Abstract
The synthesis of C-ring hydrogenated sinomenine derivatives is accomplished from sinomenine by treatment with Zn-Hg/HCl, halogenation with NIS, and Heck reactions with various acrylates. A total of nine novel sinomenine cinnamate derivatives are obtained in 84%–93% yields. The structures of all the derivatives are characterized by 1H NMR, 13C NMR, and MS spectroscopy.

Many natural products and their derivatives are Food and Drug Administration (FDA)–approved drugs. 1 Sinomenine is a natural product isolated from the stems and roots of the plant Sinomenium acutum, native to China and Japan. 2 It contains a morphinan skeleton, and some derivatives demonstrate anti-inflammatory, anti-arrhythmic, anti-oxidant, and immunosuppressive activities, especially in treatment of rheumatoid arthritis in Asia;3–7 it can also lower blood pressure. 4 In recent years, it has been reported that sinomenine can be used in the treatment of chronic pneumonia, cancer, and drug addiction. 3
However, some side effects, for example, hydrophilic–lipophilic balance (HLB), large dose requirements and the induction of hypersusceptibility,8–13 prevent it from becoming a drug directly. In order to obtain sinomenine analogues with better therapeutic effects and less side effects, structural modifications of sinomenine have attracted significant research to improve its further applications,14–16 and many of its derivatives show comprehensive immune-regulatory and anti-inflammatory functions.17,18
Structurally, sinomenine is a very suitable starting natural product for new derivatives since it possesses four fused rings and a number of chemically active groups; however, most of the structural modifications are concentrated on only one active site. Meanwhile, a few reports have focused on simultaneous modifications of two or more active sites. 19 Consequently, as a continuation of our previous efforts on the synthesis of sinomenine derivatives, 20 herein, we report the synthesis of a class of novel C-ring hydrogenated sinomenine cinnamate derivatives. These sinomenine derivatives are obtained in good yields following simultaneous modifications at the C- and A-ring.
Results and discussion
The synthetic route toward the target compounds is outlined in Scheme 1 and requires five steps. Treatment of commercial sinomenine hydrochloride (

Synthesis of C-ring hydrogenated sinomenine cinnamate derivatives.
The C-ring of (
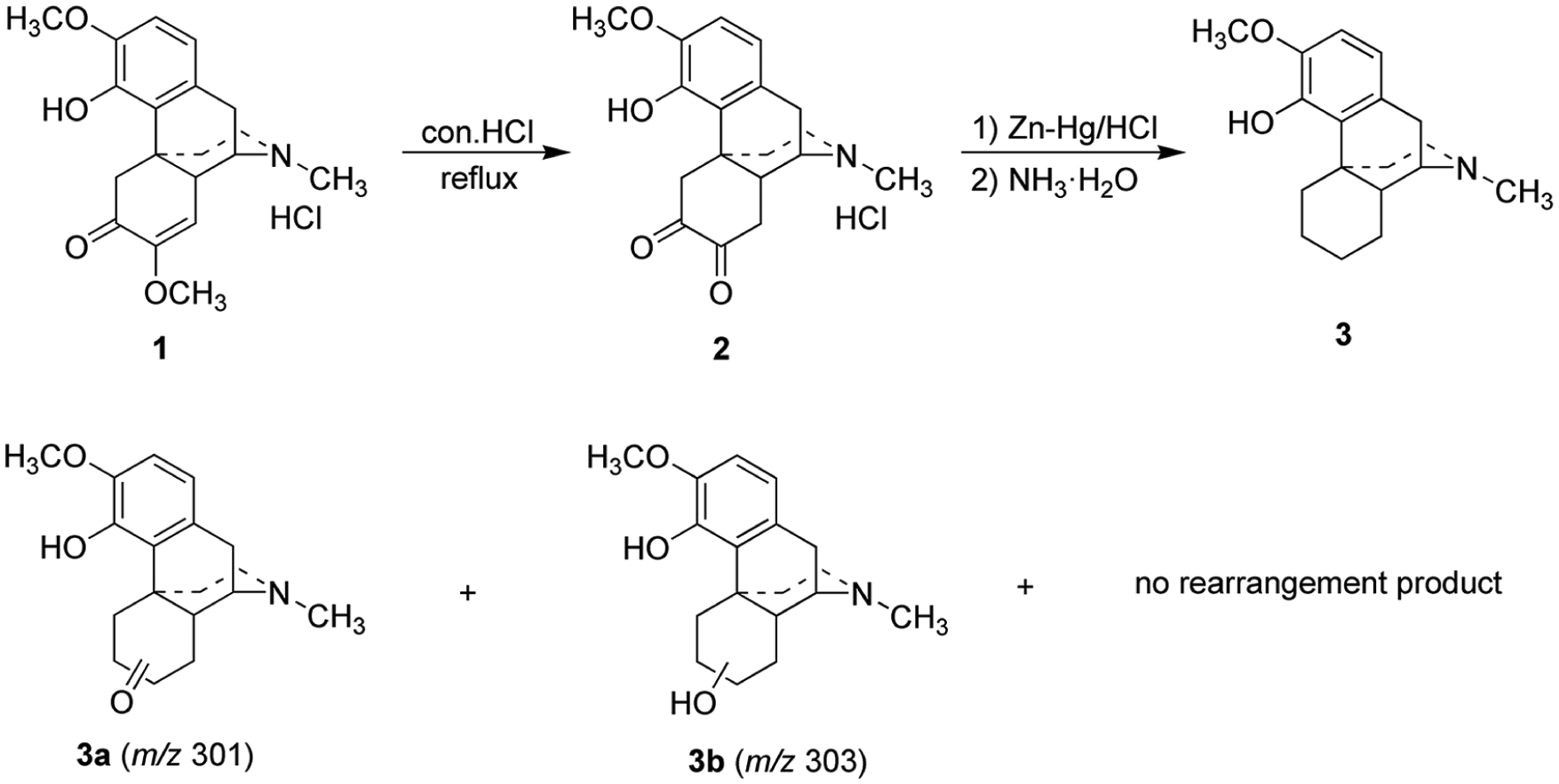
Synthesis of C-ring hydrogenated sinomenine.
