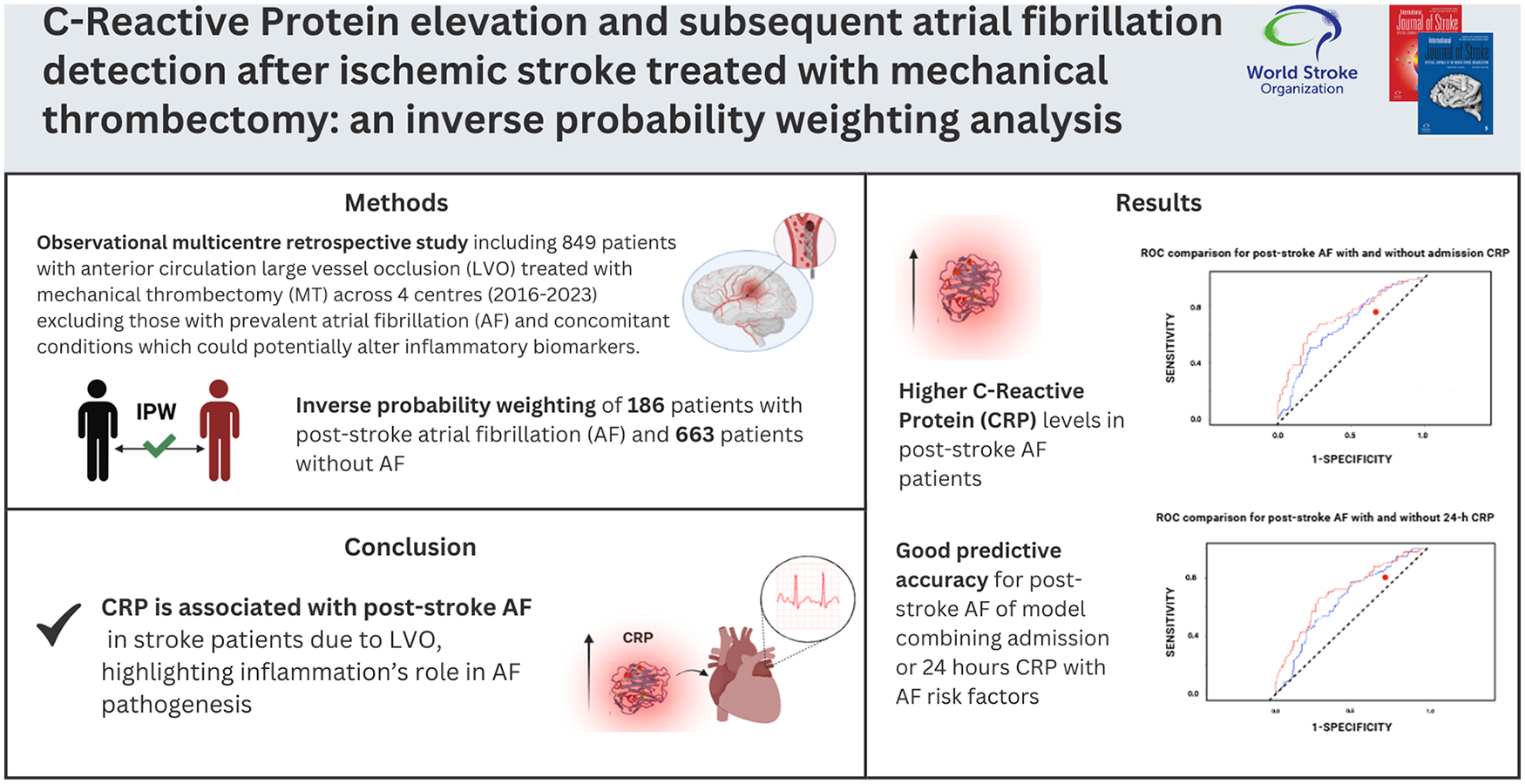
Abstract
Background and aims:
Mechanical thrombectomy (MT) is an effective treatment for acute ischemic stroke due to large vessel occlusion (LVO). Atrial fibrillation (AF) can be known before the stroke (or prevalent AF) or be newly detected after the stroke (post-stroke AF). Inflammation plays a critical role in the pathogenesis of post-stroke AF, making inflammatory markers valuable for early detection of post-stroke AF. This study investigated the predictive value of C-reactive protein (CRP) and other inflammatory biomarkers in predicting post-stroke AF in acute ischemic stroke patients treated with MT.
