Abstract
Background:
In younger patients (<60 years) with cryptogenic stroke (CS) presumed to be patent foramen ovale (PFO)-related, the standard approach involves transcatheter PFO closure combined with antithrombotic therapy. However, due to their exclusion from randomized clinical trials (RCTs), no formal recommendations exist for patients ⩾60 years. This study had two objectives (1) to compare the efficacy and safety of PFO closure versus antithrombotic therapy alone (ATA) exclusively in older patients (⩾60 years) and (2) to assess the outcomes of PFO closure in patients ⩾ 60 years versus < 60 years.
Methods:
We searched PubMed, Embase, Web of Science, and ScienceDirect databases to obtain articles in all languages from January 2004 until July 2025. The primary outcome was risk of recurrent stroke during follow-up. Secondary outcomes were risk of new-onset atrial fibrillation (AF), all-cause mortality, and in-hospital complications. PROSPERO registration ID: CRD420250652870.
Results:
Only one RCT (post hoc evaluation of the DEFENSE-PFO trial) and 11 observational studies were included. In patients aged ⩾ 60 years, risk of recurrent stroke was lower when PFO was closed compared with ATA (5.48% vs 10.05%, respectively, hazard ratio (HR) = 0.56, 95% confidence interval (CI) = 0.45–0.80, p < 0.001, I2 = 45.6%). All-cause mortality was also lower for PFO closure versus ATA (1.73% vs 7.59%, respectively, HR = 0.41; 95% CI = 0.19–0.90, p = 0.02; I2 = 43.8%). There was no difference between PFO closure and ATA in risk of new-onset AF (HR = 1.13, 95% CI = 0.53–2.44, p = 0.74). Compared with patients < 60 years, individuals ⩾ 60 years who underwent PFO closure had a higher risk of recurrent stroke (2.94% vs 1.04%, respectively, HR = 3.47; 95% CI = 1.61–7.48; p = 0.001), new-onset AF (4.86% vs 1.01%, respectively, HR = 4.12; 95% CI = 1.90–8.95; p < 0.001), and all-cause mortality during follow-up (8.32% vs 0.39%, respectively, HR = 8.24; 95% CI = 3.49–19.46; p < 0.0001). In-hospital complications after PFO closure were comparable between two age groups. Due to insufficient data, we were not able to perform a subgroup analysis based on anatomic features of PFO, antithrombotic regimen, or occluder devices.
Conclusion:
Based on available data, which is predominantly derived from observational studies, PFO closure is associated with a reduced risk of recurrent stroke compared to ATA in patients over 60 years. However, these findings are subjected to limitations, including the potential for selection bias, unmeasured confounding, and insufficient long-term follow-up. Furthermore, long-term randomized trials are essential to definitively confirm efficacy and establish clinical guidelines for PFO closure in this older population.

Introduction
In approximately 30–40% of ischemic strokes, the etiology is not identified even after a standard neurologic and cardiac workup, leading to a classification as cryptogenic stroke (CS).1,2 Among potential contributors to cryptogenic stroke, patent foramen ovale (PFO) is increasingly recognized as a key risk factor. PFO is present in approximately 25% of the adult population, and yet its prevalence is significantly higher in cryptogenic stroke patients compared to those with identifiable stroke causes. 3 This association has led to growing interest in evaluating the role of transcatheter PFO closure as a preventive strategy for reducing recurrent stroke risk in patients with cryptogenic stroke. A meta-analysis of six randomized clinical trials (RCTs) showed that transcatheter PFO closure is more effective than antithrombotic therapy alone (ATA) in reducing the risk of recurrent stroke. 4 Given these findings, current guidelines recommend transcatheter closure of PFO combined with antithrombotic therapy for select patients with cryptogenic stroke.5,6 However, the applicability of these recommendations is limited to patients under 60 years of age. Given that all RCTs, with the singular exception of DEFENSE-PFO, excluded patients aged 60 years or older, the practice of PFO closure in this older population remains contentious. In light of this, the European Stroke Organisation (ESO) Guidelines on the diagnosis and management of PFO after stroke refrain from offering a recommendation for patients exceeding 60 years of age, citing a lack of sufficient supporting evidence. 7
While no RCT has directly compared PFO closure to ATA among older patients, a subgroup analysis of DEFENSE-PFO along with several recent observational studies has suggested potential preventive benefits of PFO closure in older individuals with cryptogenic stroke.8,9 However, other studies indicate that antithrombotic therapy may be a safer alternative, offering comparable effectiveness to PFO closure in older adults.10,11 Given the limited evidence from RCTs in this population, further investigation is warranted to clarify long-term outcomes. The objective of this study was to achieve two principal aims: (1) evaluate and compare the efficacy and safety of PFO closure versus ATA specifically in older patients (⩾60 years) and (2) assess the risk of recurrent cerebrovascular ischemic events after PFO closure in patients with cryptogenic stroke, stratified by age (⩾ 60 vs < 60 years).
Methods
This systematic review and meta-analysis was conducted in accordance with the 2020 Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 12 The review protocol has been registered in the International Prospective Register of Ongoing Systematic Reviews (PROSPERO registration ID: CRD420250652870).
