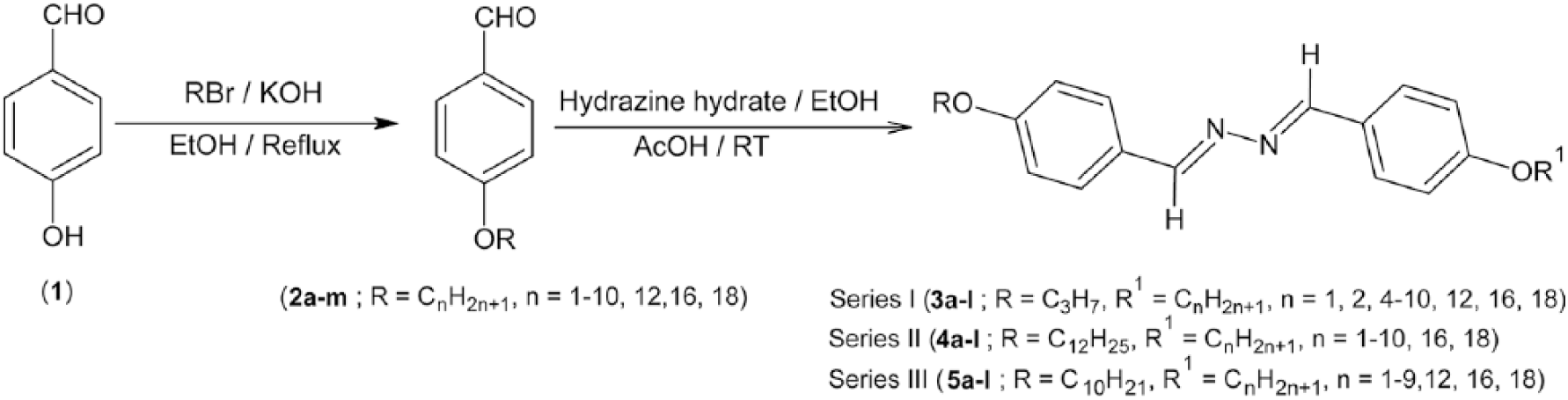
All reagents and solvents were obtained from Sigma-Aldrich Co. and were used as supplied without further purification. Solvents were distilled from the appropriate drying agent immediately prior to use. 4-(n-Alkoxy)benzaldehydes (n-alkoxy = O(CH2)
n
H, n = 1–10, 12, 16 or 18) (2a–m), {1-(4-propyloxy)-2-[4′-(n-alkoxy)]benzylidene}hydrazines (n-alkoxy = O(CH2)
n
H, n = 1,2,4–10, 12, 16 or 18) (Series I, 3a–l) and {1-[4-(n-alkoxy)]-2-(4’-dodecyloxy)benzylidene}hydrazines (n-alkoxy = O(CH2)
n
H, n = 1–10, 16 or 18) (Series II, 4a–l) were prepared according to our previous work.19–21 The purity of the prepared compounds was determined by thin-layer chromatography using silica gel as stationary phase and n-hexane/ethyl acetate (16:1) as eluent. Column chromatography was carried out using silica gel 60 (200–300 mesh). Elemental analyses (C, H and N) were determined using an EuroEA 3000 Elemental Analyzer. A Varin Cary 100 Conc. spectrophotometer was used to measure the electronic spectra of the prepared compounds in the region 200–800 nm at room temperature for a 5 × 10−5 M solution of the sample in dichloromethane. A RF-6000 Shimadzu spectrofluorimeter equipped with a ThermoNeslab RTE7 bath was used to measure the fluorescence emissions of all the prepared compounds in the solid state. IR spectra were obtained in the region 4000–600 cm−1 using an 8400S-FT–IR Shimadzu spectrophotometer. NMR spectra were recorded on a Bruker AV-II 500 MHz spectrometer (1H NMR at 500 MHz, 13C NMR at 125 MHz) in CDCl3 using TMS as internal standard. Chemical shifts (δ) are given in ppm and coupling constants (J) are given in hertz (Hz). Mass spectra of the compounds were obtained using an Orbitrap LTQ XL-Thermo Fisher scientific mass spectrometer. DSC thermograms were measured in a nitrogen atmosphere at a heating and cooling rate of 5.0°C min−1 using a Linseis STA PT-1000 equipment, which is precalibrated (the equipment was provided with an autocool system). Liquid crystalline mesophases of the compounds were obtained using a PW-BK5000R microscope equipped with a HS-400 (KER3100-08S) heating stage. The X-ray diffractograms at various temperatures were recorded on an XRD-6000 Shimadzu x-ray diffractometer equipped with an Anton Paar HTK1200 heating stage using Cu-Kα radiation (λ = 1.54178 Å).
