Abstract
At present, metalloporphyrin compounds demonstrate three main uses as anticancer sensitizers: (1) photosensitizers, (2) photothermal conversion agents, and (3) ultrasound sensitizers. Developing efficient sensitizers for cancer with excellent controllability and biocompatibility is an important goal of oncology medicine. Because of the different structural diversity of anticancer sensitizers, such sensitizers are used for treating cancers by employing a variety of tumor treatment methods such as mature photodynamic therapy, commonly used clinically photothermal therapy and promising sonodynamic therapy. Among the many sensitizers, metalloporphyrin-complex sensitizers attract wide attention due to their excellent performance in tumor treatment and diagnosis. This review briefly describes some metalloporphyrin anticancer drugs and diagnostic agents related to photodynamic, photothermal and sonodynamic therapy, and discusses the roles of metal atoms in these drugs.
Keywords
Metalloporphyrins has been utilized in the diagnosis and treatment of three treatment modalities. And the technology matures over time.

Introduction
Cancer is one of the major life and health problems faced by human beings in modern society.1–4 Although cancer treatments are in constant development, traditional treatments such as drugs, surgery, chemotherapy, radiotherapy, and immunotherapy often have strong side effects or shortcomings. More and more research studies are turning to multimode treatment strategies, and photodynamic therapy (PDT), as an important supplementary means of conventional treatment, receiving significant development.5,6
PDT utilizes a combination of light and a photosensitizer that interacts with oxygen to destroy new tumor blood vessels and the tumor’s internal environment. It is a fast-growing and promising therapy. 7 There are currently two main mechanisms (Figure 1). The type I electron transfer mechanism generates reactive oxygen species (ROS), such as superoxide compounds or hydroxyl radicals. The type II mechanism utilizes a photosensitizer that absorbs a photon and is excited to the triplet state, and then transfers the energy to an oxygen molecule, thereby generating cytotoxic singlet oxygen. The type II mechanism is considered as the main active pathway operating during PDT. Therefore, important indicators for evaluating the performances of PDT and photosensitizers are the singlet oxygen or ROS yield. 8 Through numerous studies, it has been found that metal photosensitizers have many advantages such as higher singlet oxygen yield, easy excitation, and a wide range of applications in imaging. However, in clinical practice, the therapeutic effect of photosensitizers is often not obvious or has many limitations, resulting in the use of photosensitizers in clinical imaging rather than as clinical anti cancer drugs. Therefore, the research on photosensitizers should not only focus on improving their targeting and singlet oxygen yield but also needs to fundamentally innovate the use of phototherapy drugs. 9 Only in this way can the limitation of insufficient PDT treatment be solved, and the advantages of photosensitizers in tumor diagnosis will be improved.
