are almost co-planar and that the hydrazine arms of the ligand do not bend from the molecule plane. Except for the N1–C2 bond (1.28(4) Å), the other triazine unit bond lengths are comparable (1.34(4)−1.37(4) Å), which means that the bond delocalization at this unit is not perfect.
In the structural network of

Packing of

Packing of
Conclusion
In this work, two new triazine compounds, 2,2′,2″-(1,3,5-triazine-2,4,6-triyl)tris(hydrazin-1-ium) chloride [THT·(HCl)3] and 1D-[Cd2(µ-THT)(µ-Cl)(µ-I)I2]·2(H2O) (
Experimental
Materials and instrumentation
All starting chemicals and solvents were of reagent or analytical grade and were used as received. The infrared spectra of the samples as KBr disks in the range 400–4000 cm−1 were recorded with an FTIR TENSOR 27 spectrometer. 1H NMR spectra were recorded on a Bruker Aspect 3000 instrument. The carbon, hydrogen, and nitrogen contents were determined using a Thermo Finnigan Flash Elemental Analyzer 1112 EA. The melting points were determined with a Barnstead Electrothermal 9200 electrically heated apparatus.
Preparation of THT·(HCl)3
A mixture of 2,4,6-trichloro-1,3,5-triazine (4.0 g, 21.69 mmol) and an excess amount of hydrazine hydrate (30 mL) were reacted under reflux conditions (without solvent) for 24 h by keeping the temperature at 80°C. The unreacted hydrazine hydrate was removed using a rotary evaporator to give a white powder. The product was washed twice with ethanol (20 mL). Yield 5.23 g, 66%. m.p. 197°C. Anal. calcd for C3H12Cl3N9 (280.54): C, 12.84; H, 4.31; N, 44.94; found: C, 13.01; H, 4.38; N 44.52. IR (KBr): 3275 m and 3150 m (ν N–H), 1579 s (ν triazine ring quadrant stretching and or δ NH2), 1420 s (ν triazine ring semicircle stretching), 1175 s (ν C–N), 1093 s (ν N–N), 797 m (γ triazine) cm−1. 1H NMR (300 MHz, DMSO-d6): δ = 4.02 (br s, 12H, NH, NH2+).
Preparation of [Cd2(µ-THT)(µ-Cl)(µ-I)I2]·2(H2O)
THT·(HCl)3 (0.14 g, 0.50 mmol), and CdI2 (0.32 g, 0.87 mmol) were placed in the large arms of a branched tube with a total capacity of 15 mL. A mixture of ethanol/H2O (1:1 volume) was carefully added to fill both arms. The tube was then sealed, and the ligand-containing arm was immersed in an oil bath at 60°C, while the other arm was maintained at ambient temperature.35–37 After 5 days, crystals deposited in the cooler arm and were filtered off and dried in air. Yield 0.07 g, 37%. m.p. 240°C. Anal. calcd for C3H13Cd2ClI3N9O2 (848.17): C, 4.25; H, 1.54; N, 14.86; found: C, 4.31; H, 1.55; N, 14.95. IR (KBr): 3546 m (νas O–H), 3495 m (νs O–H), 3251 m (νas NH2), 3194 m (νs NH2), 3134 m (ν N–H), 1635 s (δ H2O), 1581 and 1541 s (ν triazine ring quadrant stretching and or δ NH2), 1419 s (ν triazine ring semicircle stretching), 1170 m (ν C–N), 1088 s (ν N–N), 794 m (γ triazine), 582 w (ρr H2O), 485 m (ρw H2O) cm−1. 1H NMR (300 MHz, DMSO-d6): δ = 2.95 (s, 3H, NH), 2.15 (s, 6H, NH2).
Crystal structure determination and refinement
X-ray diffraction data for
Crystal structure data and structure refinement of complex

Selected bond lengths (Å) and angles (
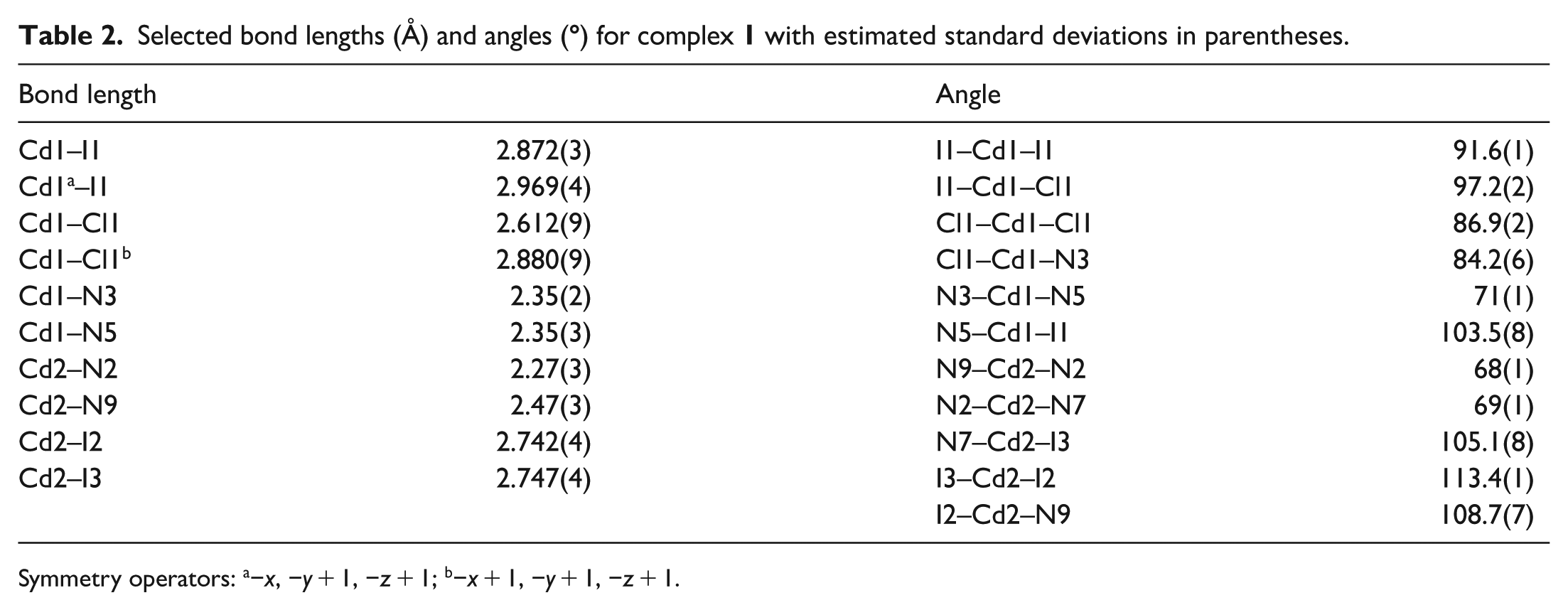
Symmetry operators: a−x, −y + 1, −z + 1; b−x + 1, −y + 1, −z + 1.
Hydrogen bond dimensions (Å and

Symmetry operators: ax, y + 1, z; b−x + 2, −y + 1, −z; cx, y − 1, z; dx + 1, y − 1, z.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