Data sources and search strategy
PubMed, Embase, Web of Science, and ScienceDirect electronic databases were searched systematically. The search string was first developed for PubMed using a combination of MeSH, Emtree, and search word terms and then was tailored to other databases. The search covered all published literature without a language filter, from 1 January 2004 to 1 July 2025. The full search string for all databases is available in Supplemental Table S1. In addition, a forward (citing) and backwards (cited) citation search of the eligible studies was performed to ensure completeness of the systematic search for potential eligible studies.
Study selection process and eligibility criteria
Two independent reviewers (C.P. and S.B.J.) performed a review of the titles and abstracts. Any discrepancy between authors was resolved with consensus. We included (a) observational studies employing a case-control, cohort, or case series design, or utilizing post hoc analysis derived from RCT that included (b) older patients (age ⩾ 60 years) with a history of cryptogenic ischemic stroke, transient ischemic attack (TIA), or other thromboembolic events and a confirmed PFO (c) who received PFO closure; and a comparison group of (d) younger patients (age < 60) and/or (e) patients over 60 years receiving ATA. We only included studies that (f) contained a follow-up of at least 2 years and (g) reported at least one of the outcomes of recurrent TIA, stroke, new-onset atrial fibrillation (AF), or mortality. Studies that defined older patients using an age cut-off below 60 years (e.g. 55 years), as well as case reports, book chapters, and conference abstracts, were excluded. A cutoff age of 60 years was chosen to align with the inclusion criteria of most trials of PFO closure13–17 and also because data for older age groups was scarce (e.g. over 70 or 80 years).
Data extraction
Two authors (C.P. and S.B.J.) independently reviewed and extracted data using a unique data-extraction sheet, which was designed for this study, and discrepancies were resolved through discussion with a third author (R.B.S.). The data items included study design and characteristics, population under study characteristics, and study endpoints. The outcome was accepted as defined in individual studies and related to the recurrence of stroke, TIA, or all-cause mortality. Data on safety included adverse events reported due to the PFO closure process.
Quality assessment
For quality assessment, two authors (C.P. and S.R.) independently analyzed the included studies. Observational studies were assessed using the Risk Of Bias In Non-randomized Studies—of Intervention (ROBINS-I) tool, while randomized controlled trials (RCTs) utilized the Cochrane Risk of Bias 2 (RoB 2) tool.18,19 Any discrepancies were resolved through discussion with a third author (S.B.J.).
Outcomes
The primary efficacy outcome was recurrent ischemic stroke during follow-up. Secondary outcomes included a composite of ischemic stroke or TIA. These outcomes were assessed to compare: (1) PFO closure versus ATA in older patients with cryptogenic stroke and (2) older versus younger patients who underwent PFO closure. The safety outcomes included (a) the risk of mortality, (b) new-onset AF during follow-up and (c) the risk of in-hospital complications in patients who underwent PFO closure.
Statistical analysis
We ran two different sets of analyses separately. First, we compared the efficacy and safety of PFO closure as an index intervention with ATA in the ⩾60-year-old population. In another analysis, we compared patients ⩾60 years as the index population with younger patients (< 60 years) to compare the efficacy and safety of PFO closure among different age groups. The effect size was hazard ratio (HR) and its 95% confidence intervals (CIs) for endpoints related to long-term follow-up. We selected HRs as they facilitate time-to-event analysis while accounting for varying follow-up durations and censoring, enhancing comparability across studies. 20 For in-hospital complications, we used the Odds Ratio (OR) and its 95% CI as the effect size. We used random effects models for all endpoints to account for possible heterogeneity among study designs. 21 Generic inverse-variance model was used to combine data of endpoints with long-term follow-up, and a generalized linear mixed model (GLMM) was used for pooling data of in-hospital complications. Heterogeneity was assessed using I2 statistics and the Cochran’s Q test. An I2 test > 50% or p-value < 0.05 was considered as high heterogeneity. In case of high heterogeneity, we detected outliers using the methods previously described in the literature. 22 A subgroup analysis based on study design was performed to separate observation studies from RCTs. In addition, a sensitivity analysis was performed by excluding studies with a high risk of bias. Due to a low number of included studies, we were not able to perform a meta-regression or publication bias assessment, because such analyses need at least 10 studies for each endpoint to ensure adequate power. 20 Additional methods regarding statistical analysis can be found in Supplementary Materials.
Results
Study selection
The search process across four major databases yielded 1422 potential studies. After removing 311 duplicates, the title and abstract of 1111 articles were checked against the eligibility criteria, which led to retaining 21 articles. One additional study met eligibility criteria and was included in the meta-analysis from citation searching. We excluded 9 full texts due to noncomparative study design or an age cut-off of < 60 years for the older group. Another study was excluded to prevent case overlap, as a newer study with extended follow-up from the same center was available 9 (Supplemental Table S2). Finally, a total of 12 studies were included in the systematic review and meta-analysis8,9,11,23–32 (Figure 1).