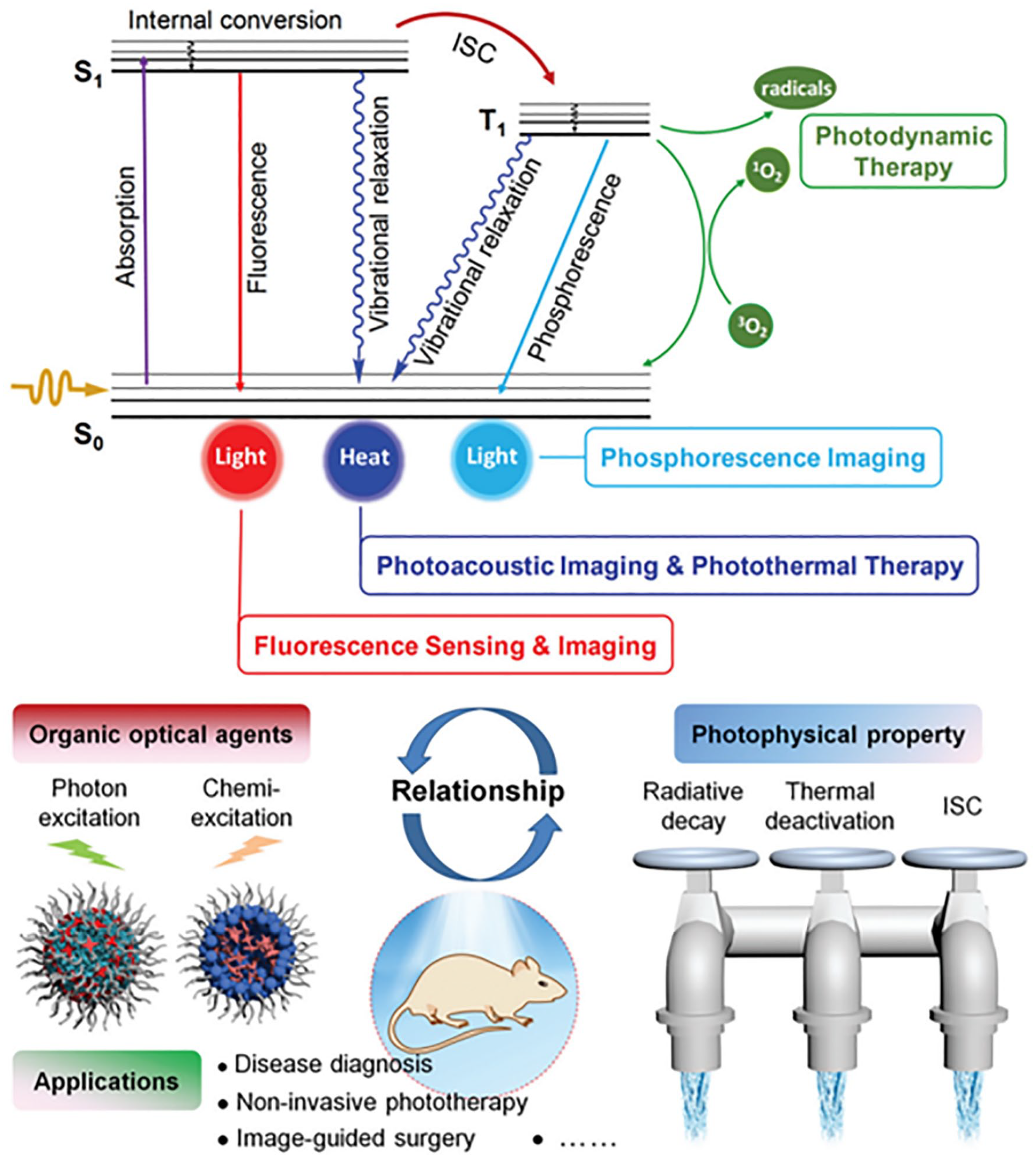
Schematic of a Jablonski diagram and its important role in guiding the design of organic optical agents with optimized phototheranostic performance. 10 Reproduced from Ref. 10 with permission. Copyright 2020, Wiley-VCH.
PDT has two obvious defects: one is that the penetration of light is not strong enough to reach deep tumor lesions; the second is that long-term use of PDT will cause the accumulation of skin toxicity. 11 Photothermal therapy (PTT) is a new type of treatment developed to solve the problem of single PDT with low efficacy. PTT can generate a certain amount of singlet oxygen and a huge quantity of heat by activating a photothermal conversion agent (PTA). The use of targeting PTAs causes significant cytotoxicity against cancer cells and affects tumor-directed therapy.12–15 PTT requires a PTA with an extremely high targeting efficiency and a high photothermal conversion efficiency, as well as high stability. PTA is more suitable for imaging technology, so PTT is regarded as a promising new technology for diagnosis and treatment. However, the shortcomings of PTT are also very obvious. There is still no effective solution for targeting tumors inside the human body, improving the low absorption rate of photothermal conversion agents by tumors, and the lack of clinically efficient PTAs.
Sonodynamic therapy (SDT) is a new type of treatment based on PDT aimed at solving the problem of the poor light penetration of PDT. By using a specific intensity of ultrasound to accurately excite the sensitizer at the tumor site, the cytotoxicity is generated and causes cancer cell damage, while normal cells around the tumor will be less affected. Ultrasound is extremely penetrating to the human body, so it can excite the sensitizer administered inside the human body. It is thought that such treatment overcomes the major defects of PDT and has the potential for clinical treatment. At present, there are two main mechanisms for SDT. 16 In addition to the generation of singlet oxygen, it is generally believed that there is a cavitation effect caused by ultrasonic pressure and ultrasound-mediated killing. The cavitation effect refers to ultrasonic waves that act on the junction of blood fluid and tissue to form a cavity. The sound intensity reaches a certain level causing oscillation and collapse of the cavity. 17 Eventually, violent chemical reactions and luminescence destroy tumor tissue blood vessels (mainly the sensitizer reacts with oxygen in the cavity to produce peroxyl radical and alkoxy radicals). Scientists use ultrasound-targeted microbubble destruction (UTMD) technology to control drug release. This technology (Figure 2) also combines certain physical therapy effects and is a safe and efficient way of targeting drug delivery. In addition to excellent tissue penetration, SDT can be monitored by a variety of imaging techniques. Among them, contrast-enhanced ultrasound (CEUS) is of great significance to the monitoring of SDT. Compared with the monitoring of PDT and PTT, the monitoring of SDT is more convenient and has a stronger positioning ability and higher sensitivity.

