Abstract
A series of extra-large micropore Sn-zeolites with different Si/Sn ratios is post-synthesized by treating the interlayer-expanded material ECNU-9-cal773 with a solution of HCl containing NH4F and SnCl4∙5H2O, named as IEZ-Sn-PLS-3(S4R). During treatment, HCl, NH4F, and SnCl4∙5H2O are hydrolyzed into H+, F+, and Sn4+ ions. The formation of a small amount of HF formed from H+ and F+ ions can corrode the Si atom of the zeolite sheets and generate hydroxy-nest defect sites, Sn4+ ions fill up the hydroxy-nests to form isolated framework Sn active sites. The structure and active sites of the obtained materials are studied by various characterization methods. IEZ-Sn-PLS-3(S4R) possesses a 14 × 12-ring (R) pore system and framework Sn active sites. With enlarged pore sizes, IEZ-Sn-PLS-3(S4R) has been proved to be efficient for catalyzing the Baeyer–Villiger oxidation of 2-admantanone using H2O2 as the oxidant. The low Si/Sn sample resulted in a higher conversion compared to typical 12-ring zeolite Sn-Beta hydrothermally synthesized in an F− system.

Introduction
Metallosilicate zeolites are a type of heterogeneous catalyst that can be used in various industrial processes such as cracking, 1 alkylation, 2 isomerization, 3 epoxidation, 4 ammoximation, 5 Baeyer–Villiger (BV) oxidation, 6 Meerwein–Ponndorf–Verley reduction, 7 and so on, according to the framework and active sites that they possess. Thus, to further broaden the application of metallosilicate zeolites and to generate novel heterogeneous catalysts with excellent catalytic activity and selectivity, the design of new zeolite structures and the development of methods for the introduction of heteroatoms as active sites into the framework of zeolites have become important factors.
For designing new zeolitic structures, the most conventional method is via direct synthesis. This can be achieved using novel organic structure as directing agents or by adding crystallization additives.8,9 This approach is most straightforward, but designing new organic structures as directing agents is often expensive and time-consuming. In contrast, the post-synthesis method is relatively easy to operate. Layered zeolites are two-dimensional materials which are different from traditional three-dimensional zeolites. The lamina of layered zeolites is connected by hydrogen bonds and easily modified, as hydrogen bonds are not as stable as chemical bonds. Among the methods for modifying lamellar precursors including intercalation,10,11 delamination,12,13 pillaring, 10 and interlayer silylation, 14 the last one, interlayer silylation is the only method that can accurately regulate the interlayer structure of layered zeolites at the molecular level. In fact, many studies have reported new zeolitic materials with interlayer-expanded structures derived by silylation.14–27 These silylated materials have ordered pore systems, and some of them possess even more ordered structures than the material generated by direct calcination of a lamellar precursor.14,17 Although their pore size is enlarged, only a few are over 12-ring (12-R). ECNU-9 is an extra-large micropore zeolite with 14 × 12-R channels prepared by interlayer silylation. Its Ti-containing samples have been proved to be efficient for catalyzing the epoxidation of alkenes. 23 If we can introduce other heteroatoms into the framework of ECNU-9, we might be able to obtain highly efficient catalysts.
Sn-zeolite has been reported to be able to catalyze the BV oxidation of ketones, 6 Meerwein–Ponndorf–Verley reduction of ketones, 7 the isomerization of glucose into fructose, 28 conversion of glyceraldehyde into alkyl lactates, 29 and to activate carbonyl groups efficiently. 30 The ionic radius of Sn4+ is much larger than Si4+ (0.71 Å vs 0.41 Å), which results in difficulties in incorporating framework heteroatom Sn into the zeolite structure. Although Sn-containing zeolites have wide catalytic applications, they are not easy to prepare. So far, most reports on this material have focused on beta topology (12-R) and there are no reported Sn-zeolites over 12-R.
In this study, we conducted post-modification of ECNU-9 by treatment with HCl solution containing NH4F and SnCl4∙5H2O to introduce Sn active sites into the framework. During the procedure, HCl, NH4F, and SnCl4∙5H2O were hydrolyzed into H+, F−, and Sn4+ ions. H+ and F− formed HF acid in a low concentration. HF molecules can corrode the Si atom of the zeolite sheets and generate hydroxy-nest defect sites. Sn4+ ions fill up the hydroxy-nests to form isolated framework Sn active sites. This process is shown in Figure 1. The 14-R extra-large micropore is stable during the post-treatment. The resulting material was named as IEZ-Sn-PLS-3(S4R). With a small crystal size, various Si/Sn ratios, and an extra-large micropore size, IEZ-Sn-PLS-3(S4R) has been shown to catalyze the BV oxidation of 2-admantanone effectively.