Methods:
This observational multicenter retrospective cohort study included 849 patients with anterior circulation LVO treated with MT across four centers from 2016 to 2023. Patients were divided into post-stroke AF and NO-AF groups, excluding those with prevalent AF. Baseline demographics, clinical and procedural variables, and inflammatory biomarkers, including CRP, were collected at admission and 24-h post-procedure. Baseline characteristics were balanced using inverse probability weighting (IPW). Logistic regression and receiver operating characteristic (ROC) analyses assessed the predictive value of CRP for post-stroke AF.
Results:
The study included 849 patients with a median age of 66 years (interquartile range (IQR) = 54–76) and 477 (56.2%) were female. Post-stroke AF was detected in 186 (21.9%) patients, while 663 (78.1%) did not experience AF during admission. In the weighted population, CRP levels, both admission and 24-h post-procedure, were higher in post-stroke AF patients. In logistic regression analysis, admission and 24-h CRP levels were associated with increased probability of post-stroke AF, respectively (odds ratio (OR) = 1.01; 95% confidence interval (CI) = 1.00–1.03, p < 0.001) and (OR = 1.02, 95% CI = 1.01–1.03, p < 0.001) following MT. We observed that the model combining age, sex, hypertension, heart failure, alcoholism, coronary artery disease, diabetes mellitus, smoking, previous transient ischemic attack (TIA), and ischemic stroke, and admission CRP (area under the curve (AUC) = 0.723, 95% CI = 0.71–0.74) and 24-h CRP (AUC = 0.704, 95% CI = 0.69–0.72) had good predictive accuracy, with optimal cutoff values of 4.25 for admission CRP and 14.69 for 24-h CRP to detect post-stroke AF. Subgroup analysis indicated CRP predictive relevance, particularly in hypertensive patients.
Conclusions:
Our findings suggest CRP is associated with post-stroke AF in stroke patients due to LVO, highlighting inflammation’s role in AF pathogenesis. Measuring CRP at admission and 24 h may enable early detection and timely anticoagulation. Incorporating CRP into clinical pathways could improve individualized risk assessment, warranting further studies to validate its predictive utility and explore additional markers.
Introduction
Mechanical thrombectomy (MT) is effective and safe in acute ischemic stroke due to large vessel occlusion (LVO) of the anterior circulation, irrespective of the cause.1,2 Atrial fibrillation (AF) constitutes a major risk factor for ischemic stroke, 3 and AF can be newly detected in close temporal proximity after the index stroke (post-stroke AF) or can be known before the stroke (prevalent AF). 4 A rapid identification of post-stroke AF in patients with acute ischemic stroke is crucial, because oral anticoagulation significantly decreases the risk of stroke recurrence and because the risk of recurrence is the highest in the acute phase.5–7 Moreover, adherence is better if prevention is started in inpatients shortly after the event.8,9 However, post-stroke AF recognition remains elusive, because 50% or more of cases may present with asymptomatic and intermittent patterns. 10 Reliable biomarkers that predict post-stroke AF could support targeted interventions, particularly in the context of stroke patients due to LVO, where rapid diagnostic insights are essential to guide optimal clinical decision-making. To date, natriuretic peptides and chronic elevation of cardiac troponin are well-known biomarkers most consistently associated with the diagnosis of post-stroke AF after acute ischemic stroke. 11 Emerging evidence suggests that systemic inflammation may play a role in the pathophysiology of post-stroke AF, with inflammatory processes potentially influencing the atrial remodeling and electrophysiological stability.4,12 C-reactive protein (CRP), a sensitive marker of systemic inflammation, is among the most studied inflammatory markers in cardiovascular and cerebrovascular disease.13,14 Elevated CRP levels have been linked to both prevalent and incident AF, suggesting a mechanistic overlap between inflammatory responses and AF onset or exacerbation. 15 This multicenter study aimed to investigate the predictive value of CRP and other inflammatory biomarkers for post-stroke AF among acute ischemic stroke patients undergoing MT. By analyzing both admission and 24-h post-procedural biomarker levels, we aimed to elucidate their potential as diagnostic tools for early identification of patients at elevated risk for post-stroke AF.