Ultrasound destroys tumor tissue blood vessels by rupturing cavities.
The scope of this review covers the latest advances in PDT, PTT, and SDT in the diagnosis and treatment of tumors. At the same time, we will focus on the research and applications of metal porphyrin complexes as sensitizers. This review selects some metal porphyrin complex sensitizers as representatives and highlights the advantages and prospects of metal complexes as sensitizers.
Applications of metalloporphyrin compounds in PDT
Since the first-generation photodynamic drug-photofrin (Figure 3) was listed by the FDA (the U.S. Food and Drug Administration) in 1995, porphyrin and its derivatives have generated interest in the pharmaceutical sciences. Such compounds are considered to be excellent photosensitizers with exceptional photostability and biocompatibility. However, due to tissue hypoxia and the limited light penetration capacity, the use of porphyrin drugs has shown insufficient treatment effects.18,19 With the continuous development of porphyrins, a variety of solutions have emerged. Considering these limitations, porphyrin compounds are being utilized as nanocarriers to improve their functionality.
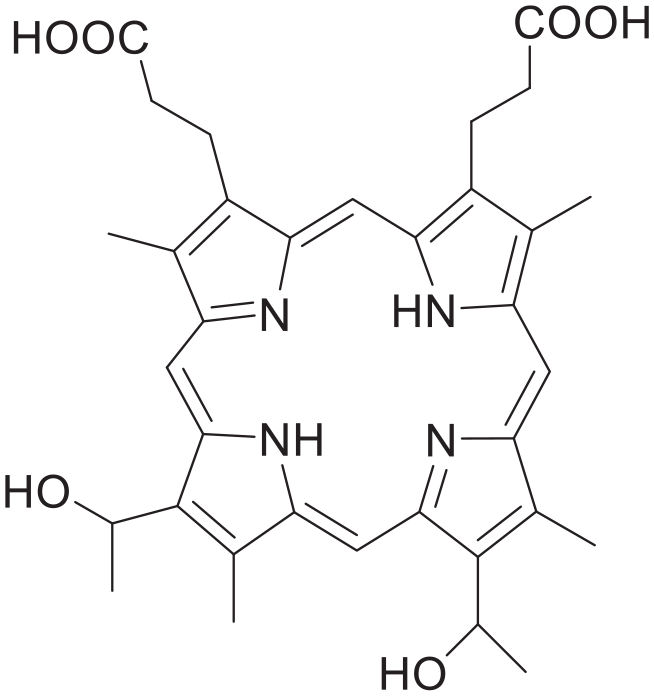
Hematoporphyrin, a component of photofrin.
The transition-metal elements often have unique physical and chemical properties. 20 In the porphyrin molecule, the metal cation is distributed in the center of the “porphyrin” with four nitrogen coordinations. 21 The combination of a metal with porphyrin can lead to excellent physiological activity and has a role in the diagnosis and treatment of surface tumors while giving traditional porphyrin compounds more features. Multiple metals stabilize the formation of chelated porphyrin nanoparticles before and after without any additional chelator. Adding metals can significantly alter a range of behaviors, such as modulating phototherapy effects; imparting responsiveness to biological stimuli; or providing contrast for magnetic resonance, positron emission spectroscopy, or surface-enhanced Raman imaging. 10 Related research has also slowly shifted from the initial zinc and copper porphyrins to structural modifications and optimization of the preparative methods.22–24 For example, Kevin et al. improved the biocompatibility and drug loading of traditional copper- and zinc-porphyrin-linked liposomes. 25 Cu-PoP fluorescence was quenched by more than 95%, while Zn-PoP undergoes only very minor self-quenching under laser irradiation. Liposomes can be released by treating with near-infrared light and the drug release rate can reach 85% after excitation, while the release rate under nonlight conditions is only about 20%, thus showing good stability that can reduce side effects. Experiments have shown that chelating copper thereby significantly reduces the phototoxicity of porphyrin compounds. On the other hand, to improve the targeting of metalloporphyrins and increase the concentration of photosensitizers in tumor tissues, nanoformulations are often used. For example, Wang et al. used the co-assembly strategy successfully. With this technology, many multifunctional nanosystems consisting of differently stacked metalloporphyrins had been developed.26,27 Among them, Zn porphyrin has the highest singlet oxygen yield among metal porphyrins, while Gd porphyrin has the highest proton relaxation efficiency. The two assembled together give mGZNs, the hemolysis rate of which is only 0.5%, and has good blood compatibility. The