Abstract
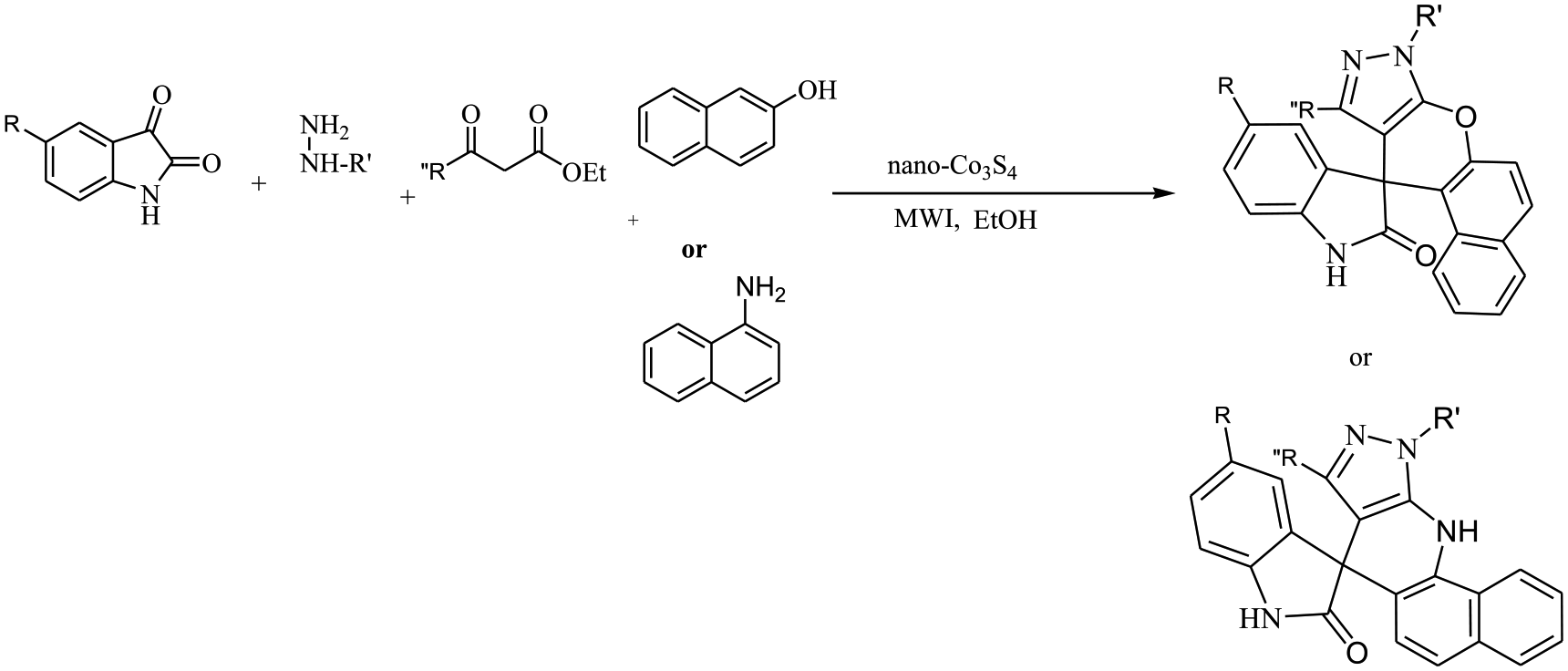
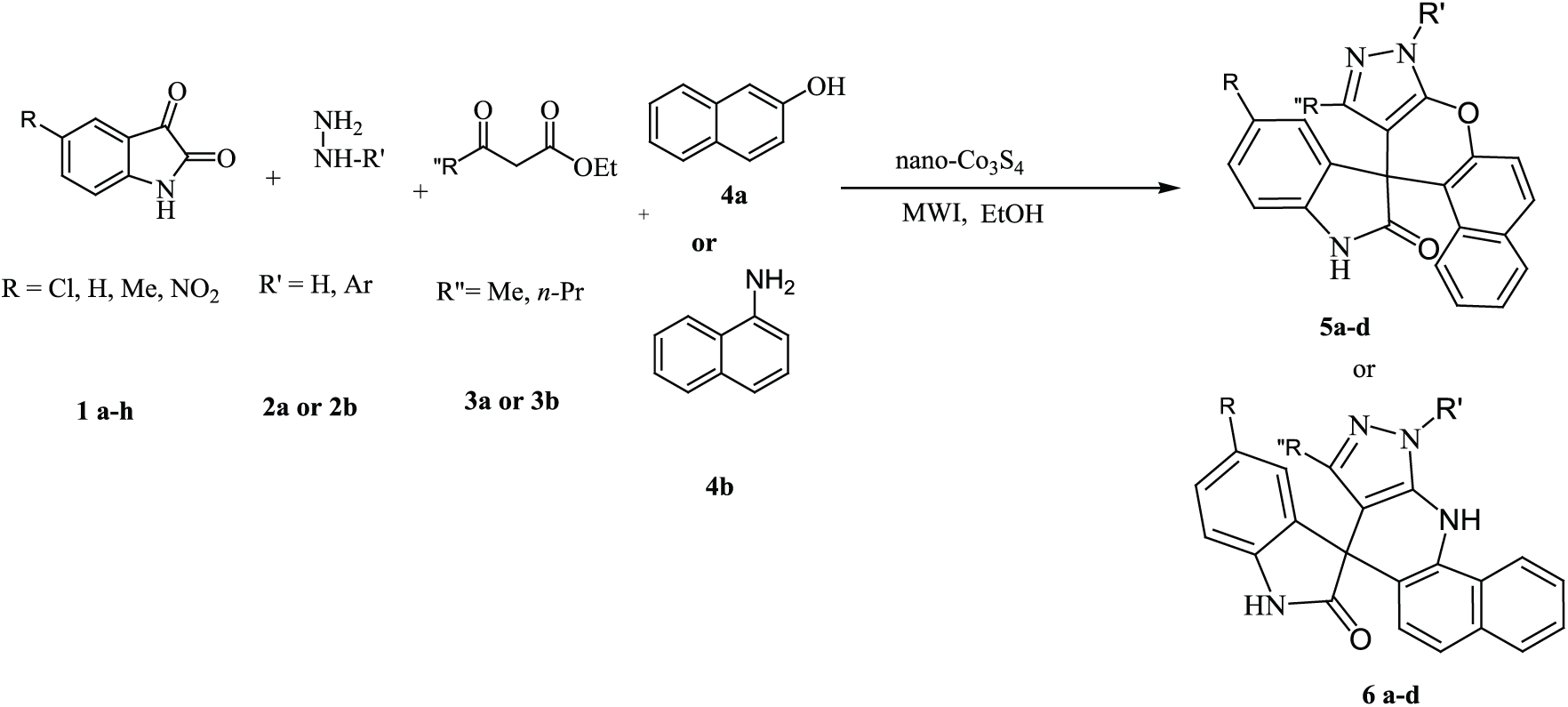
An efficient and rapid method is used for the synthesis of 10-methyl-8H-spiro[benzo[5,6]chromeno[2,3-c]pyrazole-11,3′-indol]-2′(1′H)-one and 8-methyl-10-phenyl-10,11-dihydrospiro[pyrazolo[3,4-b]benzo[h]quinolin-7,3′-indol]-2′(1′H)-one by a four-component reactions of phenylhydrazine or hydrazine hydrate, isatins, ketoesters, and naphthylamine or 2-naphthol using nano-Co3S4 under microwave irradiation.
Introduction
Spirooxindoles have emerged as a group of important heterocycles owing to their presence in a broad spectrum of natural and synthetic organic compounds,1–3 with various biological properties including antimicrobial,4–6 antitumor,7,8 and antidiabetic,
9
and can also serve as synthetic intermediates for diverse kinds of pharmaceuticals or drug precursors.
10
Several derivatives of this ring system fluorescence are considered as promising materials for application in optoelectronic devices and light-emitting diodes.11,12 These activities make spirooxindoles attractive targets in organic synthesis. The synthesis of spirooxindoles has been reported in the presence of diverse catalysts such as p-toluenesulfonic acid (p-TSA),13,14 piperidine,
15
alum (KAl(SO4)2·12H2O),
16
amino-functionalized SBA-15 (SBA-Pr-NH2),
17
nanocrystalline MgO,
18
and

Synthesis of spirooxindoles under microwave irradiations.
