Abstract
Introduction:
As a marker of chronic cerebral small vessel disease, leukoaraiosis (LA) was reported to impact the recruitment of collaterals in acute ischemic stroke (AIS). We intended to explore the impact of LA on the infarct growth rate (IGR) and clinical outcome by impaired collateral development in AIS patients with large vessel occlusion (LVO) who underwent endovascular thrombectomy (EVT).
Patients and methods:
Two hundred thirty-six AIS patients who underwent EVT were retrospectively reviewed. The severity of LA was graded using the Fazekas scale with non-contrast CT. IGR was calculated by the acute core volume on CT perfusion divided by the time from stroke onset to imaging. The collateral status after LVO was assessed using the ASITN/SIR collateral scale. The clinical outcomes after EVT were evaluated using a modified Rankin Scale (mRS). The Alberta stroke program early CT score (ASPECTS), the National Institutes of Health Stroke Scale (NIHSS) score at admission, and the modified treatment in cerebral infarction (mTICI) score after EVT were also included. Correlations between those factors were analyzed.
Results:
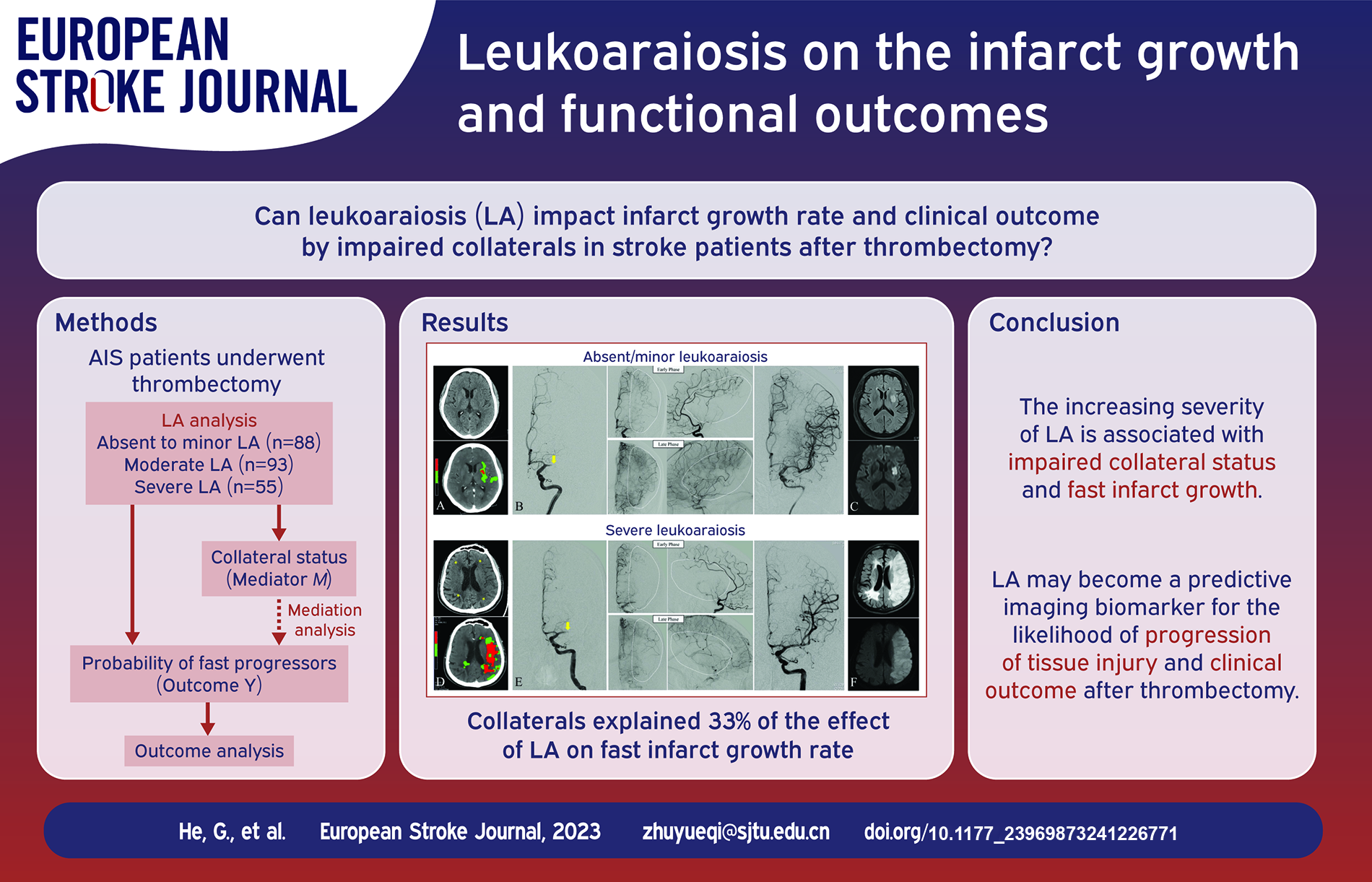
Patients with severe LA had significantly larger core volume on CTP (p = 0.022) and lower collateral grade (p < 0.001). Faster IGR was significantly associated with higher LA severity (adjusted odds ratio [aOR]: 1.53; 95% CI: 1.02–2.33; p = 0.046), higher NIHSS (aOR: 1.04; 95% CI: 1.00–1.09; p = 0.032) and impaired collaterals (aOR: 2.26; 95% CI: 1.27–4.03; p = 0.005). In mediation analysis, collaterals explained 33% of the effect of LA on fast IGR. There was correlation between the severity of LA and mRS (p = 0.007).
Discussion and Conclusion:
The increasing severity of LA is associated with impaired collateral status and fast infarct growth. These findings suggest that LA may become a predictive imaging biomarker for the likelihood of progression of tissue injury and clinical outcome after EVT in acute large vessel occlusion stroke.
Introduction
Leukoaraiosis (LA) is a prevalent neuroimaging feature of cerebral small vessel disease in the older population. 1 Chronic hypoperfusion and ischemia from lipohyalinosis, arteriolosclerosis, and fibrinoid necrosis are considered the pathogenesis of LA, leading to microvascular wall damage, luminal narrowing, and occlusion. 2 In acute ischemic stroke (AIS), patients with severe LA were found to be associated with more frequent hemorrhagic transformation and less favorable outcomes after thrombolysis.3,4 Recently, endovascular thrombectomy (EVT) has been shown to significantly increase the reperfusion rate and improve the functional outcomes of patients with AIS due to large vessel occlusive stroke.5,6 Given the accepted adverse impact of LA on the intrinsic capacity of the brain to withstand ischemia, patients with severe LA would have more rapid progression of cerebral infarction.
Collateral status plays an important role in the rate of ischemic core growth for patients with large vessel occlusions (LVO).7,8 Patients with robust leptomeningeal collaterals that last for long periods are more likely to have slow progression, while those with impaired collaterals often have an accelerated ischemic lesion evolution. 9 As LA is a reflection of various metabolic and senescent impacts on penetrating arteries of the white matter, dysfunction of the white matter arterioles would also affect the arterioles of the leptomeningeal collateral system.10,11 In the event of acute large artery occlusion, it is conceivable that compromised tissue perfusion at the macro- and micro-vascular level can result in adverse outcomes, with the downstream tissue at increased risk of faster infarction growth. Several studies have connected LA burden with the extent of leptomeningeal collaterals10,12,13; however, the relationship between LA and ischemic lesion evolution on pre-EVT imaging has not been clarified. Therefore, our aim was to investigate whether LA severity correlated with collateral development, infarct growth rate and functional outcome in AIS patients with large vessel occlusion treated with EVT.
