Abstract
Background:
The clinical utility of implantable cardiac monitors (ICMs) for atrial fibrillation (AF) detection following cryptogenic stroke or embolic stroke of undetermined source (ESUS) is well established. However, the optimal timing for ICM implantation to maximize diagnostic yield remains uncertain.
Aims:
To systematically review the literature and conduct a meta-analysis to determine whether earlier ICM implantation after cryptogenic stroke or ESUS ischemic stroke improves detection rates and reduces the time to AF diagnosis.
Summary of review:
A comprehensive search of PubMed, Embase, and Cochrane CENTRAL was conducted from inception to June 2025, without language restrictions. References of retrieved articles and relevant reviews were manually searched. We included observational studies or randomized trials reporting ICM use in patients with ESUS or cryptogenic stroke/transient ischemic attack (TIA), providing data on AF detection rates and/or timing metrics (stroke-to-ICM interval, ICM-to-AF interval). Two reviewers independently screened studies and extracted data. Disagreements were resolved by consensus or third-party adjudication. Data were extracted following Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines. Study-level AF detection rates were modeled using logit-transformed proportions and pooled using random-effects models (REML). Mixed-effects meta-regressions assessed the effect of timing (stroke-to-ICM interval) on AF detection and diagnostic delay, adjusting for ICM monitoring duration. The primary outcomes were pooled AF detection rate and mean time from ICM implantation to AF diagnosis. Timing of implantation was assessed as a continuous and categorical (early, intermediate, delayed) variable. Forty-seven studies (N = 6918 patients) were included. The pooled AF detection rate was 27.3% (95% CI: 24.6–30.2), with substantial heterogeneity (I2 = 80.8%). Early ICM implantation (<31.5 days from index event) was associated with a higher AF detection rate compared with delayed implantation (30.0% vs 23.7%; p = 0.0017), independent of monitoring duration. Stratified meta-regression confirmed that delayed implantation was associated with lower AF detection even after adjusting for ICM duration. For each additional day of delay in ICM implantation, the time from AF diagnosis increased by an additional 0.32 days on average, even after accounting for monitoring duration (p = 0.0007).
Conclusion:
These findings suggest that earlier ICM implantation enhances AF detection after ESUS or cryptogenic stroke and shortens diagnostic delay. Optimizing timing of post-stroke monitoring may improve patient selection for anticoagulation and reduce recurrent stroke risk.
CRD 420251064227.
Introduction
Covert atrial fibrillation (AF) is considered one of the most important underlying mechanisms in patients with cryptogenic or embolic stroke of undetermined source (ESUS). 1 Indeed, AF can be detected in almost 30% of cryptogenic or ESUS patients during long-term follow-up as shown by previous randomized controlled trials of prolonged cardiac monitoring like the CRYSTAL-AF(Study of Continuous Cardiac Monitoring to Assess Atrial Fibrillation After Cryptogenic Stroke), 2 EMBRACE (30-Day Cardiac Event Monitor Belt for Recording Atrial Fibrillation After a Cerebral Ischemic Event), 3 and Find-AFRANDOMISED (A Prospective, Randomized, Controlled Study to Determine the Detection of Atrial Fibrillation by Prolonged and Enhanced Holter Monitoring as Compared to Usual Care in Stroke Patients). 4 Furthermore, previous meta-analyses have also demonstrated the clinical utility of prolonged rhythm surveillance using implantable cardiac monitors (ICMs), showing improved AF detection rates compared with conventional follow-up. 5 However, despite the increasing adoption of ICMs in post-stroke care pathways, the optimal timing of device implantation remains unclear. This systematic review and meta-analysis aimed to synthesize available evidence on ICM use after cryptogenic or ESUS ischemic stroke or transient ischemic attack (TIA), with a specific focus on the timing of implantation. We assessed whether the delay between the index cerebrovascular event and ICM implantation influences AF detection rates and the time to AF diagnosis.
Methods
Study design
This systematic review and meta-analysis was registered on the International Prospective Register of Systematic Reviews (PROSPERO CRD420251064227) and reported in accordance with the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) statement guidelines. 6 The objectives of this analysis were to evaluate whether earlier implantation of ICM after a cryptogenic or ESUS stroke is associated with higher rates of AF detection and whether early implantation facilitates earlier identification of AF compared with delayed implantation of ICM. The clinical questions were formulated according to the Population–Intervention–Comparison–Outcome (PICO) framework, as reported in Supplemental Table 1.
Standard protocol approvals, registrations, and patient consents
This systematic review and meta-analysis was conducted in accordance with ethical standards for research involving human participants. Ethical approval for the conduct of this study was not required, as all data were obtained from previously published studies. For all included studies, approval by institutional or regional ethics committees was presumed based on original publications. Because this analysis did not involve direct patient contact or identifiable individual-level data, patient consent was not required. No images or other identifiable materials requiring disclosure consent were included.
Data source and search strategy
We systematically searched for peer-reviewed reports in PubMed, Embase, and Cochrane Central Register of Controlled Trials (CENTRAL) from inception to 12 June 2025. The search strategy included a combination of synonyms and MeSH/Emtree terms related to insertable cardiac monitors (“loop recorder,” “implantable loop recorder,” “insertable cardiac monitor,” ILR, ICM), ischemic or cryptogenic stroke (“ischemic stroke,” “cryptogenic stroke,” “embolic stroke of undetermined source,” ESUS), atrial arrhythmias (“atrial fibrillation,” “atrial flutter,” “subclinical atrial fibrillation”), and monitoring concepts (“monitoring,” “detection,” “diagnosis”), adapted to each database’s indexing system. We, in addition, employed a snowballing approach by manually screening the reference lists of the included studies and relevant previously published review articles. The detailed search strategy is available in Supplemental Table 2.
Eligibility criteria
For the purpose of this systematic review and meta-analysis, we included studies that met all the following eligibility criteria: (1) enrolling patients ⩾18 years of age with cryptogenic or ESUS stroke or TIA; (2) randomized controlled trials, non-randomized controlled trials, cohort studies, case series; (3) reported the variables and outcome of interests; we excluded studies involving pregnant patients and studies from which key variables of interest for the ESUS or cryptogenic stroke/TIA population could not be extracted.
Quality assessment
The risk of bias for the included observational studies was evaluated using the Newcastle–Ottawa Scale (NOS) for cohort studies (LD and GP). This tool assesses the methodological quality of non-randomized studies across three domains: selection of the study groups (maximum four stars), comparability of the cohorts (maximum two stars), and ascertainment of the outcome (maximum three stars). For the selection domain, we evaluated whether the patient population was representative of real-world cryptogenic stroke or ESUS cohorts, whether AF was ruled out at baseline, and whether ICM exposure (i.e., device type and implantation method) was clearly described. For comparability, we awarded stars to studies that adjusted for at least one key confounder (e.g., age, CHA2DS2-VASc, NIHSS, or monitoring duration), and a second star if multiple or advanced statistical adjustments were reported. The outcome domain assessed whether AF diagnosis was based on reliable and pre-specified criteria, whether follow-up duration was adequate (at least 6 months on average), and whether loss to follow-up was minimal (<20%) and clearly reported. The NOS assessment was performed independently by two reviewers (GP and LD), and discrepancies were resolved by consensus.