increase in singlet oxygen yield shows an obvious PDT effect, the mGZNs’ half-life in vivo and the high tissue retention rate of 1.68 h will be better for image-related tissues, and the signal intensity of magnetic resonance imaging (MRI) after intravenous injection of mGZNs is 1.45 times higher than before. It is worth mentioning that co-assembly increases hydrophilicity. On the other hand, Liu and colleagues used an in situ self-assembly strategy to synthesize oil chain Zn porphyrin (OC-Zn-TAPP) nanoparticles. These nanoparticles will specifically target the tumor microenvironment and can generate a large amount of oxygen through the reaction of manganese dioxide and hydrogen peroxide in the acidic tumor environment, providing basic conditions for PDT. The OC-Zn-TAPP released at the same time undergoes in situ self-assembly to form nanofibers, which greatly enhances the retention of drugs in the tumor microenvironment. The metal element in the porphyrin center not only facilitates oxygen production but also makes the self-assembly strategy more flexible. 28
In addition to the commonly used copper and zinc porphyrins, other transition-metal elements also have excellent antitumor effects. For example, Mn porphyrin promotes protein oxidation so that it has an excellent curative effect and selectivity on tumors. 29 Porphyrin complexes centered on a platinum atom have high cancer cell toxicity. At the same time, the introduction of platinum atoms strengthens the stability and makes it easier to excite the porphyrin. 30 Another strategy is to increase the tumor uptake after introducing metal elements at the porphyrin center, thereby improving the efficiency of PDT. 31 Yu et al. developed a lutetium (Lu) nanoporphyrin PEG complex: 177Lu-PPNs-10 nm (Figure 4). The radioactive elements 177Lu, 90Y, and 131I can all emit light in positron emission tomography (PET) images, so they can be used to locate cells and tissues. Because of the combination of 8-arm-PEG-NH2 and radioactive metal porphyrins (177Lu-TCPP), the photosensitizer in this compound can be efficiently absorbed by tumor cells and intracellular mitochondria. Biodistribution statistics show that 177Lu-PPNs-10 nm alone is enriched at the tumor site at 14.8 ± 1.0%id/g, which is the richest in the human body. Thus, it greatly improves the efficiency of PDT and internal radiation therapy, and has the potential for clinical application. Hu et al. successively synthesized a new type of this gallium porphyrin, which not only improved the water solubility of porphyrin but also connected cisplatin and antiplatinum. 32 The combination of the PDT effect of porphyrin and the antitumor effect of cisplatin showed better anti cancer effects.

Mitochondria uptake 177Lu-PPNs-10 nm in cancer cells.
At the same time, many metal porphyrins such as manganese porphyrin and gallium porphyrins are used as magnetic resonance contrast agents. These agents can serve as advanced PDT probes for many images. For PET imaging, Fazaeli et al. designed a 68Ga-fluorinated porphyrin complex. 33 The complex was prepared with high radiochemical purity (>99% ITLC, >99% HPLC, specific activity: 13–14 GBq/mmol). This radiolabeled porphyrin has a strong PET imaging ability and can be easily metabolized by the kidneys and livers. It is obvious that metal ions can easily enter the conjugated 18 π-electron system of the porphyrin center. Accordingly, using labeled porphyrin to display a variety of images is a classic and practical strategy.