Results and discussion
Initially, we had explored and optimized different reaction parameters for the synthesis of spirooxindoles by the condensation reaction of hydrazine hydrate, 5-chloroisatin, ethyl acetoacetate, and 2-naphthol as a model reaction. Several reactions were scrutinized using various solvents such as EtOH, CH3CN, water, or dimethylformamide (DMF). Various catalysts, solvents, and MW conditions were evaluated and the results are shown in Table 1. Yields were determined for reaction in the presence of various catalysts such as Fe3O4, ceric ammonium nitrate (CAN), NaHSO4, and Et3N, but yield of 5′-chloro-10-methyl-8H-spiro[benzo[5,6]chromeno[2,3-c]pyrazole-11,3′-indol]-2′(1′H)-one (
Optimization of the reaction conditions (catalyst, catalyst loading, solvent, and duration of reaction) for the preparation of 5′-chloro-10-methyl-8H-spiro[benzo[5,6]chromeno[2,3-c]pyrazole-11,3′-indol]-2′(1′H)-one a .

Bold: Bold values indicate the best result in reaction conditions.
MWI: microwave irradiation.
Hydrazine hydrate (1 mmol), 5-chloroisatin (1 mmol), ethyl acetoacetate (1 mmol), and 2-naphthol (1 mmol).
Isolated yield.
To investigate the scope and limitation of this catalytic process, phenylhydrazine or hydrazine hydrate, isatins, ketoesters, and 1-naphthylamine or 2-naphthol were chosen as substrates. The above-mentioned results obviously show that the present catalytic procedure is extendable to a wide variety of substrates to construct a diversity-oriented library of spirooxindoles. Investigations of the reaction scope revealed that various isatins (bearing electron-withdrawing and electron-donating groups) can be utilized in this protocol (Table 2).
Synthesis of spirooxindoles by nano-Co3S4 (15 mg) under microwave irradiation (400 W) a .
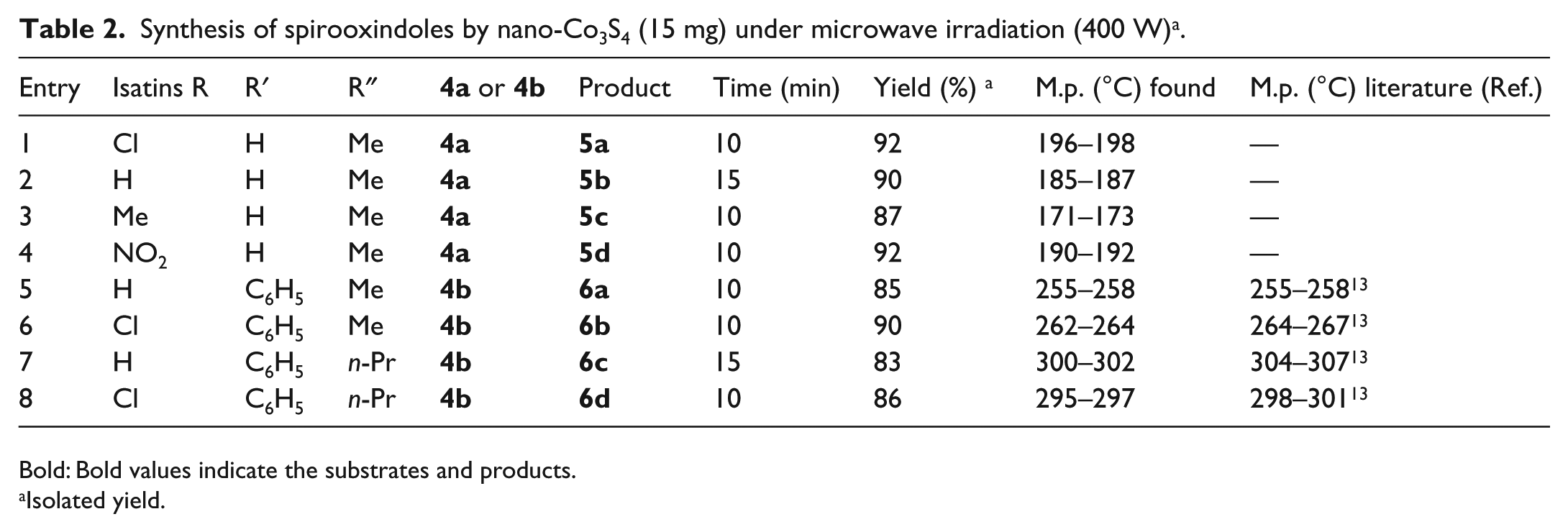
Bold: Bold values indicate the substrates and products.