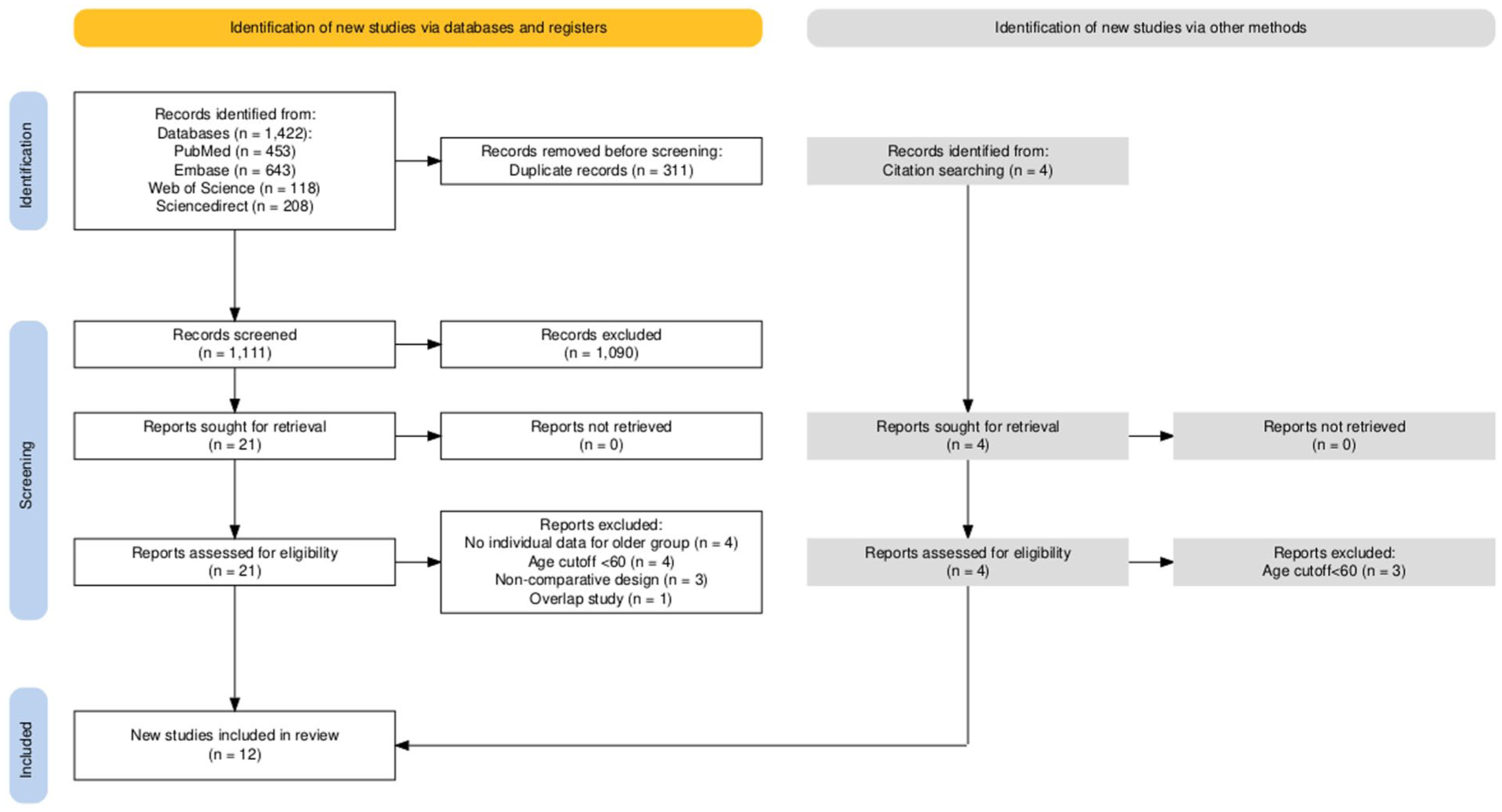
PRISMA diagram. Flow diagram summarizing the article review and selection process, including the forward and backward citation search. PRISMA = Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Study characteristics
The main characteristics of each study are summarized in Supplemental Table S3, and the patient characteristics are summarized in Supplemental Table S4. Four studies compared PFO closure versus ATA in patients over 60 years,8,25,26,28 five studies compared PFO closure in older versus younger patients,11,29–32 and three studies contained both treatment and age comparisons.9,24,27 The follow-up duration across studies varied, ranging from 2.4 to 12.6 years (average = 5.3 ± 1.8 years). While specific dosages of each antithrombotic therapy varied between studies, all studies utilized a combination of aspirin monotherapy, clopidogrel, dual antiplatelet therapy (DAPT), or anticoagulation. In all studies, antithrombotic therapy was administered to patients who underwent PFO closure. The raw data regarding efficacy and safety of each study are provided in Supplemental Table S5, and the main findings of all studies are outlined in Supplemental Table S6.
