Abstract
Selective aerobic oxidation of α-pinene to high-value products is a major challenge in chemistry. Metalloporphyrins are proved to be selective catalysts for aerobic oxidation of simple hydrocarbons. Herein, we extend this method to more complex substrates using metallodeuteroporphyrins as model catalysts. It was found that the oxidation occurs mainly on the C=C and allylic C–H bonds of α-pinene influenced by the reaction temperature, reaction time, catalyst concentration, and oxygen flow rate. Allylic C–H oxidation products are obtained with a maximum selectivity value of 78.4% using the following reaction conditions: 105°C, 7 h, 5 ppm, and 60 mL/min. The influence of the metal nuclei of the metallodeuteroporphyrins on this reaction is also investigated. It was found that metallodeuteroporphyrins with Fe3+ as the metal nucleus exhibit the highest catalytic activity.
Introduction
Due to the gradual decrease of fossil resources, there is an ever-increasing interest for the transformation of renewable resources toward more valuable products.1,2 Turpentine is one of the most important and cheapest monoterpenes.
3
As the major component of turpentine, the content of α-pinene (
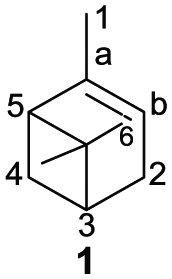
Structure and different oxidation sites of α-pinene.
α-pinene oxidation products are crucial intermediates with broad applications in the fragrance industry and in organic synthesis.6,10–17 The 2,3-epoxypinane (

Aerobic oxidation products of α-pinene derived from catalysis by MDPs.
In general, direct oxidation of α-pinene by stoichiometric oxidants such as organic and inorganic peracids, NaIO4, or H2O2 is the most efficient process for the production of oxygenated α-pinene derivatives.16–19 These procedures have raised concerns due to the production of large amounts of toxic waste and their adverse impacts on the environment. In contrast, molecular oxygen is an excellent oxidant because it is inexpensive, eco-friendly, abundant, and readily available. However, selective aerobic oxidation of α-pinene by molecular oxygen is still a major challenge in academic and industrial research because of the inertness of molecular oxygen and the complex molecular structure of α-pinene.
In recent years, considerable efforts have been made to investigate highly selective aerobic oxidation of α-pinene.10,13,20–23 Selective aerobic epoxidation of α-pinene to 2,3-epoxypinane (
During the last decade, numerous research projects have shown that metalloporphyrins (MPs) are efficient and selective catalysts for aerobic oxidation of hydrocarbons to their oxyfunctionalized products in the absence of solvents and additives (co-catalysts, reductants, stoichiometric oxidants, and other additives).24–27 Furthermore, compared with commercial catalysis in industrial processes, MP-based technology can be performed under mild conditions with significantly improved reaction conversions and product selectivities. The oxidation of simple hydrocarbons such as cyclohexane, cyclohexene, toluene, and other substrates has been widely studied.24,25 Furthermore, industrial-scale application of this biomimetic oxidation technology for aerobic oxidation of cyclohexane has been reported. 26 However, only simple hydrocarbon molecules have been investigated in most of these research studies. Selective aerobic oxidation of complex substrates with different reactive bonds in the same molecule catalyzed by MPs in solvent- and additive-free systems has not been examined frequently in previous research. 27
In the present work, an efficient metallodeuteroporphyrin (MDP)-catalyzed aerobic allylic oxidation of α-pinene (see Figure 1), containing one active C=C bond and six different C–H bonds, was established in the absence of solvents and additives. Allylic oxidation products were obtained in high selectivity because of the higher reactivity of the allylic C–H bonds of α-pinene in this aerobic oxidation system. The effects of reaction parameters and the metal nucleus of the MDPs on the yield and selectivity of the allylic oxidation products were investigated. A possible reaction mechanism for this procedure and the role of MDPs in this reaction are discussed.
Results and discussion
Aerobic oxidation of α-pinene catalyzed by MDPs
Oxidation of α-pinene was performed in solvent-free liquid substrates with molecular oxygen at atmospheric pressure. MDPs were applied as catalysts without the addition of co-catalysts, reductants, stoichiometric oxidants, or other additives. α-pinene was smoothly oxidized to high-value oxygenated products at high concentrations in this process. According to gas chromatography (GC) and gas chromatography–mass spectrometry (GC-MS) data, the oxidation product consisted of 2,3-epoxypinane (
Comparative analysis of the aerobic oxidation of α-pinene using different types of catalysts.