Synthesis of {1-[4-(n-Alkoxy)]-2-(4’-decyloxy)benzylidene}hydrazines (Series III, 5a–l); general procedure
Hydrazine hydrate (0.125 g, 2.5 mmol) was added to a mixture of 4-decyloxybenzaldehyde (2j; R = C10H21) (0.66 g, 2.5 mmol) and a 4-(n-alkoxy)benzaldehyde compound (2.5 mmol) in ethanol (30 mL). To this mixture, acetic acid (100%, 0.1 mL) was added. The reaction mixture was stirred at room temperature for 1 h, during which a precipitate formed. This was filtered off, washed with cold water and purified by column chromatography using n-hexane/ethyl acetate (19:1) as eluent. CAUTION: Hydrazine is a suspected carcinogen and therefore should be handled with appropriate precautions.
[1-(4-Methoxy)-2-(4’-decyloxy)benzylidene]hydrazine (
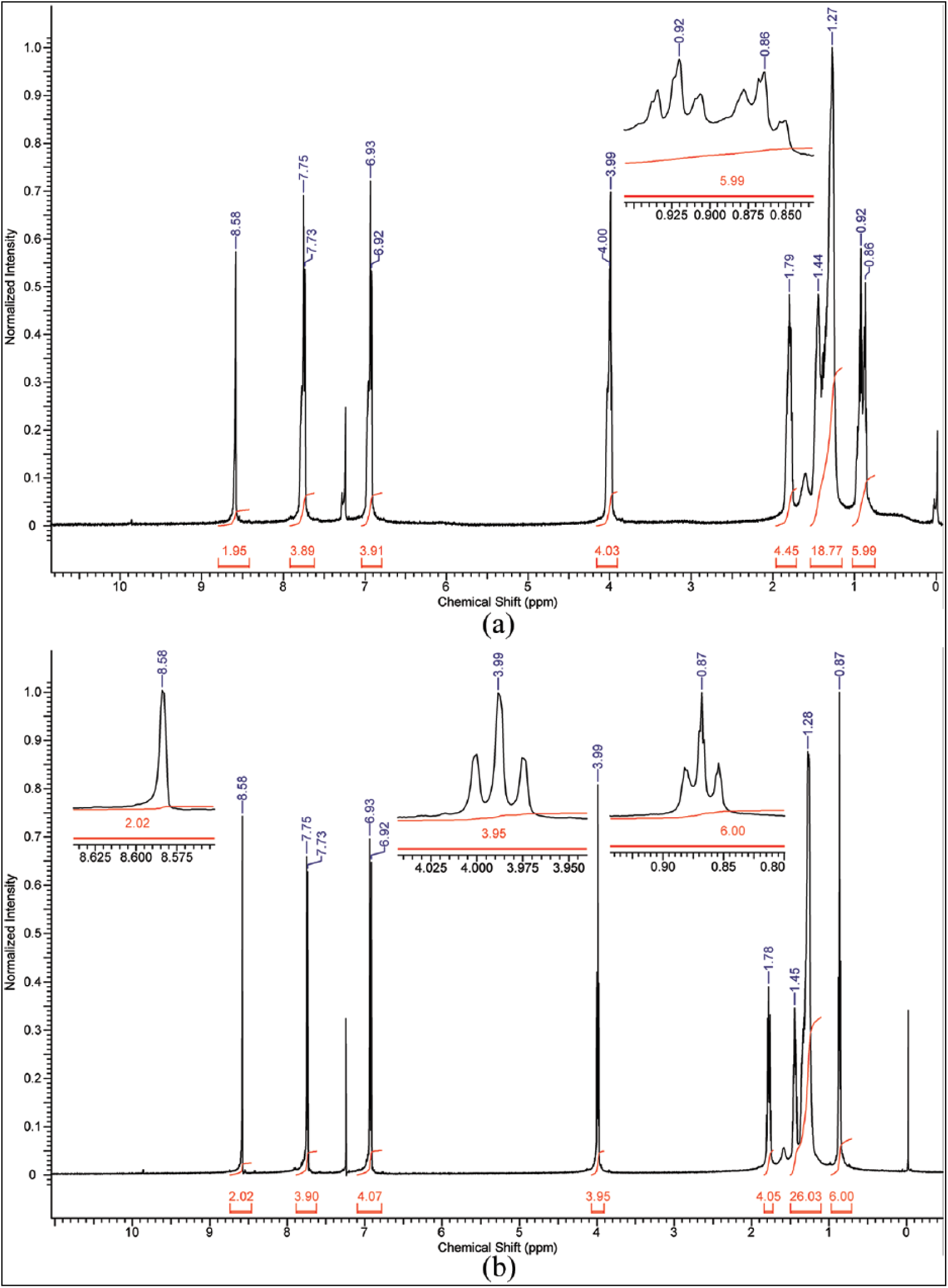
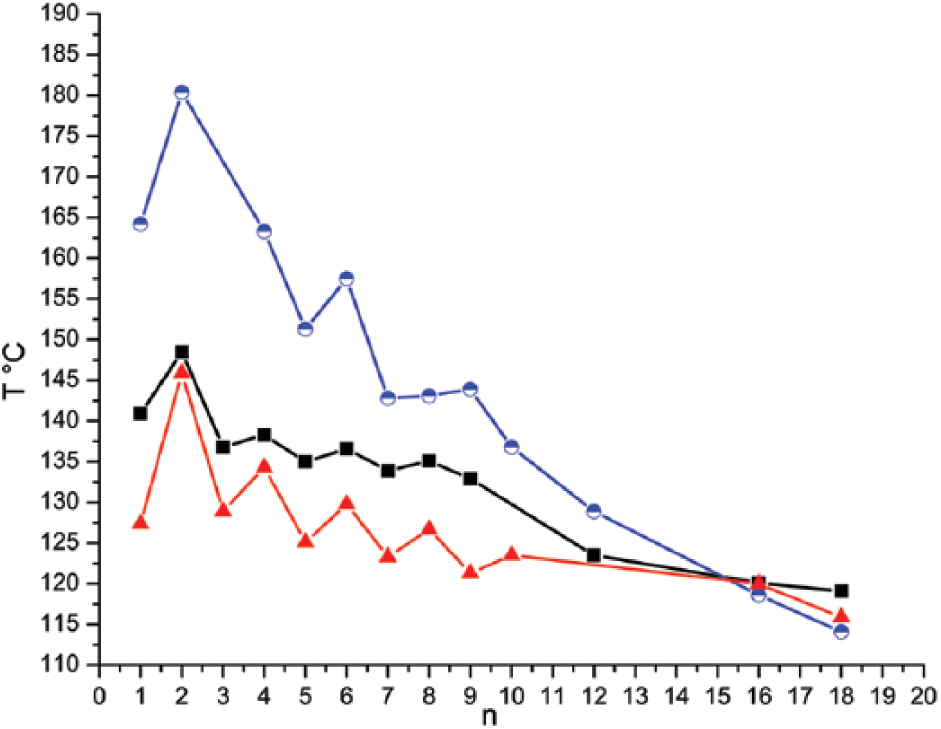
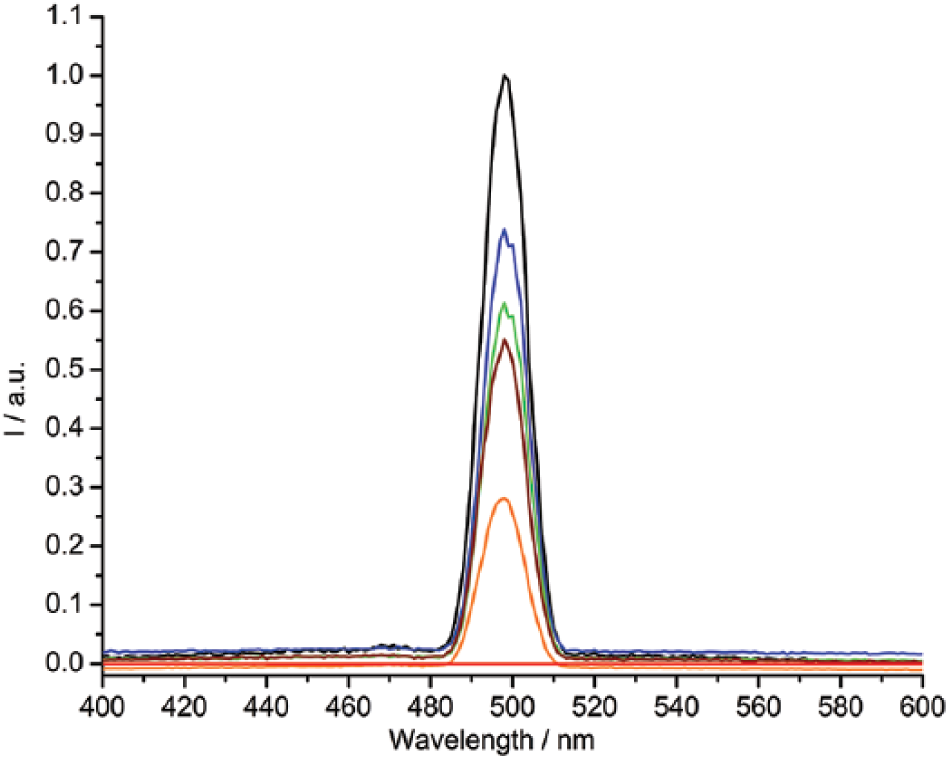
