Abstract
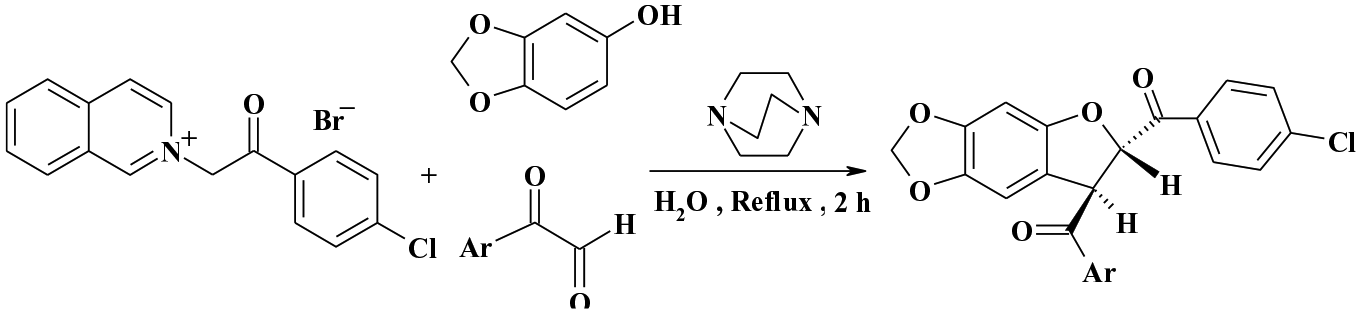
A three-component one-pot condensation of 2-[2-(4-chlorophenyl)-2-oxoethyl)]isoquinolinium bromide with benzo[1,3]dioxol-5-ol and an arylglyoxal in the presence of catalytic amounts of 1,4-diaza-bicyclo[2.2.2]octane in refluxing water gave a series of seven trans-(4-chlorophenyl)-7-aryl-6,7-dihydro[1,3]dioxolo[4,5-f][1]benzofuran-6-yl)methanones in excellent yield and in a short time.
Keywords
Introduction
Multicomponent reactions (MCRs), by virtue of their convergence, productivity, facile execution and generally high yields, have attracted much attention in the area of combinatorial chemistry.1–3 Many important heterocyclic syntheses are MCRs.
Dihydrofurans are important hetrocycles commonly found in a large variety of naturally occurring substances. 4 The development of new and efficient methods for their synthesis remains an area of current interest, and a whole series of new synthetic methods have appeared in the literature.5–8 The synthesis of dihydrofurans has been reported by the condensation of an aryl aldehyde, a cyclic 1,3-diketone, 2-bromo-1-phenylethanone or 4-nitrobenzyl bromide and pyridine in the presence of 10 mol% sodium hydroxide in refluxing aqueous solution. 9 Recently, organic reactions in aqueous media have attracted a great deal of attention 10 as a result of increasing interest in the concepts of sustainability and green chemistry. 11 Also, the latter synthesis used a procedure first described by Wang et al. 12 in which they synthesised a series of dihydrofurans via the pyridine-catalysed reaction of phenacyl bromides with a series of aromatic aldehydes and 4-hydroxycoumarin in the presence of catalytic amounts of NaOH or Et3N. Although many of the reported methods are effective, some of them suffer from disadvantages such as harsh reaction conditions, use of hazardous solvents, long reaction times, complex working and purification procedures, high catalyst loadings and moderate yields. Therefore, the development of a simple, mild and efficient method is still needed. In this work, we used 1,4-diaza-bicyclo[2.2.2]octane (DABCO) as an efficient catalyst to overcome these limitations.
DABCO has been used in many organic preparations as a good solid catalyst.
13
DABCO has received considerable attention as an inexpensive, eco-friendly, highly reactive, easy to handle and non-toxic basic catalyst for various organic transformations, affording the corresponding products in excellent yields with high selectivity. The reactions are environmentally friendly and the catalyst can be recycled in some cases.
14
As a part of our current studies on the development of new routes in organic synthesis,15,16 in this study, we have used the procedure of Wang et al.
12
and Khan et al.
9
to synthesise another class of dihydrofurans via a three-component condensation reaction of 2-[2-(4-chlorophenyl)-2-oxoethyl)]isoquinolinium bromide