Risk of bias and quality assessment
Using the ROBINS-I tool, ten of the included observational studies were assessed as having an overall moderate risk of bias, while two studies were identified with a serious risk of bias8,26 (Supplemental Figure S1). The latter two studies were not based on a thorough and well-documented etiological work-up for cryptogenic stroke, but rather on large administrative registries of patients with ischemic stroke and an atrial shunt, which could correspond to a PFO or an Atrial Septal Defect (ASD) due to the same International Classification of Diseases (ICD) diagnosis code. These registries provided insufficient data on several critical factors, including stroke severity and the rationale behind treatment decisions. The remaining studies also had a moderate risk of confounding bias, as they lacked sufficient data on stroke severity, high-risk versus low-risk PFO features, and the reasons behind treatment choices between antiplatelet/anticoagulant therapy and closure. The only RCT in our series—the DEFENSE-PFO study—was assessed as also having some concerns for risk of bias based on quality scores by RoB 2 tool due to concerns regarding deviation from intended intervention and bias in selection of reported results (Supplemental Figure S2).
PFO closure versus ATA among ⩾ 60 years
For the primary analysis (risk of recurrent stroke), we analyzed data from seven studies (six observational and one RCT (post hoc analysis of DEFENSE-PFO trial)), comparing 1610 patients who underwent PFO closure and 4925 patients who received ATA. Stroke occurred in 5.48% (95% CI = 3.45–8.62) of the closure group versus 10.05% (95% CI = 6.00–16.38) of the ATA group. The pooled HR was 0.56 (95% CI = 0.43–0.74, p < 0.0001; I² = 45.6%), indicating a significantly lower stroke risk with closure in patients ⩾ 60 years. In a subgroup analysis stratified by study design, the pooled HR for the observational studies was 0.57 (95% CI = 0.44–0.75, p < 0.0001). The single RCT in our series yielded an HR of 0.16 (95% CI = 0.02–1.62, p = 0.12) (Figure 2(a)). A sensitivity analysis, conducted after the exclusion of two serious-risk studies,8,26 reinforced these findings with a pooled HR of 0.38 (95% CI = 0.15–0.98, p = 0.04) (Supplemental Figure S3). No study was found to be an outlier.
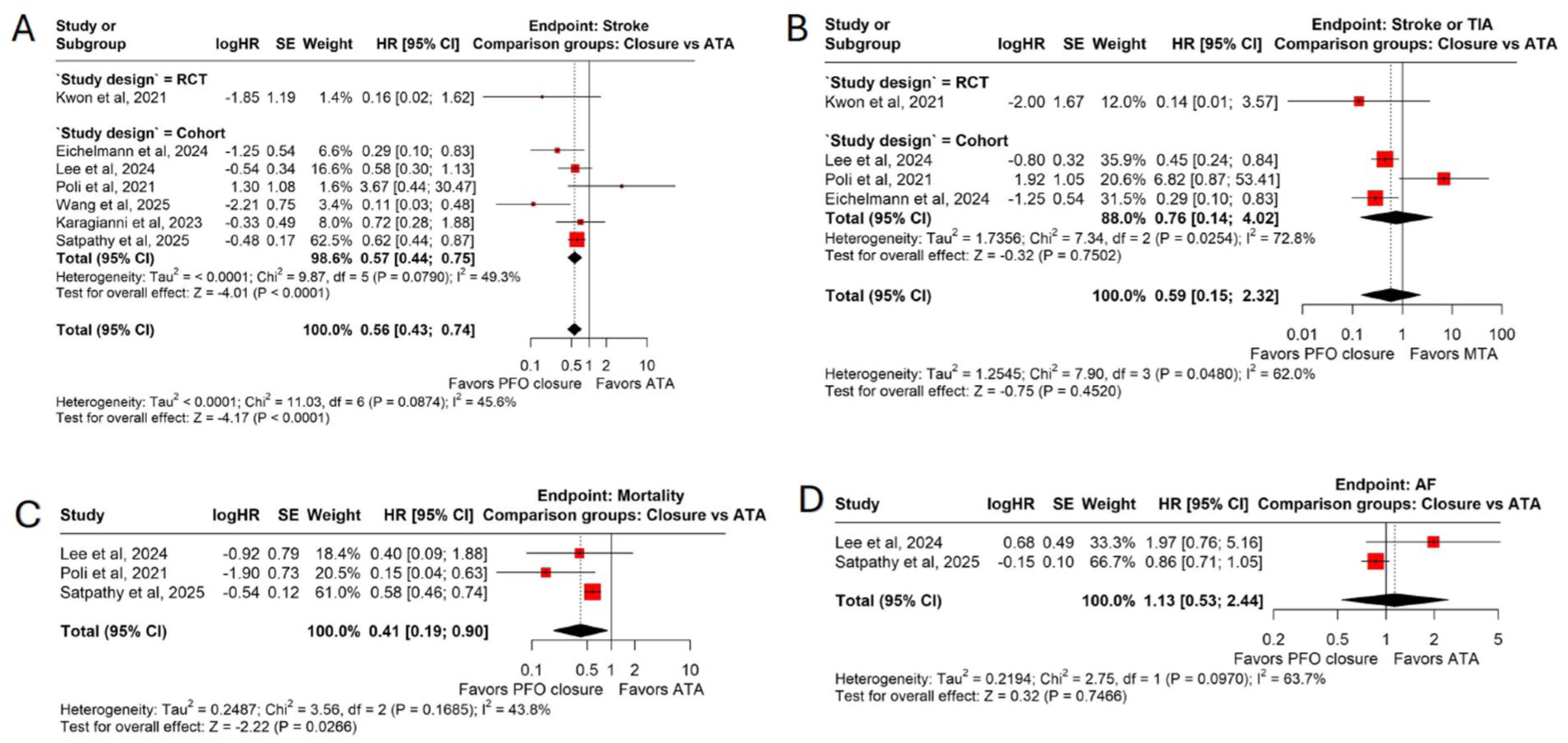
PFO closure compared with ATA in patients over 60 years. Forest plot comparing the efficacy and safety of PFO closure with ATA in patients over 60 years with cryptogenic stroke. (a) Stroke, (b) TIA or stroke, (c) new-onset AF, and (d) all-cause mortality. PFO = patent foramen ovale; ATA = antithrombotic therapy alone; TIA = transient ischemic attack; HR = hazard ratio; CI = confidence interval; SE = standard error.