Methods
Study design and patients
This retrospective cohort study included all acute stroke patients aged 18 years or older consecutively treated with MT for anterior circulation LVO in four thrombectomy centers: Charing Cross Hospital, Imperial College Healthcare NHS Trust, London (UK); St George’s University of London, London (UK); Udine University Hospital, Udine (Italy) and Boston University (USA); between 1 January 2016 and 30 March 2023 with local stroke registries available.16–18 The study was conducted in accordance with the recommendations of the World Medical Assembly (Helsinki 1964 and later revisions). Patients were stratified into two groups: those with newly detected AF during the hospital stay (post-stroke AF) in close temporal proximity after the index stroke and those with no AF detected or known before the index stroke (NO-AF). This approach ensures alignment with the primary aim of the study, which is to evaluate the predictive role of inflammatory biomarkers, without assuming their relevance a priori. We excluded from the analysis patients with AF already known before the index stroke (prevalent AF) (Supplemental Figure 1). All patients were managed according to standardized clinical protocols, and relevant clinical and laboratory data were collected prospectively upon admission.
Patient inclusion and exclusion criteria for the analysis
The criteria for patients’ selection were: (1) age ⩾18 years; (2) National Institutes of Health Stroke Scale (NIHSS) score 6 or more; (3) Alberta Stroke Program Early CT score (ASPECTS) 5 or more (17); (4) LVO sites: distal internal carotid artery, middle cerebral artery segments M1 or M2; (5) initiation of the MT had to be possible within 6 h after the stroke onset; and (6) pre-stroke modified Rankin Scale (mRS) score of 0 to 2. Intravenous thrombolysis (IVT) with intravenous tissue plasminogen activator (tPA) was administered in all patients who presented within 4.5 h of stroke symptom onset without contraindications according to the guidelines. For this analysis, we excluded stroke patients with basilar artery occlusion and patients that met DAWN or DEFUSE 3 eligibility criteria.19,20 Patients with pre-stroke mRS scores greater than 2 were excluded in accordance with established MT guidelines, which recommend treatment primarily for patients with good pre-stroke functional status.1,21,22 In addition, this criterion aligns with the treatment protocols at our participating centers, where MT is not performed on patients with pre-stroke mRS > 2. Moreover, for the purpose of this analysis, we excluded patients with concomitant condition which could potentially alter the inflammation biomarkers, in particular: (1) ongoing effect of immunomodulatory or immunosuppressive drugs, (2) ongoing infections or infections developed within 48 h after admission, (3) chronic inflammatory diseases, (4) hematological disorders, (5) cancers (active and/or under treatment), (6) major traumas or surgical procedures in the previous 28 days, (7) acute myocardial infarction (AMI) with or without ST elevation, (8) severe liver or kidney dysfunction (estimated glomerular filtration rate (eGFR) < 30 mL/min), and (9) recent transfusion (< 7 days before admission) or transfusion done within 24 h after admission.
Data collection
Please see Supplemental Material.
Statistical analysis
Categorical variables are presented as count and percentage, and continuous variables are presented as mean and standard deviation or median and interquartile range according to normal distribution.
We used inverse probability weighting (IPW) to balance the baseline characteristics of the exposed and unexposed cohorts (post-stroke AF vs. NO-AF group), aiming to reduce the confounding factors on study outcomes and to ensure that these variables did not act as confounders in subsequent analyses. A detailed methodological explanation of the IPW estimation process is available in the Supplemental Methods. In brief, weights were obtained by calculating the probability of being in the group with post-stroke AF while controlling for a set of relevant variables that could have influenced the outcome. The weights obtained were then used to balance the baseline covariates, therefore creating a pseudo-population independent of the measured confounders (i.e. pseudo-randomization). 23 Statistical comparisons were performed in the weighted population between patient groups using the χ2 test, Fisher’s exact test, Student’s t-test, and Mann–Whitney U as indicated for dichotomous or continuous variables.