Methods
Study design and population
The institutional review board approved the study. Informed consent was provided by patients or their family members. Between January 2020 and June 2022, consecutive patients (n = 327) with acute LVO stroke involving the internal carotid artery (ICA) terminus or proximal middle cerebral artery (MCA), who underwent non-contrast CT (NCCT) and CT perfusion (CTP) within 16 h of symptom onset, were registered. All enrolled patients had a National Institutes of Health Stroke Scale (NIHSS) score of at least 6 at admission and a score of 0 or 1 on the modified Rankin scale (mRS) before the onset of stroke. Patients with wake-up stroke, unknown onset time, and severe motion artifact on CT images were excluded. All patients received mechanical thrombectomy treatment and successful reperfusion was defined as mTICI 2b or 3. A good functional outcome was defined as a mRS score of 2 or less at 90 days. Baseline clinical characteristics and demographic information were extracted from the medical records.
Imaging protocol
NCCT and CTP images were obtained using a 640-slice uCT960+ scanner (United Imaging Healthcare, Shanghai, China). The NCCT scan was performed from the foramen magnum to the vertex to exclude cerebral hemorrhage and assess the Alberta Stroke Program Early CT Score (ASPECTS) with the following parameters: rotation speed, 0.5 s; pitch, 0.8938:1; detector configuration, 320 × 0.5 mm; collimation, 40 mm; tube voltage 120 kV; and tube current 300 mAs.
CTP was used to evaluate the ischemic core and hypoperfusion volume, with the scan parameters as follows: tube voltage 100 kV, FOV 300 mm × 300 mm, 160 mm coverage of the brain, matrix 512 × 512, and rotation time 0.5 s. Patients were placed in the supine position. Fifty milliliter of contrast medium (Ultravist 370; Bayer HealthCare, Berlin, Germany) and 30 mL of normal saline were injected via the cubital vein using a double-piston syringe at an injection rate of 6 mL/s through an 18 G intravenous indwelling needle. CTP acquisition was performed 5 s after injection of the contrast, and the scan duration was 55.5 s. The tube current settings were as follows: from 5 to 5.5 s after injection, the tube current was 150 mAs, and the scan interval was 3 s; from 11 to 15.5 s after injection, the tube current was 75 mAs, and the scan interval was 2 s; from 17 to 27.5 s after injection, the tube current was 150 mAs, and the interval was 2 s; from 30 to 36.5 s after injection, the tube current was 75 mAs, and the scan interval was 2 s; from 40 to 60.5 s after injection, the tube current was 75 mAs, and the interval was 5 s. The acquisition data were reconstructed into images with a 5 mm slice thickness and a 5 mm interslice interval for the CTP analysis.
Imaging analysis
NCCT and CTP images were obtained using a 640-slice uCT960+ scanner (United Imaging Healthcare, Shanghai, China). Detailed imaging acquisition protocols were provided in Supplemental Materials. Leukoaraiosis was classified using the Fazekas criteria on baseline NCCT which divides the white matter into periventricular and deep white matter, and each region was given a grade from 0 to 3 depending on the size and confluence of lesions. 14 Before further analysis, we assessed the intra-rater CT-MRI agreements using 100 paired NCCT and MRI scans in a consecutive cohort of non-acute stroke patients as part of this study, with a short delay (within 1 month) between CT and MRI. Considering that the Fazekas scale on NCCT in the previous report had a limited interrater agreement, 10 we defined the Fazekas scale scores of 0–2 as absent to minor LA and scores of 3–4 and 5–6 as moderate and severe LA, respectively. Two experienced neuro-interventional radiologists (GC He and LM Wei) blinded to the clinical history interpreted the CT and MRI images. In cases of disagreement, the score was adjudicated by a third neuro-interventional radiologist (YQ Zhu).
Collateral status was evaluated according to the American Society of Interventional and Therapeutic Neuroradiology/Society of Interventional Radiology (ASITN/SIR) collateral scale based on digital subtraction angiography (DSA), which includes the following five categories: grade 0 (no collaterals visible to the ischemic site); grade 1 (slow collaterals to the periphery of the ischemic site with the persistence of some of the defect); grade 2 (rapid collaterals to the periphery of the ischemic site with the persistence of some of the defect and to only a portion of the ischemic territory); grade 3 (collaterals with slow but complete angiographic blood flow of the ischemic bed by the late venous phase); and grade 4 (complete and rapid collateral blood flow to the vascular bed in the entire ischemic territory by retrograde perfusion). 15 Impaired collaterals were defined as ASITN/SIR grade ⩽2.
The CTP-derived ischemic core and hypoperfused tissue were determined using MIStar automated software (Apollo Medical Imaging, Melbourne, Australia). MIStar uses a delay and dispersion-corrected singular value decomposition algorithm to deconvolve the tissue enhancement curve. 16 Previously validated thresholds were applied to measure the volume of the acute perfusion lesion (relative Delay Time ⩾3 s) and acute ischemic core (relative cerebral blood flow ⩽30% within the perfusion lesion). 17 The IGR was determined using the baseline CTP-derived ischemic core volume divided by the time between symptom onset and the CTP. Essentially, it assumed that core volume was zero just before symptom onset and that core volume grew in a nearly linear pattern within 24 h of stroke onset according to Lin et al. 18 The optimal IGR cutoff was identified by maximizing the sum of the sensitivity and specificity to correlate best with favorable outcome and to improve its the predictability. Patients were stratified into slow progressors if IGR< the cutoff value and fast progressors if IGR⩾ the cutoff value.
Statistical analysis
Baseline characteristics and procedural results were compared using the Mann-Whitney test, Kruskal-Wallis test, or Chi-squared test as appropriate. A receiver operating characteristic curve was used to identify the ability of IGR to differentiate patients with and without functional independence. The IGR cutoff was identified by maximizing the Euden’s J (a composite of sensitivity and specificity). To determine associations with the fast progressors (as dependent variable), we used ordinal multivariable logistic regression adjusted for potential baseline confounder variables. Mediation analysis was performed using Baron and Kenny’s template 19 and Vanderweele’s method. 20 All pathways were assessed using multivariable logistic regression analyses adjusted for the set of baseline variables. All analyses were conducted using open-source R software (version 4.0.3; R Foundation for Statistical Computing). A two-sided p-value less than 0.05 was considered to be statistically significant.
Results
A total of 236 patients (men, 138 [55.6%]) were included in the final analysis and were eligible for EVT after clinical and imaging selection. (Figure 1). The median age was 74 years (interquartile range [IQR], 65–82 years). The period of symptom onset to admission within 6 h was found in 73.7% (174 of 236) patients; it was 6–16 h in 26.3% (62 of 236) patients. The median NIHSS score at admission was 14 (IQR 9–19). The median time from symptom onset to first imaging was 3.9 h (IQR 2.0–6.4 h) and the median CTP-derived ischemic core volume was 19 mL (IQR 5–43 mL).
