Abstract
A series of 12 novel methyl- and halogen-substituted bischalcones containing an azo linkage was synthesized by a Claisen–Schmidt condensation between (E)-1-{4-[(4-acetyl-2-hydroxyphenyl)diazenyl]phenyl}ethanone and various araldehydes using microwave irradiation and conventional procedures and the results were compared.
Introduction
Chalcone fragments feature widely in both natural and synthetic products. They are significant intermediates in organic synthesis.
1
Bischalcones are natural products found in the Anacardiacea family. Both biflavonoids and bischalcones are plentiful in the Rhus genus. Rhuschalcones II–VI have been separated from the root bark of Rhus pyroides, and synthetic derivatives have been shown to have vigorous antiplasmodial and moderate antiproliferative and antiprotozoal activities. In addition, Rhuschalcone VI (

Natural and synthetic bischalcones.
Some natural products, pharmaceuticals, organic functional materials, dyes and significant synthetic intermediates have azo groups in their structures. Some azo analogues demonstrate miscellaneous biological activities, making them of interest to the pharmaceutical industry. It has been reported that they are effective as anti-proliferation agents in lung cancer and as antibacterials and carbonic anhydrase inhibitors.3,9 Following our recent report of the synthesis of an azo-linked bischalcone with a sulfonamide 10 and a quinoline 11 and considering the pharmacological importance of both bischalcones and azo compounds, we set out to synthesize a new series of bischalcones possessing different substitution patterns on an azo framework. Here, we describe their synthesis using microwave (MW) irradiation and conventional procedures and compare the results.
Results and discussion
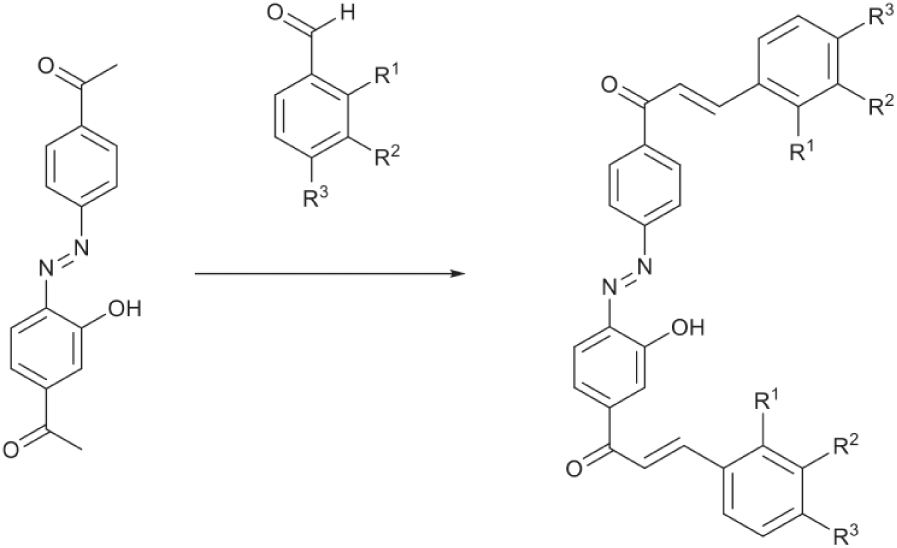
Our starting point for the synthesis of a series of azo-bridged bischalcones was the preparation of the azo-bridged bisacetophenone, (E)-1-{4-[(4-acetyl-2-hydroxyphenyl)diazenyl]phenyl}ethanone (

Synthesis of azo-bridged bisacetophenone (3).

Synthesis of new bischalcones (5a-l).
We found that a shorter reaction time and a lower or higher MW power gave rise to lower conversion rates, while increasing the reaction time or MW power resulted in fragmentation of the target product, as revealed by TLC analysis. We then used these optimal MW conditions to synthesize 11 other azo-bridged bischalcones (
Yields of azo-bridged chalcones