From these studies, 34 we can see that the development of metal porphyrin photosensitizers follows four main directions: (1) modifying and improving the metal porphyrin structure is meant to obtain a single compound with better targeting and higher bioavailability. (2) Inserting different metal elements in the center of porphyrin to change its functions and adapt to different requirements of diagnosis and treatment. (3) By making porphyrin nano preparations, using the enhanced permeability and retention (EPR) effect effect aims to further improve drug targeting and drug loading. (4) Preparing a variety of composite materials by making use of the excellent properties of other materials to make up for the limitations of porphyrin compounds. After these directions, the performance of metalloporphyrin photosensitizers has been improved, with examples being selective enrichment of tumors, improved singlet oxygen yields in anoxic environments, better image expression, and low skin toxicity. However, the distance to them becoming clinical first-line medicines still needs revolutionary breakthroughs or new technology. 35
Applications of metalloporphyrin compounds in PTT
Significant progress has been made in PDT. Further studies showed that after enhancing the near-infrared absorption of a variety of porphyrins, these porphyrin drugs exhibit a photothermal conversion effect, which can kill tumors by heat. PTT is also very helpful for medical imaging, and to a certain extent, the therapeutic effect of porphyrin drugs has been enhanced. Photothermal-combined therapy gradually replaces traditional PDT with its high therapeutic efficiency and better images.36–38 Porphyrin drugs also have excellent applications in PTT. The traditional Zn and Cu porphyrins have also been redeveloped. Wang et al. redesigned a Zn porphyrin metal–organic framework (MOF), which has good monodispersity and a very uniform pore size distribution. 39 This simple nano reagent does not need to add complex multicomponents to achieve PDT and PTT. For synergistic treatment, photoactive porphyrin ligands were successfully introduced into the Zn-TCPP structure, and nanoparticles with both photodynamic and photothermal properties were constructed. DPBF was added after 650 nm laser irradiation for 5 min, and the significant decrease in the DPBFs fluorescence at 480 nm indicated that a large amount of singlet oxygen had been produced. Using a 650nm laser to irradiate a 100-μm concentration of Zn-TCPP nMOF, the solution temperature rises from 23 °C to 42 °C within 10 min, with excellent light-to-heat conversion efficiency (14.4). Li et al. used the copper ion d-d energy band transition to design an ultrathin copper porphyrin metal framework to improve the insufficient absorption of a porphyrin MOF in the near-infrared region (NIR), 40 because Cu(II) has the effect of absorbing NIR. Therefore, this ultrathin material can quickly respond to NIR light and can be used for PDT/PTT cooperative treatment. Inspired by in-depth research and development, the MOFs can achieve specificity by changing the metal ions and ligands. Wang et al. developed a MOF with iron atoms as the porphyrin center, which could be used for PTT and photoacoustic imaging. 41 It demonstrates obvious PTT effects under 808 nm NIR irradiation, and the Fe(III) center can catalyze the decomposition of endogenous H2O2, thereby generating more singlet oxygen, which can be used for photoacoustic imaging.
Mn porphyrin has been a key compound in PTT due to its high photothermal conversion ability and good MR imaging contrast.42,43 Zhang et al. have designed an Mn porphyrin MOF, in which Zr4+ and Mn porphyrin self-assemble. 44 MOFs have been used to transport gases such as NO due to their excellent porosity. NO is an unstable toxic gas. To achieve tumor treatment, it must be precisely controlled and released. The resulting NMOFs were then used to conjugate with S-nitrosothiol (SNO), a type of heat unstable NO donor. Therefore, MRI-guided NO and photothermal synergistic therapy was realized with the NMOF platform. Under the excitation of NIR light, this complex can control the release of NO and produce a good PTT effect. The NMOF-SNO (Figure 5) solution with a concentration of 400 μg mL−1 was rapidly heated from room temperature to 54 °C after being exposed to near-infrared light for 8 min. Over five cycles of laser irradiation NMOF-SNO, its temperature change remains constant showing excellent photothermal stability. T1-weighted MRI was performed to estimate the ability of NMOF-SNO for clinical diagnostic applications. The NMOFs exhibited the r1 value of 26.9 mM−1 s−1, higher than that of previous Mn-porphyrin composites, and have a larger pore size (Mn ions are more accessible to water molecules), so it is very suitable for MR imaging.