Isolated yield.
The reusability is one of the significant properties of this catalyst. The reusability of nano-Co3S4 was studied for the reaction of hydrazine hydrate, 5-chloroisatin, ethyl acetoacetate, and 2-naphthol, and it was found that product yields decreased to a small extent on each reuse (run 1, 92%; run 2, 92%; run 3, 91%; run 4, 91%; run 5, 90%; run 6, 90%). After completion of the reaction (thin layer chromatography (TLC)), acetone was added. The catalyst was insoluble in acetone, and it could therefore be recycled by an easy filtration. The solvent was then evaporated, and the solid obtained was recrystallized from ethanol to afford the spirooxindoles. The recovered nano-Co3S4 was washed five to seven times with ethanol and dried at room temperature for 20 h.
In conclusion, we have developed a straightforward and efficient method for the synthesis of spirooxindoles using nano-Co3S4 under MWIs. The method offers several advantages including high atom economy, easy availability, high yields, shorter reaction times, reusability of the catalyst, low catalyst loading, and utilization of microwave as clean procedure.
Experimental
Reagent-grade chemicals were purchased from Sigma-Aldrich or Merck and were used without further purification. The products were isolated and characterized by physical and spectral data. NMR spectra were obtained on a Bruker AVANCE 400 MHz spectrometer (1H NMR at 400 Hz and 13C NMR at 100 Hz) in dimethyl sulfoxide (DMSO)-d6 using tetramethylsilane (TMS) as an internal standard. Chemical shifts (δ) are given in parts per million and coupling constants (J) are given in Hertz. The infrared (IR) spectra were recorded on Fourier transform infrared (FT-IR) Magna 550 apparatus using KBr plates. Melting points were determined on Electrothermal 9200 and are not corrected. The elemental analyses (C, H, N) were obtained from a Carlo ERBA Model EA 1108 analyzer. We used the Milestone microwave (MLS GmbH-ATC-FO 300; Microwave Labstation) for synthesis.
Preparation of nano-Co3S4
In a typical preparation, molar ratios (1:3) of Co(CH3COO)2·4H2O to thioglycolic acid were mixed in 200 mL distilled water with stirring. After 30 min, stirring the final solution was sealed in a 250-mL Teflon-lined stainless steel autoclave and heated at 150 °C for 48 h in an electric oven. The autoclave was allowed to cool to room temperature, and the product was washed with distilled water and absolute ethanol several times to remove excessive reactants and by-products, followed by drying in an electric oven at 70 °C for 12 h (See Electronic supplementary information).
General procedure for the synthesis of spirooxindoles
A mixture of isatin derivatives (1 mmol), phenylhydrazine or hydrazine hydrate (1 mmol), alkyl acetoacetate (1 mmol), naphthalene amine or 2-naphthol (1 mmol) and nano-Co3S4 (15 mg) were irradiated under microwave at 400 W for the appropriate times. The reaction was monitored by TLC (hexane/ethyl acetate 8:2). After cooling, the reaction mixture was dissolved in acetone and the mixture was stirred for 2 min. The suspended solution was filtered and the heterogeneous catalyst was recovered. The acetone was evaporated and the solid that separated out was filtered and recrystallized with ethanol to get pure product. The structures of the products were fully established on the basis of their 1H NMR, 13C NMR, FT-IR spectra, and melting points.
Physical and spectroscopic data
5'-Chloro-10-methyl-8H-spiro[benzo[5,6]chromeno[2,3-c]pyrazole-11,3'-indol]-2'(1′H)-one (
10-Methyl-8H-spiro[benzo[5,6]chromeno[2,3-c]pyrazole-11,3'-indol]-2'(1'H)-one (
5'-Methyl-10-methyl-8H-spiro[benzo[5,6]chromeno[2,3-c]pyrazole-11,3'-indol]-2'(1'H)-one (
5'-Nitro-10-methyl-8H-spiro[benzo[5,6]chromeno[2,3-c]pyrazole-11,3'-indol]-2'(1′H)-one
Supplemental Material
Electronic_supplementary___information – Supplemental material for Preparation of spirooxindoles catalyzed by nano-Co3S4 under microwave irradiations
Supplemental material, Electronic_supplementary___information for Preparation of spirooxindoles catalyzed by nano-Co3S4 under microwave irradiations by Sheida Khojasteh-Khosro and Hossein Shahbazi-Alavi in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
