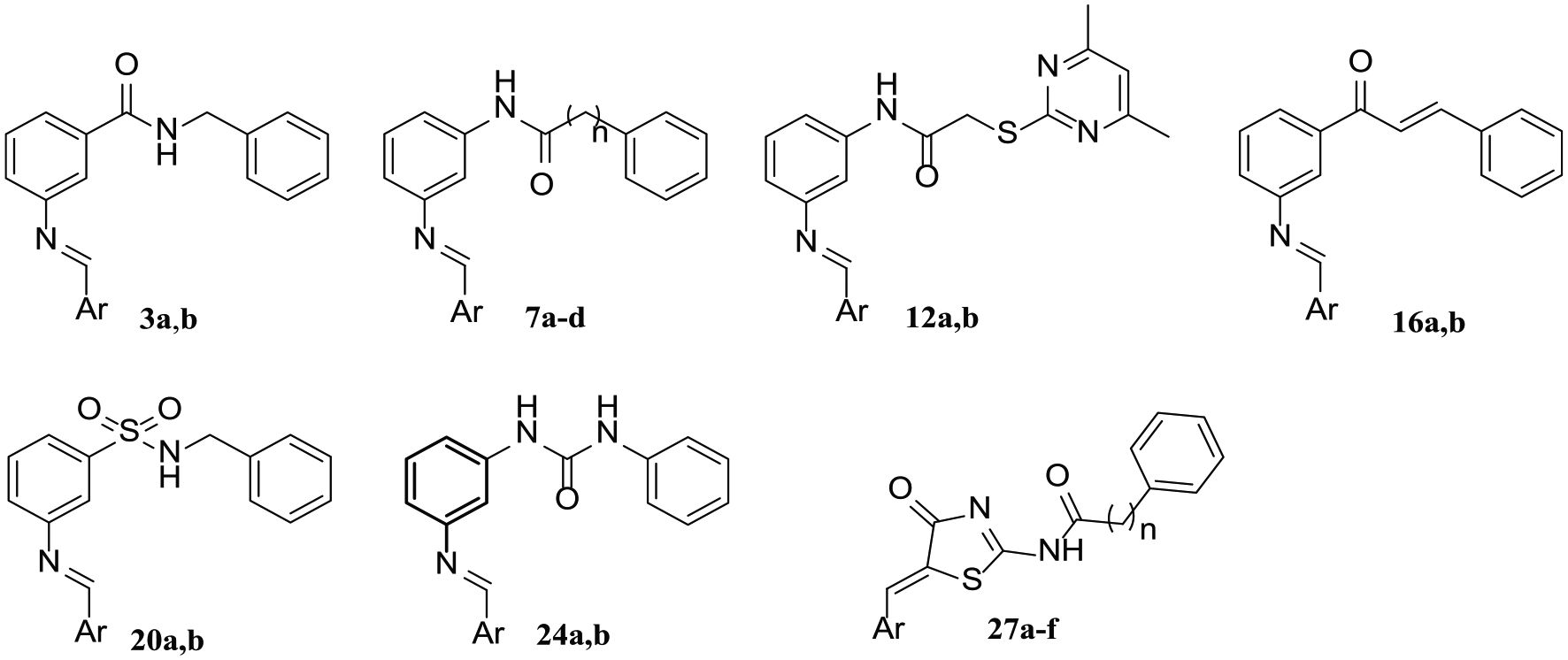
General procedure for the preparation of the Schiff bases 7a–d, 12a,b, 16a,b, 20a,b and 24a,b
A solution of the appropriate amine 6a,b, 11, 15, 19, 23 (2.35 mmol) in absolute ethanol (20 mL) and salicylaldehyde (0.28 mL, 2.59 mmol) or 2-hydroxy-1-naphthaldehyde (0.41 g, 2.35 mmol) was heated under reflux for 4–15 h at 70°C in the presence of a catalytic amount of glacial acetic acid. The reaction mixture was left to cool, the precipitated crystals were filtered and recrystallized from an appropriate solvent(s).