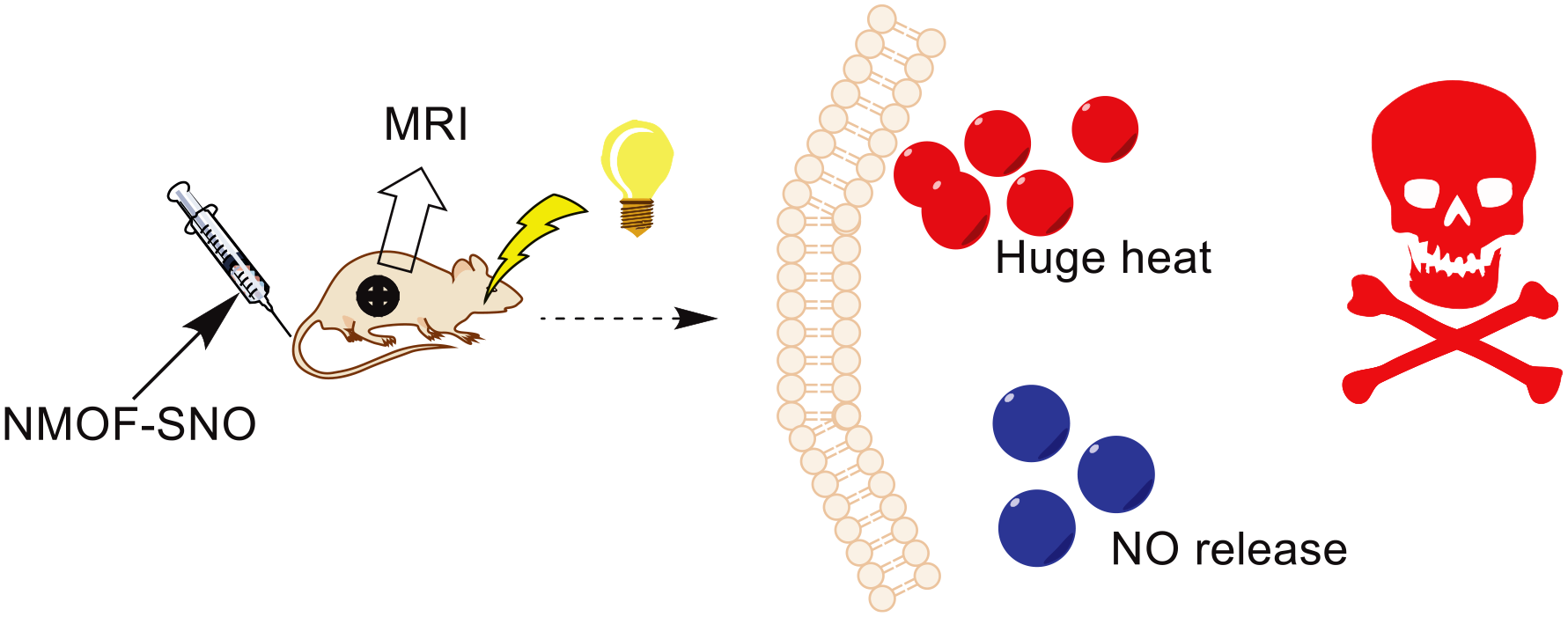
Schematic diagram of the mechanism of NMOF-SNO.
In addition to MOFs, the application of metal porphyrin compounds in photothermal treatment results in a high photothermal conversion rate after conversion into nanoformulations.45,46 Bing Guo et al. have designed a nano-porphyrin-based copolymer. 47 The molecules are packed and the density is extremely high, so they have good photostability and high extinction coefficients. The highest photothermal conversion efficiency is 63.8 at 800 nm. Further in vivo and in vitro experiments proved that this co-polymer can be taken up by cancer cells efficiently and has excellent biocompatibility. This multifunctional nanoparticle was developed for tumor imaging and treatment.
We can see that MOFs have a wide range of applications in photothermal conversion agents. Metal porphyrins demonstrate excellent performance when they are converted into MOFs. The MOF takes into account the photothermal conversion efficiency of PTT and the guiding role of medical imaging. At present, metal porphyrins are mainly made into MOF photothermal conversion agents, but it is worth mentioning that nonmetalloporphyrin Ce6 is also widely used in photothermal treatment.48–50 Furthermore, research on PTAs with high efficiency, nontoxic targeting, and excellent biocompatibility is key to whether PTT can become a routine clinical diagnosis and treatment method for tumors.