5a; R = C10H21, R1 = CH3): Prepared from 4-methoxybenzaldehyde (2a; R = CH3) (0.34 g) to give (5a; R = C10H21, R1 = CH3); light yellow solid; yield 60%; m.p. (clearing temperature) 141°C; Rf = 0.43; UV-Vis (λ/nm) (εmax/dm3 mol−1 cm−1): (241) (3700) (n–σ*), (332) (13800) (π–π*); fluorescence (λmaxexc/nm) (λmaxems/nm): (248.0) (497.9); IR (υ/cm−1): 3057 (w, υ (C–H) aromatic), 2955 (w, υas (C–H) of CH3 group), 2918 (s, υas (C–H) of CH2 group), 2873 (w, υs (C–H) of CH3 group), 2850 (m, υs (C–H) of CH2 group), 1624 (s, υ (C=N)), 1604, 1575 and 1508 (s, m, s, υ (C=C) aromatic), 1249 (vs, υ (C–O)); 1H NMR (δ/ppm): 0.96 (3H, t, J = 7.0 Hz, CH3), 1.19–1.36 (14H, m, CH2), 1.79 (2H, m, CH2), 3.83 (3H, s, OCH3), 4.05 (2H, t, J = 6.9 Hz, OCH2), 6.88 (4H, d, J = 8.8 Hz, aromatic protons), 7.73 (4H, d, J = 8.8 Hz, aromatic protons), 8.59 (2H, s, CH=N); 13C NMR (δ/ppm): 10.5 (CH3), 22.2 (CH2), 26.6 (CH2), 29.4 (CH2), 29.8 (CH2), 31.8 (CH2), 56.2 (OCH3), 69.4 (OCH2), 114.2 (aromatic C–H), 126.7 (aromatic C), 130.4 (aromatic C–H), 161.2 (CH=N), 161.2 (aromatic C–O); MS m/z: 394. Anal. calcd for C25H34N2O2: C, 76.10; H, 8.69; N, 7.10; found: C, 76.03; H, 8.54; N, 7.01%.