Data for the secondary outcome of recurrent stroke or TIA were available in four studies (three observational and one RCT). The analysis found no difference between PFO closure (5.76%; 95% CI = 2.34–13.44) versus ATA (10.76%; 95% CI = 4.83–22.28) among ⩾ 60 years (HR = 0.59, 95% CI = 0.15–2.32, p = 0.45; I2 = 62.0%). The subgroup analysis based on study design showed an insignificant difference among PFO closure and ATA for both observational studies (HR = 0.76, 95% CI = 0.14–4.02, p = 0.75; I2 = 62.0%) and the single RCT in the series (HR = 0.14, 95% CI = 0.01–3.57, p = 0.23) (Figure 2(b)). There was no outlier study for this analysis.
Data regarding all-cause mortality was available in three observational studies. The mortality was lower with PFO closure (1.73%; 95% CI = 0.56–5.24) compared to ATA (7.59%; 95% CI = 4.36 vs 12.9) with HR of 0.41 (95% CI = 0.19–0.90, p = 0.02; I2 = 43.8%) (Figure 2(c)). A sensitivity analysis, conducted after the exclusion of one serious-risk study, 8 reinforced these findings with a pooled HR of 0.24 (95% CI = 0.08–0.67, p = 0.007) (Supplemental Figure S4). There was no outlier study for this analysis.
Data regarding new-onset AF was available in only two observational studies. The rate of new-onset AF was comparable among both groups with no outlier study (HR = 1.13, 95% CI = 0.53–2.44, p = 0.74; I2 = 63.7%) (Figure 2(d)).
PFO closure among ⩾ 60 years versus < 60 years
For our primary outcome (risk of recurrent stroke), we analyzed data from four observational studies, comparing 579 patients ⩾ 60 years with 2152 patients < 60 years, all of whom underwent PFO closure. The absolute risk of recurrent stroke was 2.94% (95% CI = 1.84–4.67%) in patients ⩾ 60 years and 1.04% (95% CI = 0.55–1.96) in patients < 60 years, respectively. A random-effects analysis yielded a pooled HR of 3.47 (95% CI = 1.61–7.48, p = 0.001; I2 = 0%) for the primary outcome, showing a higher incidence of recurrent stroke among patients aged ⩾ 60 years compared to those < 60 years (Figure 3(a)). In our secondary analysis, the absolute risk of stroke or TIA was 5.41% (95% CI = 2.59–10.96) in patients ⩾ 60 years versus 1.89% (95% CI = 1.01–3.53) with a pooled HR of 2.68 (95% CI =1.40–5.13, p = 0.002; I² = 15.5%) (Figure 3(b)). In addition, the rate of new-onset AF was higher among ⩾ 60 years with an absolute risk of 4.86% (95% CI = 3.12–7.49) versus 1.01% (95% CI = 0.25–4.01) and HR of 4.12 (95% CI = 1.90–8.95, p < 0.001; I2 = 33.9%) (Figure 3(c)).