Applications of metalloporphyrin compounds in SDT
SDT is very similar to PDT and PTT, which both use singlet oxygen to kill cancer cells, but the three treatment methods have different excitation methods. 51 SDT can concentrate ultrasound to reach malignant tumors deep in tissues, which overcomes the shortcomings of traditional PDT and the major flaw of PTT. Compared with traditional PDT and PTT images, SDT can use more imaging methods and is considered more accurate. Therefore, SDT is regarded as a promising integrated method of diagnosis and treatment. The first commercially available photosensitizer, hematoporphyrin, was found to significantly reduce the survival rate of various cancer cells after the activation of ultrasound, opening the door for the application of porphyrin compounds in SDT. A variety of metal porphyrins have also been developed as acoustic sensitizers.52,53 ATX-70 (Figure 6) is a type of gallium porphyrin compound. 54 Yumitaa et al. found that the mechanism of SDT is similar to PDT.55–58 After exposing tumors in mice to a 3 W/cm2 ultrasound intensity environment, following injection of 2.5 mg/kg or higher concentration of ATX-70, it was found that the tumor volume had reduced by half after 3 days, and the inhibition rate of tumor cells became higher as the dose increased. In subsequent research, it was found that both plane waves and focused waves produced significant antitumor effects, but when using focused waves, the cavitation threshold should be avoided to prevent damage to the tissues surrounding. At the same time, it was found that ATX-70 can be enriched in the kidney, and the renal concentration is an order of magnitude higher than that in the plasma. This enrichment can selectively reduce damage to the surrounding tissues. ATX-70 has been proven to have excellent biodistribution selectivity. Although it is not widely used in clinical treatment, the emergence of ATX-70 has brought additional hope to ultrasound therapy.

A type of gallium porphyrin- ATX-70.
SDT can also take advantage of the anisotropy of different metal porphyrins. 59 Huang et al. used hollow mesoporous organosilica nanoparticles (HMONs) to hybridize disulfide bonds into the framework of HMONs (increasing the biodegradation reaction in tumor tissues) and connect them to protoporphyrin (PPIX) chelated manganese ions. 60 The porphyrin compound MnPpIX (Figure 7) represents a biodegradable, highly biocompatible, higher loaded, and activated controllable multifunctional ultrasonic sensitizer HMONs-MnPpIX, which has high SDT efficiency. The hollow mesoporous structure design guarantees the compound which can be used for MR imaging, and the paramagnetic Mn significantly improves the MRI performance of the compound. The relaxation r1 is as high as 9.43 mm−1 s−2, which is more than two times higher than the clinical Gd drugs. It can be used for SDT guidance and monitoring. HMONs-MnPpIX has achieved excellent results from both in vitro cell experiments and in vivo tumor growth experiments. Therefore, additional metal complex porphyrins have been tested for their performance and mechanism of SDT. The experimental results of Giuntini et al. proved that the ultrasonic activity of porphyrins depends to a large extent on the presence of metal ions. 61 From an in-depth study of the supporting mechanism behind the generation of cytotoxic substances, it is believed that acoustic cavitation leads to the generation of singlet oxygen by the active substance. Similarly, there are studies using aminopropionic acid (which can enhance the effect of ultrasonic sensitizers and can be used for the identification of singlet oxygen) to explore the SDT mechanism, while cancer cells in an aminopropionic acid environment have undergone sonodynamically induced apoptosis. Acoustic cavitation is a possible core of the SDT mechanism, but still needs further confirmation. Hence, a series of water-soluble Fe(III), Zn(II) porphyrins, and Pd(II) porphyrins were exposed to light conditions or ultrasonic environments to determine the correlation between porphyrin-mediated ROS generation efficiency and sonodynamic activity.

(a) MnPPIX and (b) Structure of HMONs-MnPpIX-PEG.
Apart from the above preparations, researchers have combined SDT and microbubbles (MBs) based on the excellent delivery performance of MBs, the sound-controlled release, and good diagnostic effects. Sinoporphyrin Sodium (DVDMS) is a new generation of photosensitizer developed based on photofrin, with high photosensitivity activity.62,63 In addition to its excellent performance in PDT, it has also been found to have the effect of mediating SDT in recent studies. Hu et al. have further discussed the absorption mode of DVDMS in detail. 63 The pH at the tumor site is lower than that of normal tissues, and low pH is more conducive to the absorption of DVDMS. Furthermore, DVDMS greatly improves the shortcomings of the poor water solubility of porphyrin drugs, and its skin retention is greatly reduced compared with other sensitizers. The authors confirmed that DVDMS, like other porphyrins, is mainly distributed in mitochondria through subcellular localization. An ultrasonic intensity gradient experiment proved that the sonotoxicity of DVDMS is proportional to the ultrasonic intensity. Liu et al. used MBs embedded with DVDMS to conduct cell viability experiments and apoptosis experiments. 64 They found that tumor cells are slightly inhibited under the intensity of 0.36 and 0.54 W/cm2 ultrasound irradiation. When MBs were introduced, the treatment effect was significantly improved. In the absence of SDT intervention, Hematoxylin & Eosin (H&E) staining showed that the MBs group had almost no effect on the surrounding tissues of the tumor. When SDT was used, the inhibition rate of the cancer cells can reach 60%. At the same time, metalloporphyrin MBs, which have both CEUS and MRI imaging capabilities, have been developed.65,66 Chen and co-workers developed MnP-MBs, a type of nanoparticles that can be converted by ultrasound at the tumor site to achieve rapid tumor MRI contrast enhancement. 65 Only a low Mn injection dosage can produce excellent MRI images within 30 min. Such MRI images can eliminate the shortcomings of low ultrasound imaging sensitivity due to background scattering. According to the authors, this is also the first contrast agent (CA) that combines ultrasound imaging and MRI. On using a 7.0 T magnetic field to evaluate the contrast enhancement behavior of MRI, there is a linear relationship between 1/T1 and Mn concentration. This relationship can be expressed as r1 = 1.61 mM−1 s−1. Therefore, MnP could serve as an efficient MRI CA.