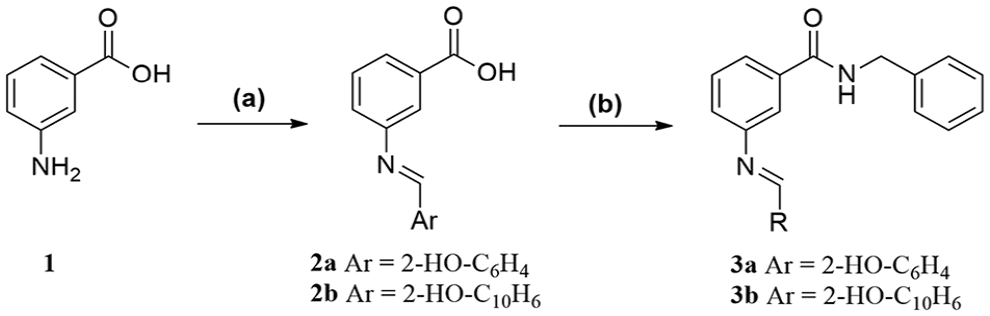
N-(3-((2-Hydroxybenzylidene)amino)phenyl)benzamide (7a): The title compound separated as yellow crystals, recrystallized from n-butanol, m.p. 175–177°C, yield 0.61 g (83%). IR ν (cm−1): 3445 (OH), 3308 (NH), 3055–3032 (CH aromatic), 1647 (C=O), 1620 (C=N), 1595–1558 (C=C). 1H NMR (DMSO-d6, 400 MHz): δ 6.98–7.01 (m, 2H, aromatic H), 7.19 (d, J = 7.96 Hz, 1H, aromatic H), 7.42–7.47 (m, 2H, aromatic H), 7.54–7.62 (m, 3H, aromatic H), 7.69–7.74 (m, 2H, aromatic H), 7.88 (s, 1H, aromatic H), 7.99 (d, J = 7.36 Hz, 2H, aromatic H), 8.98 (s, 1H, CH=N), 10.38 (br s, 1H, NH, D2O exchangeable), 13.06 (br s, 1H, OH, D2O exchangeable). 13C NMR (DMSO-d6, 100 MHz): δ 114.1, 116.6, 117.1, 119.3, 119.70, 119.74, 128.1, 128.9, 130.1, 132.2, 133.0, 133.9, 135.2, 140.7, 148.9, 160.8 (aromatic carbons), 163.8 (C=N), 166.2 (C=O). MS (EI): m/z (%) = 316 (M+, 54.45), 105 (100). Anal. calcd for C20H16N2O2 (316.36): C, 75.93; H, 5.10; N, 8.86; found: C, 76.14; H, 5.23; N, 9.15%.
N-(3-(((2-Hydroxynaphthalen-1-yl)methylene)amino)phenyl)benzamide (7b): The product separated as yellow crystals, recrystallized from n-butanol, m.p. 221–223°C, yield 0.74 g (86%). IR ν (cm−1): 3460 (OH), 3323 (NH), 1653 (C=O), 1622 (C=N), 1593–1531 (C=C). 1H NMR (DMSO-d6, 400 MHz): δ 7.03 (d, J = 9.20 Hz, 1H, aromatic H), 7.37 (t, J = 8.16 Hz, 1H, aromatic H), 7.49–7.63 (m, 6H, aromatic H), 7.76 (br s, 1H, aromatic H), 7.81 (d, J = 7.92 Hz, 1H, aromatic H), 7.94 (d, J = 9.16 Hz, 1H, aromatic H), 7.96 (s, 1H, aromatic H), 8.01 (d, J = 7.48 Hz, 2H, aromatic H), 8.48 (d, J = 8.44 Hz, 1H, aromatic H), 9.66 (s, 1H, CH=N), 10.40 (br s, 1H, NH, D2O exchangeable), 15.78 (br s, 1H, OH, D2O exchangeable). 13C NMR (DMSO-d6, 100 MHz): δ 108.9, 113.3, 115.4, 118.8, 120.6, 122.8, 124.0, 127.1, 128.1, 128.7, 128.9, 129.5, 130.4, 132.2, 133.6, 135.2, 137.6, 140.8, 144.5, 155.8 (aromatic carbons), 166.2 (C=N), 171.4 (C=O). MS (EI): m/z (%) = 366 (M+, 89.97), 59 (100). Anal. calcd for C24H18N2O2 (366.42): C, 78.67; H, 4.95; N, 7.65; found: C, 79.01; H, 5.12; N, 7.88%.
N-(3-((2-Hydroxybenzylidene)amino)phenyl)-2-phenylacetamide (7c): The title compound was obtained as yellow crystals, recrystallized from n-butanol, m.p. 195–197°C, yield 0.61 g (78%). IR ν (cm−1): 3450 (OH), 3260 (NH), 3047–3026 (CH aromatic), 2993–2920 (CH aliphatic), 1661 (C=O), 1622 (C=N), 1589–1533 (C=C). 1H NMR (DMSO-d6, 400 MHz): δ 3.67 (s, 2H, CH2), 6.97–7.00 (m, 2H, aromatic H), 7.12 (d, J = 7.84 Hz, 1H, aromatic H), 7.26 (t, J = 6.40 Hz, 1H, aromatic H), 7.32–7.44 (m, 6H, aromatic H), 7.49 (d, J = 8.20 Hz, 1H, aromatic H), 7.67–7.71 (m, 2H, aromatic H), 8.92 (s, 1H, CH=N), 10.32 (br s, 1H, NH, D2O exchangeable), 13.02 (br s, 1H, OH, D2O exchangeable). 13C NMR (DMSO-d6, 100 MHz): δ 43.8 (CH2), 112.8, 116.2, 117.1, 118.0, 119.6, 119.7, 127.0, 128.8, 129.6, 130.2, 133.0, 133.8, 136.3, 140.7, 149.0, 160.7 (aromatic carbons), 163.8 (C=N), 169.8 (C=O). MS (EI): m/z (%) = 330 (M+, 100). Anal. calcd for C21H18N2O2 (330.39): C, 76.34; H, 5.49; N, 8.48; found: C, 76.49; H, 5.67; N, 8.67%.