MW: microwave.
Reaction conditions: To a stirred mixture of compound
Reaction conditions: To a stirred mixture of compound
Isolated yields.
An important feature of the structural characterization of chalcones
Some other selected spectral data of other compounds are discussed in the following. For compound
1H and 13C NMR data of all chalcones
Conclusion
In conclusion, a novel series of bischalcones linked by an azo group was synthesized using both a conventional and a MW method. The structures of all compounds were confirmed by FTIR, NMR and MS spectral data and elemental analysis.
Experimental
The compounds 4-aminoacetophenone, hydrochloric acid, NaNO2, 3-hydroxyacetophenone, NaOH, sodium acetate and all aldehydes were obtained from Sigma or Merck and used as received without further purification. All solvents were purchased from Merck and freshly distilled. NMR spectra were obtained on a Varian Mercury spectrometer (1H NMR at 200 Hz and 13C NMR at 50 Hz) in CDCl3 using tetramethylsilane (TMS) as internal standard. Chemical shifts (δ) are given in ppm and coupling constants (J) are given in hertz (Hz). Mass spectrometric analysis was carried out on an Agilent 1260 Infinity Series quadrupole time-of-flight (Q-TOF) liquid chromatography–mass spectrometry (LC/MS; electrospray ionization (ESI)/MS; for compound
Synthesis of (E)-1-{4-[(4-acetyl-2-hydroxyphenyl)diazenyl]phenyl}ethanone (3 )
4-Aminoacetophenone (2 g, 0.0148 mol) was dissolved in 5 × 10–3 L of concentrated HCl. The solution was cooled down in an ice–salt bath, and then, a cold solution of NaNO2 (1 g, 0.0148 mol) in 1 × 10–2 L of water was slowly added. The reaction mixture that resulted was stirred for 3 h at 0–5 °C. The resulting diazonium salt was cooled in an ice–salt bath. 3-Hydroxyacetophenone (2 g, 0.0148 mol) was dissolved in a dilute NaOH solution and cooled in an ice–salt bath, and then, the cold diazonium solution was added to this cooled solution dropwise at 0–5 °C. After stirring the reaction mixture for 4 h, the pH of the reaction mixture was adjusted to 4–5 by the addition of a saturated sodium acetate solution. Subsequently, it was stirred for 2 h. The precipitate was filtered off, dried and recrystallized from EtOH/H2O (1:1): yield (4.10 g), m.p. 206–207 °C (lit. 3 206–207 °C).
Conventional method for synthesis of chalcones 5a–l
(E)-1-{4-[(4-Acetyl-2-hydroxyphenyl)diazenyl]phenyl}ethanone (
MW method for synthesis of chalcones 5a–l
(E)-1-{4-[(4-Acetyl-2-hydroxyphenyl)diazenyl]phenyl}ethanone (
(E)-1-{3-Hydroxy-4-[(E)-{4-[(E)-3-o-tolylacryloyl]phenyl}diazenyl]phenyl}-3-o-tolylprop-2-en-1-one (
(E)-1-{3-Hydroxy-4-[(E)-{4-[(E)-3-m-tolylacryloyl]phenyl}diazenyl]phenyl}-3-m-tolylprop-2-en-1-one (
(E)-1-{3-Hydroxy-4-[(E)-{4-[(E)-3-p-tolylacryloyl]phenyl}diazenyl]phenyl}-3-p-tolylprop-2-en-1-one (
(E)-3-(2-Fluorophenyl)-1-{4-[(E)-{4-[(E)-3-(2-fluorophenyl)acryloyl]-2-hydroxyphenyl}diazenyl]phenyl}prop-2-en-1-one (
(E)-3-(3-Fluorophenyl)-1-{4-[(E)-{4-[(E)-3-(3-fluorophenyl)acryloyl]-2-hydroxyphenyl}diazenyl]phenyl}prop-2-en-1-one (
(E)-3-(4-Fluorophenyl)-1-{4-[(E)-{4-[(E)-3-(4-fluorophenyl)acryloyl]-2-hydroxyphenyl}diazenyl]phenyl}prop-2-en-1-one (
(E)-3-(2-Chlorophenyl)-1-{4-[(E)-{4-[(E)-3-(2-chlorophenyl)acryloyl]-2-hydroxyphenyl}diazenyl]phenyl}prop-2-en-1-one (
(E)-3-(3-Chlorophenyl)-1-{4-[(E)-{4-[(E)-3-(3-chlorophenyl)acryloyl]-2-hydroxyphenyl}diazenyl]phenyl}prop-2-en-1-one (
(E)-3-(2,4-Dichlorophenyl)-1-{4-[(E)-{4-[(E)-3-(2,4-dichlorophenyl)acryloyl]-2-hydroxyphenyl}diazenyl]phenyl}prop-2-en-1-one (
(E)-3-(2-Bromophenyl)-1-{4-[(E)-{4-[(E)-3-(2-bromophenyl)acryloyl]-2-hydroxyphenyl}diazenyl]phenyl}prop-2-en-1-one (
(E)-3-(3-Bromophenyl)-1-{4-[(E)-{4-[(E)-3-(3-bromophenyl)acryloyl]-2-hydroxyphenyl}diazenyl]phenyl}prop-2-en-1-one (
(E)-3-(4-Bromophenyl)-1-{4-[(E)-{4-[(E)-3-(4-bromophenyl)acryloyl]-2-hydroxyphenyl}diazenyl]phenyl}prop-2-en-1-one (
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work received financial support from The Research Fund of Karadeniz Technical University (BAP, Project No. 2012/9181).
