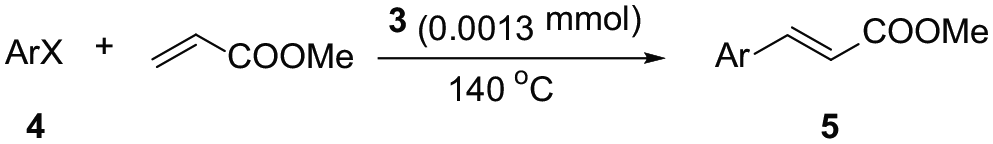
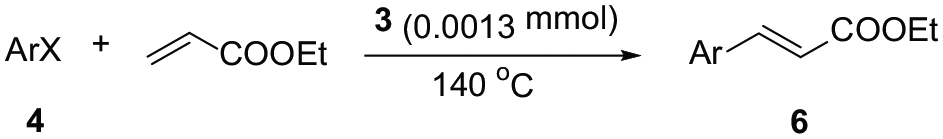
Abstract
A new bis-N-heterocyclic carbene palladium complex, (C13H9N2F2)2PdCl2, is synthesized by a three-step reaction and characterized by 1H NMR and 13C NMR spectroscopy as well as by X-ray crystallography. This new bis-N-heterocyclic carbene palladium complex has excellent stability and is capable of efficiently catalyzing the Mizoroki–Heck coupling reaction of aryl halides with acrylates.
Introduction
In recent years, the catalytic cross-coupling reactions of N-heterocyclic carbene (NHC) metal complexes have become a popular research topic.1,2 Compared with traditional phosphine and nitrogen ligands,3,4 NHC ligands have the following advantages: they are electron rich and strong electron donors, they strongly coordinate to metals, they are relatively stable in synthesized metal complexes5–20 and easy to modify, and so on.21,22 They are typically represented by an NHC-Pd complex which exhibits excellent reactivity in catalytic reactions, mainly due to the strong σ electron-donating properties of the NHC, which makes the central Pd atom more susceptible to oxidative addition at the inactive bond.
There are many reports on the catalytic cross-coupling reactions of NHC-Pd complexes.23–27 In 1991, Arduengo et al. 21 first synthesized a stable NHC complex and determined its molecular structure. Seven years later, Herrmann synthesized a bidentate NHC-Pd catalyst that exhibited higher activity for brominated aromatic hydrocarbons. 