Outcomes, data extraction
The outcomes of interest were as follows: (1) the proportion of AF detected by ICMs following ESUS/cryptogenic ischemic stroke or TIA; (2) the time from stroke/TIA to ICM implantation; and (3) the time from ICM implantation to AF detection. Data extraction was independently conducted by two investigators (LD and GP) using a standardized data collection form. Any discrepancies were resolved through discussion, and a third reviewer (MF) was consulted in case of persistent disagreement. From each included study, the following baseline information was extracted: study design and country, sample size, publication year, patient characteristics (including mean or median age, sex distribution, CHA2DS2-VASc score, CHADS2 score, NIHSS score), device model, duration of ICM monitoring, AF detection rate, stroke recurrence, and follow-up duration.
Statistical analysis
All statistical analyses were conducted using R version 4.3.2, primarily utilizing the metafor and dplyr packages. A random-effects meta-analysis was performed using the restricted maximum likelihood (REML) estimator to account for between-study heterogeneity. Proportions of AF detection following ESUS or cryptogenic ischemic stroke/TIA were calculated using logit-transformed proportions (PLO) and pooled using inverse-variance weighting. Heterogeneity was assessed using τ2 (between-study variance), I2 (the proportion of total variability attributable to heterogeneity), and R2 (the proportion of heterogeneity explained by moderators). Sensitivity analyses included leave-one-out procedures to assess the influence of individual studies on the overall estimates, and Egger’s test was used to evaluate small-study effects. Funnel plots were visually inspected to explore potential publication bias. To evaluate the potential impact of unmeasured confounding on the association between the timing of insertable cardiac monitor implantation and AF detection, we calculated E-values for selected variables, including age, female sex, and CHA2DS2-VASc score. To examine the impact of timing on AF detection, mixed-effects meta-regression models were fitted with the time from stroke/TIA to ICM implantation as the primary exposure. Timing was modeled both as a binary variable (early vs delayed) and as categorical based on tertiles across studies. All models were adjusted for ICM monitoring duration and included as a continuous covariate. A separate meta-regression was also conducted to evaluate whether the time from ICM implantation to AF detection was influenced by the delay in ICM implantation, adjusting for monitoring duration. When medians were reported instead of means for key variables (e.g., time from stroke to ICM or time to AF detection), values were converted to approximate means using established transformation methods (e.g., the Wan method), assuming near-symmetrical distributions.
Results
The systematic literature search initially identified 2096 records. After removing 593 duplicates, 1563 titles and abstracts were screened, leading to the exclusion of 838 records that did not meet inclusion criteria. A total of 725 full-text articles were assessed for eligibility, of which 678 were excluded for reasons such as inadequate outcome reporting or overlapping populations. Ultimately, 47 studies were included in the final quantitative synthesis. The study selection process, including reasons for exclusion at each stage, is illustrated in the PRISMA flow diagram (Supplemental Figure 1).