A three-component condensation reaction of 2-[2-(4-chlorophenyl)-2-oxoethyl)]isoquinolinium bromide
Results and discussion
One of the starting materials for our synthesis, compound
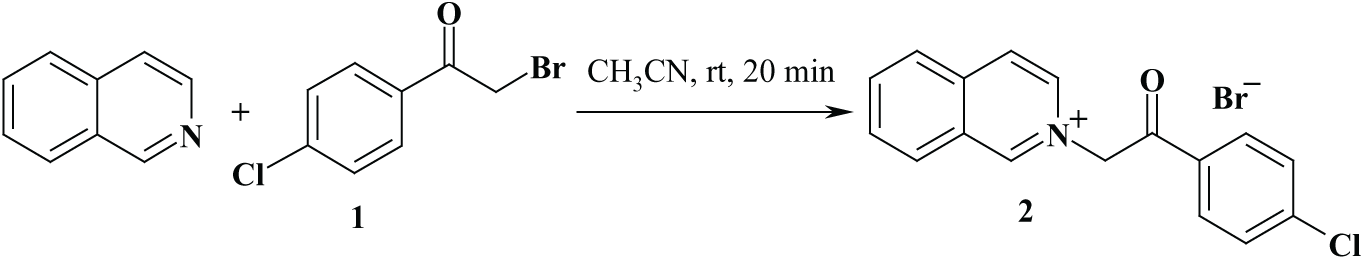
Synthesis of 2-[2-(4-chlorophenyl)-2-oxoethyl)]isoquinolinium bromide (
A one-pot three-component reaction of 2-[2-(4-chlorophenyl)-2-oxoethyl)]isoquinolinium bromide
Yields of a series of trans-(4-chlorophenyl)-7-aryl-6,7-dihydro[1,3]dioxolo[4,5-f][1]benzofuran-6-yl)methanones (

Yields refer to the pure isolated products.
The structures of compounds
A proposed mechanism for this reaction is shown in Scheme 3. The formation of the product can be explained as follows. The 2-[2-(4-chlorophenyl)-2-oxoethyl)]isoquinolinium bromide

Suggested mechanism for formation of compound
Experimental
Melting points were determined with an electrothermal 9100 apparatus. Elemental analyses were performed using a Heraeus CHN-O-Rapid analyser. Mass spectra were recorded on a FINNIGAN-MAT 8430 mass spectrometer operating at an ionisation potential of 70 eV. IR spectra were recorded on a Shimadzu IR-470 spectrometer. 1H and 13C NMR spectra were recorded on a Bruker DRX-400 Avance spectrometer in CDCl3 using tetramethylsilane (TMS) as internal standard. 2-[2-(4-chlorophenyl)-2-oxoethyl)]isoquinolinium bromide
General procedure
To a magnetically stirred solution of 2-[2-(4-chlorophenyl)-2-oxoethyl)]isoquinolinium bromide
trans-(4-Chlorophenyl)-7-(4-nitrobenzoyl)-6,7-dihydro[1,3]dioxolo[4,5-f][1]benzofuran-6-yl)methanone (
trans-(4-Chlorophenyl)-7-(4-fluorobenzoyl)-6,7-dihydro[1,3]dioxolo[4,5-f][1]benzofuran-6-yl)methanone (
trans-(4-Chlorophenyl)-7-(2-bromobenzoyl)-6,7-dihydro[1,3]dioxolo[4,5-f][1]benzofuran-6-yl)methanone (
trans-(4-Chlorophenyl)-7-(4-chlorobenzoyl)-6,7-dihydro[1,3]dioxolo[4,5-f][1]benzofuran-6-yl)methanone (
trans-(4-Chlorophenyl)-7-(2-chloroenzoyl)-6,7-dihydro[1,3]dioxolo[4,5-f][1]benzofuran-6-yl)methanone (
trans-(4-Chlorophenyl)-7-(4-hydroxybenzoyl)-6,7-dihydro[1,3]dioxolo[4,5-f][1]benzofuran-6-yl)methanone (
trans-(4-Chlorophenyl)-7-(4-bromophenyl)-6,7-dihydro[1,3]dioxolo[4,5-f][1]benzofuran-6-yl)methanone (
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