28 His project was then developed to give a NHC-Pd catalyst containing a bis-NHC that achieved the coupling of a chlorinated aromatic hydrocarbon at room temperature in high yield. In 2005, Singh et al. synthesized a monodentate NHC-Pd complex by a simple method. This NHC-Pd complex could effectively catalyze the Suzuki–Miyaura cross-coupling reaction of highly hindered chlorinated aromatic hydrocarbons, benzyl chloride, and allyl chlorides. 29 Recently, Lin et al. 30 synthesized a series of NHC-Pd complexes that catalyze the Mizoroki–Heck reaction, which was achieved with very high yields using 1 mol% of the catalyst. Zhong et al. 31 synthesized a new NHC-Pd complex that catalyzed the Suzuki–Miyaura reaction in an aqueous solvent at room temperature, a good yield being obtained by using 1 mol% of their catalyst.
Among the NHC metal complexes that have been reported so far, most structures are monoligands, while bis-ligands are relatively rare.32,33 A new NHC ligand containing a F atom, which is helpful in forming hydrogen bonds, was designed and synthesized in this work. This new bis-ligand complex, whose stability was greatly enhanced by the presence of the hydrogen bond and the bis-ligand structure, was obtained after complexing with palladium. A single crystal of the complex was left in the air at room temperature and standard atmospheric pressure for 1 month without any resulting changing in its properties. The crystal structure of the complex and its catalytic activity in Heck coupling reactions were then studied.
Results and discussion
Synthesis and crystal structure
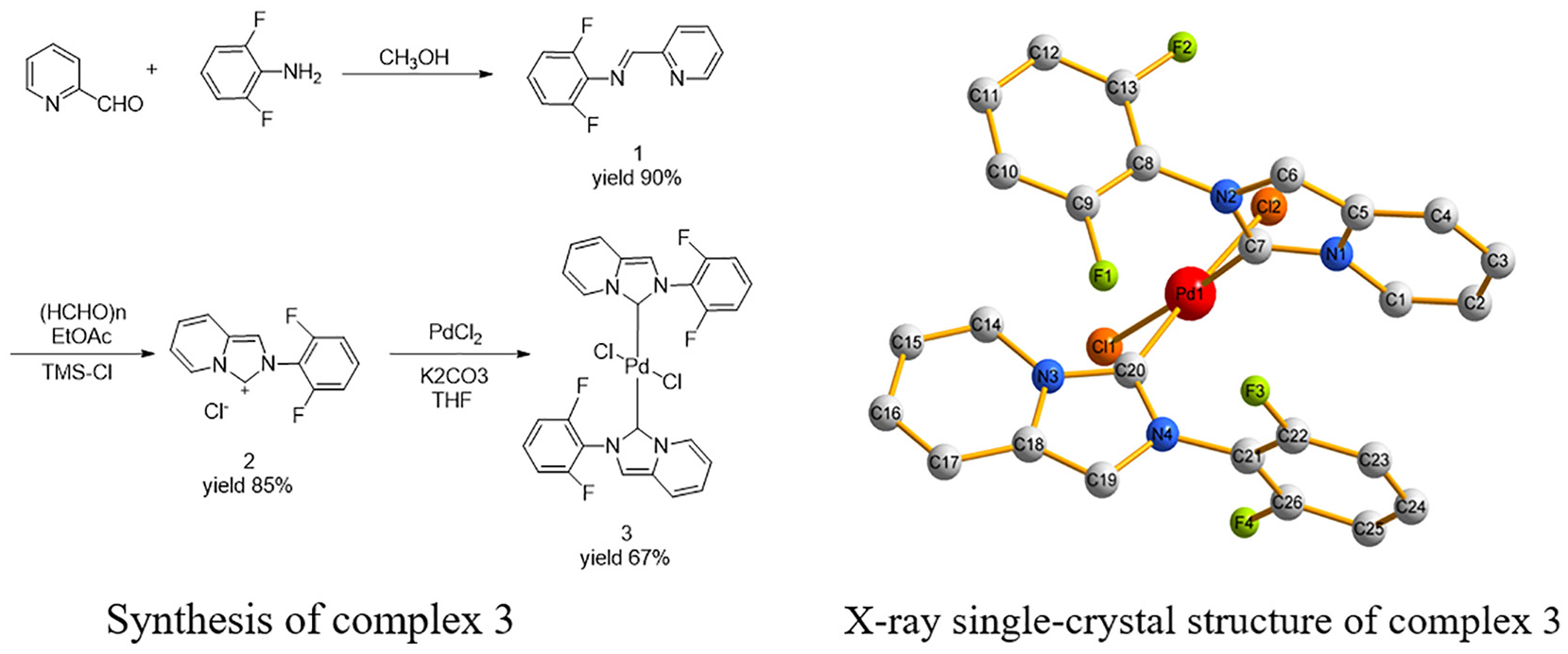
The synthesis of the new (C13H9N2F2)2PdCl2 complex is depicted in Scheme 1. Reacting