Baseline characteristics of the included studies
Supplemental Table 3 summarizes the characteristics of the 47 studies included in our analysis, enrolling 6918 patients who underwent ILR monitoring after ischemic stroke/TIA. Most were single-center observational studies, either prospective or retrospective in design, while a subset were multicentre cohorts conducted across various countries including the United States, Japan, Italy, Germany, the United Kingdom, France, and others. The studies spanned publication years from 2013 to 2025, reflecting a growing interest in post-stroke cardiac monitoring over the past decade. A wide range of ICMs were used across studies, with the most common devices being the Reveal LINQ and Reveal XT (Medtronic). Other ICMs included Confirm Rx (Abbott) and various models of the Biomonitor series (Biotronik). Supplemental Table 4 presents baseline characteristics from 47 included studies reporting on patients with ESUS or cryptogenic stroke/TIA undergoing ICM implantation. The number of patients per study ranged from 22 to 500. AF detection rates varied between 10% and 70%. The time from index event to ICM implantation ranged from 3 days to more than 800 days, and the time from ICM implantation to AF detection ranged from 1 day to more than 500 days, where reported. Follow-up durations ranged from 221 to 1603 days. Reported mean or median ages ranged from 60 to 76 years. The proportion of female patients ranged from 18% to 76%. CHA2DS2-VASc scores were reported in 27 studies and NIHSS scores in 13 studies. Stroke recurrence data were available in 18 studies, with reported rates ranging from 0% to 21%. Moreover, definitions of AF varied across studies, with most adopting a minimum episode duration of ⩾2 min, while others used shorter (e.g., >30 s) or longer (e.g., ⩾5 min) thresholds.
Pooled proportion of atrial fibrillation detection across studies
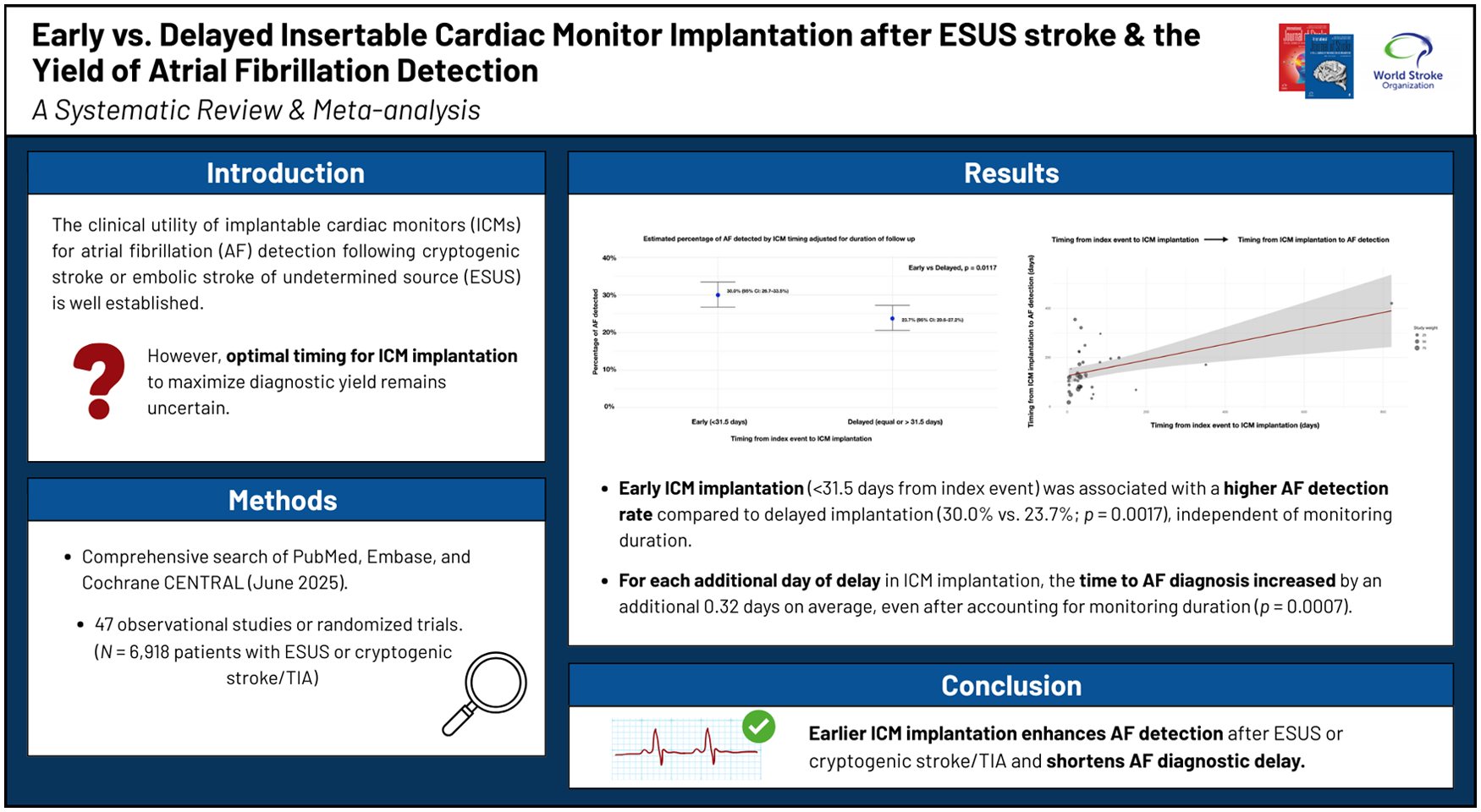
We performed a random-effects meta-analysis to estimate the overall proportion of AF detected by ICM in patients after a cryptogenic stroke or ESUS index event. A total of 47 studies were included, enrolling 6918 patients who underwent ILR monitoring after ischemic stroke. Across these studies, 1873 patients were diagnosed with AF. The pooled AF detection rate was estimated at 27.3% (95% CI: 24.6–30.2). Between-study heterogeneity was considerable, with an I2 of 80.8%, suggesting substantial clinical and methodological differences among the included studies (Figure 1). Individual study estimates varied widely, ranging from 10.3% (Toyoda et al. 7 ) to 70.0% (Cuadrado-Godia et al. 8 ) Several studies reported AF detection rates above 40%, including Melis et al.,9,10 (45.7%), Gambino et al. (41.7%), and Kitsiou et al. 11 (41.5%). Conversely, detection rates were below 15% in studies such as Toyoda et al. 7 (10.3%) and Yushan et al. 12 (12.0%). We performed a leave-one-out sensitivity analysis to assess the influence of individual studies on the overall estimate (Supplemental Figure 2). The pooled AF detection proportion remained stable across iterations, and