[1-(4-Ethoxy)-2-(4′-decyloxy)benzylidene]hydrazine (
5b; R = C10H21, R1 = C2H5): Prepared from 4-ethoxybenzaldehyde (2b; R = C2H5) (0.38 g) to give (5b; R = C10H21, R1 = C2H5); light yellow solid; yield 61%; m.p. (clearing temperature) 149°C; Rf=0.46; UV-Vis (λ/nm) (εmax/dm3 mol−1 cm−1): (242) (3220) (n–σ*), (333) (13940) (π–π*); fluorescence (λmaxexc/nm) (λmaxems/nm): (248.0) (498.0); IR (υ/cm−1): 3066 (w, υ (C–H) aromatic), 2956 (w, υas (C–H) of CH3 group), 2920 (s, υas (C–H) of CH2 group), 2874 (w-m, υs (C–H) of CH3 group), 2850 (m, υs (C–H) of CH2 group), 1622 (m, υ (C=N)), 1605, 1575 and 1508 (s, m, m, υ (C=C) aromatic), 1247 (vs, υ (C–O)); 1H NMR (δ/ppm): 0.91 (3H, t, J = 7.0 Hz, CH3), 1.08 (3H, t, J = 7.0 Hz, CH3), 1.20–1.44 (14H, m, CH2), 1.84 (2H, m, CH2), 3.95 (2H, t, J = 6.9 Hz, OCH2), 4.08 (2H, t, J = 6.9 Hz, OCH2), 6.93 (4H, d, J = 8.9 Hz, aromatic protons), 7.74 (4H, d, J = 8.9 Hz, aromatic protons), 8.58 (2H, s, CH=N); 13C NMR (δ/ppm): 10.7 (CH3), 14.81 (CH3), 22.6 (CH2), 26.3 (CH2), 29.3 (CH2), 29.7 (CH2), 31.8 (CH2), 63.3 (OCH2), 69.2 (OCH2), 114.4 (aromatic C–H), 126.4 (aromatic C), 130.3 (aromatic C–H), 161.2 (CH=N), 161.5 (aromatic C–O); MS m/z: 408. Anal. calcd for C26H36N2O2: C, 76.43; H, 8.88; N, 6.86; found: C 76.24, H 8.72, N 6.73%.
[1-(4-Propyloxy)-2-(4′-decyloxy)benzylidene]hydrazine (
5c; R = C10H21, R1 = C3H7): Prepared from 4-propyloxybenzaldehyde (2c; R = C3H7) (0.41 g) to give (5c; R = C10H21, R1 = C3H7); light yellow solid; yield 67%; m.p. (clearing temperature) 138°C; Rf = 0.49; UV-Vis (λ/nm) (εmax/dm3 mol−1 cm−1): (242) (5180) (n–σ*), (333) (26650) (π–π*); fluorescence (λmaxexc/nm) (λmaxems/nm): (248.0) (495.0); IR (υ/cm−1): 3067 (w, υ (C–H) aromatic), 2955 (m, υas (C–H) of CH3 group), 2918 (s, υas (C–H) of CH2 group), 2872 (w-m, υs (C–H) of CH3 group), 2851 (m, υs (C–H) of CH2 group), 1622 (m, υ (C=N)), 1605, 1574 and 1508 (s, m, s, υ (C=C) aromatic), 1248 (vs, υ (C–O)); 1H NMR (δ/ppm): 0.82 (3H, t, J = 7.3 Hz, CH3), 0.96 (3H, t, J = 7.3 Hz, CH3), 1.19–1.33 (14H, m, CH2), 1.75 (4H, m, CH2), 3.90 (2H, t, J = 6.6 Hz, OCH2), 3.93 (2H, t, J = 6.6 Hz, OCH2), 6.89 (4H, d, J = 8.8 Hz, aromatic protons), 7.70 (4H, d, J = 8.8 Hz, aromatic protons), 8.54 (2H, s, CH=N); 13C NMR (δ/ppm): 10.5 (CH3), 14.1 )CH3), 22.4 (CH2), 26.04 (CH2), 29.3 (CH2), 29.6 (CH2), 32.0 (CH2), 68.2 (OCH2), 69.6 (OCH2), 114.8 (aromatic C–H), 126.8 (aromatic C), 130.1 (aromatic C–H), 161.1 (CH=N), 161.7 (aromatic C–O); MS m/z: 422. Anal. calcd for C27H38N2O2: C, 76.74; H, 9.06; N, 6.63; found: C, 76.54; H, 9.01; N, 6.50%.