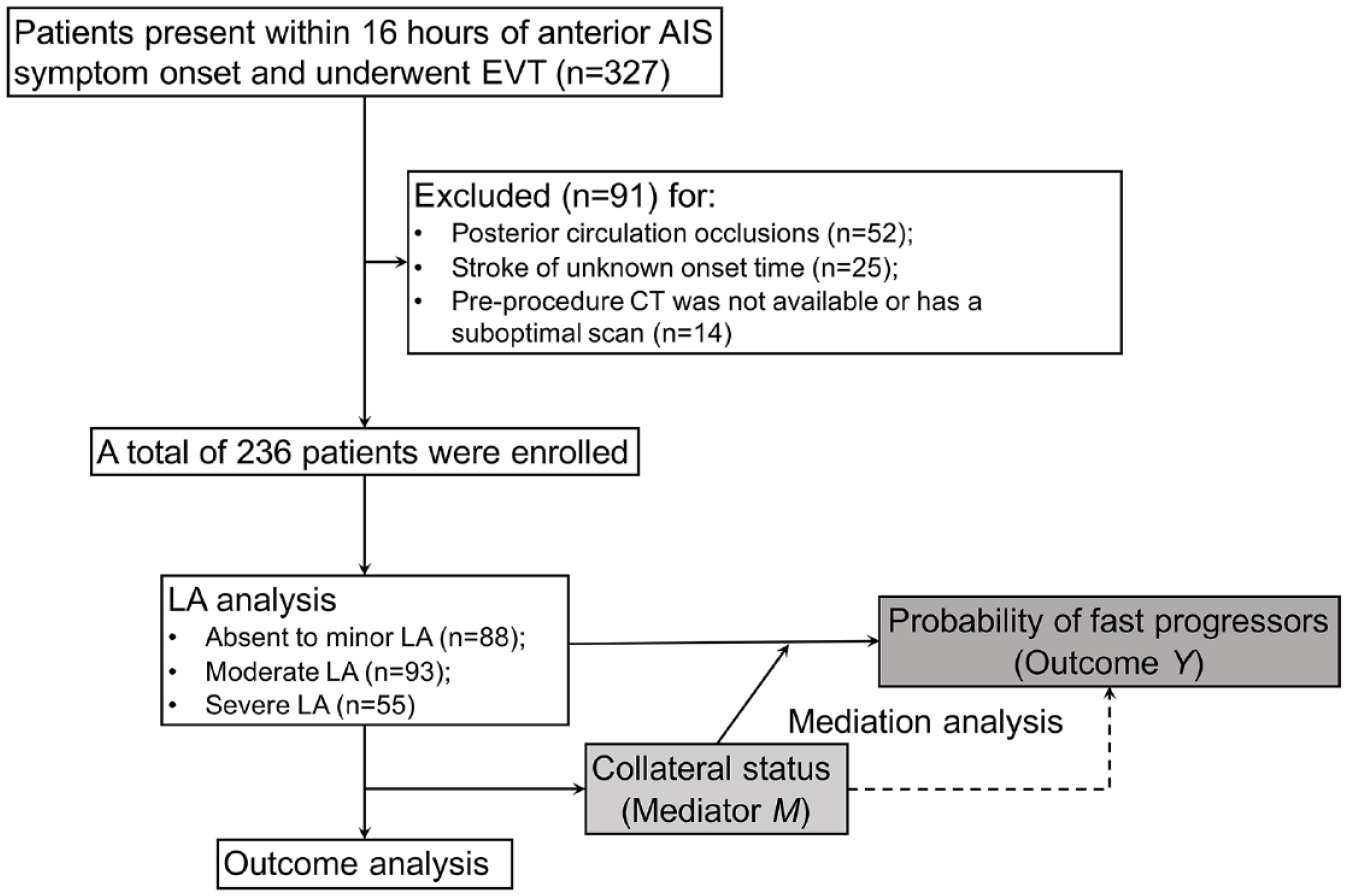
Flow diagram of the patient inclusion process in this study.
Severity distribution of LA from NCCT
Preliminary LA analysis in a consecutive cohort of non-acute stroke patients (n = 100) indicated substantial agreement of weighted κ for the total Fazekas score between CT and MRI (0.71 for rater 1 with 95% CI: 0.65–0.77 and 0.66 for rater 2 with 95% CI: 0.57–0.75). (Figure 2) On NCCT, when a three-level scale based on the Fazekas score was applied, absent to minor LA was found in 37.3% (88 of 236) patients, moderate LA was found in 39.4% (93 of 236) patients, and severe LA was found in 23.3% (55 of 236) patients. The weighted κ for LA analysis was 0.73 (95% CI: 0.67–0.78), showing a substantial interobserver agreement.
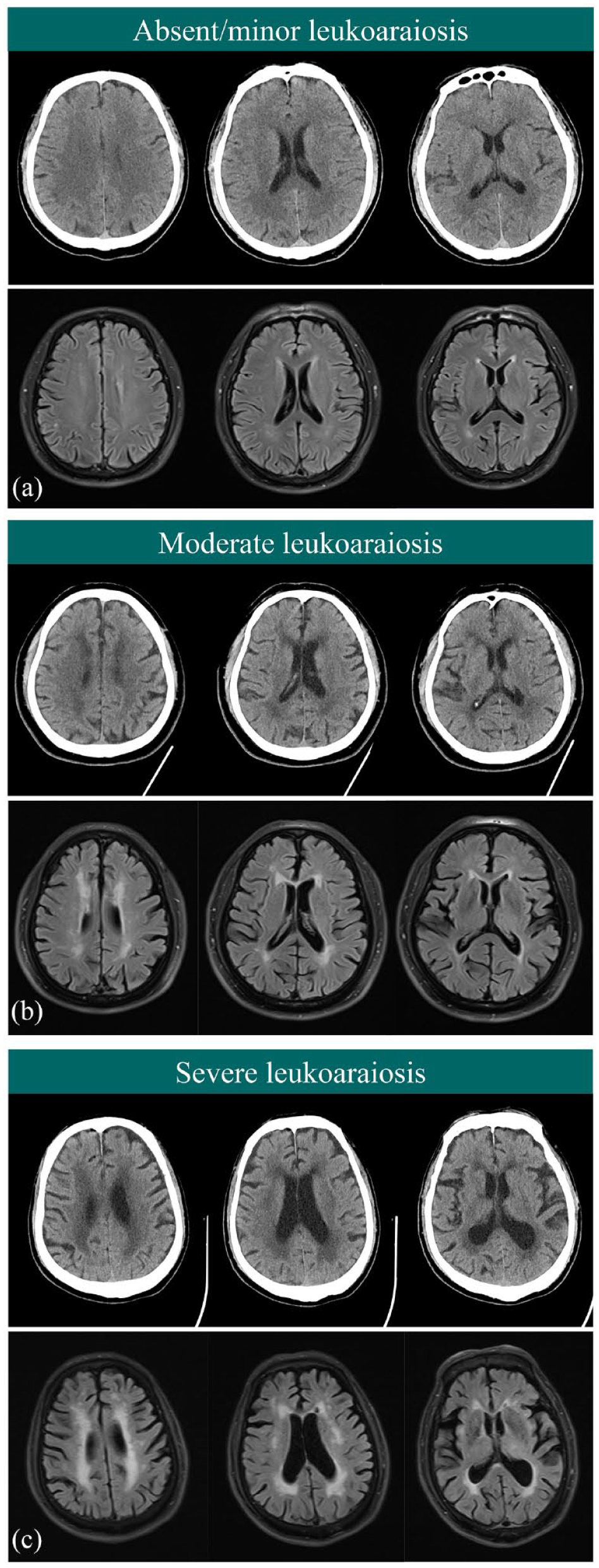
Tripartite classification of LA on the baseline NCCT using the Fazekas criteria with a reference to MR FLAIR images: (a) absent to minor LA, (b) moderate LA, and (c) severe LA.