Following IPW, the only variables left unbalanced were inflammatory markers. The selection of covariates for regression models began with a detailed comparison of these markers between the post-stroke AF and NO-AF groups in the weighted cohort, as reported in Table 1. Variables showing significant differences in this analysis were prioritized for inclusion in the univariate and multivariate models. Logistic regression models were constructed using the inflammatory markers identified as significant in the univariate analysis to determine predictors of post-stroke AF.
Blood test on admission and after 24 h in the weighted population.
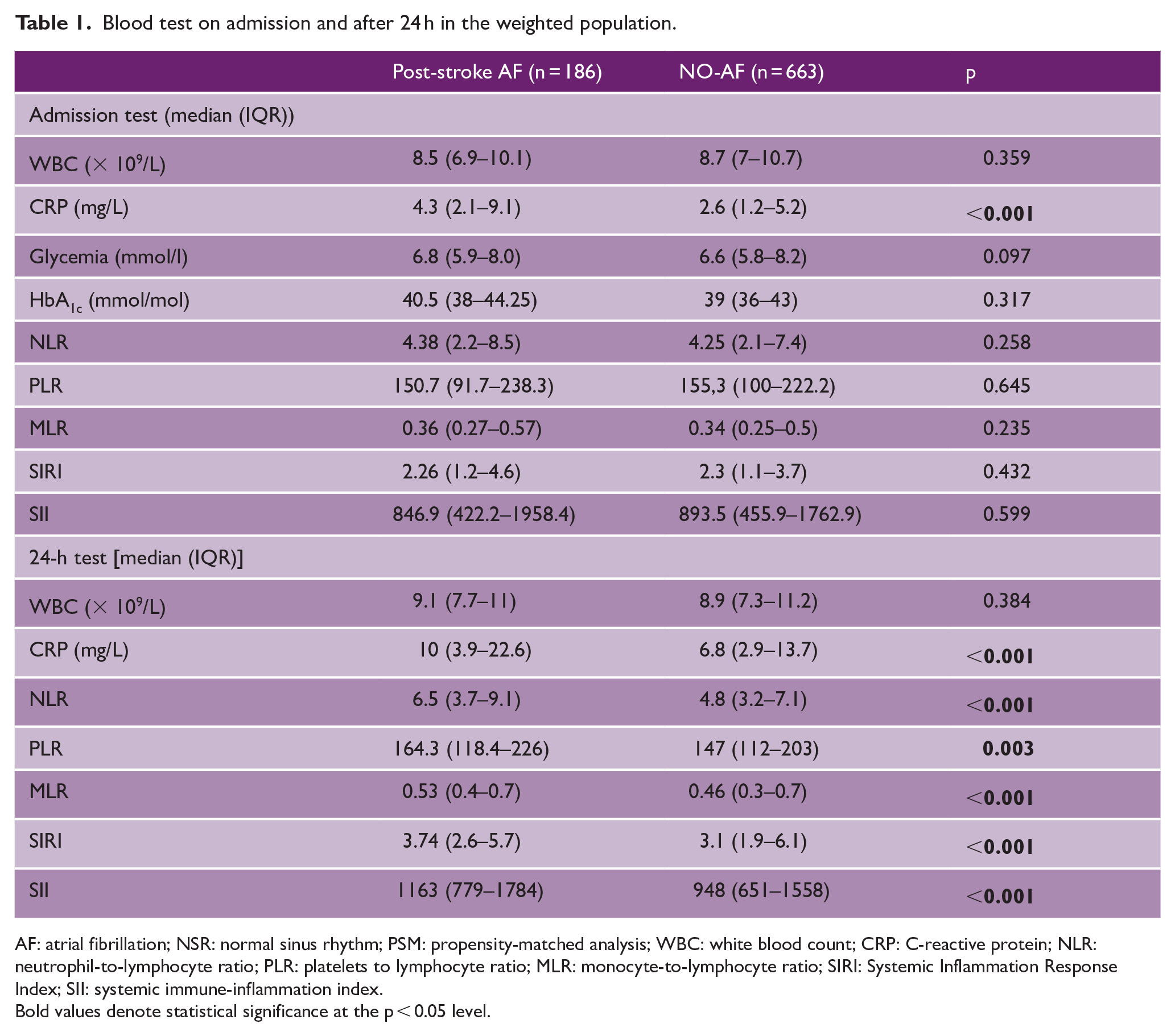
AF: atrial fibrillation; NSR: normal sinus rhythm; PSM: propensity-matched analysis; WBC: white blood count; CRP: C-reactive protein; NLR: neutrophil-to-lymphocyte ratio; PLR: platelets to lymphocyte ratio; MLR: monocyte-to-lymphocyte ratio; SIRI: Systemic Inflammation Response Index; SII: systemic immune-inflammation index.