[1-(4-Butyloxy)-2-(4′-decyloxy)benzylidene]hydrazine (
5d; R = C10H21, R1 = C4H9): Prepared from 4-butyloxybenzaldehyde (2d; R = C4H9) (0.45 g) to give (5d; R = C10H21, R1 = C4H9); light yellow solid; yield 64%; m.p. (clearing temperature) 140°C; Rf = 0.52; UV-Vis (λ/nm) (εmax/dm3 mol−1 cm 1): (241) (3221) (n–σ*), (331) (13780) (π–π*); fluorescence (λmaxexc/nm) (λmaxems/nm): (248.0) (497.1); IR (υ/cm−1): 3066 (w, υ (C–H) aromatic), 2956 (w-m, υas (C–H) of CH3 group), 2920 (s, υas (C–H) of CH2 group), 2872 (w-m, υs (C–H) of CH3 group), 2850 (m, υs (C–H) of CH2 group), 1622 (m, υ (C=N)), 1604, 1573 and 1508 (s, m, m, υ (C=C) aromatic), 1247 (vs, υ (C–O)); 1H NMR (δ/ppm): 0.88 (3H, t, J = 7.0 Hz, CH3), 0.94 (3H, t, J = 7.0 Hz, CH3), 1.25–1.52 (16H, m, CH2), 1.80 (4H, m, CH2), 4.00 (2H, t, J = 6.8Hz, OCH2), 4.04 (2H, t, J = 6.8Hz, OCH2), 6.97 (4H, d, J = 8.8 Hz, aromatic protons), 7.79 (4H, d, J = 8.8 Hz, aromatic protons), 8.59 (2H, s, CH=N); 13C NMR (δ/ppm): 13.8 (CH3), 19.2 (CH3), 22.8 (CH2), 26.1 (CH2), 29.2 (CH2), 29.4 (CH2), 29.7 (CH2), 31.9 (CH2), 67.9 (OCH2), 68.2 (OCH2), 114.7 (aromatic C–H), 126.7 (aromatic C), 130.2 (aromatic C–H), 161.1 (CH=N), 161.7 (aromatic C–O); MS m/z: 436. Anal. calcd for C28H40N2O2: C, 77.02; H, 9.23; N, 6.42; found: C, 77.13; H, 9.15; N, 6.34%.
[1-(4-Pentyloxy)-2-(4′-decyloxy)benzylidene]hydrazine (
5e; R = C10H21, R1 = C5H11): Prepared from 4-pentyloxybenzaldehyde (2e; R = C5H11) (0.48 g) to give (5e; R = C10H21, R1 = C5H11); light yellow solid; yield 60%; m.p. (clearing temperature) 135°C; Rf = 0.54; UV-Vis (λ/nm) (εmax/dm3 mol−1 cm−1): (242) (3140) (n–σ*), (332) (16500) (π–π*); fluorescence (λmaxexc/nm) (λmaxems/nm): (248.0) (498.0); IR (υ/cm−1): 3074 (w, υ (C–H) aromatic), 2956 (w, υas (C–H) of CH3 group), 2922 (s, υas (C–H) of CH2 group), 2872 (w, υs (C–H) of CH3 group), 2854 (m, υs (C–H) of CH2 group), 1622 (m, υ (C=N)), 1604, 1573 and 1508 (s, m, s, υ (C=C) aromatic), 1249 (vs, υ (C–O)); 1H NMR (δ/ppm): 0.86 (3H, t, J = 7.1 Hz, CH3), 0.92 (3H, t, J = 7.1 Hz, CH3), 1.22–1.51 (18H, m, CH2), 1.79 (4H, m, CH2), 3.99 (2H, t, J = 6.8 Hz, OCH2); 4.03 (2H, t, J = 6.8 Hz, OCH2), 6.925 (4H, d, J = 8.8 Hz, aromatic protons); 7.74 (4H, d, J = 8.8 Hz, aromatic protons), 8.58 (2H, s, CH=N); 13C NMR (δ/ppm): 12.1 (CH3), 18.1 (CH3), 22.7 (CH2), 26.4 (CH2), 29.3 (CH2), 29.5 (CH2), 29.6 (CH2), 32.0 (CH2), 67.9 (OCH2), 68.2 (OCH2), 114.7 (aromatic C–H), 126.7 (aromatic C), 130.3 (aromatic C–H), 161.1 (CH=N), 161.7 (aromatic C–O); MS m/z: 450. Anal. calcd for C29H42N2O2: C, 77.29; H, 9.39; N, 6.22; found: C, 77.20; H, 9.22; N, 6.04%.