Comparison of different variables among various categories of LA
In comparison to the patients with moderate LA or severe LA, the patients with absent to minor LA had lower median age, lower rates of coronary artery disease, smaller core volume on CTP, lower IGR, higher median value of ASITN/SIR grade, and a significantly higher frequency of good collaterals. Detailed patient characteristics comparison for the different levels of LA was summarized in Table 1. No significant difference was found in the puncture to reperfusion time, the successful recanalization (mTICI 2b-3), and the symptomatic intracerebral hemorrhage among the groups with different LA severity score.
General demographics and clinical differences stratified by LA status.
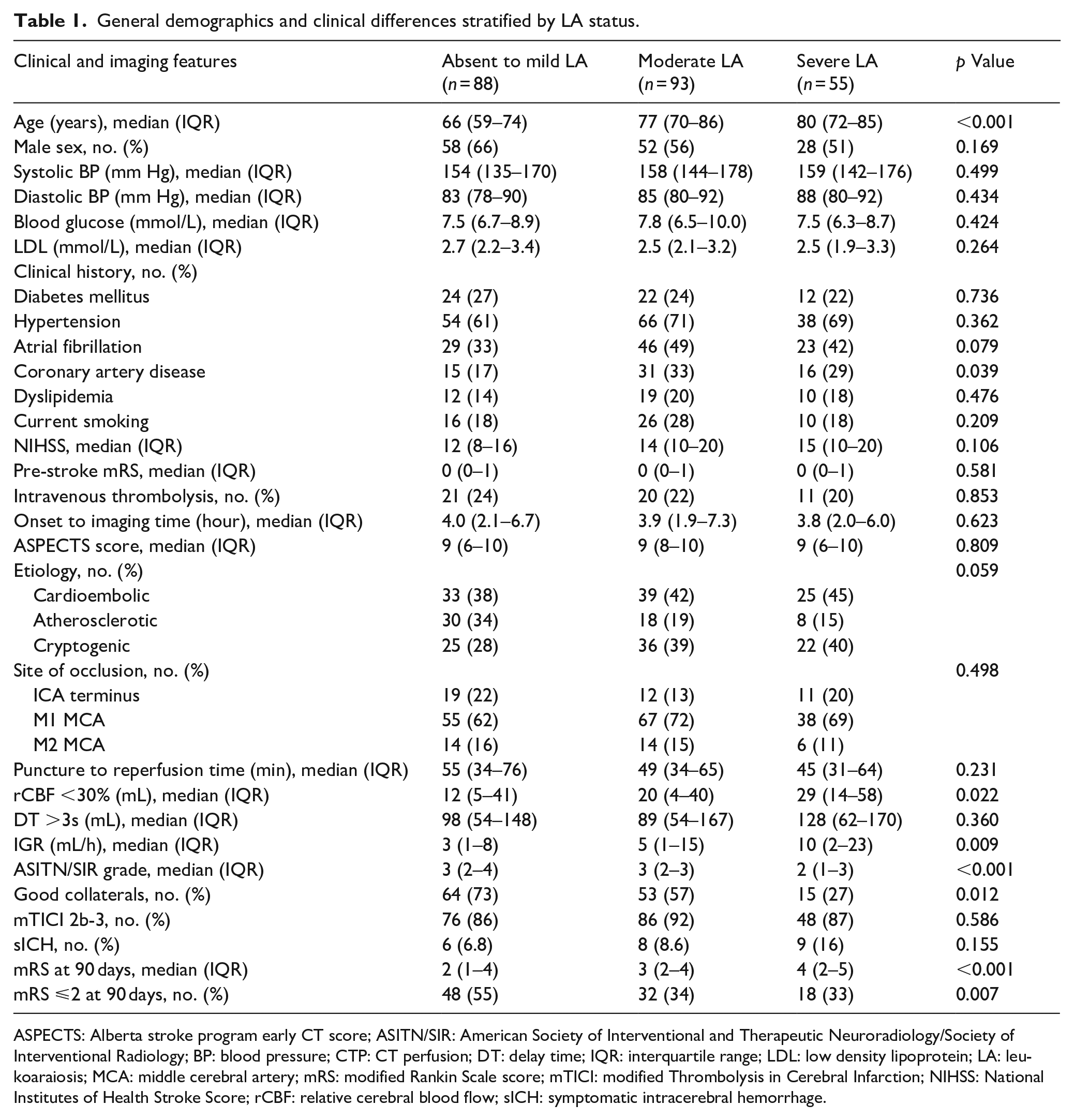
ASPECTS: Alberta stroke program early CT score; ASITN/SIR: American Society of Interventional and Therapeutic Neuroradiology/Society of Interventional Radiology; BP: blood pressure; CTP: CT perfusion; DT: delay time; IQR: interquartile range; LDL: low density lipoprotein; LA: leukoaraiosis; MCA: middle cerebral artery; mRS: modified Rankin Scale score; mTICI: modified Thrombolysis in Cerebral Infarction; NIHSS: National Institutes of Health Stroke Score; rCBF: relative cerebral blood flow; sICH: symptomatic intracerebral hemorrhage.
Correlation between IGR and other variables
The optimal IGR cutoff was 5.8 mL/h (IQR, 1.3–16.0 mL/h) with a range of 0–141 mL/h for favorable clinical outcome. Slow progressors had higher ASITN/SIR grades on DSA (3 [IQR 2–4] vs 2 [IQR 1–3], p < 0.001), higher frequency of good collaterals (67% [89/132] vs 41% [43/104], p < 0.001), and higher rates of atherosclerotic strokes (30% [40/132] vs 15% [16/104], p = 0.028) as compared to fast progressors. On the contrary, the fast progressors had higher median NIHSS (16 [IQR 10–21] vs 12 [IQR 8–18], p = 0.003), shorter onset to imaging time (2.5 [IQR 1.5–5.3] vs 5.2 [IQR 2.7–8.4] h, p < 0.001), larger core volume on CTP (48 [IQR 33–88] vs 8 [IQR 2–15] mL, p < 0.001), and larger penumbra volume on CTP (144 [IQR 84–216] vs 78 [IQR 45–132] mL, p < 0.001) (Table 2, Figure 3). After adjusting for age, sex and etiology, the binomial logistic regression analysis showed that LA status (adjusted odds ratio [aOR] 1.53, [95% CI 1.02–2.33], p = 0.046), baseline NIHSS (aOR 1.04, [95% CI 1.00–1.09], p = 0.032), and impaired collaterals (aOR 2.26, [95% CI 1.27–4.03], p = 0.005) were independently associated with fast IGR.