Reaction conditions: α-pinene (100 g), FeClDPDME (5 mg), 105°C, ambient pressure, flow rate of oxygen = 60 mL/min, 7 h.
Reaction conditions: α-pinene (100 g), 105°C, ambient pressure, flow rate of oxygen = 60 mL/min, 7 h.
It has been reported that α-pinene could also be oxidized by molecular oxygen in the absence of any catalyst. 31 In order to obtain the data regarding the influence of MDPs on this procedure, the selectivity of various oxidation products without any catalysts was investigated, and the results are also listed in Table 1. The results showed that similar products were obtained in this aerobic oxidation process. However, compared with the reaction without a catalyst, the oxidation under the catalysis of MDPs was restricted to allylic C–H bonds, indicating that these reactions proceeded in a different manner and that MDPs played an important role as site specific catalysts in this reaction.
Reaction mechanism of the aerobic oxidation of α-pinene via the catalysis by MDPs
Aerobic oxidation of hydrocarbons in the presence and absence of catalysts has been widely investigated in previous research studies. It is widely accepted that these processes are initiated and propagated by iron-oxy radicals.24,31–37 These radicals remove hydrogen atoms in substrate molecules, yielding the corresponding resonance-stabilized alkyl radicals. In aerobic oxidations catalyzed by ferric porphyrins (FePs), high-valence radical intermediates generated according to the process shown in Scheme 1 are regarded as the active species.24,34–36 Although these intermediates were not characterized directly in this work because of the limitations of our detection conditions, the formation of high-valence FeP radical species has been proved by Groves and Watanabe38,39 and Decker et al., 40 and has been successfully tracked by ultraviolet–visible (UV-Vis) spectra. The UV-Vis spectra of the FeClDPDME interaction with oxidants in dimethyl sulfoxide (DMSO) were recorded and are shown in Figure 3. The results demonstrated that the absorption peak of FeClDPDME was red-shifted from 321 to 390 nm gradually and the Q bands absorption peaks at 558 nm and 586 nm were strengthened after the bubbling of molecular oxygen. Such observations were consistent with the formation of [PFeIV=O]+ from the activation of molecular oxygen by ferric(III) porphyrins.38,39

The formation of high-valence radical intermediates in FeP-catalyzed oxidation reactions.

The UV-Vis spectrum of the FeClDPDME interactions with molecular oxygen.
Even though the details for the formation of oxygenated hydrocarbon products are not well established at this stage, an increasing number of scientists agree on the hypothesis that a so-called oxygen rebound step is involved in FeP-catalyzed aerobic oxidation reactions.24,34 In this consensus mechanism, high-valence FeP radicals remove hydrogen atoms that are components of relatively weak C–H bonds (e.g. allylic C–H) in substrate molecules, yielding caged pair of resonance-stabilized alkyl radicals and high-valence FePs, which collapse to an alcohol or over-oxidized ketone products via a complex oxygen transfer process (Scheme 2(a)). 24 The interaction of high-valence FeP radicals with C=C bonds leads to the formation of a caged pair of alkyl cation radicals with high-valence FeP radicals and collapses by free-radical interactions to give an epoxide through an electron-transfer procedure (Scheme 2(b)). 41 It has been reported that the methyl group at the a-site of α-pinene would sterically hinder the approach of other molecules and hamper their addition to this site. 31 Perhaps, this is the most probable reason for the high site specificity of allylic C–H bonds in this catalytic oxidation system.
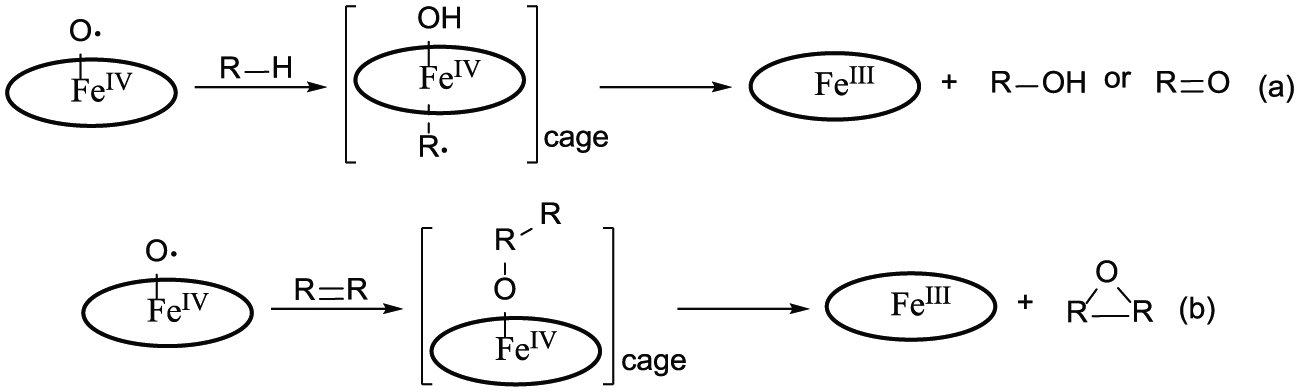
Reaction mechanism for the FeP-catalyzed aerobic oxidation of (a) alkanes and (b) alkenes.
Quantum chemical calculation results from previous investigations showed that removal of hydrogen from α-pinene mainly occurs at the 1 and 2 sites of the molecule and produces two resonance-stabilized radicals (Figure 4).
31
In most cases, secondary C–H radicals are much more stable than the radicals derived from primary C–H bonds. Thus, the selectivity of verbenyl-oxidation products (