The conversion of ECNU-9 into IEZ-Sn-PLS-3(S4R) by isomorphous substitution of Si4+ with Sn4+. Yellow atoms represent Sn atoms.
Results and discussion
Figure 2 gives the X-ray diffractions (XRDs) of ECNU-9 and its post-treated materials with different Sn contents. Interlayer-expanded material ECNU-9, prepared from silylation using 1,3,5,7-tetramethylcyclosilane and which possessed a single 4-ring structure (S4R) silane, exhibited sharp and intense reflections (Figure 2(a)), indicating that it possessed an ordered structure. The positions of its characteristic diffraction peaks were the same as reported by Yang et al. 23 in 2018, confirming that we had obtained the ECNU-9 structure. After calcination, the XRD reflections of ECNU-9 did not change obviously (Figure 2(b)), suggesting that the calcination process did not damage the structure of this material. A weak broad band appeared at 20°–30° for the ECNU-9 materials, indicating that it was slightly disordered in structure. This may be caused by uneven insertion of silane reagents into a few parts of ECNU-9. We chose to modify the calcined sample because the uncalcined material ECNU-9 contained methyl groups introduced by silylation reagents, which will block the channels and hinder the introduction of heteroatomic Sn. Hence, we placed ECNU-9-cal773 into HCl solution containing NH4F and different amounts of SnCl4∙5H2O to generate the Sn-zeolite IEZ-Sn-PLS-3(S4R) with varying Sn contents. According to the XRD patterns, the products had good crystalline structures, since their XRDs were sharp and intense (Figure 2(c)–(f)), indicating that the structure of ECNU-9-cal773 was maintained during the modification process.
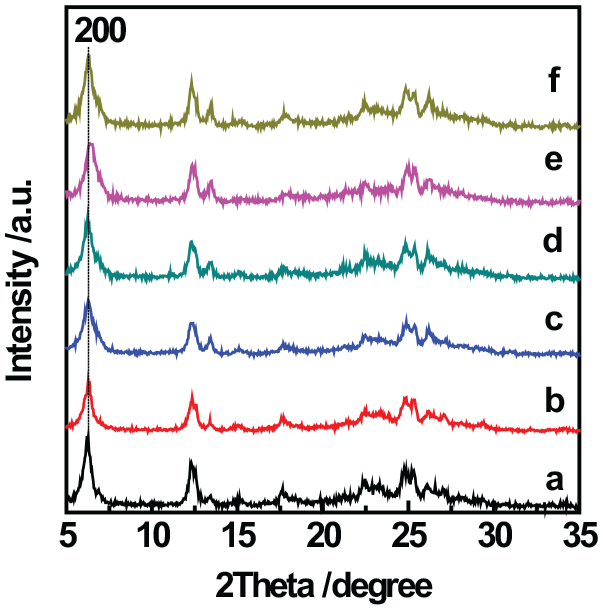
XRDs of (a) ECNU-9, (b) ECNU-9-cal773, (c) IEZ-Sn-PLS-3-30(S4R), (d) IEZ-Sn-PLS-3-50(S4R), (e) IEZ-Sn-PLS-3-70(S4R), and (f) IEZ-Sn-PLS-3-90(S4R).
Figure 3 shows the representative scanning electron microscope (SEM) images of ECNU-9, ECNU-9-cal773, IEZ-Sn-PLS-3-30(S4R), and IEZ-Sn-PLS-3-50(S4R). These four materials had the same rod-like morphology and had crystal sizes of nanoscale, c.a. 50–150 nm. This indicated that the post-treatment did not affect the morphology of the products and that the Sn-containing materials had high crystallinity and phase purities.