General demographics and clinical differences between slow and fast progressors.
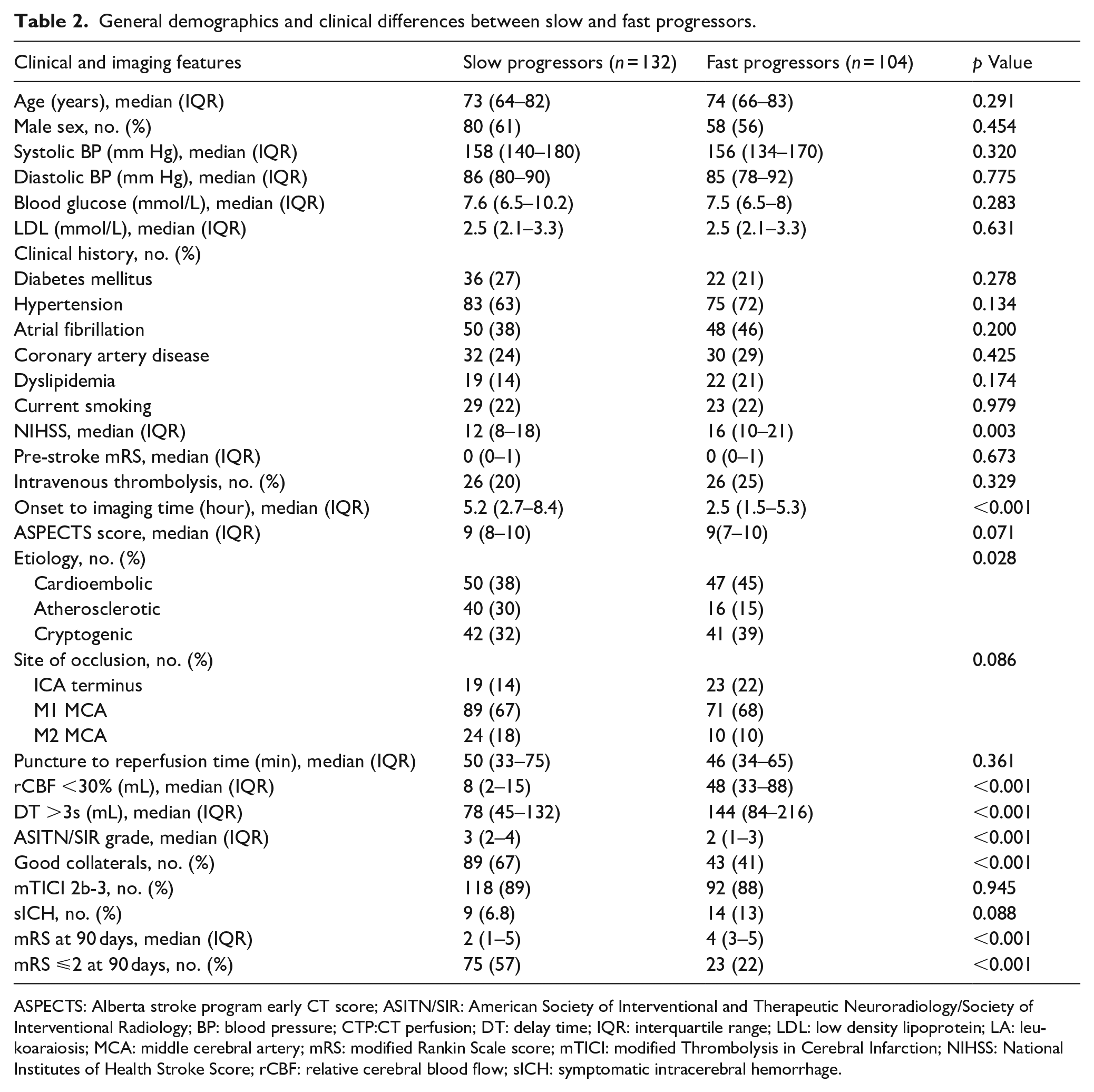
ASPECTS: Alberta stroke program early CT score; ASITN/SIR: American Society of Interventional and Therapeutic Neuroradiology/Society of Interventional Radiology; BP: blood pressure; CTP:CT perfusion; DT: delay time; IQR: interquartile range; LDL: low density lipoprotein; LA: leukoaraiosis; MCA: middle cerebral artery; mRS: modified Rankin Scale score; mTICI: modified Thrombolysis in Cerebral Infarction; NIHSS: National Institutes of Health Stroke Score; rCBF: relative cerebral blood flow; sICH: symptomatic intracerebral hemorrhage.
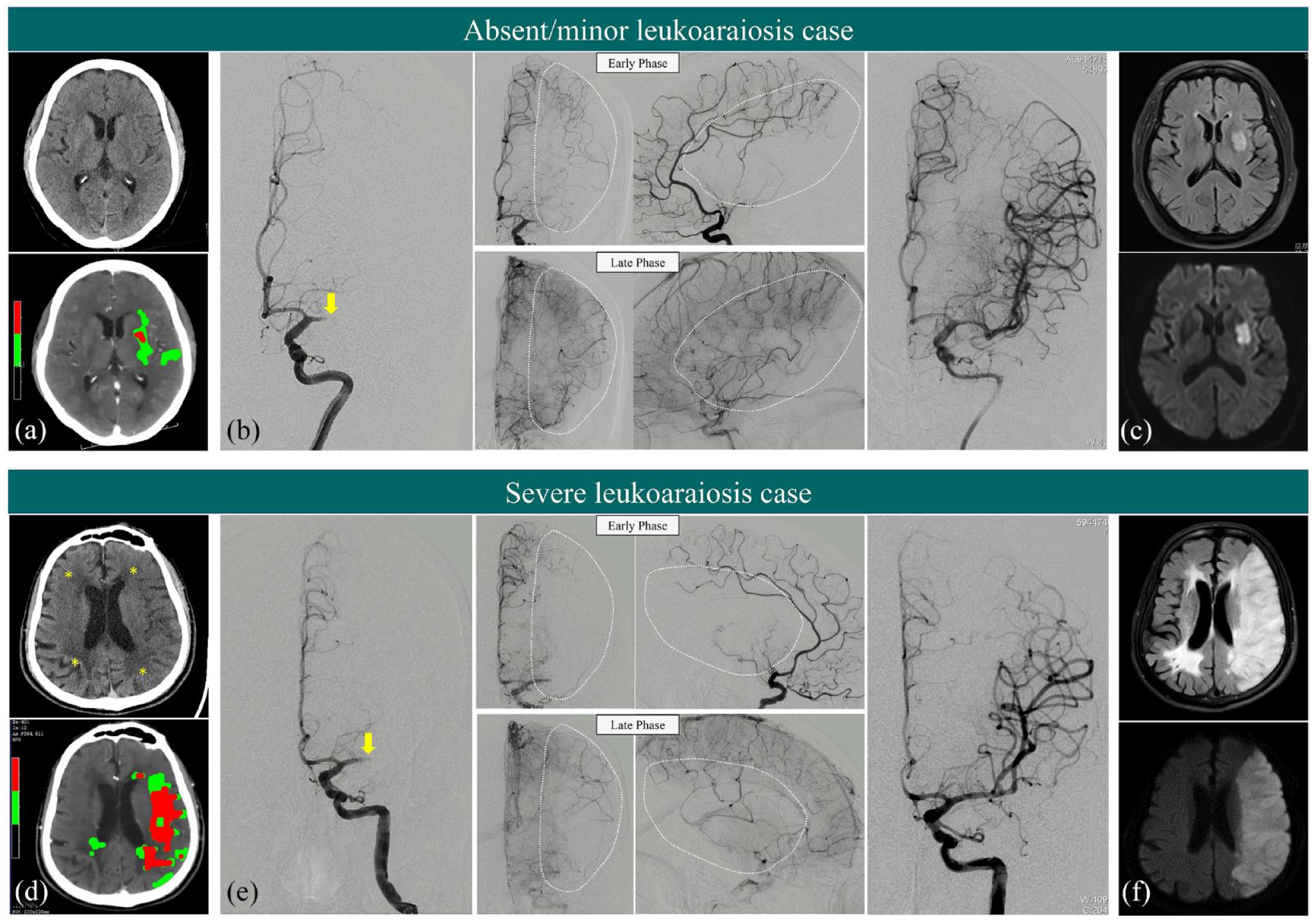
A Cardioembolic stroke patient with minor LA with an IGR of 0.2 mL/h: ((a) core: 1 ml; onset to imaging time: 5 h) and a Cardioembolic stroke patient with severe LA (asterisk) with an IGR of 19.5 mL/h, ((d) core: 42 ml; onset to imaging time: 2.15 h) on NCCT and baseline CTP. DSA imaging shows a proximal left M1 occlusion (yellow arrow) and successful thrombectomy with mTICI 3 flow restoration in both cases (b and e). The minor LA has slowed but complete angiographic blood flow of the ischemic