no single study had a disproportionate effect on the overall results or heterogeneity estimates, confirming the robustness of the findings. The results remained highly consistent across iterations, with predicted AF detection proportions ranging narrowly from 26.7% to 27.9%, and confidence intervals showing minimal variation. Visual inspection of the funnel plot showed no clear asymmetry, and Egger’s test did not provide statistically evidence of small-study effects (z = −0.69, p = 0.490; Supplemental Figure 3).
Forest plot of atrial fibrillation (AF) detection rates across the 47 included studies. Each point represents the proportion of patients with AF detected by insertable cardiac monitor (ICM) during follow-up. Horizontal lines denote 95% confidence intervals. Studies are ordered by AF detection rate.
Timing from index event to ICM implant and AF detection
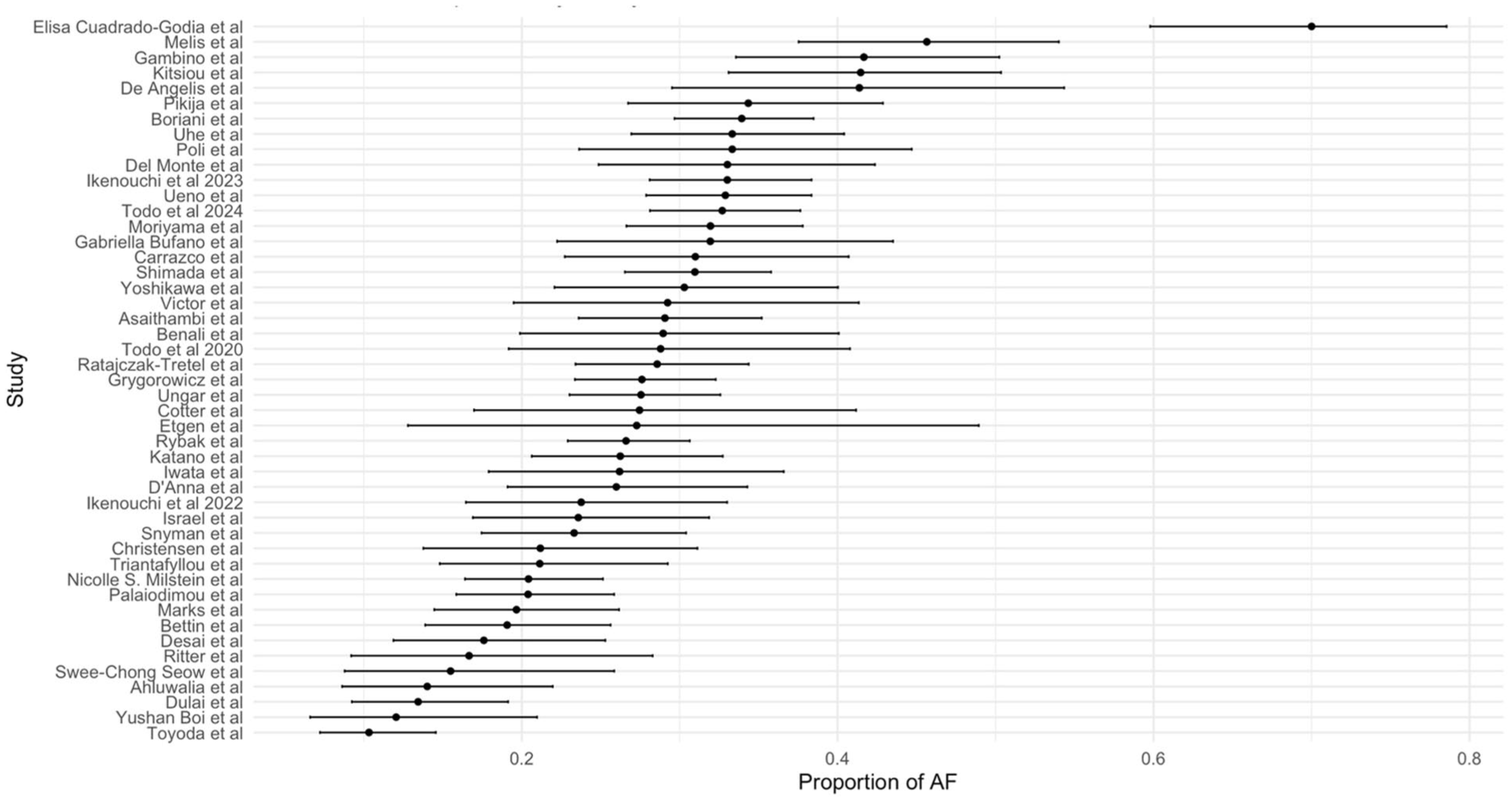
We conducted a mixed-effects meta-regression to investigate whether the timing of ICM implantation—categorized as early versus delayed—was associated with AF detection rates, while adjusting for the duration of ILR monitoring. The grouping was based on the median time from the index stroke/TIA to ICM implantation across studies, which was 31.5 days. Studies with a median ILR implantation time below 31.5 days were classified as the early group, whereas those with a median time equal to or greater than 31.5 days were classified as the delayed group. This analysis included 45 studies with complete data as two studies13,14 did not report the duration of the ICM monitoring. We found that the median ICM monitoring duration was longer in the delayed group (707 days, IQR: 592–974) compared with the early group (382, IQR: 364–465) (p < 0.001). The meta-regression analysis adjusted for the duration of the ICM monitoring showed that timing of ICM implantation emerged as a predictor of AF detection (β = −0.3198, p = 0.0117), indicating that delayed implantation was associated with lower log-odds of AF detection compared with early implantation, even after adjusting for monitoring duration (Figure 2). The model explained 41.95% of the between-study heterogeneity (R2 = 41.95%) and reduced residual heterogeneity, with an I2 of 80.84%. Based on model predictions at the mean ICM monitoring duration, the estimated percentage of AF detection was 30.0% (95% CI: 26.7–33.5) in the early group and 23.7% (95% CI: 20.6–27.2) in the delayed group (Figure 2). There were no differences in the distribution or proportions of key variables between the early and delayed ICM groups, including age, sex, and CHA2DS2-VASc score. See supplemental results for additional analysis. To assess the potential impact of unmeasured confounding by these variables, we calculated E-values for each. The E-value for age was 1.16, for female sex was 1.60, and for CHA2DS2-VASc score was 1.27. The E-value for CHA2DS2-VASc marginally exceeded the commonly used threshold of 1.25.
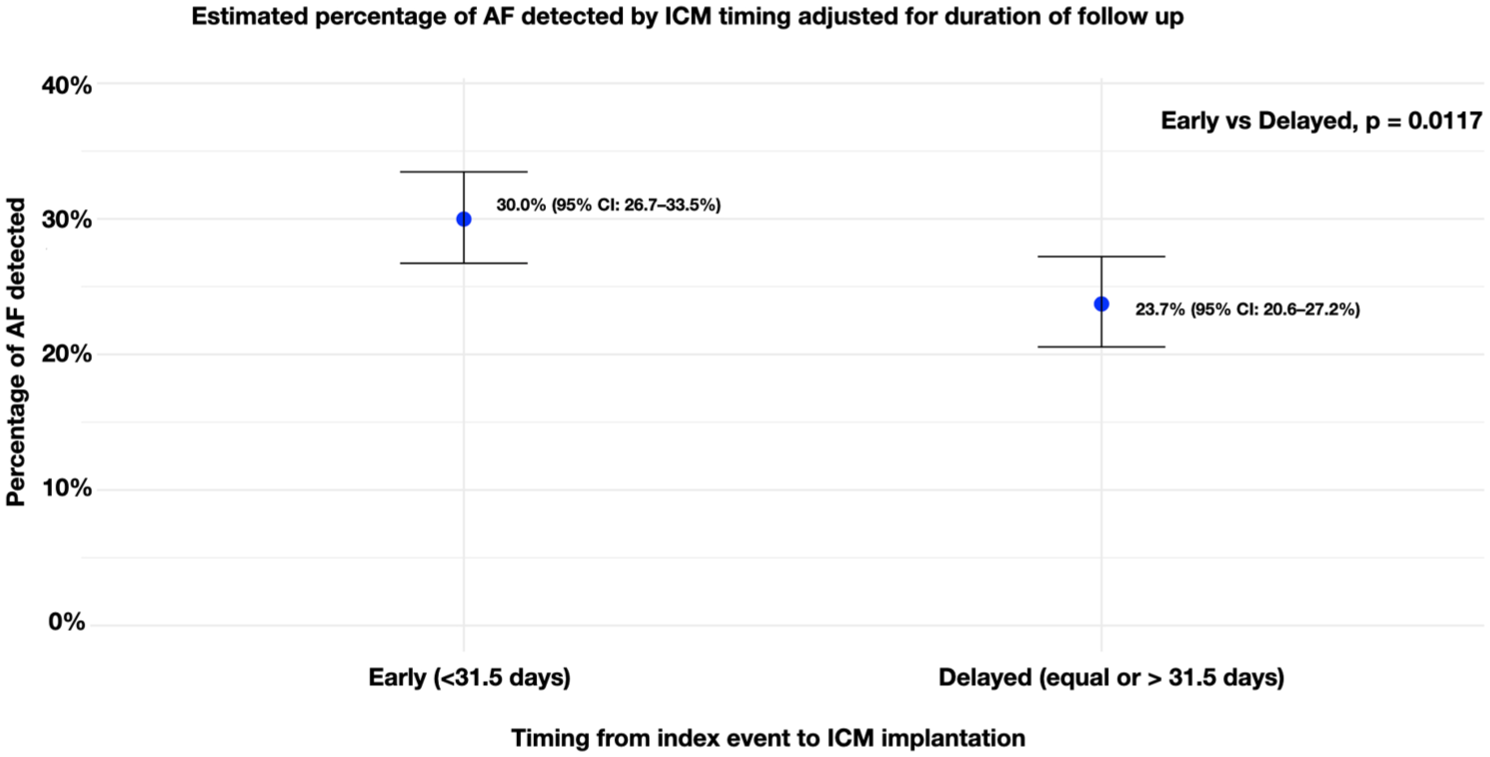
Pooled proportion of AF detection by timing of ICM implantation (early vs delayed), adjusted for monitoring duration. Patients receiving ICM implantation within 31.5 days from the index event had a higher AF detection rate compared with those with delayed implantation (30.0% vs 23.7%, p = 0.0117).