Bold values denote statistical significance at the p < 0.05 level.
The diagnostic values of factors, solely or in combination, to predict post-stroke AF were tested with area under the receiver operating characteristic curves (AUC-ROC) in the weighted population. The predictive model included a prespecified set of relevant variables that could have influenced the outcome according to the literature. 11 Receiver operating characteristic (ROC) curves were compared using the DeLong test.
Subgroup analysis of the post-stroke AF outcome in the weighted population was performed on prespecified subgroups: age (<75 years or ⩾ 75 years), presence of arterial hypertension, heart failure, or coronary artery disease. We used a generalized linear model with admission CRP or 24-h CRP, subgroup variable, and their interaction term as independent variables to determine the homogeneity in the effect of admission CRP or 24-h CRP by each subgroup variable. The p-value was presented for the interaction term. Finally, marginal effects analysis was employed to visualize and examine predicted probabilities of post-stroke AF across the range of admission CRP or 24-h CRP within each subgroup. Statistical significance was set at a p-value of <0.05.
R Studio (version 2024.4) was used for statistical analysis.
Results
Our analysis included 849 patients with acute ischemic stroke due to LVO treated with MT (Supplemental Figure 1). Of note, 32 patients (3.8%) were previously on treatment with oral anticoagulant because of previous deep venous thrombosis. Among the included patients, during the admission, 663 (78.1%) patients did not have a diagnosis of AF (NO-AF) while 186 (21.9%) had post-stroke AF. Table 2 reports unweighted and weighted results for baseline characteristics. Overall, good balance was obtained for all major baseline variables of interest (Supplemental Figures 2 and 3 and Table 1). Supplemental Table 1 reports the procedural features in the weighted population. Supplemental Table 2 reports the outcome distribution between the two weighted groups. The 90-day mortality rate in our cohort was 11.9%, with 12.4% in post-stroke AF group and 11.8% in the NO-AF group (p = 0.924).
Baseline characteristics.