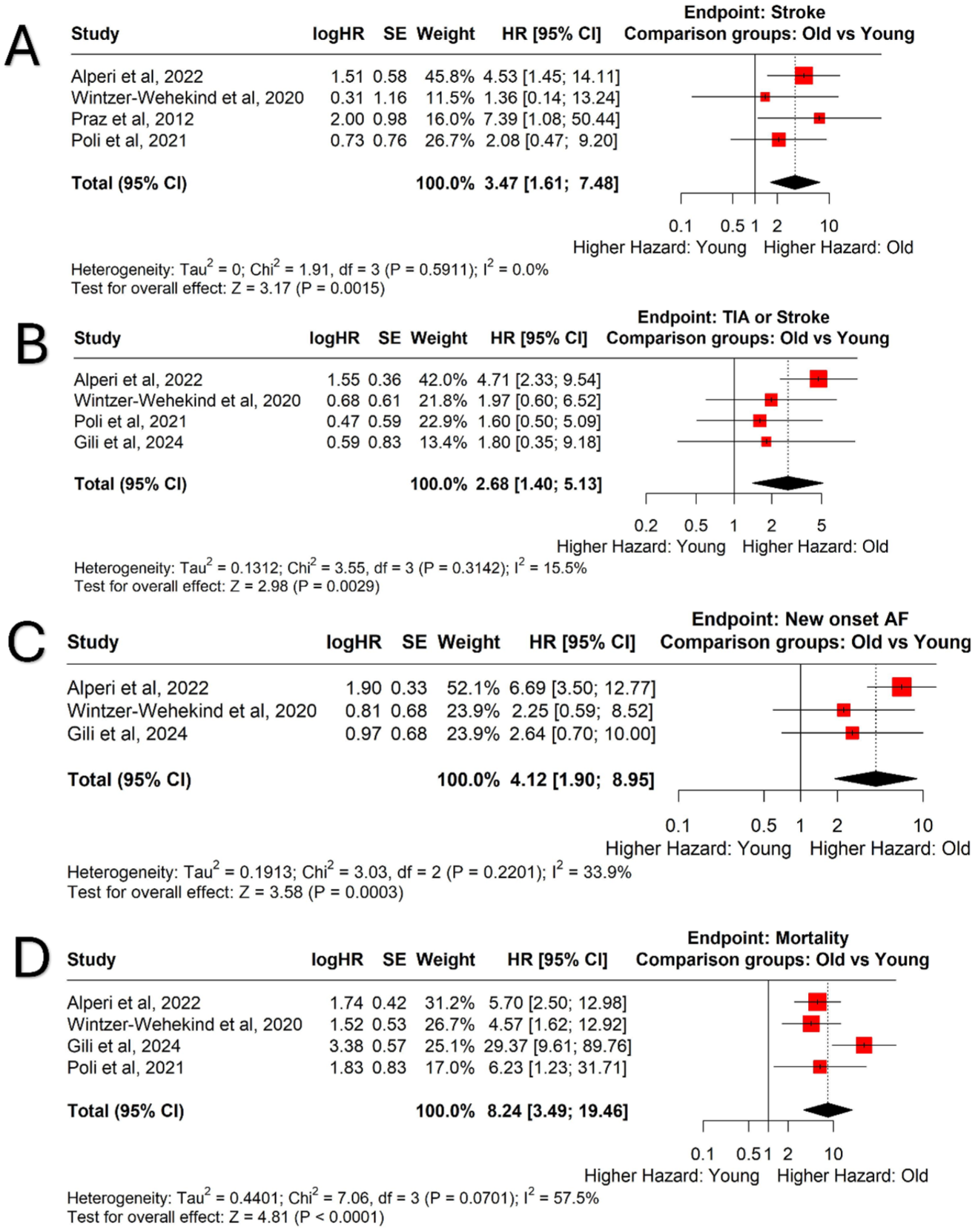
PFO closure in patients over 60 years compared with younger patients. Forest plot comparing the efficacy and safety of PFO closure in older patients with cryptogenic stroke compared with the young patients. (a) Stroke, (b) TIA or stroke, (c) new-onset atrial fibrillation, and (d) all-cause mortality. PFO = patent foramen ovale; TIA = transient ischemic attack; HR = hazard ratio; CI = confidence interval; SE = standard error.
The rate of all-cause mortality during follow-up was higher among the ⩾ 60 years with an absolute risk of 8.32% (95% CI = 3.4–20.79) versus 0.39% (95% CI = 0.06–2.37) and HR of 8.24 (95% CI = 3.49–19.46, p < 0.0001; I2 = 57.5%) (Figure 3(d)). The results became more robust when we excluded one outlier study (Gili et al.), with HR being 5.36 (95% CI = 2.94–9.76, p < 0.0001; I2 = 0%). Notably, the rate of all in-hospital complications, including periprocedural AF, was similar among the below and over 60 years (Supplemental Figures S5 to S10). Each complication is discussed in detail in Supplemental Table S7.
Discussion
A central question in clinical practice is whether patients aged ⩾ 60 years with a PFO and a history of cryptogenic stroke derive meaningful benefit from PFO closure, or whether ATA remains the preferred strategy. Based primarily on retrospective cohort studies, our findings suggest that PFO closure is associated with a reduced risk of future ischemic stroke, without a significant increase in the incidence of new-onset AF compared to ATA. When stratified by age, patients ⩾ 60 years who underwent PFO closure experienced notably higher rates of recurrent stroke (~3% vs ~1%), recurrent stroke or TIA (5.4% vs 1.9%), new-onset AF (4.8% vs 1%), and all-cause mortality (8.3% vs 0.4%) compared to those < 60 years, despite comparable rates of periprocedural complications across both age groups.