We can see that SDT has been developed significantly since it was discovered in the 1990s. In theory, using SDT, it is entirely possible to solve the defects of insufficient PDT/PTT penetration ability. Using SDT, we can see that metal porphyrins have a wide range of applications. Related research also shows that metal atoms can improve the excitation of porphyrins by ultrasound. Therefore, metal porphyrins are a hot research topic in SDT treatment. One of the obvious shortcomings of SDT is that there is no effect on cavity viscera, and the other is the damage to normal tissue outside the lesion area. Hence, the development and improvement of anticancer activity and the ability of these compounds to penetrate tissues become the focus of further research.
Summary
PDT, PTT, and SDT have significant applicability. Clinically, PDT has been widely used in the treatment of human surface tumors. PTT has made breakthroughs in the direction of photothermal imaging and can be used for monitoring the treatment of tumors, while SDT is a promising treatment that can be combined with traditional treatments. The mechanisms of these three treatment methods are similar. Compared with radiotherapy and chemotherapy, they have higher targeting, lower toxicity (dark toxicity), and more abundant chemical structures. Photothermal conversion agents and ultrasound sensitizers can often play a therapeutic role by directly using photosensitizers to only change the treatment mode or to change the structure of the photosensitizer, hence these drugs are very similar in structure. In such compounds, porphyrins are the most studied and widely applied drugs. However, further improvements are still necessary to further improve the singlet oxygen production efficiency and the capacities of the sensitizers, so to breakthrough the limit of the hypoxic environment in the body to achieve higher curative effects. With porphyrin compounds, the introduction of metal atoms can increase the probability of the absorption transition from S0→T1 and the crossover between S1→T1 systems. Therefore, metal porphyrin compounds have received extensive attention. They have the following advantages:
They can be excited more efficiently than nonmetallic porphyrins and therefore lead to a higher singlet oxygen yield.
After the introduction of a metal atom, the excitation wavelength of the corresponding compound is red shifted, which is more in line with the requirements of the excitation source during treatment.
Different metal atoms have different characteristics, making metal porphyrin compounds more widely applicable.
The introduction of certain elements will increase the accuracy and breadth of the image, which is advantageous for tumor monitoring.
Outlook
We can expect that in the future there will be many more metal porphyrin compounds or their nano preparations that have enough specificity to respond to different light, heat, and acoustic signals. And with the development of these types of treatments in terms of light (heat or sound) sources, light (heat or sound) transmission methods, and new molecular technologies, they have great potential to become routine clinical treatments. All of these conditions can improve the therapeutic efficacy of sensitizers, but the ability of sensitizers to be used as medicine requires reference to their ability to bind images. We hope to diagnose with a more accurate image, which can help with drug delivery and real-time monitoring, to reduce the amount of sensitizers and the side effects of these treatments, especially the accumulation of skin toxicity after long-term medication. Recently, new light source technologies have appeared including optical fibers and chemiluminescence in vivo. It is both a difficult and attractive subject how to use these technologies to make the treatments applicable to systemic tumors. In the field of PTT and SDT, the emergence of new technologies in vivo is also required. In the direction of cancer treatment, the invention of new technologies in vivo and the enhancement of the therapeutic ability of sensitizers are equally important. The combination of the above-mentioned new advances in diagnosis and treatment will undoubtedly promote the further development of these three treatment methods.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We are grateful to the Hunan Provincial Department of Education Key Project (18A244), the Hunan Provincial Innovation Foundation For Postgraduate Research (CS20200954), the Hunan University Students Innovation and Entrepreneurship Training Program (S202010555206), and the University of South China Students Innovation and Entrepreneurship Training Program (X202010555394).