To further investigate the association between the timing of ICM implantation and AF detection, we categorized the interval between the index event and ILR implantation into three groups based on tertiles of the distribution across the 45 included studies (two studies were excluded from this analysis as they did not report ICM monitoring duration). Specifically, studies with a median time from stroke to ILR implantation of less than 27.7 days were categorized as early, those between 27.7 and 62.3 days as intermediate, and those with more than 62.3 days as delayed. We observed a gradual increase in the median duration of monitoring from early to delayed implantation (387, 510, and 569 days, respectively). We then conducted a mixed-effects meta-regression including these three timing categories as dummy variables and adjusting for ILR monitoring duration. Based on model predictions at the median ICM monitoring duration, the estimated percentage of AF detection was 30.8% (95% CI: 26.8–35.2) in the early group, 28.5% (95% CI: 24.7–32.7) in the intermediate group, and 22.0% (95% CI: 18.6–25.8) in the delayed group (Figure 3). These estimates were adjusted for ICM duration using a random-effects meta-regression model with logit-transformed proportions and REML estimation. In the first model, using early implantation as the reference group, the delayed group was associated with lower log-odds of AF detection (β = −0.459, 95% CI: −0.752 to −0.166, p = 0.0022), while the intermediate group showed no difference (β = −0.111, 95% CI: −0.389 to 0.169, p = 0.438). Longer ICM monitoring duration was independently associated with increased AF detection (β = 0.0011, p < 0.0001). The model explained 45.8% of the between-study heterogeneity (R2 = 45.8%), with substantial residual heterogeneity (I2 = 79.5%).
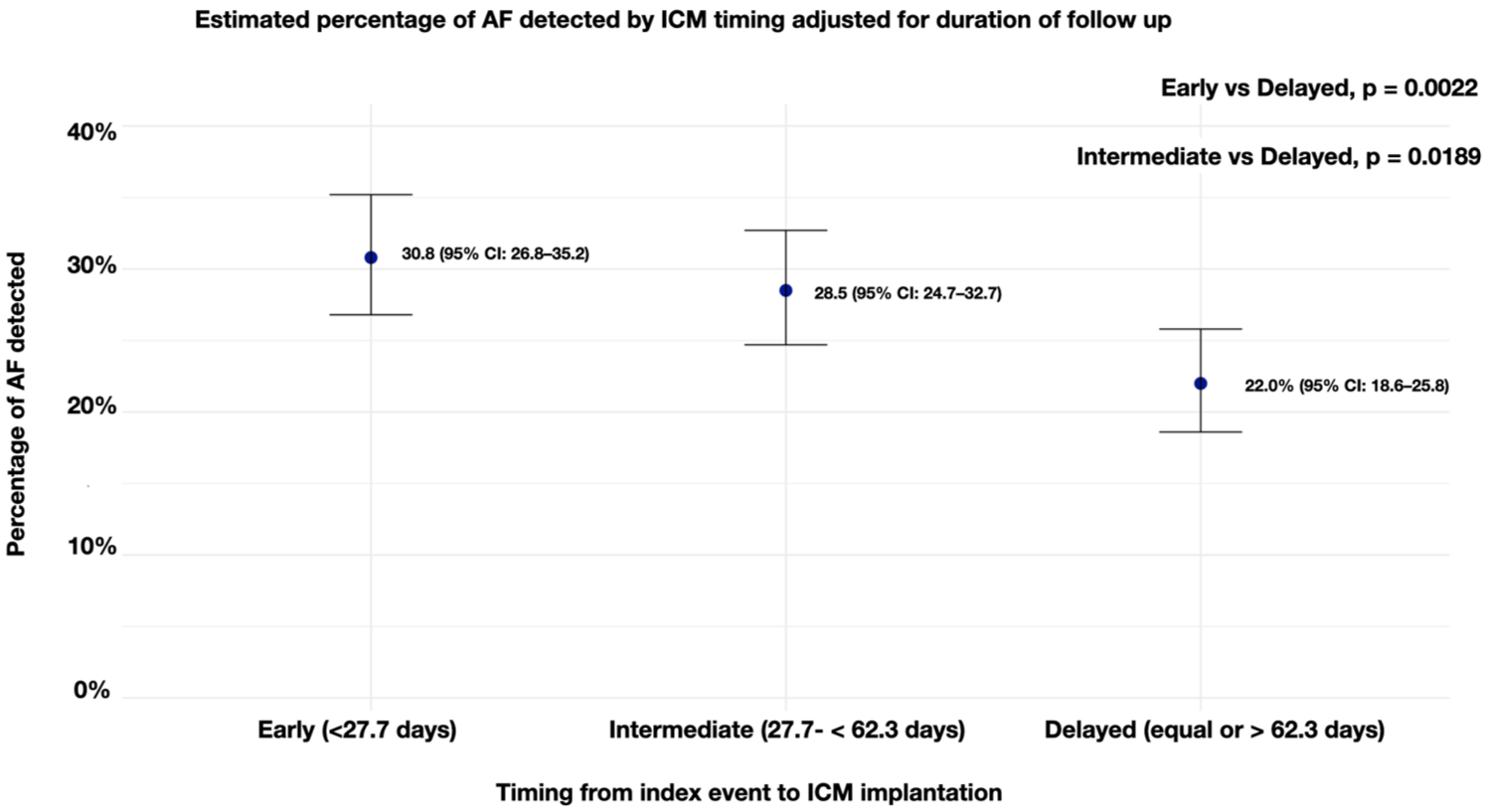
Pooled proportion of AF detection stratified by tertiles of ICM implantation timing, adjusted for monitoring duration. The detection rate was highest in the early group (<27.7 days, 30.8%) and progressively declined in the intermediate (27.7–62.3 days, 28.5%) and delayed (⩾62.3 days, 22.0%) groups. Early and intermediate groups both showed higher detection rates compared with the delayed group (p = 0.0022 and p = 0.0189, respectively).
Association between timing of ICM implantation and AF detection delay
To explore whether the timing of ICM implantation influences the time to AF detection, we conducted a meta-regression with time from ICM implant to AF detection as the outcome, and time from index event to ICM implant as a continuous predictor, adjusting for the duration of ICM monitoring. The model showed a statistically positive association between later ICM implantation and longer time to AF detection (β = 0.32; 95% CI: 0.14–0.51; p = 0.0007), even after adjusting for ICM monitoring duration. This suggests that each day of delay in ICM implantation was associated, on average, with an additional 0.32 days until AF diagnosis. The amount of heterogeneity explained by the model was moderate (R2 = 23.1%), while residual heterogeneity remained high (I2 = 100%) (Figure 4).