According to the results reported in the literature,
21
we used NIS to carry out the halogenation reaction of C-ring hydrogenated sinomenine
There are two methods for the synthesis of cinnamate structural units starting from
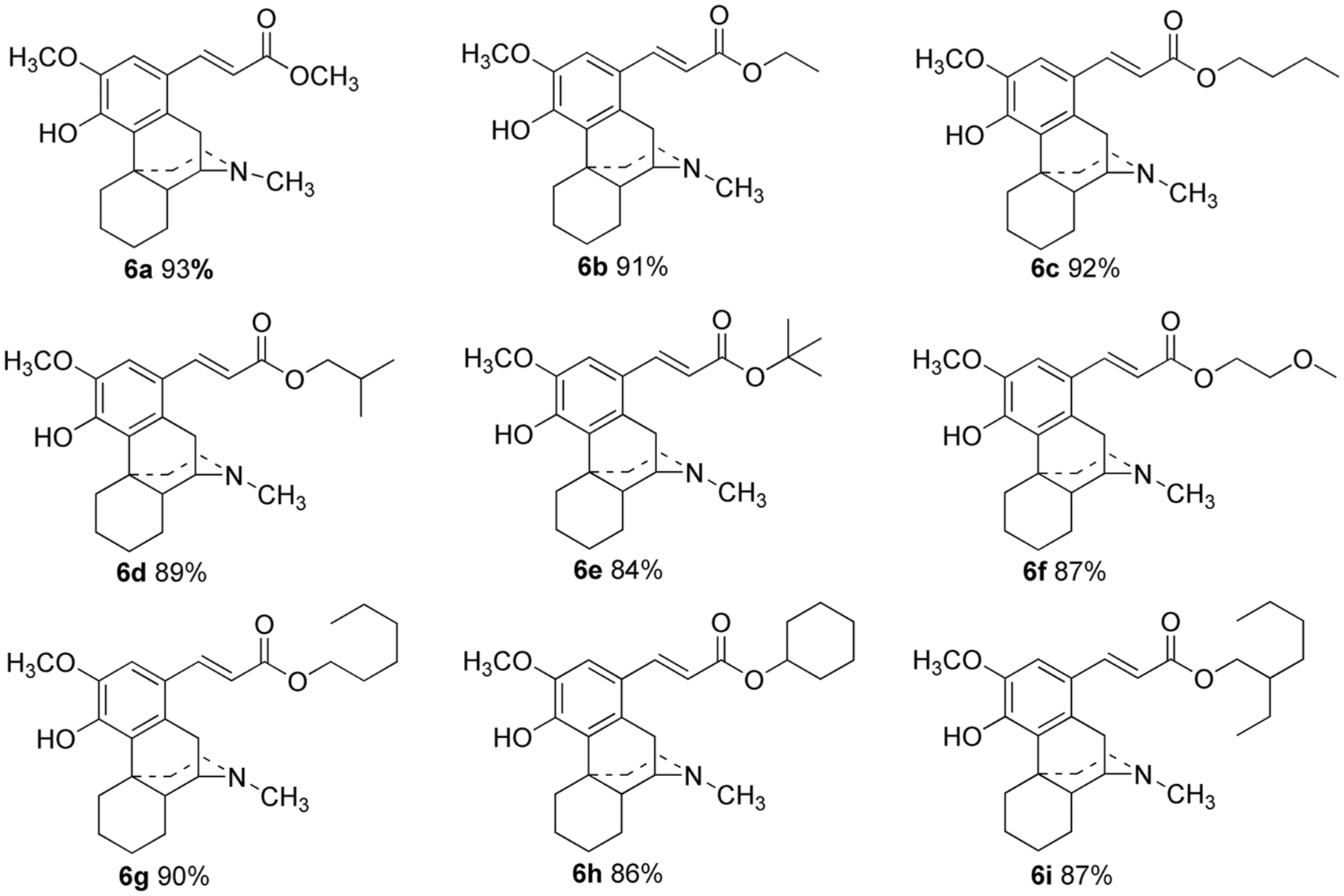
C-ring hydrogenated sinomenine cinnamate derivatives.