bed by the late venous phase (D, ASITN/SIR grade 3), while severe LA compromises poor collaterals to the periphery of the ischemic site with persistence of some of the defect (E, ASITN/SIR grade 1). The final infarct core and LA severity is confirmed on the follow-up flair and DWI MR scans (c and f).
In mediation analyses, the total effect on fast progressors is composed of the direct effect of LA severity (+9%, p = 0.040) and the mediated indirect effect of collateral status (+5%, p = 0.010). An increase in LA severity by one grade was associated with an increase in the probability of fast IGR by 14% (p = 0.006). The effect of LA status on fast progressors was partially mediated by impaired collaterals, which explained 33% (p = 0.016) of the overall effect. Thus, patients with higher LA severity had an increased risk for fast IGR (Table 3). Patients with lower LA severity had a lesser chance of being fast progressors with higher ASITN/SIR grades. (Figure 4) Subgroup analysis was provided in Supplemental Materials.
Results of the mediation analysis.
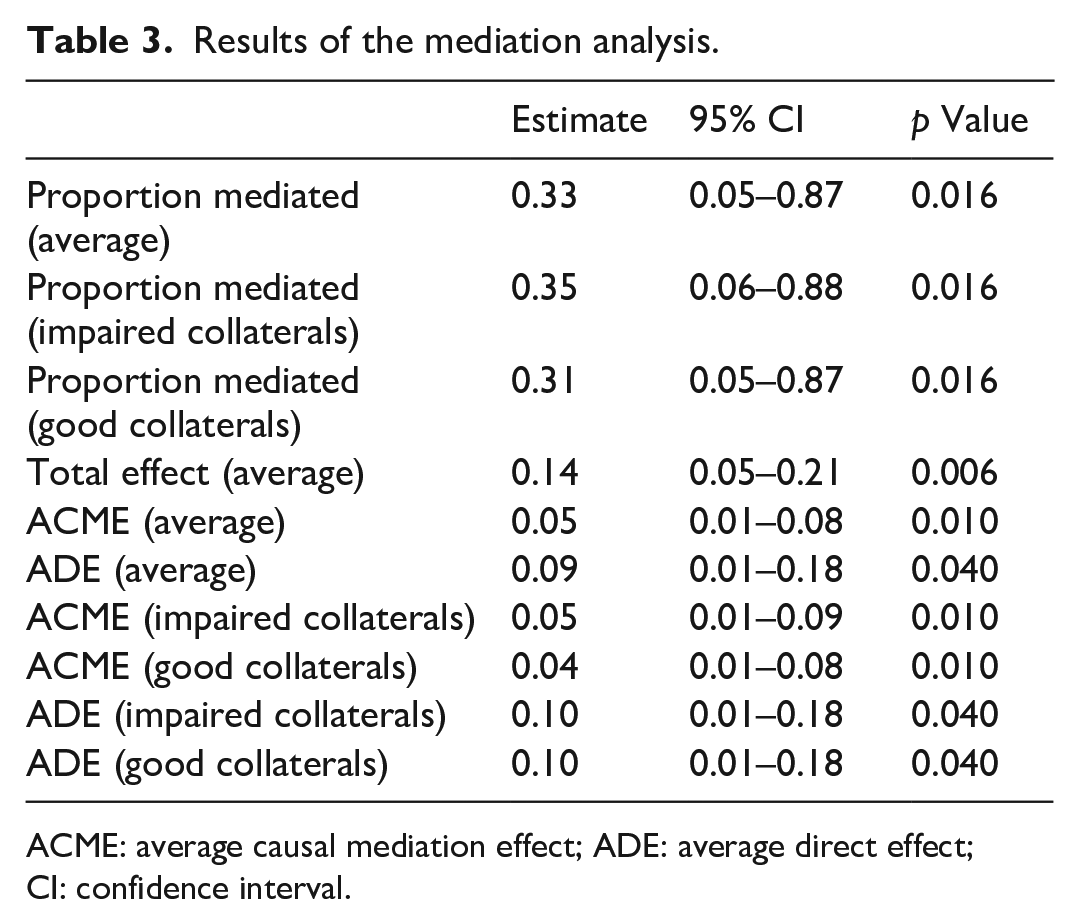
ACME: average causal mediation effect; ADE: average direct effect; CI: confidence interval.