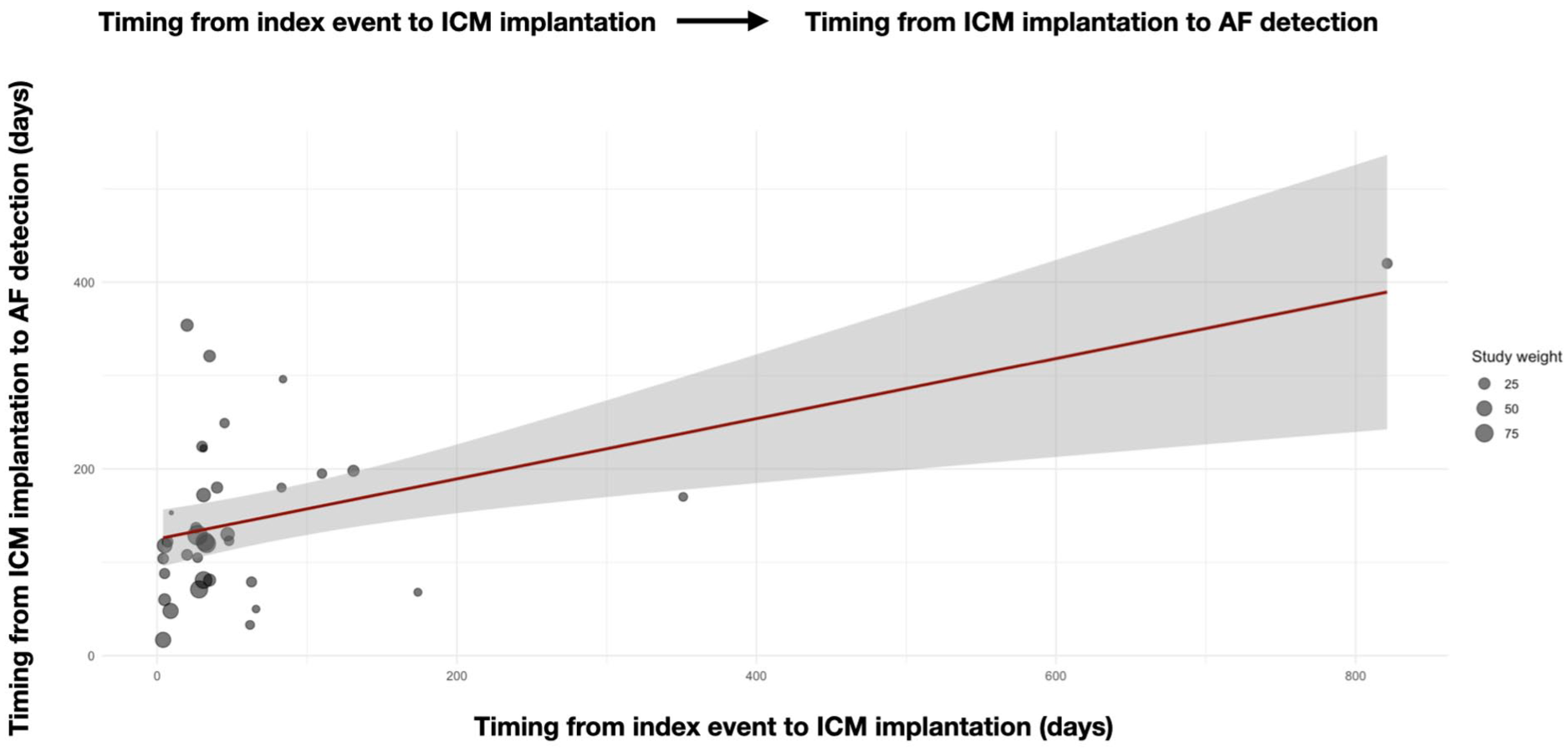
Bubble plot of meta-regression assessing the association between timing of ICM implantation and time to AF detection. Each bubble represents a study, with size proportional to its weight in the meta-regression. Later ICM implantation was associated with longer time to AF diagnosis (β = 0.32; 95% CI: 0.14–0.51; p = 0.0007), even after adjustment for ICM monitoring duration.
Risk of bias
Overall, the methodological quality of the included studies was moderate to high. According to the Newcastle–Ottawa Scale (NOS), total scores ranged from 4 to 9 out of a maximum of nine stars. Most studies received full scores in the selection domain, indicating good representativeness of the ESUS/cryptogenic population and adequate description of ICM procedures. However, the comparability domain revealed greater variability, with several studies failing to adjust for important confounders. The outcome domain was generally well addressed, with most studies providing sufficient follow-up (⩾6 months) and clear criteria for AF diagnosis. A graphical summary of NOS domain scores across studies is presented in Supplemental Figure 4. Based on this assessment, the overall risk of bias was judged to be low to moderate.
Discussion
Our systematic review and meta-analysis, which included 47 studies and 6918 patients with ESUS or cryptogenic TIA/stroke, demonstrates that the timing of ICM implantation influences the diagnostic yield for AF detection. Indeed, we showed that earlier implantation of ICMs following the index cerebrovascular event was associated with higher AF detection rates and shorter times to diagnosis. These findings might support the notion that delays in initiating prolonged cardiac monitoring may reduce the clinical yield of ICMs and potentially defer opportunities for preventive anticoagulation.
One of the major elements of novelty of our analysis is that when stratifying studies based on the timing of ICM implantation—defined as early (<31.5 days from index event) vs delayed (⩾31.5 days)—we observed a higher pooled AF detection rate in the early group. Building on this, our stratified meta-regression using tertiles of implantation timing further reinforced the importance of early ICM placement. When categorizing studies into early (<14 days), intermediate (14–34 days), and delayed (⩾35 days) implantation groups and adjusting for ICM monitoring duration, we found that delayed implantation was consistently associated with lower AF detection rates even when compared to the intermediate group. In interpreting these findings, it is also important to consider the potential influence of baseline cardiovascular risk profiles. Although initially developed for patients with atrial fibrillation, the CHA2DS2-VASc score has been increasingly recognized as a marker of atrial cardiopathy and an independent predictor of new-onset AF in cryptogenic stroke or ESUS populations undergoing prolonged monitoring. Its inclusion as a potential confounder in our analysis reflects this broader predictive role beyond established AF. Moreover, while less invasive and less costly modalities such as prolonged Holter or external loop monitoring remain important for initial screening, further evidence consistently show that implantable loop recorders provide the highest diagnostic yield for delayed or paroxysmal AF.2,15 Therefore, our findings should be interpreted within the context of patients in whom ILR implantation is clinically indicated, suggesting that earlier initiation may maximize detection and shorten diagnostic delay.
Our findings are aligned with emerging pathophysiological and clinical insights suggesting that AF may be particularly active or detectable in the acute phase following a cerebrovascular event. 16 The concept of “atrial fibrillation detected after stroke” (AFDAS) has gained traction as a distinct clinical entity characterized by a temporally heightened vulnerability to arrhythmia in the days to weeks after stroke or TIA.17,18 In this context, a recent meta-analysis by Sposato et al. provided critical supporting evidence. 19 Their work included more than 7000 patients from both randomized trials and observational studies, 28% of them cryptogenic cases, undergoing serial ECGs, continuous inpatient ECG monitoring, continuous inpatient cardiac telemetry, in-hospital Holter monitoring, mobile cardiac outpatient telemetry, external loop recording, and implantable loop recording after an index event. The authors demonstrated that the odds of detecting AFDAS are over fivefold higher when continuous cardiac monitoring is initiated at a mean of 27 days after stroke than a mean of 75 days. Moreover, previous studies suggested that the early initiation of cardiac monitoring is crucial for improving atrial fibrillation diagnosis after stroke and TIA, with more than 70% of cases being diagnosed within the first 3 days after admission. 20 It is unknown whether the higher AF diagnostic yield immediately after a cerebrovascular event might be explained by the pathophysiology of AFDAS. However, deferring monitoring might lead to miss transient or paroxysmal episodes of AFDAS that occur predominantly in the immediate post-stroke period. 16 In our analysis, the higher detection rate of AF observed with early monitoring may also reflect the underlying stroke pathophysiology. When AF is causally related to the index ischemic event, it is often intermittently active and thus more likely to be captured shortly after stroke. This interpretation underscores that the timing of monitoring and the pre-test probability of cardioembolic stroke are intrinsically linked, highlighting the importance of patient selection and timing in optimizing AF detection after ESUS or cryptogenic stroke.