N-(3-{[(2-Hydroxynaphthalen-1-yl)methylidene]amino}phenyl)-2-phenylacetamide (7d): The product was obtained as yellow crystals, recrystallized from n-butanol, m.p. 194–196°C, yield 0.72 g (80%). IR ν (cm−1): 3441 (OH), 3287 (NH), 3055–3028 (CH aromatic), 2943 (CH aliphatic), 1662 (C=O), 1627 (C=N), 1589–1535 (C=C). 1H NMR (DMSO-d6, 400 MHz): δ 3.69 (s, 2H, CH2), 7.01 (d, J = 9.16 Hz, 1H, aromatic H), 7.26–7.42 (m, 8H, aromatic H), 7.51–7.55 (m, 2H, aromatic H), 7.78–7.81 (m, 2H, aromatic H), 7.94 (d, J = 9.24 Hz, 1H, aromatic H), 8.45 (d, J = 8.48 Hz, 1H, aromatic H), 9.60 (s, 1H, CH=N), 10.35 (br s, 1H, NH, D2O exchangeable), 15.71 (br s, 1H, OH, D2O exchangeable). 13C NMR (DMSO-d6, 100 MHz): δ 43.8 (CH2), 108.8, 112.0, 115.0, 117.6, 120.5, 122.8, 124.0, 127.0, 127.1, 128.71, 128.74, 128.8, 129.5, 129.6, 130.5, 133.5, 136.2, 137.6, 140.8, 144.5, 155.7 (aromatic carbons), 170.0 (C=N), 171.6 (C=O). MS (EI): m/z (%) = 380 (M+, 100). Anal. calcd for C25H20N2O2 (380.45): C, 78.93; H, 5.30; N, 7.36; found: C, 78.70; H, 5.49; N, 7.68%.
2-((4,6-Dimethylpyrimidin-2-yl)thio)-N-(3-((2-hydroxybenzylidene)amino)phenyl) acetamide (12a): The title compound separated as yellow crystals, crystallized from methanol, m.p. 139–140°C, yield 0.60 g (65%). IR ν (cm−1): 3479 (OH), 3263 (NH), 3082 (CH aromatic), 2970 (CH aliphatic), 1666 (C=O), 1605 (C=N), 1577–1543 (C=C). 1H NMR (DMSO-d6, 400 MHz): δ 2.34 (s, 6H, 2 CH3), 4.07 (s, 2H, CH2), 6.96–7.00 (m, 3H, aromatic H), 7.13 (d, J = 7.72 Hz, 1H, aromatic H), 7.38–7.47 (m, 3H, aromatic H), 7.67–7.70 (m, 2H, aromatic H), 8.92 (s, 1H, CH=N), 10.39 (br s, 1H, NH, D2O exchangeable), 13.00 (br s, 1H, OH, D2O exchangeable). 13C NMR (DMSO-d6, 100 MHz): δ 23.8 (2 CH3), 36.0 (CH2), 112.9, 116.2, 116.5, 117.1, 118.0, 119.6, 119.7, 130.3, 133.0, 133.8, 140.5, 149.0, 160.7, 163.8, 167.3, 167.4 (aromatic carbons), 169.7 (C=N), 192.0 (C=O). MS (EI): m/z (%) = 392 (M+, 44.38), 181 (100). Anal. calcd for C21H20N4O2S (392.48): C, 64.27; H, 5.14; N, 14.28; found: C, 64.49; H, 5.26; N, 14.49%.