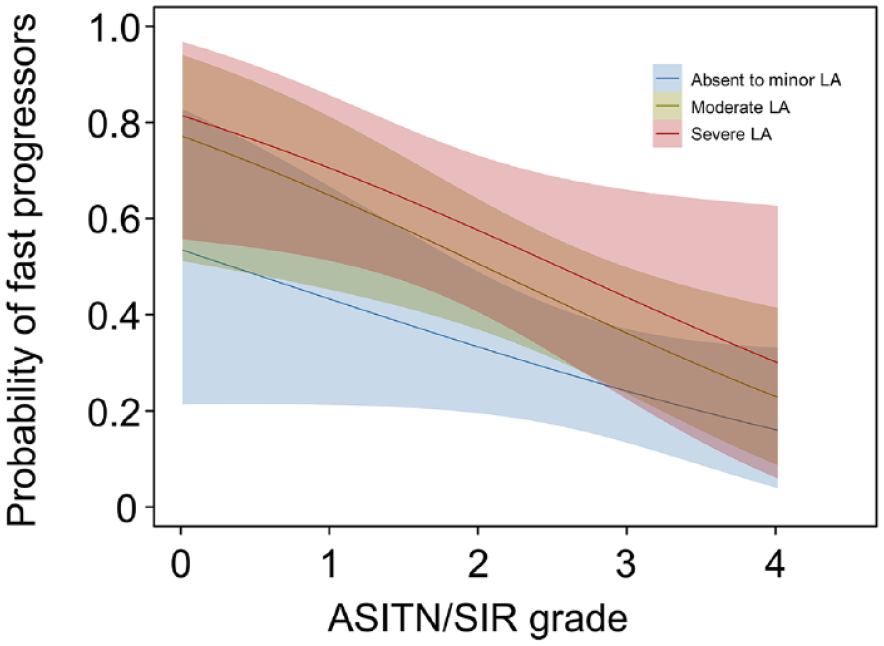
Probability curves to predict fast progressors according to LA severity and ASITN/SIR grade.
Prediction of clinical outcome by LA severity
Of the 236 patients, 41.5% (98 of 236) had an mRS score of 0–2 at 90 days. Lower LA severity was found to be associated with the occurrence of good clinical outcomes (p = 0.007). Patients with moderate and severe LA had a median mRS score of 4 (IQR 3–5) at 90 days, whereas patients with absent to minor LA had a median mRS score of 3 (IQR 2–5), respectively. (p < 0.001) (Figure 5) In mediation analysis among LA status, collaterals and good functional outcome, neither the direct nor the indirect effect was significant (Supplemental Table 1).
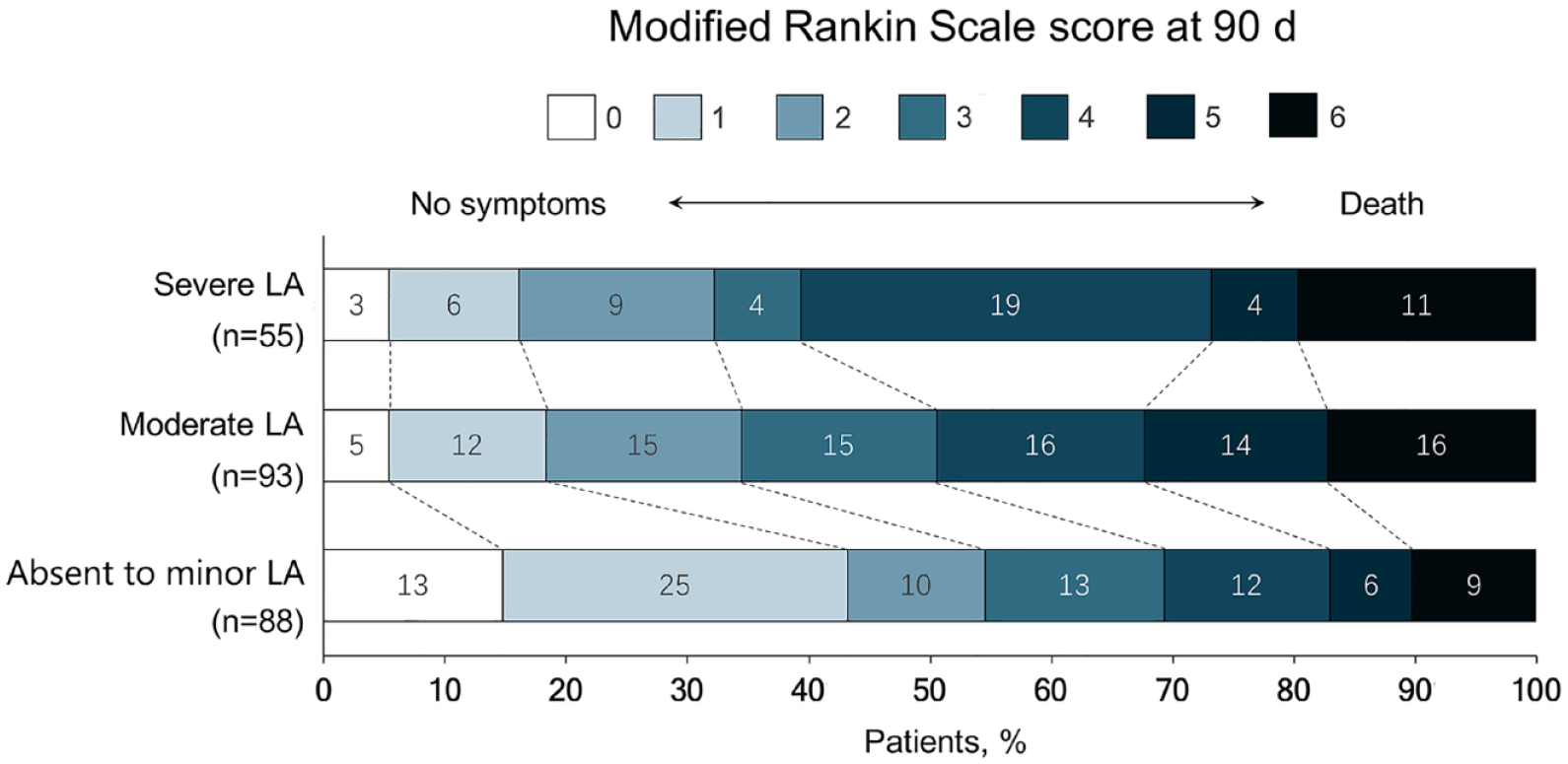
Distribution of modified Rankin Scale score at 90 days according to LA severity.
Discussion
The main finding of this study was that LA increased infarct growth rate in acute LVO stroke through impaired collateral circulation. Meanwhile, higher LA severity was associated with unfavorable functional outcomes following mechanical thrombectomy after 90 days. These findings support the underlying hypothesis that increasing severity of LA is an imaging biomarker for impaired cerebrovascular reserve and may manifest as limited vasodilatory recruitment of leptomeningeal collaterals to protect penumbral tissue from ischemia during acute LVO.
It is well recognized that LA is associated with hypoperfusion ischemia and chronic arteriolar sclerosis. 21 Chronic exposure to hypertension would increase shear stress, stiffen the myogenic tone of vessels, impair endothelial function, and diminish the vasodilatory capacity of leptomeningeal collaterals. 22 Cerebral LA could reflect chronic microcirculatory hypertension as an imaging biomarker. However, as the leptomeningeal collateral system is composed of small arteries and arterioles, it is also conceivable that chronic hypoxia-ischemia may also facilitate the development of leptomeningeal collateral circulation and maturation of preexisting collaterals. 23 Thus, varied relationships between LA and collaterals have been demonstrated in previous studies. White matter assessment changes using quantitative volume analysis or Fazekas score by Sanossian et al., 13 Eker et al. 24 and Derraz et al. 25 showed no correlation between LA and the collateral extent. Our findings are consistent with reports from other groups (Giurgiutiu et al., 12 Mark et al., 10 and Lin et al. 26 ) which demonstrated that LA was detrimental to collateral circulation. We noticed that the above studies with negative results included more patients with milder stroke or less severe LA. For instance, in the Derraz study, the median LA volume was 3.76 mL (IQR 1.09–11.81 mL), whereas in the Giurgiutiu study, the median LA volume was 7.24 mL (IQR 2.2–8.8 mL). In the Sanossian study, 93% of patients were grade 0–1 in the deep white matter (DWM) area and 92% of patients were grade 0–1 in the periventricular white matter (PWM) area, compared with only 37% of patients in our study having a total Fazekas score less than 2.