[1-(4-Hexyloxy)-2-(4’-decyloxy)benzylidene]hydrazine (
5f; R = C10H21, R1 = C6H13): Prepared from 4-hexyloxybenzaldehyde (2f; R = C6H13) (0.52 g) to give (5f; R = C10H21, R1 = C6H13); light yellow solid; yield 62%; m.p. (clearing temperature) 137°C; Rf = 0.57; UV-Vis (λ/nm) (εmax/dm3 mol−1 cm−1): (242) (3250) (n–σ*), (331) (17950) (π–π*); fluorescence (λmaxexc/nm) (λmaxems/nm): (248.0) (498.1); IR (υ/cm–1): 3068 (w, υ (C–H) aromatic), 2955 (w-m, υas (C–H) of CH3 group), 2920 (s, υas (C–H) of CH2 group), 2876 (w, υs (C–H) of CH3 group), 2850 (m, υs (C–H) of CH2 group), 1622 (m, υ (C=N)), 1604, 1575 and 1508 (s, m, m, υ (C=C) aromatic), 1246 (vs, υ(C–O)); 1H NMR (δ/ppm): 0.90 (6H, t, J = 7.0 Hz, CH3), 1.30–1.51 (20H, m, CH2), 1.81 (4H, m, CH2), 3.99 (2H, t, J = 6.9 Hz, OCH2), 6.94 (4H, d, J = 8.9 Hz, aromatic protons), 7.76 (4H, d, J = 8.9 Hz, aromatic protons), 8.57 (2H, s, CH=N); 13C NMR (δ/ppm): 10.7 (CH3), 15.0 (CH3), 22.5 (CH2), 26.5 (CH2), 29.4 (CH2), 29.5 (CH2), 29.9 (CH2), 31.9 (CH2), 67.9 (OCH2), 68.2 (OCH2), 114. 8 (aromatic C–H), 126.7 (aromatic C), 130.3 (aromatic C–H), 161.1 (CH=N), 161.6 (aromatic C–O); MS m/z: 464. Anal. calcd for C30H44N2O2: C, 77.54; H, 9.54; N, 6.03; found: C, 77.33; H, 9.42; N, 5.84%.
[1-(4-Heptyloxy)-2-(4’-decyloxy)benzylidene]hydrazine (
5g; R = C10H21, R1 = C7H15): Prepared from 4-heptyloxybenzaldehyde (2g; R = C7H15) (0.55 g) to give (5g; R = C10H21, R1 = C7H15); light yellow solid; yield 63%; m.p. (clearing temperature) 134 °C; Rf = 0.60; UV-Vis (λ/nm) (εmax/dm3 mol−1 cm−1): (241) (3340) (n–σ*), (332) (13670) (π–π*); fluorescence (λmaxexc/nm) (λmaxems/nm): (248.0) (496.9); IR (υ/cm−1): 3074 (w, υ (C–H) aromatic), 2955 (w-m, υas (C–H) of CH3 group), 2920 (s, υas (C–H) of CH2 group), 2873 (w, υs (C–H) of CH3 group), 2850 (m, υs (C–H) of CH2 group), 1622 (m, υ (C=N)), 1604, 1575 and 1508 (s, m, m, υ (C=C) aromatic), 1249 (vs, υ (C–O)); 1H NMR (δ/ppm): 0.90 (6H, t, J = 7.1 Hz, CH3), 1.30–1.52 (22H, m, CH2), 1.81 (4H, m, CH2), 4.00 (4H, t, J = 6.8 Hz, OCH2), 6.94 (4H, d, J = 8.8 Hz, aromatic protons), 7.74 (4H, d, J = 8.8 Hz, aromatic protons), 8.58 (2H, s, CH=N); 13C NMR (δ/ppm): 10.3 (CH3), 14.6 (CH3), 22.3 (CH2), 26.7 (CH2), 29.3 (CH2), 29.5 (CH2), 29.8 (CH2), 31.9 (CH2), 67.9 (OCH2), 68.3 (OCH2), 114.8 (aromatic C–H), 126.7 (aromatic C), 130.3 (aromatic C–H), 161.1 (CH=N), 161.7 (aromatic C–O); MS m/z: 478. Anal. calcd for C31H46N2O2: C, 77.78; H, 9.69; N, 5.85; found: C, 77.64; H, 9.61; N 5.73%.