SEM images of (a) ECNU-9, (b) ECNU-9-cal773, (c) IEZ-Sn-PLS-3-30(S4R), and (d) IEZ-Sn-PLS-3-50(S4R).
The transmission electron microscope (TEM) images of IEZ-Sn-PLS-3-50(S4R) are given in Figure 4 as a representative example. Figure 4(a) and (b) display the crystalline structure of IEZ-Sn-PLS-3-50(S4R) along the b and c axes, respectively. This sample exhibited clear lattice fringes with a highly ordered arrangement of pores along these two directions without aggregation of Sn species. This result proved more directly that the post-modification process for introducing heteroatom Sn did not cause structural damage to the material, and that the introduced Sn species did not exist in aggregate form but in isolated form.

TEM images of IEZ-Sn-PLS-3-50(S4R) along the (a) [010] and (b) [001] directions.
After the characterization of the structure, the active sites of the resulting Sn-containing material were examined by UV-Vis and by vacuum infrared pyridine adsorption taking IEZ-Sn-PLS-3-50(S4R) as a representative. UV-Vis spectroscopy is a frequently used technique to investigate heteroatom-containing zeolites, and it can be used to analyze the coordination state of the transition metal ions in metallosilicates, especially Ti, Sn, and Zr. 31 The UV-Vis spectrum of IEZ-Sn-PLS-3-50(S4R) is shown in Figure 5. The main absorbance peak at 220 nm is characteristic of the Sn species in the framework position with a tetrahedrally coordinated state, confirming the existence of framework Sn heteroatoms. This band was often observed around 210 nm in the case of Sn-Beta zeolites. 6 This difference suggested that the energy level of Sn or the ligand in IEZ-Sn-PLS-3(S4R) may be different from conventional Sn-Beta zeolites. In addition, the spectral peak of the material was relatively broad, being indicative of the existence of a small amount of extra-framework Sn species with a hexa-coordinated state.

UV-Vis spectrum of IEZ-Sn-PLS-3-50(S4R).
Vacuum infrared pyridine adsorption spectra can be used to analyze the Brønsted and Lewis acid sites in zeolites in terms of both acid content and acid strength. Pyridine desorption at different temperatures was carried out using IEZ-Sn-PLS-3-50(S4R) (Figure 6). The absorbance at 1450 cm−1 which was attributed to Lewis acid sites, and at 1490 cm−1, which was attributed to both Brønsted and Lewis acid sites, remained even after desorption at a high temperature of 723 K. 32 Since these two absorbances were related to the vibration modes of the pyridine molecules adsorbed on the Lewis acid sites, they can serve as proof for the existence of Lewis acid sites in this stanosilicate material.

Vacuum infrared pyridine adsorption spectra of ECNU-9-cal773 after pyridine adsorption at 298 K for 30 min and desorption at (a) 523 K for 1 h and IEZ-Sn-PLS-3-50(S4R) after pyridine adsorption at 298 K for 30 min and desorption at (b) 423 K for 1 h, (c) 523 K for 1 h, (d) 623 K for 1 h, and (e) 723 K for 1 h.
The textural properties of ECNU-9-cal773 and IEZ-Sn-PLS-3(S4R) are given in Table 1. The SBET and Vtotal values were similar for ECNU-9-cal773 and its modification-derived Sn-containing materials, serving as further confirmation of the maintenance of the structure during post-treatment. All the materials exhibited type-I adsorption/desorption isotherms, indicative of the presence of microporous structure (see Figure 7). The Sn source that we used in the starting mixture was partially incorporated in the products, since the Si/Sn ratios of the products were higher than that in the starting mixture.

N2 adsorption isotherms of different materials.
The textural properties and BV oxidation of 2-admantanone with H2O2 catalyzed by the different zeolites. a