The present study demonstrates that severe LA is an independent predictor of faster infarct growth. Previous studies indicated a correlation between LA volume and other risk factors for elevated ischemic core growth rate, such as aging, chronic hypertension, diabetes mellitus, hypercholesterolemia, and coronary artery disease.27,28 In patients with severe LA, both the resting cerebral blood flow (up to 30%) 29 and oxygen availability to brain tissue 30 were significantly reduced, suggesting a decreased resilience of the brain parenchyma to emerging ischemia. Moreover, LA might increase platelet aggregability 31 and induce hypercoagulability, 32 which could make the ischemic area less prone to reperfusion therapy and increase final infarct volume. As leptomeningeal collateral was the major determinant of the survival rate of ischemic penumbra, 33 severe LA could impede collateral development and promote infarct evolution. However, in present study, age was found significantly increased with the severity of LA status; thus, it might become a strong confounder for the analysis despite statistical adjustment. When independent factors were investigated for the prediction of IGR, p-values of LA status and NIHSS were significant but at the borderline, which indicated their predicted power might be potentially influenced by age. We believe further study with larger number of patients should be applied to verify their prediction ability. Moreover, we identified a relationship between LA severity, collateral status, and fast IGR through mediation analysis, in which collateral circulation explained 33% of the effect of LA severity on fast progressors. More definitive conclusions regarding the relationship between LA and tissue fate can be derived from analyses of larger data sets in which there are serial imaging data at multiple time points to assess the presence and timing of recanalization of arterial occlusion.
The relationship between LA severity and clinical outcome after EVT was inconsistent between different studies. Zhu et al. 34 showed that patients with higher LA burden had an increased risk for death or major disability after AIS. Similarly, in a multicenter prospective observational study, Mistry et al. 35 found that patients with moderate-to-severe LA had significantly worse functional outcomes at 90 days. More recently, in a single-center study involving 366 patients with AIS, patients demonstrated higher mRS scores with increasing LA volumes, although no significant associations between LA and 90-day mortality was found. 36 In contrast, Mechtouff et al. 37 demonstrated no correlation between LA burden (determined with the Fazekas score) and clinical outcome, parenchymal hemorrhage risk, or reperfusion rate. This inconsistency might arise from varying criteria for LA assessment, the number of prognostic variables included, and other differences in demographics. Similar to most studies, we also demonstrated that the LA severity was associated with clinical outcomes at 90 days.
Our study has several limitations. First, the retrospective design had an inherent selection bias, as we only included patients with known stroke onset time who underwent mechanical thrombectomy with a pre-stroke mRS scale of 0–1 and NIHSS score ⩾6. Patients with lower baseline NIHSS and underlying better collaterals might be excluded from EVT. Second, we assessed LA using the CT-based Fazekas score rather than MRI becomes one of the main weaknesses of present study due to a moderate inter-rater agreement between CT and MRI-based scoring systems of white matter lesions. We believe more precise methods such as volumetric quantification may be required in further study. Third, the evaluation of leptomeningeal collateral flow using the ASITN/SIR scores on DSA was limited by the lack of contralateral anterior and posterior circulation vessel injections. Lastly, other unknown factors might also affect our results. Since the present study was not designed to establish the risk-versus-benefit ratio of reperfusion therapies in relation to LA, treatment should not be withheld from eligible patients solely because of severe LA.
Conclusions
Increasing severity of LA is associated with impaired collateral status, fast infarct growth, and poor clinical outcomes in acute LVO stroke after EVT. These findings suggest that LA may become a predictive imaging biomarker for the likelihood of progression of tissue injury and clinical outcome after EVT in acute large vessel occlusion stroke. Further larger confirmatory prospective studies are warranted to confirm our findings.
Abbreviations
LA = leukoaraiosis, AIS = acute ischemic stroke, IGR = infarct growth rate, LVO = large vessel occlusion, EVT = endovascular thrombectomy, NCCT = non-contrast CT, CTP = computed tomography perfusion, ASITN/SIR = the American Society of Interventional and Therapeutic Neuroradiology/Society of Interventional Radiology, mRS = modified Rankin Scale, ASPECTS = Alberta Stroke Program Early CT Score, NIHSS = National Institutes of Health Stroke Score, mTICI = modified thrombolysis in cerebral infarction, ICA = internal carotid artery, MCA = proximal middle cerebral artery, IQR = interquartile range, DSA = digital subtraction angiography, aOR = adjusted odds ratio.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241226771 – Supplemental material for Impact of leukoaraiosis on the infarct growth rate and clinical outcome in acute large vessel occlusion stroke after endovascular thrombectomy
Supplemental material, sj-docx-1-eso-10.1177_23969873241226771 for Impact of leukoaraiosis on the infarct growth rate and clinical outcome in acute large vessel occlusion stroke after endovascular thrombectomy by Guangchen He, Hui Fang, Bo Xue, Liming Wei, Haitao Lu, Jiangshan Deng and Yueqi Zhu in European Stroke Journal
Supplemental Material
sj-docx-2-eso-10.1177_23969873241226771 – Supplemental material for Impact of leukoaraiosis on the infarct growth rate and clinical outcome in acute large vessel occlusion stroke after endovascular thrombectomy
Supplemental material, sj-docx-2-eso-10.1177_23969873241226771 for Impact of leukoaraiosis on the infarct growth rate and clinical outcome in acute large vessel occlusion stroke after endovascular thrombectomy by Guangchen He, Hui Fang, Bo Xue, Liming Wei, Haitao Lu, Jiangshan Deng and Yueqi Zhu in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: (1) New interdisciplinary research Project of Shanghai Municipal Health Commission (No.2022JC022). (2) Shanghai Jiao Tong University “Medical and Research” Program (ZH2018ZDA19).
Ethical approval
The ethics committee of Shanghai Sixth People’s Hospital Affiliated to Shanghai Jiao Tong University School of Medicine approved this study (2018-002-1).
Informed consent
Informed consent was provided by patients or their family members.
Guarantor
Yueqi Zhu.
Contributorship
(1) Guangchen He, Hui Fang and Bo Xue: Acquisition of data, analysis and interpretation of data and drafting of the manuscript
(2) Yueqi Zhu: Study concept and design, critical revision of the manuscript for important intellectual content, and study supervision.
(3) Liming Wei, Haitao Lu, Jiangshan Deng: acquisition of data, revision of the manuscript
(4) Guangchen He: statistical analysis
Data availability
This study’s data are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