Conclusion
In summary, we have developed a practical approach to synthesize novel C-ring hydrogenated sinomenine cinnamate derivatives with excellent yields via Heck reactions. This approach has several merits including no by-products, easy purification, and high yields. All compounds were characterized by 1H NMR, 13C NMR, and ESI-MS (nuclear magnetic resonance (NMR) spectra for all new compounds are in the Supporting Information). These new compounds may be beneficially used in future drug development.
Experimental
All regents, including sinomenine, were purchased from Macklin and China National Pharmaceutical Group Corporation, and all commercial reagents were used directly without further purification. All reactions were monitored by TLC with GF254 silica gel plates under a UV lamp (254 nm) or with I2 detection. Column chromatography purifications were performed on silica gel (300–400 mesh) with CH2Cl2/CH3OH/NH3·H2O (200:10:1–400:10:1, v/v) as eluents. Melting points were measured with a Jinke SGWX-4B melting point apparatus and are uncorrected. All NMR spectra were recorded on a Bruker AV-II 300 MHz NMR spectrometer using tetramethylsilane as an internal standard, operating at 300 MHz for 1H NMR, and 75 MHz for 13C NMR. CDCl3 was used as the solvent, and chemical shifts (δ) are reported in parts per million (ppm). Mass spectra were obtained on an Agilent 6100 mass spectrometer.
Synthesis of C-ring hydrogenated sinomenine 3
Concentrated HCl (3 mL) was diluted to 60 mL; HgCl2 (4 g, 14.7 mmol) was added; and the mixture poured into a 500 mL round-bottom flask containing Zn (60 g, 0.92 mol). After shaking for 5 min, water was decanted and the solid washed once with concentrated HCl to give zinc amalgam. Sinomenine hydrochloride (
(9α,13α,14α)-4-Hydroxy-3-methoxy-17-methyl morphinan (
Synthesis of 1-iodo-C-ring hydrogenated sinomenine 4
C-ring hydrogenated sinomenine
(9α,13α,14α)-1-Iodo-4-hydroxy-3-methoxy-17-methyl morphinan (
Synthesis of C-ring hydrogenated sinomenine cinnamate derivatives 6a-i ; general procedure
1-Iodo-C-ring hydrogenated sinomenine
(9α,13α,14α)-1-Methylacrylate-4-hydroxy-3-methoxy-17-methyl morphinan (
(9α,13α,14α)-1-Ethylacrylate-4-hydroxy-3-methoxy-17-methyl morphinan (
(9α,13α,14α)-1-Butylacrylate-4-hydroxy-3-methoxy-17-methyl morphinan (
(9α,13α,14α)-1-Isobutylacrylate-4-hydroxy-3-methoxy-17-methyl morphinan (
(9α,13α,14α)-1-(tert-Butylacrylate)-4-hydroxy-3-methoxy-17-methyl morphinan (
(9α,13α,14α)-1-(2-Methoxyethylacrylate)-4-hydroxy-3-methoxy-17-methyl morphinan (
(9α,13α,14α)-1-Hexylacrylate-4-hydroxy-3-methoxy-17-methyl morphinan (
(9α,13α,14α)-1-Cyclohexylacrylate-4-hydroxy-3-methoxy-17-methyl morphinan (
(9α,13α,14α)-1-(2-Ethyl-hexylacrylate)-4-hydroxy-3-methoxy-17-methyl morphinan (
Supplemental Material
3.Supplementary_information-2 – Supplemental material for Synthesis of C-ring hydrogenated sinomenine cinnamate derivatives via Heck reactions
Supplemental material, 3.Supplementary_information-2 for Synthesis of C-ring hydrogenated sinomenine cinnamate derivatives via Heck reactions by Hongmei Pan, Tong Lu, Xuedan Wu, Chuan Jiang, Chengwen Gu, Kehua Zhang and Jie Jin in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Anhui University Natural Science Research Project (KJ2019A0770, KJ2019JD19) and the Science and Technology Innovation Foundation for College Students (C19000).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