2-((4,6-Dimethylpyrimidin-2-yl)thio)-N-(3-(((2-hydroxynaphthalen-1-yl)methylene) amino)phenyl)acetamide (12b): The obtained product separated as yellow crystals, crystalized from n-butanol, m.p. 204–206°C, yield 0.70 g (67%). IR ν (cm−1): 3444 (OH), 3294 (NH), 3059–3032 (CH aromatic), 2997–2920 (CH aliphatic), 1666 (C=O), 1620 (C=N), 1578–1535 (C=C). 1H NMR (DMSO-d6, 400 MHz): δ 2.35 (s, 6H, CH3), 4.09 (s, 2H, CH2), 6.98 (s, 1H, aromatic H), 7.01 (d, J = 9.20 Hz, 1H, aromatic H), 7.34 (t, J = 7.20 Hz, 1H, aromatic H), 7.44–7.47 (m, 3H, aromatic H), 7.55 (t, J = 7.08 Hz, 1H, aromatic H), 7.76 (br s, 1H, aromatic H), 7.80 (d, J = 7.48 Hz, 1H, aromatic H), 7.94 (d, J = 9.20 Hz, 1H, aromatic H), 8.45 (d, J = 8.44 Hz, 1H, aromatic H), 9.61 (s, 1H, CH=N), 10.41 (br s, 1H, NH, D2O exchangeable), 15.70 (br s, 1H, OH, D2O exchangeable). 13C NMR (DMSO-d6, 100 MHz): δ 23.7 (2 CH3), 35.9 (CH2), 108.8, 112.0, 115.0, 116.6, 117.6, 120.5, 122.8, 124.1, 127.1, 128.8, 129.5, 130.6, 133.5, 137.7, 140.5, 144.4, 155.6, 167.5, 169.6 (aromatic carbons), 171.7 (C=N), 193.4 (C=O). MS (EI): m/z (%) = 442 (M+, 70.74), 153 (100). Anal. calcd for C25H22N4O2S (442.54): C, 67.85; H, 5.01; N, 12.66; found: C, 67.53; H, 5.24; N, 12.48%.
(2E)-1-{3-[(2-Hydroxybenzylidene)amino]phenyl}-3-phenylprop-2-en-1-one (16a): The title compound separated as pale yellow crystals on cooling the reaction mixture, recrystallized from 90% ethanol, m.p. 115–117°C, yield 0.61 g (79%). IR ν (cm−1): 3419 (OH), 3062–3022 (CH aromatic), 1662 (C=O), 1618 (C=N), 1607–1574 (C=C). 1H NMR (DMSO-d6, 400 MHz): δ 7.02 (t, J = 8.12 Hz, 2H, aromatic H), 7.44–7.49 (m, 4H, aromatic H), 7.65–7.74 (m, 3H, aromatic H), 7.81 (d, J = 15.60 Hz, 1H, ethylenic H), 7.93–7.95 (m, 2H, aromatic H), 8.03 (d, J = 15.60 Hz, 1H, ethylenic H), 8.09 (d, J = 7.44 Hz, 1H, aromatic H), 8.18 (s, 1H, aromatic H), 9.08 (s, 1H, CH=N), 12.90 (br s, 1H, OH, D2O exchangeable). 13C NMR (DMSO-d6, 100 MHz): δ 117.1, 119.7, 119.8, 121.6, 122.5, 126.7, 127.2, 129.2, 129.4, 129.5, 130.4, 131.2, 133.2, 134.0, 135.1, 139.3, 144.9, 149.3, 160.8, 165.1 (aromatic carbons), 189.3 (C=N), 192.3 (C=O). MS (EI): m/z (%) = 327 (M+, 100). Anal. calcd for C22H17NO2 (327.38): C, 80.71; H, 5.23; N, 4.28; found: C, 80.53; H, 5.60; N, 4.32%.
(2E)-1-(3-{[(2-Hydroxynaphthalen-1-yl)methylidene]amino}phenyl)-3-phenylprop-2-en-1-one (16b): The obtained product was filtered while hot and crystallized from n-butanol as yellow crystals, m.p. 203–205°C, yield 0.66 g (74%). IR ν (cm−1): 3414 (OH), 3059–3028 (CH aromatic), 1655 (C=O), 1620 (C=N), 1605–1574 (C=C). 1H NMR (DMSO-d6, 400 MHz): δ 7.07 (d, J = 9.12 Hz, 1H, aromatic H), 7.38 (t, J = 8.00 Hz, 1H, aromatic H), 7.49–7.53 (m, 3H, aromatic H), 7.58 (t, J = 8.00 Hz, 1H, aromatic H), 7.70 (t, J = 8.00 Hz, 1H, aromatic H), 7.81–7.84 (m, 2H, ethylenic H and aromatic H), 7.95–7.99 (m, 4H, aromatic H), 8.04 (d, J = 15.52 Hz, 1H, ethylenic H), 8.05 (d, J = 7.92 Hz, 1H, aromatic H), 8.33 (s, 1H, aromatic H), 8.56 (d, J = 8.48 Hz, 1H, aromatic H), 9.78 (s, 1H, CH=N), 15.63 (br s, 1H, OH, D2O exchangeable). 13C NMR (DMSO-d6, 100 MHz): δ 109.2, 121.0, 121.1, 122.3, 122.6, 124.1, 125.6, 126.8, 127.3, 128.7, 129.1, 129.4, 129.5, 130.6, 131.3, 133.6, 135.1, 137.6, 138.9, 139.5, 145.0, 145.4, 157.5, 170.4 (aromatic carbons), 189.4 (C=N), 193.3 (C=O). MS (EI): m/z (%) = 377 (M+, 100). Anal. calcd for C26H19NO2 (377.44): C, 82.74; H, 5.07; N, 3.71; found: C, 82.41; H, 5.39; N, 3.98%.