BV: Baeyer–Villiger; BET: Brunauer–Emmett–Teller; ICP: inductively coupled plasma.
Detailed conditions for the reaction: Sn-zeolite (50 mg), H2O2 (50%) (4 mmol), 2-adamantanone (2 mmol), C6H5NO3 (10 mL), 363 K, and 1 h.
Specific surface area (BET method)
Total volume (BET method, N2 adsorption, and P/P0 = 0.9).
Determined by ICP.
The BV oxidation of 2-admantanone was conducted to test the catalytic performance of the materials prepared above. We examined the effect of the solvent on the reaction (Table 2). Nitrobenzene (C6H5NO3) was the best of all the tested solvents; thus, we used C6H5NO3 as the solvent for further tests. The overall trend for the conversion of 2-admantanone with all the IEZ-Sn-PLS-3(S4R) samples was that the conversion decreased with the increase in the Si/Sn ratio (Table 1). This was because of the reduction of the total Sn active sites. The best conversion of the IEZ-Sn-PLS-3(S4R) materials reached 57% which was higher than the Sn-Beta zeolite hydrothermally prepared from the F− system. Although the Sn content for IEZ-Sn-PLS-3(S4R) was higher than Sn-Beta, this result still confirmed that the material we prepared was an efficient catalyst for the BV oxidation of 2-admantanone. The extra-large micropore sizes of the IEZ-Sn-PLS-3(S4R) samples can reduce steric hindrance and be beneficial to substrate diffusion, which may contribute to its comparable excellent catalytic activity.
BV oxidation of 2-admantanone with H2O2 catalyzed by IEZ-Sn-PLS-3-50(S4R) in different solvents. a

BV: Baeyer–Villiger.
Detailed conditions for the reactions: Sn-zeolite (50 mg), H2O2 (50%) (4 mmol), 2-adamantanone (2 mmol), solvent (10 mL), 363 K, and 4 h.
Conclusion
Novel Sn-containing ferrierite (FER) type zeolites IEZ-Sn-PLS-3(S4R) possessing adjustable Sn contents have been post-synthesized by treating the interlayer-expanded material ECNU-9-cal773 in HCl solution containing NH4F and SnCl4∙5H2O. We characterized the structures and Sn active sites of the resulting Sn-zeolites, which were found to possess 14 × 12-R extra-large micropore system and framework Sn heteroatoms. This is the first report on Sn-zeolite with 14 × 12-R extra-large micropore. With an extra-large micropore size, IEZ-Sn-PLS-3(S4R) materials have been confirmed to be efficient solid catalysts for the BV oxidation of bulk 2-admantanone using H2O2 as the oxidant. With the help of the extra-large pore size, the high Sn content samples of IEZ-Sn-PLS-3(S4R) can give higher conversion than the typical large micropore zeolite Sn-Beta synthesized in the F− system.
Experimental
Preparation of IEZ-Sn-PLS-3(S4R) materials
The interlayer-expanded structure ECNU-9 was prepared according to a literature report. 23 After being calcined at 773 K for 6 h in air, we obtained ECNU-9-cal773. Next, ECNU-9-cal773 was treated with HCl solution containing NH4F and SnCl4∙5H2O at 323 K for 5 h to produce IEZ-Sn-PLS-3(S4R) materials. The reacting mixture had a composition of SiO2:H+:F−:Sn4+:H2O of 1.0:0.4:1.2:(0.011–0.033):83.33 (using HCl, NH4F, and SnCl4∙5H2O as reagents). The resulting material was named IEZ-Sn-PLS-3-n(S4R) (where n is the molar ratio of Si/Sn in the reacting mixture).
Preparation of Sn-Beta
For comparison, Sn-Beta was synthesized using a fluorine system. 31 The reactant had a molar ratio of SiO2:HF:TEAOH:SnCl4:H2O of 1.0:0.54:0.56:0.005:7.5 together with de-aluminated Beta as seeds.
Reactions
The BV oxidations were run in a 50-mL flask with oil bath heating under stirring. The reaction conditions are shown in the footnotes of Tables 1 and 2.
Characterization
The following equipment was used for characterization. XRD: X’ Pert PRO MPD. SEM: Hitachi S-4800 microscope. TEM: FEI G2F30, inductively coupled plasma (ICP), VISTA-MPX atomic emission spectrometer. N2 adsorption/desorption: BEL-MAX gas/vapor adsorption equipment. Fourier-transform infrared spectroscopy (FTIR): Nicolet Nexus 670. UV-Vis: PerkinElmer Lambda 35 spectrophotometer.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this paper: This work was supported by the National Natural Science Foundation of China (Grant No. 21802002), Jinlin City Science and Technology Bureau (No. 20200104077), and Central Government to Guide Local Science and Technology Development Funds, Jinlin Province Basic Research Project (No. 202002006JC).