Synthesis of complex

X-ray single-crystal structure of complex
Crystal data and structure refinement for complex

Selected bond lengths (Å) and bond angles (°).

Symmetry transformations used to generate equivalent atoms: (a) −x + 2, −y, −z; (b) −x, −y + 1, −z.
The crystal structure of complex
Complex

View of the hydrogen bonding of F(3) in two molecules of complex

View of the hydrogen bond of F(4) in the chain structure of complex
Catalytic activity
The catalytic activity of the palladium complex
Optimization of the reaction conditions for the coupling of 4-bromotoluene with phenylboronic catalyzed by complex

DMSO: dimethyl sulfoxide; DMA: dimethylacetamide; DMF: dimethylformamide.
Reactions were carried out using
Isolated yields.
The scope of this reaction was then investigated under the optimal conditions and the results are summarized in Tables 4 and 5. Using methyl acrylate as a fixed substrate, various aryl halides were subjected to the coupling reaction. When the substituent on the aryl group was electron-withdrawing, such as in 1-bromo-4-nitrobenzene and 4-bromobenzaldehyde, high yields (93%, 92%) were obtained (Table 4, entries 1 and 2). Electron-donating substituents, such as those in 4-methylbromobenzene and 4-methoxybromobenzene (Table 4, entries 4 and 5), led to moderate yields (78%, 36%) being obtained (Table 3, entries 4 and 5). Even after lowering the loading, the catalyst was still quite effective for the reaction of the 4-methyliodobenzene (Table 4, entry 6). For 4-nitrochlorobenzene, much lower reactivity was observed (Table 4, entry 7) even with a higher loading of catalyst
Heck reactions of aryl halides and methyl acrylate
a
catalyzed by complex

DMA: dimethylacetamide.
Reactions were carried out using
Isolated yields.
Reactions were carried out using
Reaction was carried out using
Heck reactions of aryl halides and ethyl acrylate
a
catalyzed by complex

DMA: dimethylacetamide.
Reactions were carried out using
Isolated yields.
Reaction was carried out using
Reaction was carried out using
We found that ethyl acrylate was a better substrate for the reaction than methyl acrylate. Again the results showed that the reaction was sensitive to the electronic properties of the phenyl substituents. Much higher yields were obtained when electron-withdrawing groups such as nitro and formyl were present on the phenyl rings. In contrast, lower yields were obtained when electron-donating groups such as methyl and methoxyl were present on the phenyl rings (see Table 5). All the products in Tables 3–5 had been reported.34–36
Conclusion
A new bis-NHC palladium complex, (C13H9N2F2)2PdCl, has been synthesized by a three-step reaction and characterized by 1H NMR and 13C NMR spectroscopy as well as by X-ray crystallography. The bis-NHC palladium complex has excellent stability and enriches the diversity of NHC ligands and will aid further development of new NHC-Pd complexes. The catalytic activity of complex
Experimental
1H NMR and 13C NMR spectra were collected on a Bruker Avance III 400 MHz spectrometer with CDCl3 or DMSO as the solvent and tetramethylsilane (TMS) as the internal reference. Chemicals and reagents were obtained from commercial sources and used directly.
Synthesis of compound 1 [(E)-N-(2,6-difluorophenyl)-1-(pyridin-2-yl)methanimine]
To a 100-mL three-neck round-bottom flask, pyridine-2-carbaldehyde (2.00 g, 18.69 mmol) and methanol (6 mL) were added under an N2 atmosphere.37,38 Next, a mixed solution of 2,6-difluoroaniline (2.40 g, 18.60 mmol) in methanol (6 mL) was added dropwise. The mixture was stirred for 6 h at room temperature. On cooling in an ice bath, a solid was precipitated, and the solvent was removed under reduced pressure to give product
Synthesis of compound 2 (2-(2,6-difluorophenyl)imidazo[1,5-a]pyridin-2-ium chloride)
Product
Synthesis of complex 3
Under a N2 atmosphere, to a 10-mL Schlenk flask, palladium chloride (66.51 mg, 0.37 mmol), potassium carbonate (103.72 mg, 0.75 mmol), product
Heck reaction procedure
A Schlenk flask was filled with the aryl halide (1.3 mmol), methyl acrylate (1.3 mmol), Na2CO3 (2.6 mmol), complex
X-ray crystallography
A single crystal of complex
Supplemental Material
supporting – Supplemental material for Synthesis and characterization of a new bis-NHC palladium complex and its catalytic activity in the Mizoroki–Heck reaction
Supplemental material, supporting for Synthesis and characterization of a new bis-NHC palladium complex and its catalytic activity in the Mizoroki–Heck reaction by Can Feng, Cheng-xin Liu, Yu-fang Wang, Jin Cui and Ming-jie Zhang in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Tianjin (17JCQNJC05500), P. R. China.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.