N-Benzyl-3-((2-hydroxybenzylidene)amino)benzenesulfonamide (20a): The title product was obtained as pale yellow crystals, recrystallized from methanol/chloroform mixture (5:1), m.p. 153–154°C, yield 0.70 g (81%). IR ν (cm−1): 3475 (OH), 3302 (NH), 3063 (CH aromatic), 2980 (CH aliphatic), 1620 (C=N), 1610 (NH bending), 1570 (C=C), 1350, 1150 (SO2). 1H NMR (DMSO-d6, 400 MHz): δ 4.05 (d, J = 5.48 Hz, 2H, CH2), 7.00–7.04 (m, 2H, aromatic H), 7.20–7.30 (m, 5H, aromatic H), 7.45 (t, J = 7.76 Hz, 1H, aromatic H), 7.65–7.66 (m, 2H, aromatic H), 7.72–7.74 (m, 3H, aromatic H), 8.23 (t, J = 5.72 Hz, 1H, NH, D2O exchangeable), 8.97 (s, 1H, CH=N), 12.62 (br s, 1H, OH, D2O exchangeable). 13C NMR (DMSO-d6, 100 MHz): δ 46.6 (CH2), 111.6, 117.2, 117.7, 119.7, 125.6, 127.9, 128.1, 128.6, 128.7, 129.9, 130.9, 133.0, 136.9, 137.9, 142.5, 149.4, 160.6, 165.2 (aromatic carbons), 192.5 (C=N). MS (EI): m/z (%) = 366 (M+, 100). Anal. calcd for C20H18N2O3S (366.44): C, 65.56; H, 4.95; N, 7.65; found: C, 65.80; H, 5.12; N, 7.83%.
N-Benzyl-3-(((2-hydroxynaphthalen-1-yl)methylene)amino)benzenesulfonamide (20b): The product separated as yellow crystals, recrystallized from n-butanol, m.p. 182–184°C, yield 0.85 g (87%). IR ν (cm−1): 3417 (OH), 3244 (NH), 3059–3028 (CH aromatic), 2935 (CH aliphatic), 1620 (C=N), 1574 (C=C), 1350, 1150 (SO2). 1H NMR (DMSO-d6, 400 MHz): δ 4.07 (d, J = 6.32 Hz, 2H, CH2), 7.09 (d, J = 9.16 Hz, 1H, aromatic H), 7.19–7.27 (m, 5H, aromatic H), 7.40 (t, J = 7.20 Hz, 1H, aromatic H), 7.60 (t, J = 8.32 Hz, 1H, aromatic H), 7.66–7.73 (m, 2H, aromatic H), 7.83–7.90 (m, 3H, aromatic H), 8.00 (d, J = 9.16 Hz, 1H, aromatic H), 8.23 (t, J = 6.32 Hz, 1H, NH, D2O exchangeable), 8.53 (d, J = 8.48 Hz, 1H, aromatic H), 9.69 (s, 1H, CH=N), 15.39 (br s, 1H, OH, D2O exchangeable). 13C NMR (CDCl3, 100 MHz): δ 47.4 (CH2), 109.1, 118.4, 119.3, 121.1, 123.9, 124.6, 125.5, 127.9, 128.0, 128.4, 128.7, 129.4, 130.4, 132.9, 136.0, 137.0, 141.7, 147.3, 157.2, 168.1 (aromatic carbons), 193.3 (C=N). MS (EI): m/z (%) = 416 (M+, 100). Anal. calcd for C24H20N2O3S (416.50): C, 69.21; H, 4.84; N, 6.73; found: C, 69.03; H, 4.97; N, 6.94%.
1-{3-[(2-Hydroxybenzylidene)amino]phenyl}-3-phenylurea (24a): The title compound separated as yellow crystals, recrystallized from ethanol, m.p. 227–229°C, yield 0.51 g (65%). IR ν (cm−1): 3290 (OH), 3200, 3125 (2 NH), 3059–3035 (CH aromatic), 1732 (C=O), 1647 (NH bending), 1620 (C=N), 1593–1558 (C=C). 1H NMR (DMSO-d6, 400 MHz): δ 6.98–7.05 (m, 4H, aromatic H), 7.27–7.31 (m, 3H, aromatic H), 7.38 (t, J = 7.88 Hz, 1H, aromatic H), 7.41–7.48 (m, 3H, aromatic H), 7.59 (s, 1H, aromatic H), 7.69 (d, J = 7.60 Hz, 1H, aromatic H), 8.73 (br s, 1H, NH, D2O exchangeable), 8.81 (br s, 1H, NH, D2O exchangeable), 8.94 (s, 1H, CH=N), 13.08 (br s, 1H, OH, D2O exchangeable). 13C NMR (DMSO-d6, 100 MHz): δ 111.8, 114.8, 117.1, 118.8, 119.7, 122.5, 127.1, 129.3, 129.8, 130.3, 133.0, 133.8, 136.9, 141.2, 149.1, 153.0 (aromatic carbons), 160.7 (C=N), 163.8 (C=O). MS (EI): m/z (%) = 331 (M+, 10.43), 93 (100). Anal. calcd for C20H17N3O2 (331.38): C, 72.49; H, 5.17; N, 12.68; found: C, 72.68; H, 5.40; N, 12.50%.