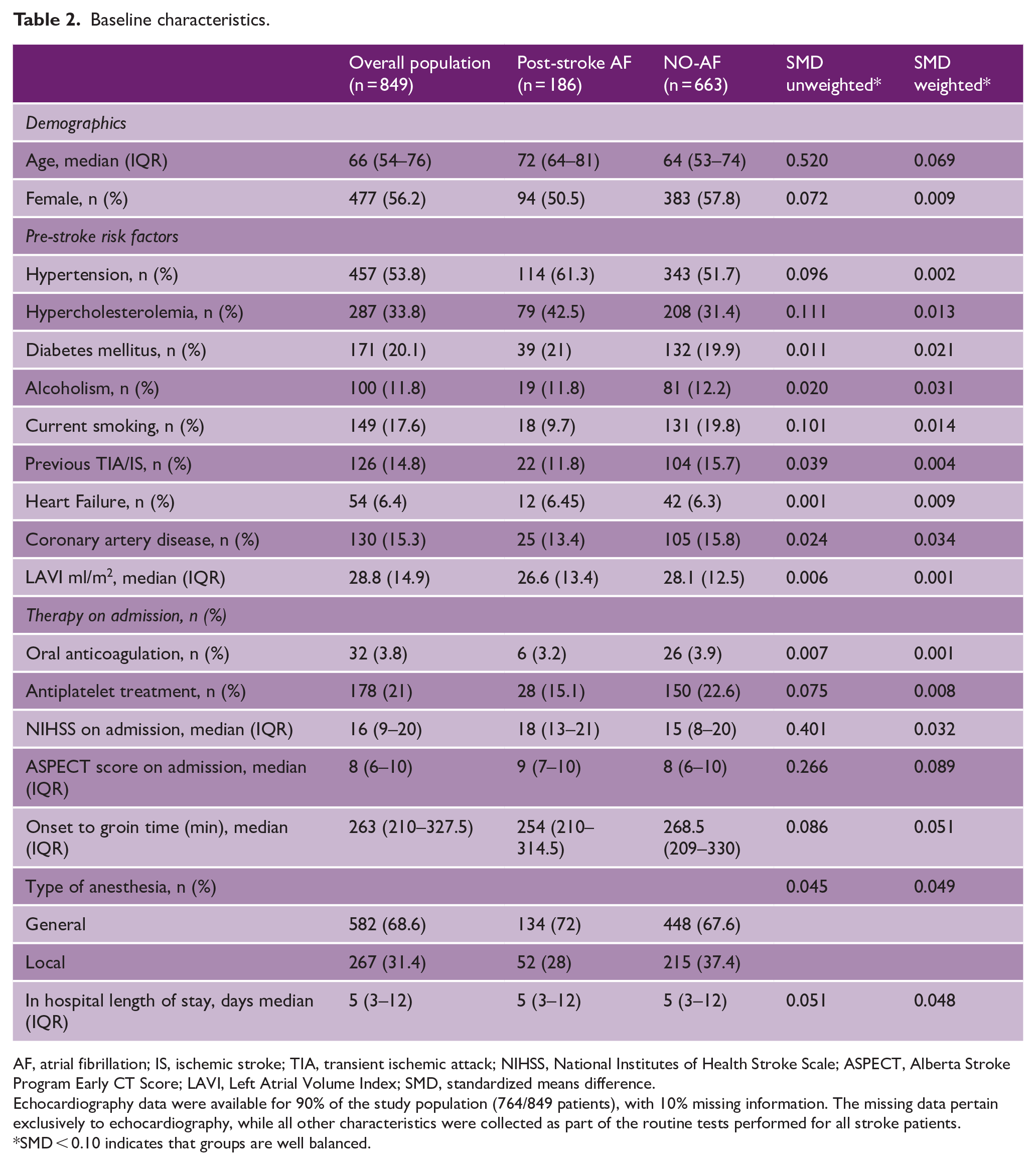
AF, atrial fibrillation; IS, ischemic stroke; TIA, transient ischemic attack; NIHSS, National Institutes of Health Stroke Scale; ASPECT, Alberta Stroke Program Early CT Score; LAVI, Left Atrial Volume Index; SMD, standardized means difference.
Echocardiography data were available for 90% of the study population (764/849 patients), with 10% missing information. The missing data pertain exclusively to echocardiography, while all other characteristics were collected as part of the routine tests performed for all stroke patients.
SMD < 0.10 indicates that groups are well balanced.
Table 1 reports blood tests collected on admission and at 24 h from the index event in the weighted population (Figure 1). CRP on admission was significantly different between the two groups (p < 0.001). At 24 h from the index event, CRP (p < 0.001), neutrophil-to-lymphocyte ratio (NLR) (p < 0.001), platelet-to-lymphocyte ratio (PLR) (p = 0.003), monocyte-to-lymphocyte ratio (MLR) (p < 0.001), systemic inflammation response index (SIRI) (p < 0.001), and systemic immune-inflammation index (SII) (p < 0.001) were significantly different between post-stroke AF and NO-AF patients (Figure 2). The rest of the blood test on admission and at 24 h from the index event are reported in Supplemental Table 3.