The only RCT in our analysis was a subgroup analysis of the DEFENSE-PFO trial, which assessed the outcomes of PFO closure versus ATA in 34 patients over 60 years old with high-risk PFO. 27 At the 2-year follow-up, four ischemic events were recorded, all occurring in the antithrombotic therapy group (p = 0.07). Notably, when the analysis was limited to patients over 70 years, the difference reached statistical significance (p = 0.03). Based on these findings, the authors recommended PFO closure for elderly patients with high-risk PFO, while acknowledging the limitation of a small sample size. On the contrary, Poli et al. 24 did not find any difference in the risk of recurrent ischemic stroke among high-risk PFO patients over 60 years who underwent PFO-closure compared to those receiving antithrombotic treatment alone. Our analysis suggests that, based on currently available evidence, PFO closure significantly reduces the risk of ischemic stroke in patients over 60 years compared to ATA. However, this conclusion is heavily reliant on the body of evidence being predominantly observational and is strongly influenced by the recent study by Satpathy et al., 8 which accounts for 62.5% of the weight in our meta-analysis. This study, conducted among U.S. Medicare beneficiaries with stroke and atrial shunt (PFO or ASD), lacks critical details regarding PFO anatomy, stroke severity, antithrombotic regimen, and etiological work-up, limiting its interpretability. To address this, we performed a sensitivity analysis which confirmed the robustness of our primary findings. Nonetheless, the absence of statistical significance in our secondary analysis for the combined endpoint of stroke or TIA underscores the need for further data to validate the effectiveness of PFO closure in older patients. It is also important to note that the clinical diagnosis of TIA remains uncertain, and several studies opted not to include TIA in their reported endpoints, resulting in a reduced number of studies available for this specific analysis.
Several scoring systems are used to guide patient selection for PFO closure in patients with cryptogenic stroke. Risk of Paradoxical Embolism (RoPE) score is a commonly used scoring system in this regard. 33 It incorporates patient characteristics such as age, vascular risk factors, and imaging findings. Patients with a high RoPE score (⩾ 7) are more likely to have a PFO-related stroke and therefore have greater potential benefit from PFO closure. The RoPE score is inherently age-dependent, meaning older patients consistently receive lower scores, with a maximum achievable score of 6 for individuals over 60 years. However, it is informative to see whether even older patients with typically lower RoPE scores still benefit from PFO closure. In this regard, two studies conducted observed-versus-expected risk assessments using the RoPE score to evaluate PFO closure in older patients.11,30 Notably, the mean RoPE score in both these studies was 4.6 ± 1.1. Wintzer-Wehekind et al. found that older patients undergoing PFO closure had a significantly lower occurrence of ischemic events (5.5%) over 2 years compared to their expected risk based on the RoPE score (10.6%, p < 0.001). This beneficial finding was supported by Alperi et al., who reported an even lower observed event rate of 3.3%.
The anatomical and functional characteristics of a PFO are also crucial clinical markers for patient selection, differentiating between high-risk and low-risk PFOs to guide closure decisions. 34 Lee et al. 25 compared 130 patients ⩾ 60 years with a history of cryptogenic stroke who underwent PFO closure to 130 matched patients on antithrombotic therapy. Over a median follow-up of 3.9 years, PFO closure was significantly associated with a reduced risk of the composite endpoint of ischemic stroke or TIA (p = 0.012), although it did not show a significant benefit for ischemic stroke alone (p = 0.107). However, for high-risk PFOs, closure significantly lowered both the composite endpoint (p = 0.006) and recurrent ischemic stroke (p = 0.035). Findings from Lee et al. suggest that while PFO closure may reduce recurrent embolic events in older patients, its effectiveness in preventing recurrent ischemic stroke largely depends on the presence of high-risk PFO features. This notion is further supported by a recent subgroup analysis by Wang et al., 9 which found that PFO closure significantly reduced risk only in older patients with high-risk PFO features (p = 0.03) but not in those without such features (p = 0.08).
Another useful scoring system is PFO-Associated Stroke Causal Likelihood (PASCAL) classification system, which combines the RoPE score items with high-risk anatomical features of PFO. However, a limitation of the PASCAL score is its applicability to older patients; it is not suitable for individuals over 60 years, as their maximum possible RoPE score is six, falling below the PASCAL system’s RoPE cutoff of seven for attributing PFO-related risk. Therefore, there is a need to develop or validate alternative scoring systems or diagnostic algorithms that are specifically tailored for older adults with cryptogenic stroke and PFO or to refine the PASCAL system itself to account for age-related differences in RoPE scores and PFO characteristics. 9 This would ensure that treatment decisions for PFO-associated stroke in patients over 60 are based on appropriate risk stratification tools.
The observation of lower mortality in the PFO closure group compared to ATA in our study warrants further discussion. The lower mortality observed in the PFO closure group is likely attributable to residual or unmeasured confounding rather than a direct therapeutic benefit of the closure procedure. This is primarily because the inclusion of only three retrospective studies and a small sample size limits the generalizability of these findings and introduces random variation. More specifically, the observed difference may be an artifact of age-related disparities, as the patients selected for PFO closure are often younger and healthier, a finding supported by the study by Poli et al. 24 In addition, our findings pertain to all-cause mortality, which may not accurately capture the specific effect of the intervention itself. This limitation is underscored by the fact that key confounders, such as stroke severity and cause of death, were not consistently collected across the included studies. For example, in the analysis by Lee et al., a favorable HR for all-cause mortality contrasted with an unfavorable HR for vascular death, suggesting that unmeasured factors beyond the intervention itself were influencing outcomes.