1-(3-{[(2-Hydroxynaphthalen-1-yl)methylidene]amino}phenyl)-3-phenylurea (24b): The title compound separated as yellow crystals, recrystallized from ethanol, m.p. 262–265°C, yield 0.56 g (58%). IR ν (cm−1): 3541 (2 NH), 3398 (OH), 3067 (CH aromatic), 1732 (C=O), 1628 (C=N), 1593–1535 (C=C). 1H NMR (DMSO-d6, 400 MHz): δ 7.04 (d, J = 9.16 Hz, 1H, aromatic H), 7.18 (t, J = 9.16 Hz, 1H, aromatic H), 7.39–7.43 (m, 3H, aromatic H), 7.51–7.57 (m, 3H, aromatic H), 7.81–7.90 (m, 4H, aromatic H), 7.96 (d, J = 9.20 Hz, 1H, aromatic H), 8.07 (s, 1H, aromatic H), 8.46 (d, J = 8.44 Hz, 1H, aromatic H), 9.66 (s, 1H, CH=N), 10.90 (br s, 1H, NH, D2O exchangeable), 10.97 (br s, 1H, NH, D2O exchangeable), 15.66 (br s, 1H, OH, D2O exchangeable). 13C NMR (DMSO-d6, 100 MHz): δ 108.9, 113.2, 116.8, 118.8, 120.6, 121.0, 122.6, 124.1, 125.2, 127.2, 128.7, 129.3, 129.6, 130.5, 133.6, 137.6, 138.0, 139.2, 144.8, 156.1, 158.9 (aromatic carbons), 159.2 (C=N), 171.1 (C=O). MS (EI): m/z (%) = 410 (M±1, 9.74), 92 (100). Anal. calcd for C24H19N4O3 (411.44): C, 75.57; H, 5.02; N, 11.02; found: C, 75.79; H, 5.13; N, 11.34%.
2-[(4,6-Dimethylpyrimidin-2-yl)sulfanyl]-N-(3-nitrophenyl)acetamide (10): A suspension of 2-mercapto-4,6-dimethylpyrimidine hydrochloride (9) (1.76 g, 10 mmol) and potassium carbonate (2.76 g, 20 mmol) in absolute ethanol (20 mL) was heated under reflux for 30 min. Then, 2-Chloro-N-(3-nitrophenyl)acetamide (8) (2.15 g, 10 mmol) was added, and the mixture was heated under reflux for another 3 h. The obtained residue was filtered and washed several times with water (3 × 10 mL) and recrystallized from ethanol. The product separated as yellowish-white crystals, m.p. 138–139°C, yield 2.36 g (74%). IR ν (cm−1): 3259 (NH), 3094–3024 (CH aromatic), 2978–2924 (CH aliphatic), 1678 (C=O), 1585–1535 (C=C). 1H NMR (DMSO-d6, 400 MHz): δ 2.32 (s, 6H, 2 CH3), 4.09 (s, 2H, CH2), 6.95 (s, 1H, aromatic H), 7.61 (t, J = 8.20 Hz, 1H, aromatic H), 7.90–7.93 (m, 2H, aromatic H), 8.62 (s, 1H, aromatic H), 10.73 (br s, 1H, NH, D2O exchangeable). 13C NMR (DMSO-d6, 100 MHz): δ 23.8 (2 CH3), 36.0 (CH2), 113.6, 116.6, 118.2, 125.5, 130.7, 140.6, 148.4, 167.4, 167.9 (aromatic carbons), 169.6 (C=O). MS (EI): m/z (%) = 318 (M+, 5.77), 181 (100). Anal. calcd for C14H14N4O3S (318.35): C, 52.82; H, 4.43; N, 17.60; found: C, 52.69; H, 4.65; N, 17.87%.
N-(3-Aminophenyl)-2-[(4,6-dimethyl pyrimidin-2-yl)sulfanyl]acetamide (11): A suspension of 10 (1.02 g, 3.23 mmol) in ethanol (9 mL) was slowly added to a solution of stannous chloride dihydrate (SnCl2·2H2O) (2.55 g, 11.31 mmol) and 37% hydrochloric acid (3 mL) while cooling in an ice bath. After 30 min, the reaction mixture allowed to reach room temperature and stirred for a further 48 h. After completion of the reaction, the solution was diluted with water (100 mL) and neutralized with ammonia. The resulting solution was extracted with chloroform, washed with water and dried over anhydrous sodium sulfate. The obtained aniline derivative was crystallized from ethanol.