Two resonance-stabilized radicals formed upon abstraction of a proton from α-pinene.
Optimization of the working conditions
MPs are important biomimetic catalysts with bright prospects in industrial applications. In order to obtain the optimum reaction conditions, FeClDPDME was selected as a model to investigate the influence of temperature, time, catalyst concentration, and oxygen flow rate on this reaction.
Table 2 lists the
Comparison of the aerobic oxidation of α-pinene at different reaction temperatures.

Reaction conditions: α-pinene (100 g), MPs (5 mg), ambient pressure, flow rate of oxygen = 60 mL/min, 7 h.
Reaction conditions: α-pinene (100 g), ambient pressure, flow rate of air = 60 mL/min, 7 h.
To identify the influence of the external reaction temperature on the selectivity of MDPs, aerobic oxidation of α-pinene at various reaction temperatures in the presence and absence of catalysis was investigated (Table 2). The results show that the selectivity of various oxidation products under the catalysis of MDPs at low reaction temperature was similar to those obtained without catalysis. The selectivity ratio between epoxidation products and allylic C–H oxidation products in the absence of MDPs did not change with variation of the external reaction temperature. However, the selectivity of allylic C–H oxidation products in MDP-catalyzed reactions increased significantly when the reaction temperatures were raised and reached a maximum value of 78.4% at a reaction temperature of 105°C, indicating that the external reaction temperature exerted a tremendous influence on the selectivity of MDPs.
The conversion of α-pinene and the selectivity of various oxidation products as a function of reaction time at 105°C were evaluated and are illustrated in Figure 5(a) and (b). The results show that the

The effects of reaction time on the (a)
In our previous research, catalyst concentration was found to be a critical factor in biomimetic reactions catalyzed by MDPs.
45
Table 3 shows the conversion of α-pinene and the selectivity of various oxidation products over the FeClDPDME concentration from 1 to 9 ppm. The results indicated that the conversion of α-pinene and the selectivity of the allylic C–H oxidation products were enhanced with increased FeClDPDME concentrations when the catalyst concentration was lower than 5 ppm. Higher concentrations of FeClDPDME have been reported to lead to the formation of inactive μ-oxo MP dimers.
27
Thus, when more catalyst was added, the efficient concentration of the active catalyst was reduced and caused a decrease of the
Comparison of aerobic oxidation of α-pinene at different catalyst concentrations.

Reaction conditions: α-pinene (100 g), 105°C, ambient pressure, flow rate of air = 60 mL/min, 7 h.
The effect of oxygen flow rate on the conversion and product selectivity was also investigated. As is illustrated in Figure 6, the conversion of α-pinene was enhanced with an increase of the flow rate of oxygen, especially at an oxygen flow rate range from 40 to 60 mL/min. However, when the oxygen flow rate was higher than 80 mL/min, the selectivity of products