Regarding the safety of PFO closure in older patients, Merkler et al. 10 reported a significantly higher rate of in-hospital complications among older patients compared with patients < 60 years (10.9% vs 4.9%). In contrast, our meta-analysis found no significant difference in risk of in-hospital complications including periprocedural AF, cardiac adverse events, deep vein thrombosis or peripheral embolism, and minor or major vascular complications. However, the risk of new-onset AF in the follow-up was higher among ⩾ 60 years compared with < 60 years (4.8% vs 1%). This suggests that while PFO closure may be safe in the short term, long-term cardiovascular risks, particularly AF, could have important implications for post-procedural management in older patients. Therefore, we recommend routine post-procedural AF monitoring in older patients, such as ambulatory electrocardiogram (ECG), to facilitate early detection and prompt intervention for new-onset AF.
Several methodological limitations warrant discussion when interpreting our findings. A key concern is the heterogeneity in the clinical definition of a “cryptogenic” event across the studies, which may introduce variability and influence the pooled results. A significant and related confounding factor is the lack of standardized guidance on long-term antithrombotic medication following PFO closure. Since the duration and type of post-procedural medical therapy (dual antiplatelet, single antiplatelet, or anticoagulant therapy) were not consistently reported, variations in long-term treatment may have a substantial, unmeasured impact on recurrence rates and could confound the true effect of the PFO closure procedure itself.
In addition, differences in baseline characteristics, comorbidities, and RoPE scores could affect the observed effectiveness of PFO closure, warranting cautious interpretation of the findings. Despite contacting all corresponding authors of the included studies to obtain individual patient data, we were ultimately unable to acquire this information from all authors—a limitation common to many meta-analyses. Therefore, due to insufficient data, we were unable to perform a subgroup analysis based on RoPE scores or high-risk PFO features. This data limitation also extended to antithrombotic regimens, precluding a meaningful comparison between antiplatelet and anticoagulation therapies—a critical distinction, as recent evidence suggests that direct oral anticoagulants may provide greater benefits than antiplatelet therapy in patients with embolic stroke of undetermined source and PFO. 35 Also, the comparison between different occluder devices was not feasible. Finally, the long-term comparison between PFO closure and ATA requires further investigation, as available data remain limited. Notably, the mean follow-up time across studies was 5.6 years, which is not sufficient to assess the long-term effects of PFO closure.
Conclusion
Our analysis suggests that PFO closure may reduce the risk of recurrent ischemic stroke in patients aged ⩾ 60 years, particularly those with high-risk anatomical features. However, the evidence base remains predominantly observational and is influenced by studies with limited clinical detail, underscoring the need for more rigorous, age-specific research. Current scoring systems such as PASCAL are limited in their applicability to older adults, highlighting the need for refined risk stratification tools tailored to this population.
Further long-term, randomized trials are essential to fully understand the long-term effects of PFO closure and optimize management strategies in this complex and often underrepresented population. Encouragingly, it has been shown that the sample size for RCTs involving older patients with high-risk PFO is feasible, 36 and at least two ongoing trials, CLOSE-2 37 and STOP trials, 38 are currently recruiting older participants. CLOSE-2 is a three-arm trial comparing PFO closure to antiplatelet therapy alone and oral anticoagulant therapy to antiplatelet therapy in patients aged 60–80 years with high-risk PFO, and the ongoing STOP trial compares PFO closure to optimal medical treatment (left at the discretion of the investigator) without requiring large shunt or ASA and with no upper age limit.
Supplemental Material
sj-docx-1-wso-10.1177_17474930251392333 – Supplemental material for Outcomes of patent foramen ovale closure in patients over 60 years with cryptogenic stroke: A systematic review and meta-analysis
Supplemental material, sj-docx-1-wso-10.1177_17474930251392333 for Outcomes of patent foramen ovale closure in patients over 60 years with cryptogenic stroke: A systematic review and meta-analysis by Seyed Behnam Jazayeri, Cattien Phan, Sherief Ghozy, Soumya Ravichandran, Ana H Maleki, Oana M Dumitrascu, Kunal Agrawal, Royya Modir, Thomas Hemmen, Sven Poli, Christine S Zuern, Daniela Trabattoni, Dawn Meyer, Brett Meyer and Bavarsad Shahripour in International Journal of Stroke
Footnotes
Acknowledgements
None.
Author contributions
R.B.S. conceived the study and supervised the project. C.P. wrote the search strategy. S.B.J., C.P., and S.R. extracted data and assessed the quality of studies. S.G., A.H.M., and S.B.J. participated in data analysis. S.P., C.S.Z., and D.T. participated in the acquisition of raw data and revising the manuscript. S.B.J. and O.M.D. wrote the first draft. K.A., R.M., T.H., D.M., and B.M. participated in critical revision of the article for important intellectual content. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Informed consent
Not applicable.
Data availability
The data will be made available upon reasonable request from the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