General procedure for the preparation of compounds 27a–f
To a mixture of the appropriate aldehyde (2.27 mmol) and thiazolidinone derivative 22a,b (2.27 mmol) in absolute ethanol (15 mL), fused sodium acetate (0.18 g, 2.27 mmol) was added. The reaction mixture was heated under reflux for 4 h, then transferred into a beaker and left to cool. The obtained precipitate was filtered, washed, dried and recrystallized from ethanol.
N-[5-(2-Hydroxybenzylidene)-4-oxo-4,5-dihydro-1,3-thiazol-2-yl]benzamide (27a): The title compound was obtained as pale brown crystals, m.p. 243–244°C, yield 0.46 g (62%). IR ν (cm−1): 3387 (OH), 3155 (NH), 3062 (CH aromatic), 1701 (2 C=O), 1628 (NH bending), 1597–1539 (C=C). 1H NMR (DMSO-d6, 400 MHz): δ 6.99–7.04 (m, 2H, aromatic H), 7.35 (t, J = 7.24 Hz, 1H, aromatic H), 7.51–7.59 (m, 3H, aromatic H), 7.67 (t, J = 7.32 Hz, 1H, aromatic H), 8.07 (s, 1H, CH=C), 8.18 (d, J = 7.44 Hz, 2H, aromatic H), 10.59 (br s, 1H, OH, D2O exchangeable), 13.08 (br s, 1H, NH, D2O exchangeable). 13C NMR (DMSO-d6, 100 MHz): δ 116.7, 120.2, 120.7, 122.9, 129.1, 129.6, 129.8, 130.0, 133.1, 133.8, 158.0 (aromatic carbons), 169.0, 176.0 (2 C=O). MS (EI): m/z (%) = 324 (M+, 1.38), 77 (100). Anal. calcd for C17H12N2O3S (324.35): C, 62.95; H, 3.73; N, 8.64; found: C, 63.11; H, 3.86; N, 8.88%.
N-{5-[(2-Hydroxynaphthalen-1-yl)methylidene]-4-oxo-4,5-dihydro-1,3-thiazol-2-yl}benzamide (27b): The product was obtained as dark-brown crystals, m.p. 180–182°C, yield 0.62 g (73%). IR ν (cm−1): 3483 (OH), 3314 (NH), 3067 (CH aromatic), 1697 (2 C=O), 1628 (NH bending), 1578–1531 (C=C). 1H NMR (DMSO-d6, 400 MHz): δ 7.28 (d, J = 9.08 Hz, 1H, aromatic H), 7.41 (t, J = 8.00 Hz, 1H, aromatic H), 7.55–7.67 (m, 2H, aromatic H), 7.72 (t, J = 7.72 Hz, 1H, aromatic H), 7.89–7.97 (m, 2H, aromatic H), 8.05 (d, J = 7.92 Hz, 1H, aromatic H), 8.14 (d, J = 7.20 Hz, 1H, aromatic H), 8.20–8.22 (m, 2H, aromatic H), 9.27 (s, 1H, CH=C), 10.94 (br s, 1H, NH), 12.89 (br s, 1H, OH). 13C NMR (DMSO-d6, 100 MHz): δ 113.2, 118.6, 123.7, 123.8, 127.8, 128.3, 128.6, 128.9, 128.9, 129.0, 129.1, 129.6, 129.7, 129.8, 132.1, 132.5, 133.4, 135.7, 135.9 (aromatic carbons), 154.5, 172.5 (2 C=O). Anal. calcd for C21H14N2O3S (374.41): C, 67.37; H, 3.77; N, 7.48; found: C, 67.12; H, 3.89; N, 7.63%.
N-[4-Oxo-5-(thiophen-2-ylmethylidene)-4,5-dihydro-1,3-thiazol-2-yl]benzamide (27c): The title compound was obtained as yellowish-green crystals, m.p. 224–226°C, yield 0.61 g (85%). IR ν (cm−1): 3391 (NH), 3028 (CH aromatic), 1724 (C=O), 1697 (C=O), 1647 (NH bending), 1589–1551 (C=C). 1H NMR (DMSO-d6, 400 MHz): δ 7.33 (t, J = 4.32 Hz, 1H, thiophene H), 7.58 (t, J = 7.44 Hz, 2H, aromatic H), 7.68 (t, J = 7.32 Hz, 1H, aromatic H), 7.75 (d, J = 3.36 Hz, 1H, thiophene H), 8.07–8.09 (m, 2H, CH=C and thiophene H), 8.20 (d, J = 7.32 Hz, 2H, aromatic H), 13.13 (br s, 1H, NH, D2O exchangeable). 13C NMR (DMSO-d6, 100 MHz): δ 122.2, 127.6, 129.2, 129.5, 129.8, 133.9, 134.3, 135.0, 135.9, 137.9 (aromatic carbons and thiophene carbons), 168.7, 176.0 (2 C=O). Anal. calcd for C15H10N2O2S2 (314.38): C, 57.31; H, 3.21; N, 8.91; found: C, 57.60; H, 3.49; N, 9.12%.