Effects of oxygen flow rate on the
Effect of the central metal on the catalytic activity of MDPs
As reported in our previous work,27,45 MDPs complexed with various metal ions presented very different catalytic activities. Table 4 summarizes the data obtained from the aerobic oxidation of α-pinene under the catalysis of various MDPs. The results show that the
Comparison of the aerobic oxidation of α-pinene in the presence of different metals.
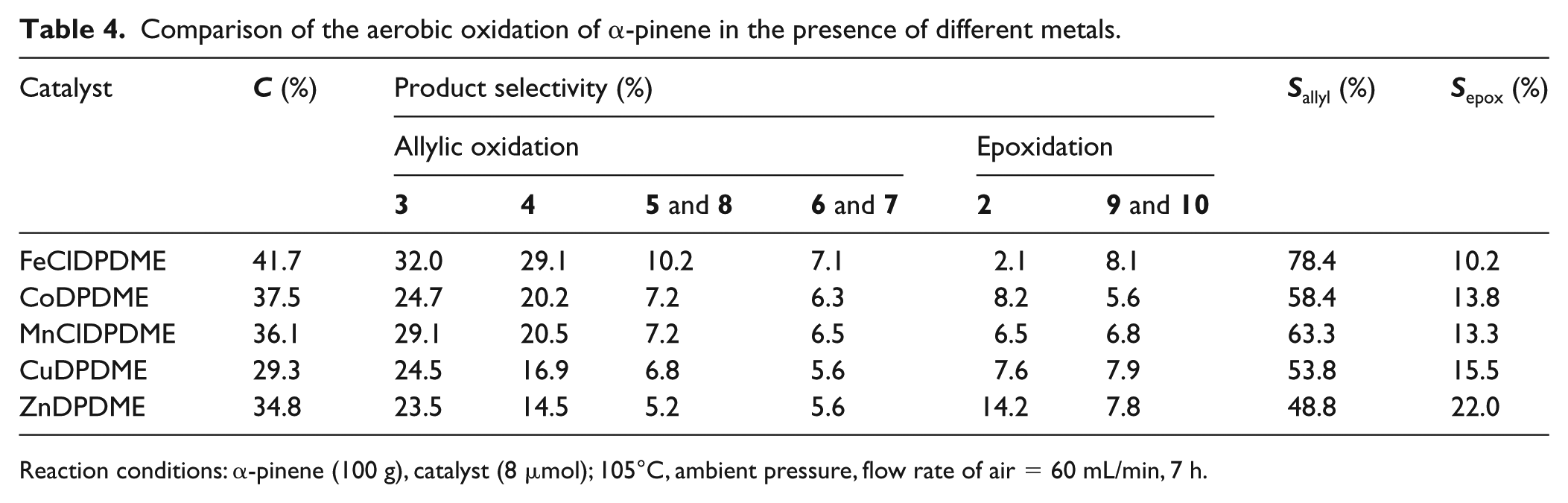
Reaction conditions: α-pinene (100 g), catalyst (8 μmol); 105°C, ambient pressure, flow rate of air = 60 mL/min, 7 h.
Under the optimized reaction conditions, FeClDPDME was found to be the most efficient catalyst with the highest conversion and selectivity values, while the lowest yield of allylic oxidation products was obtained with the CuDPDME catalyst. The catalytic activity of MDPs follows the sequence: FeClDPDME > CoDPDME≈MnClDPDME > ZnDPDME > CuDPDME, similar to the conclusions obtained in previous studies.27,46 It has been reported that the catalytic activity and selectivity of MPs are influenced by the stabilities and redox potentials of the central metals.27,28,46,47 It is conceivable that these factors may explain the higher catalytic activity of FeClDPDME in comparison to the other catalysts.
Conclusion
Aerobic oxidation of α-pinene catalyzed by MDPs in the absence of solvents and additives at ambient pressure was studied. The
Experimental
Instruments and equipment
1 H NMR spectra were obtained on a Bruker Avance III 500 MHz spectrometer (Bruker, Germany) using CDCl3 or DMSO-d6 as solvents, and all chemical shifts were compared to tetramethylsilane (TMS) as the reference. Infrared (IR) spectra were recorded using a Thermo Nicolet IS10 IR instrument (Thermo, USA) using the attenuated total reflection method. ESI-MS/MS results were recorded on a Finnigan TSQ Quantum ultra AM mass spectrometer (Finnigan, USA) with an electrospray ionization (ESI) source. UV-Vis spectra were recorded on a Lambda-35 UV-spectrofluorimeter (Perkin-Elmer, USA). GC data were acquired on a Shimadzu GC-2014AF (Shimadzu, Japan) equipped with a 0.25 mm i.d. × 30 m quartz capillary column and a hydrogen flame ionization detector. GC-MS analysis was performed on an Agilent 6890N/5973N GC-MS instrument (Agilent, USA) equipped with a 0.25 mm i.d. × 30 m quartz capillary column.
Chemicals and reagents
Hemin (purity 98.5%) was obtained from Tianjin Institute of Life Sciences Applications (China). Other analytical grade chemicals were obtained commercially and were used without further purification. Organic solvents were dried before use. α-pinene (96.2%) was obtained from Zhuzhou Sonbon Forest Chemical Co., and redistilled to over 98.0% before use. Metallodeuteroporphyrin dimethyl esters (MDPDMEs,

Chemical structure of the MDPs. M = FeCl (
Aerobic oxidation procedure
Aerobic oxidation of α-pinene was carried out in a 250 mL four-necked glass flask containing a reflux condenser, a thermometer, and a breather pipe. The mixture was agitated and heated with an electromagnetic heating stirrer apparatus. Air or oxygen was pumped into the reactor through an air bottle under continuous stirring and the flow rate was regulated and measured by a needle valve and a rotameter, respectively. In a typical procedure, α-pinene (100 g) and the appropriate amount of MDP were added. When the reaction temperature reached 60–130°C, oxygen was added to the mixture with a flow rate of 10–100 mL/min. Samples were taken from the reaction mixture every 1 h and quantified by GC using n-nonane as an inert, internal standard. Hydroperoxide yields were determined by double injection before and after the treatment of the reaction sample with triphenylphosphine. The components of the acquired samples were identified by GC-MS.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful for the financial support from the National Natural Science Foundation of China (31600466) and the Fundamental Research Funds of the Research Institute of Forest New Technology (CAFYBB2018SZ012).
