Abstract
Background:
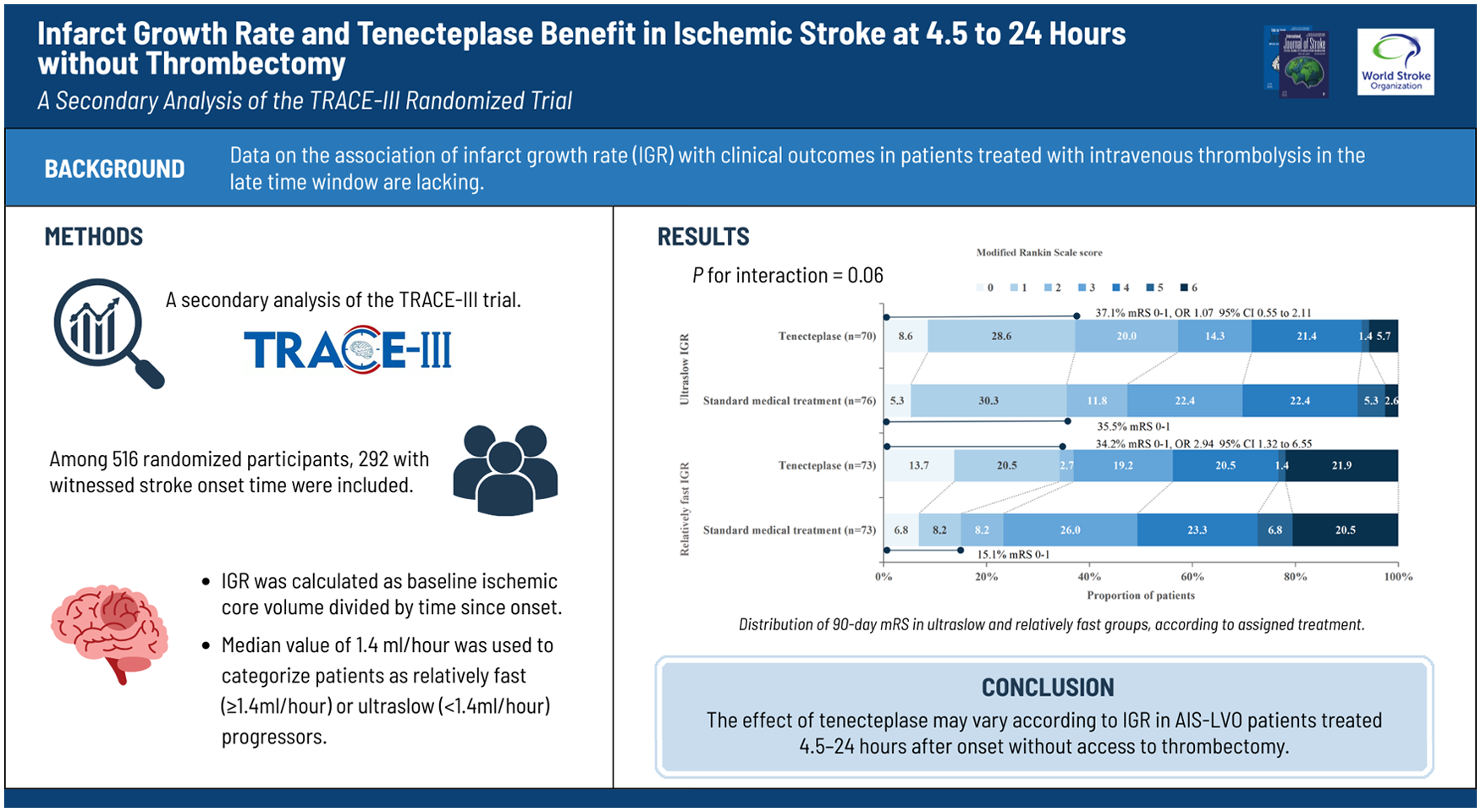
We aimed to explore whether the benefits of tenecteplase within 4.5 to 24 h would be modified by the infarct growth rate (IGR).
Methods:
This study is a secondary analysis of the Tenecteplase Reperfusion Therapy in Acute Ischemic Cerebrovascular Events–III (TRACE-III) trial, a Phase 3, multicenter, prospective, open-label, randomized, blinded-outcome-assessment trial. In the TRACE-III trial, patients with large-vessel occlusion at 4.5 to 24 h without thrombectomy were enrolled at 58 centers in China and randomly assigned to receive 0.25 mg/kg tenecteplase or standard medical treatment. Of these, patients with witnessed stroke onset time were analyzed. The IGR was calculated as baseline ischemic core volume divided by time since onset. The primary outcome was the modified Rankin Scale (mRS) 0-1 at 90 days. The treatment effect of tenecteplase versus standard medical treatment was assessed in two groups based on the median IGR cut point. A multiplicative interaction term for IGR*treatment was used to test for effect modification.
Results:
In 292 eligible patients, the median IGR was 1.4 ml/h. A total of 146 patients with IGR < 1.4 ml/h were classified as ultraslow IGR (Tenecteplase, 70; Standard medical treatment, 76), and 146 patients with IGR ⩾ 1.4 ml/h were classified as slow IGR (Tenecteplase, 73; Standard medical treatment, 73). The rate of no disability (mRS ⩽ 1) was significantly increased with tenecteplase in slow progressors (34.2% vs 15.1%; OR = 2.94, 95% CI 1.32 to 6.55; P = 0.009). The functional status was similar in ultraslow progressors (mRS 0-1: 37.1% vs 35.5%; OR = 1.07, 95% CI 0.55 to 2.11; P = 0.84). A P value for the interaction between IGR and treatment was 0.06. The incidence of sICH and mortality did not differ significantly between the two treatment regimens in the IGR groups.
Conclusion:
Patients with late-window ischemic stroke who are not eligible for endovascular thrombectomy may derive greater benefit from tenecteplase compared with standard medical treatment among those with relatively faster infarct growth, although this finding should be considered exploratory.
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