In our analysis, each day of delay in ICM implantation from the index event was associated with an average increase of 0.33 days in the time to AF detection, even after adjustment for monitoring duration. This result should not be interpreted in absolute temporal terms, as the pooled estimate reflects heterogeneous definitions of “early” versus “delayed” implantation across studies. Rather, it reinforces the broader concept that earlier initiation of cardiac monitoring—typically within the first weeks after stroke—is associated with a higher likelihood of detecting AF during the period of increased atrial vulnerability following the index event. This result might suggest that earlier implantation of the device may not only improve overall AF detection rates but also accelerate the time to diagnosis. As anticoagulation in stroke patients with AF is the most effective strategy for secondary stroke prevention, the present findings have potentially important clinical implications. 21 Earlier identification of AF may facilitate more timely initiation of anticoagulant therapy, thereby minimizing the period during which patients remain at risk of recurrent cardioembolic events. In contrast, delayed ICM implantation may hinder the detection of brief, early post-stroke arrhythmic episodes—many of which are asymptomatic—ultimately limiting opportunities for effective intervention. These results reinforce the importance of prompt cardiac rhythm monitoring as a key component of individualized secondary prevention strategies in patients with ischemic stroke of undetermined source.
As expected, longer ICM monitoring duration was independently associated with higher AF detection rates, consistent with the established advantage of prolonged rhythm surveillance in this population. This further supports the overall utility of ICMs in post-stroke care pathways and highlights the diagnostic yield associated with sustained monitoring over time.
A major challenge emerging from our analysis is the lack of a uniform definition for AF episode duration across studies. The threshold used to define AF varied substantially, ranging from >30 s 22 to ⩾3 min. 23 This inconsistency likely contributes to the wide variability in reported detection rates and limits the comparability of findings between cohorts. Establishing a standardized, clinically meaningful duration threshold for AF episodes is, therefore, essential to improve consistency in future research, facilitate meta-analytic synthesis, and ensure that AF detection after stroke is interpreted within a uniform diagnostic framework.
From a clinical standpoint, recent data from the ALONE-AF trial 24 further emphasize the importance of precise rhythm monitoring. In that study, continuation of NOAC therapy after apparently successful AF ablation did not confer additional benefit, underscoring the uncertainty surrounding the prognostic meaning of brief or intermittent AF episodes. When discontinuation of oral anticoagulant therapy is considered, incorporating regular rhythm monitoring to detect AF recurrence is crucial, given the poor correlation between post-ablation AF burden and symptoms. In the trial setting, continuous invasive monitoring remains the gold standard for post-ablation surveillance, although prolonged intermittent monitoring with ambulatory ECG devices represents a practical alternative. Although this context differs from post-stroke AF detection, it reinforces a common principle across clinical scenarios—namely, that consistent, high-resolution rhythm monitoring is key to informing individualized anticoagulation strategies.
Our study has several strengths. All patients included across the 47 studies (n = 6918) met established definitions of cryptogenic stroke or ESUS. This homogeneous population enhances the clinical relevance and interpretability of our findings. By synthesizing data from these studies, we were able to explore both absolute AF detection rates and time to diagnosis using multiple analytic approaches, including stratified and continuous meta-regressions adjusted for ICM monitoring duration. Our findings were robust across sensitivity analyses and supported by consistent patterns in both binary and continuous exposure models. Moreover, we applied established statistical methods for proportion meta-analysis, heterogeneity exploration, and effect modification, lending methodological rigor to our estimates. Despite these strengths, several limitations should be acknowledged. First, the included studies were mostly observational and heterogeneous in design, population characteristics, and reporting standards. Although we employed rigorous data extraction procedures and conducted multiple sensitivity analyses, residual confounding and potential publication bias cannot be entirely excluded. Second, in a subset of studies, key timing variables (e.g., stroke-to-ICM interval or ICM-to-AF detection) were reported as medians rather than means. While we applied validated transformation methods (e.g., Wan et al.) to approximate means from medians, such conversions may introduce estimation error, particularly in the context of skewed distributions. Third, data on anticoagulation initiation timing or uptake following AF diagnosis were inconsistently available, limiting our ability to evaluate the downstream clinical implications of early detection. Studies differed in how they defined and reported AF episodes—particularly with respect to minimal duration thresholds required for AF classification—which may have influenced AF detection rates and introduced heterogeneity across studies. An important consideration is that patients who underwent delayed ICM implantation may represent a selected subgroup of the initially eligible population—specifically, those who had not already experienced AF detection or recurrent stroke by the time of implantation. This introduces the possibility of attrition bias, whereby individuals at higher baseline risk are systematically excluded from later cohorts due to early clinical events. As a result, the lower AF detection rate observed in the delayed group may not solely reflect the effect of timing but could be partially driven by this depletion of higher-risk individuals over time. At the same time, such delays in ICM implantation likely reflect real-world clinical practice, as the majority of included studies were observational and conducted across diverse international settings. While this introduces some methodological heterogeneity, it also enhances the external validity of our findings and helps contextualize the observed variability in ICM timing. Together, these factors underscore the importance of early monitoring—not only to maximize diagnostic yield but also to avoid missing critical windows of AF vulnerability in the acute post-stroke phase. In addition, a broader selection bias may exist at the population level, as ICM implantation was generally offered to older patients with higher CHA2DS2-VASc scores or features suggestive of atrial cardiopathy. Although a few cohorts reported recurrent stroke, heterogeneity in outcome definitions and limited data on anticoagulation after AF detection precluded estimation of outcome-based metrics such as the number of patients who would need ICM implantation to prevent one recurrent stroke. Nevertheless, the higher detection rate observed with early monitoring may not solely reflect a temporal biological effect. It may also result, at least in part, from selection and attrition mechanisms, as patients with stroke-related AF are more likely to be identified shortly after the index event, while those at lower risk remain arrhythmia-free over time. This interpretation is consistent with the possibility of selection bias discussed above and supports the view that both biological and methodological factors may jointly contribute to the observed difference between early and delayed implantation. An additional limitation is the variability in sample size across the included studies, ranging from 22 to 500 patients. This imbalance may have contributed to the heterogeneity observed in the pooled estimates. However, rigorous data extraction procedures, the use of random-effects models, and multiple sensitivity analyses were applied to mitigate this issue, and the consistency of findings across methods supports the robustness of our conclusions. Most included cohorts had a mean age below 75 years, reflecting the demographic profile of ESUS populations typically enrolled in cardiac monitoring studies. Consequently, the generalizability of our findings to very elderly or frail patients may be limited, as age-related differences in arrhythmia burden, comorbidities, and competing risks could influence both AF detection and clinical outcomes. Finally, our meta-regression analyses relied on study-level aggregate data rather than individual patient data (IPD), which restricts the precision of effect estimates and limits the capacity to assess interaction effects or confounding at the patient level. Future IPD meta-analyses may help overcome these limitations and further clarify the temporal dynamics between monitoring initiation and clinical outcomes.