N-[5-(2-Hydroxybenzylidene)-4-oxo-4,5-dihydro-1,3-thiazol-2-yl]-2-phenylacetamide (27d): The product was obtained as light brown crystals, m.p. 267–268°C, yield 0.53 g (69%). IR ν (cm−1): 3263 (OH), 3128 (NH), 3032 (CH aromatic), 2927 (CH aliphatic), 1724 (C=O), 1663 (C=O), 1582–1555 (C=C). 1H NMR (DMSO-d6, 400 MHz): δ 3.89 (s, 2H, CH2), 6.94–6.98 (m, 2H, aromatic H), 7.27–7.37 (m, 6H, aromatic H), 7.45 (d, J = 7.68 Hz, 1H, aromatic H), 8.08 (s, 1H, CH=C), 10.54 (br s, 1H, NH, D2O exchangeable), 13.02 (br s, 1H, OH, D2O exchangeable). 13C NMR (DMSO-d6, 100 MHz): δ 43.5 (CH2), 116.7, 120.2, 120.9, 124.9, 127.5, 128.9, 129.3, 129.9, 130.0, 130.8, 133.0, 134.4 (aromatic carbons), 158.0, 174.0 (2 C=O). Anal. calcd for C18H14N2O3S (338.38): C, 63.89; H, 4.17; N, 8.28; found: C, 63.71; H, 4.38; N, 8.05%.
N-{5-[(2-hydroxynaphthalen-1-yl)methylidene]-4-oxo-4,5-dihydro-1,3-thiazol-2-yl}-2-phenylacetamide (27e): The title compound was obtained as dark-brown crystals, m.p. 200–201°C (decomposition), yield 0.55 g (63%). IR ν (cm−1): 3395 (OH), 3186 (NH), 3062–3032 (CH aromatic), 2981 (CH aliphatic), 1717 (2 C=O), 1631 (NH bending), 1593–1535 (C=C). 1H NMR (DMSO-d6, 400 MHz): δ 3.91 (s, 2H, CH2), 7.22–7.37 (m, 3H, aromatic H), 7.46–7.53 (m, 2H, aromatic H), 7.62–7.74 (m, 2H, aromatic H), 7.87–7.90 (m, 2H, aromatic H), 8.11–8.19 (m, 2H, aromatic H), 9.02 (s, 1H, CH=C), 10.15 (br s, 1H, NH, D2O exchangeable), 13.15 (br s, 1H, OH, D2O exchangeable). 13C NMR (DMSO-d6, 100 MHz): δ 43.5 (CH2), 112.0, 112.6, 114.0, 125.1, 127.1, 127.6, 127.7, 128.3, 128.7, 128.8, 128.9, 129.1, 129.7, 129.9, 130.0, 130.3, 131.1, 131.8, 133.3, 135.2 (aromatic carbons), 150.2, 178.9 (2 C=O). Anal. calcd for C22H16N2O3S (388.44): C, 68.03; H, 4.15; N, 7.21; found: C, 68.21; H, 4.33; N, 7.49%.
N-[4-Oxo-5-(thiophen-2-ylmethylidene)-4,5-dihydro-1,3-thiazol-2-yl]-2-phenylacetamide (27f): The product was obtained as brown crystals, m.p. 280–281°C, yield 0.55 g (74%). IR ν (cm−1): 3113 (NH), 3028 (CH aromatic), 2908 (CH aliphatic), 1720 (C=O), 1686 (C=O), 1589–1558 (C=C). 1H NMR (DMSO-d6, 400 MHz): δ 3.90 (s, 2H, CH2), 7.28–7.36 (m, 6H, aromatic H and thiophene H), 7.71 (d, J = 3.40 Hz, 1H, thiophene H), 8.01 (d, J = 4.92 Hz, 1H, thiophene H), 8.06 (s, 1H, CH=C), 13.11 (br s, 1H, NH, D2O exchangeable). 13C NMR (DMSO-d6, 100 MHz): δ 43.5 (CH2), 124.1, 127.5, 128.4, 128.9, 129.6, 129.9, 130.0, 133.8, 134.3, 135.8, 138.5 (aromatic carbons and thiophene carbons), 172.9, 180.4 (2 C=O). Anal. calcd for C16H12N2O2S2 (328.40): C, 58.52; H, 3.68; N, 8.53; found: C, 58.74; H, 3.85; N, 8.79%.