[1-(4-Octyloxy)-2-(4’-decyloxy)benzylidene]hydrazine (
5h; R = C10H21, R1 = C8H17): Prepared from 4-octyloxybenzaldehyde (2h; R = C8H17) (0.59 g) to give (5h; R = C10H21, R1 = C8H17); light yellow solid; yield 62%; m.p. (clearing temperature) 135°C; Rf = 0.62; UV-Vis (λ/nm) (εmax/dm3 mol−1 cm−1): (241) (3350) (n–σ*), (331) (13790) (π–π*); fluorescence (λmaxexc/nm) (λmaxems/nm): (248.0) (498.0); IR (υ/cm–1): 3057 (w, υ (C–H) aromatic), 2955 (w-m, υas (C–H) of CH3 group), 2918 (s, υas (C–H) of CH2 group), 2878 (w, υs (C–H) of CH3 group), 2850 (m, υs (C–H) of CH2 group), 1622 (m, υ (C=N)), 1604, 1573 and 1506 (s, m, m, υ (C=C) aromatic), 1247 (vs, υ (C–O)); 1H NMR (δ/ppm): 0.88 (6H, t, J = 7.0Hz, CH3), 1.25–1.48 (24H, m, CH2), 1.79 (4H, m, CH2); 4.01 (4H, t, J = 6.8 Hz, OCH2), 6.93 (4H, d, J = 8.8 Hz, aromatic protons), 7.74 (4H, d, J = 8.8 Hz, aromatic protons), 8.57 (2H, s, CH=N); 13C NMR (δ/ppm): 10.3 )CH3), 14.9 )CH3), 22.4 (CH2), 26.6 (CH2), 29.35 (CH2), 29.52 (CH2), 29.90 (CH2), 31.92 (CH2), 67.91 (OCH2), 68.38 (OCH2), 114.72 (aromatic C–H), 126.7 (aromatic C), 130.2 (aromatic C–H), 161.1 (CH=N), 161.6 (aromatic C–O); MS m/z: 492. Anal. calcd for C32H48N2O2: C, 78.00; H, 9.82; N, 5.69; found: C, 77.88; H, 9.73; N, 5.62%.
[1-(4-Nonyloxy)-2-(4’-decyloxy)benzylidene]hydrazine (
5i; R = C10H21, R1 = C9H19): Prepared from 4-nonyloxybenzaldehyde (2i; R = C9H19) (0.62 g) to give (5i; R = C10H21, R1 = C9H19); light yellow solid; yield 64%, m.p. (clearing temperature) 132 °C; Rf = 0.64; UV-Vis (λ/nm) (εmax/dm3 mol−1 cm−1): (242) (3870) (n–σ*), (332) (14250) (π–π*); fluorescence (λmaxexc/nm) (λmaxems/nm): (248.0) (497.0); IR (υ/cm–1): 3052 (w, υ (C–H) aromatic), 2956 (w-m, υas (C–H) of CH3 group), 2918 (s, υas (C–H) of CH2 group), 2872 (w, υs (C–H) of CH3 group), 2848 (m, υs (C–H) of CH2 group), 1622 (m, υ (C=N)), 1604, 1575 and 1508 (s, m, m, υ (C=C) aromatic), 1247 (vs, υ (C–O)); 1H NMR (δ/ppm): 0.87 (6H, t, J = 7.0Hz, CH3), 1.25–1.48 (26H, m, CH2), 1.78 (4H, m, CH2), 3.99 (4H, t, J = 6.9 Hz, OCH2), 6.925 (4H, d, J = 8.5 Hz, aromatic protons), 7.74 (4H, d, J = 8.5 Hz, aromatic protons), 8.58 (2H, s, CH=N); 13C NMR (δ/ppm): 10.3 (CH3), 15.0 (CH3), 22.4 (CH2), 26.7 (CH2), 29.4 (CH2), 29.5 (CH2), 29.9 (CH2), 31.9 (CH2), 67.9 (OCH2), 68.4 (OCH2), 114.7 (aromatic C–H), 126.7 (aromatic C), 130.3 (aromatic C–H), 161.1 (CH=N), 161.6 (aromatic C–O); MS m/z: 506. Anal. calcd for C33H50N2O2: C, 78.21; H, 9.94; N, 5.53; found: C, 78.03; H, 9.72; N, 5.44%.
[1-(4-Dodecyloxy)-2-(4′-decyloxy)benzylidene]hydrazine (
5j; R = C10H21, R1 = C12H25): Prepared from 4-decyloxybenzaldehyde (2j; R = C12H25) (0.73 g) to give (5j; R = C10H21, R1 = C12H25); light yellow solid; yield 64%; m.p. (clearing temperature) 124°C; Rf = 0.68; UV-Vis (λ/nm) (εmax/dm3 mol−1 cm−1): (240) (7100) (n–σ*), (333) (26070) (π–π*); fluorescence (λmaxexc/nm) (λmaxems/nm): (248.0) (499.1); IR (υ/cm-1): 3057 (w, υ (C–H) aromatic), 2950 (w-m, υas (C–H) of CH3 group), 2918 (s, υas (C–H) of CH2 group), 2878 (w, υs (C–H) of CH3 group), 2855 (s, υs (C–H) of CH2 group), 1622 (m, υ(C=N)), 1600, 1573 and 1508 (s, m, m, υ (C=C) aromatic), 1247 (vs, υ (C–O)); 1H NMR (δ/ppm): 0.91 (6H, t, J = 7.0 Hz, CH3), 1.03 (3H, t, J = 7.0 Hz, CH3), 1.31–1.52 (32H, m, CH2), 1.80 (4H, m, CH2); 4.00 (4H, t, J = 6.8 Hz, OCH2), 6.96 (4H, d, J = 8.8 Hz, aromatic protons), 7.76 (4H, d, J = 8.8 Hz, aromatic protons), 8.59 (2H, s, CH=N); 13C NMR (δ/ppm): 10.3 (CH3), 14.9 (CH3), 22.3 (CH2), 26.7 (CH2), 29.3 (CH2), 29.5 (CH2), 29.8 (CH2), 31.9 (CH2), 67.9 (OCH2), 68.3 (OCH2), 114.7 (aromatic C–H), 126.7 (aromatic C), 130.3 (aromatic C–H), 161.1 (CH=N), 161.6 (aromatic C–O); MS m/z: 548. Anal. calcd for C36H56N2O2: C, 78.78; H, 10.28; N, 5.10; found: C, 78.61; H, 10.20; N, 5.03%.