In conclusion, this study underscores that earlier ICM implantation following cryptogenic or ESUS acute ischemic stroke is associated with both higher detection rates and shorter time to diagnosis of AF. These findings highlight the importance of prioritizing early ICM implantation as an integral component of secondary stroke prevention strategies. Future studies are warranted to validate these associations and explore whether earlier AF detection translates into reduced stroke recurrence and improved patient outcomes.
Supplemental Material
sj-docx-1-wso-10.1177_17474930251404336 – Supplemental material for Early versus delayed insertable cardiac monitor implantation after ESUS stroke and the yield of atrial fibrillation detection: A systematic review and meta-analysis
Supplemental material, sj-docx-1-wso-10.1177_17474930251404336 for Early versus delayed insertable cardiac monitor implantation after ESUS stroke and the yield of atrial fibrillation detection: A systematic review and meta-analysis by Lucio D’Anna, Gabriele Prandin, Matteo Foschi, Samir Abu-Rumeileh, Francesco Favruzzo, Renato Simonetti, Lorenzo Barba, Giovanni Merlino, Francesco Bax, Arsalan Faiz, Markus Otto, Jianqun Guan, Asha Barnard, Lydia Jeffrey, Jake Dagan, Tsering Dolkar, Jonathan Hayton, Mariarosaria Valente, Gian Luigi Gigli, Simona Sacco, Maurizio Paciaroni, Arvind Chandratheva, Robert Simister, Soma Banerjee and Phang Boon Lim in International Journal of Stroke
Supplemental Material
sj-jpeg-5-wso-10.1177_17474930251404336 – Supplemental material for Early versus delayed insertable cardiac monitor implantation after ESUS stroke and the yield of atrial fibrillation detection: A systematic review and meta-analysis
Supplemental material, sj-jpeg-5-wso-10.1177_17474930251404336 for Early versus delayed insertable cardiac monitor implantation after ESUS stroke and the yield of atrial fibrillation detection: A systematic review and meta-analysis by Lucio D’Anna, Gabriele Prandin, Matteo Foschi, Samir Abu-Rumeileh, Francesco Favruzzo, Renato Simonetti, Lorenzo Barba, Giovanni Merlino, Francesco Bax, Arsalan Faiz, Markus Otto, Jianqun Guan, Asha Barnard, Lydia Jeffrey, Jake Dagan, Tsering Dolkar, Jonathan Hayton, Mariarosaria Valente, Gian Luigi Gigli, Simona Sacco, Maurizio Paciaroni, Arvind Chandratheva, Robert Simister, Soma Banerjee and Phang Boon Lim in International Journal of Stroke
Supplemental Material
sj-jpg-3-wso-10.1177_17474930251404336 – Supplemental material for Early versus delayed insertable cardiac monitor implantation after ESUS stroke and the yield of atrial fibrillation detection: A systematic review and meta-analysis
Supplemental material, sj-jpg-3-wso-10.1177_17474930251404336 for Early versus delayed insertable cardiac monitor implantation after ESUS stroke and the yield of atrial fibrillation detection: A systematic review and meta-analysis by Lucio D’Anna, Gabriele Prandin, Matteo Foschi, Samir Abu-Rumeileh, Francesco Favruzzo, Renato Simonetti, Lorenzo Barba, Giovanni Merlino, Francesco Bax, Arsalan Faiz, Markus Otto, Jianqun Guan, Asha Barnard, Lydia Jeffrey, Jake Dagan, Tsering Dolkar, Jonathan Hayton, Mariarosaria Valente, Gian Luigi Gigli, Simona Sacco, Maurizio Paciaroni, Arvind Chandratheva, Robert Simister, Soma Banerjee and Phang Boon Lim in International Journal of Stroke
Supplemental Material
sj-jpg-4-wso-10.1177_17474930251404336 – Supplemental material for Early versus delayed insertable cardiac monitor implantation after ESUS stroke and the yield of atrial fibrillation detection: A systematic review and meta-analysis
Supplemental material, sj-jpg-4-wso-10.1177_17474930251404336 for Early versus delayed insertable cardiac monitor implantation after ESUS stroke and the yield of atrial fibrillation detection: A systematic review and meta-analysis by Lucio D’Anna, Gabriele Prandin, Matteo Foschi, Samir Abu-Rumeileh, Francesco Favruzzo, Renato Simonetti, Lorenzo Barba, Giovanni Merlino, Francesco Bax, Arsalan Faiz, Markus Otto, Jianqun Guan, Asha Barnard, Lydia Jeffrey, Jake Dagan, Tsering Dolkar, Jonathan Hayton, Mariarosaria Valente, Gian Luigi Gigli, Simona Sacco, Maurizio Paciaroni, Arvind Chandratheva, Robert Simister, Soma Banerjee and Phang Boon Lim in International Journal of Stroke
Supplemental Material
sj-pdf-2-wso-10.1177_17474930251404336 – Supplemental material for Early versus delayed insertable cardiac monitor implantation after ESUS stroke and the yield of atrial fibrillation detection: A systematic review and meta-analysis
Supplemental material, sj-pdf-2-wso-10.1177_17474930251404336 for Early versus delayed insertable cardiac monitor implantation after ESUS stroke and the yield of atrial fibrillation detection: A systematic review and meta-analysis by Lucio D’Anna, Gabriele Prandin, Matteo Foschi, Samir Abu-Rumeileh, Francesco Favruzzo, Renato Simonetti, Lorenzo Barba, Giovanni Merlino, Francesco Bax, Arsalan Faiz, Markus Otto, Jianqun Guan, Asha Barnard, Lydia Jeffrey, Jake Dagan, Tsering Dolkar, Jonathan Hayton, Mariarosaria Valente, Gian Luigi Gigli, Simona Sacco, Maurizio Paciaroni, Arvind Chandratheva, Robert Simister, Soma Banerjee and Phang Boon Lim in International Journal of Stroke
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SB is a key opinion leader for RAPIDAI. RS is partially funded by the UCLH Biomedical Research Center. All other authors declare no conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Data availability
Data are available upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