[1-(4-Hexadecyloxy)-2-(4’-decyloxy)benzylidene]hydrazine (
5k; R = C10H21, R1 = C16H33): Prepared from 4-hexadecyloxybenzaldehyde (2k; R = C16H33) (0.87 g) to give (5k; R = C10H21, R1 = C16H33); light yellow solid; yield 62%; m.p. (clearing temperature) 120°C; Rf = 0.71; UV-Vis (λ/nm) (εmax/dm3 mol−1 cm−1): (241) (4080) (n–σ*), (333) (17740) (π–π*); fluorescence (λmaxexc/nm) (λmaxems/nm): (248.0) (498.1); IR (υ/cm−1): 3066 (w, υ (C–H) aromatic), 2955 (w-m, υas (C–H) of CH3 group), 2918 (s, υas(C–H) of CH2 group), 2871 (w, υs (C–H) of CH3 group), 2850 (m, υs (C–H) of CH2 group), 1624 (m, υ (C=N)), 1606, 1575 and 1510 (s, m, m, υ (C=C) aromatic), 1251 (vs, υ (C–O)); 1H NMR (δ/ppm): 0.87 (6H, t, J = 7.0 Hz, CH3), 1.21–1.45 (40 H, m, CH2), 1.79 (4H, m, CH2), 3.99 (4H, t, J = 6.8 Hz, OCH2), 6.93 (4H, d, J = 8.8 Hz, aromatic protons), 7.75 (4H, d, J = 8.8 Hz, aromatic protons), 8.59 (2H, s, CH=N); 13C NMR (δ/ppm): 10.4 (CH3), 14.8 (CH3), 22.4 (CH2), 26.7 (CH2), 29.3 (CH2), 29.6 (CH2), 30.0 (CH2), 31.9 (CH2), 67.9 (OCH2), 68.3 (OCH2), 114.7 (aromatic C–H), 126.7 (aromatic C), 130.3 (aromatic C–H), 161.1 (CH=N), 161.6 (aromatic C–O); MS m/z: 604. Anal. calcd for C40H64N2O2: C, 79.42; H, 10.66; N, 4.63; found: C, 79.36; H, 10.57; N, 4.55%.
[1-(4-Octadecyloxy)-2-(4’-decyloxy)benzylidene]hydrazine (
5l; R = C10H21, R1 = C18H35): Prepared from 4-octadecyloxybenzaldehyde (2l; R = C18H35) (0.94 g) to give (5l; R = C10H21, R1 = C18H35); light yellow solid; yield 64%, m.p. (clearing temperature) 119°C; Rf = 0.73; UV-Vis (λ/nm) (εmax/dm3 mol−1 cm−1): (244) (3980) (n–σ*), (334) (17200) (π–π*); fluorescence (λmaxexc/nm) (λmaxems/nm): (248.0) (498.3); IR (υ/cm–1): 3070 (w, υ (C–H) aromatic), 2955 (w-m, υas (C–H) of CH3 group), 2918 (s, υas (C–H) of CH2 group), 2878 (w, υs (C–H) of CH3 group), 2848 (s, υs (C–H) of CH2 group), 1624 (m, υ (C=N)), 1606, 1575 and 1508 (s, m, m, υ (C=C) aromatic), 1251 (vs, υ (C–O)); 1H NMR (δ/ppm): 0.87 (6H, t, J = 7.0 Hz, CH3), 1.32–1.53 (44H, m, CH2), 1.80 (4H, m, CH2), 3.98 (4H, t, J = 6.9 Hz, OCH2), 6.93 (4H, d, J = 8.8 Hz, aromatic protons), 7.76 (4H, d, J = 8.8 Hz aromatic protons), 8.57 (2H, s, CH=N); 13C NMR (δ/ppm): 10.3 (CH3), 14.9 (CH3), 22.4 (CH2), 26.7 (CH2), 29.3 (CH2), 29.6 (CH2), 29.9 (CH2), 31.9 (CH2), 67.9 (OCH2), 68.3 (OCH2), 114.7 (aromatic C–H), 126.8 (aromatic C), 130.3 (aromatic C–H), 161.1 (CH=N), 161.5 (aromatic C–O); MS m/z: 633. Anal. calcd for C42H68N2O2: C, 79.69; H, 10.83; N, 4.43; found: C, 79.60; H, 10.68; N, 4.29%.