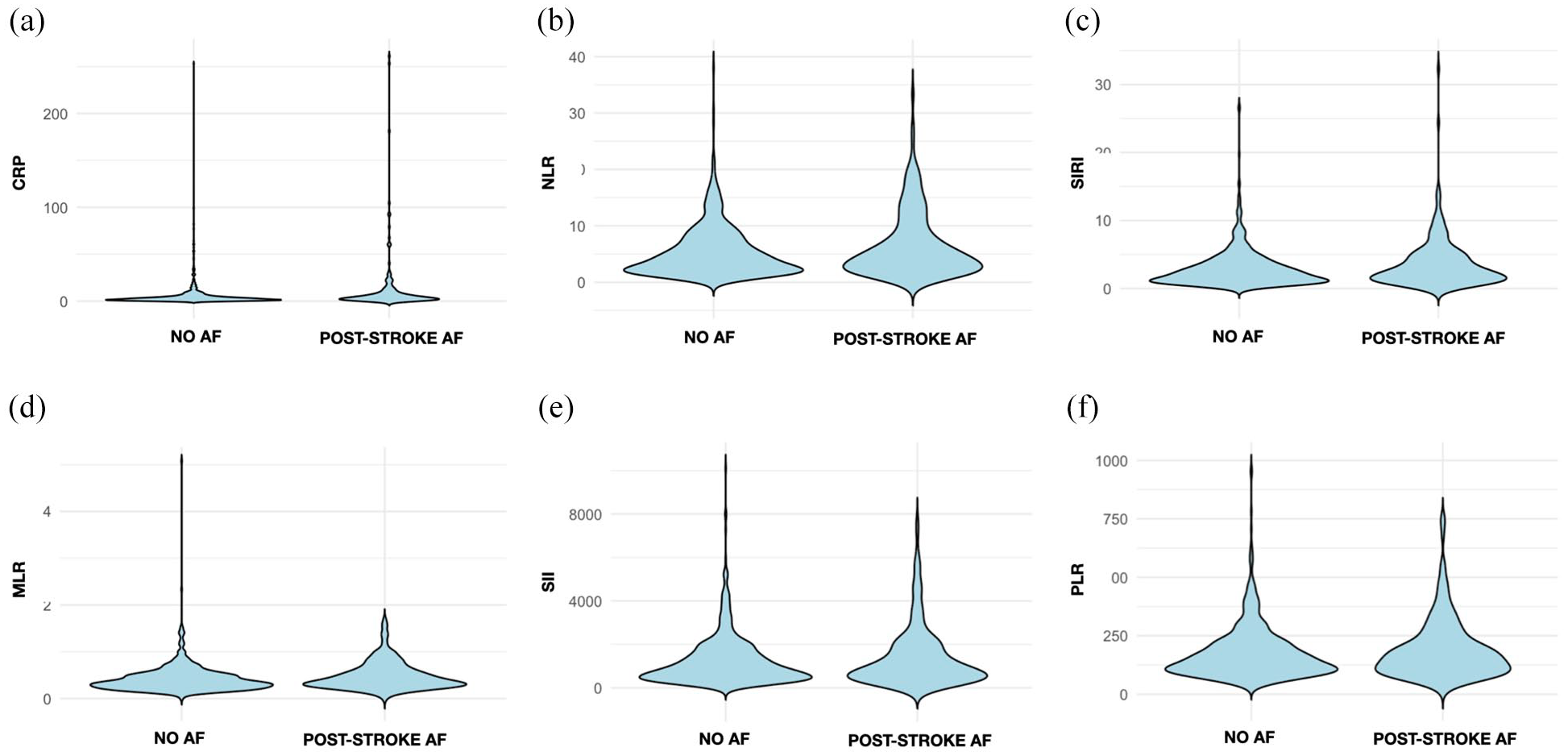
Violin plots showing the distribution of blood tests collected on admission in the weighted population. C-reactive protein (CRP), neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), monocyte-to-lymphocyte ratio (MLR), systemic inflammation response index (SIRI), and systemic immune-inflammation index (SII).
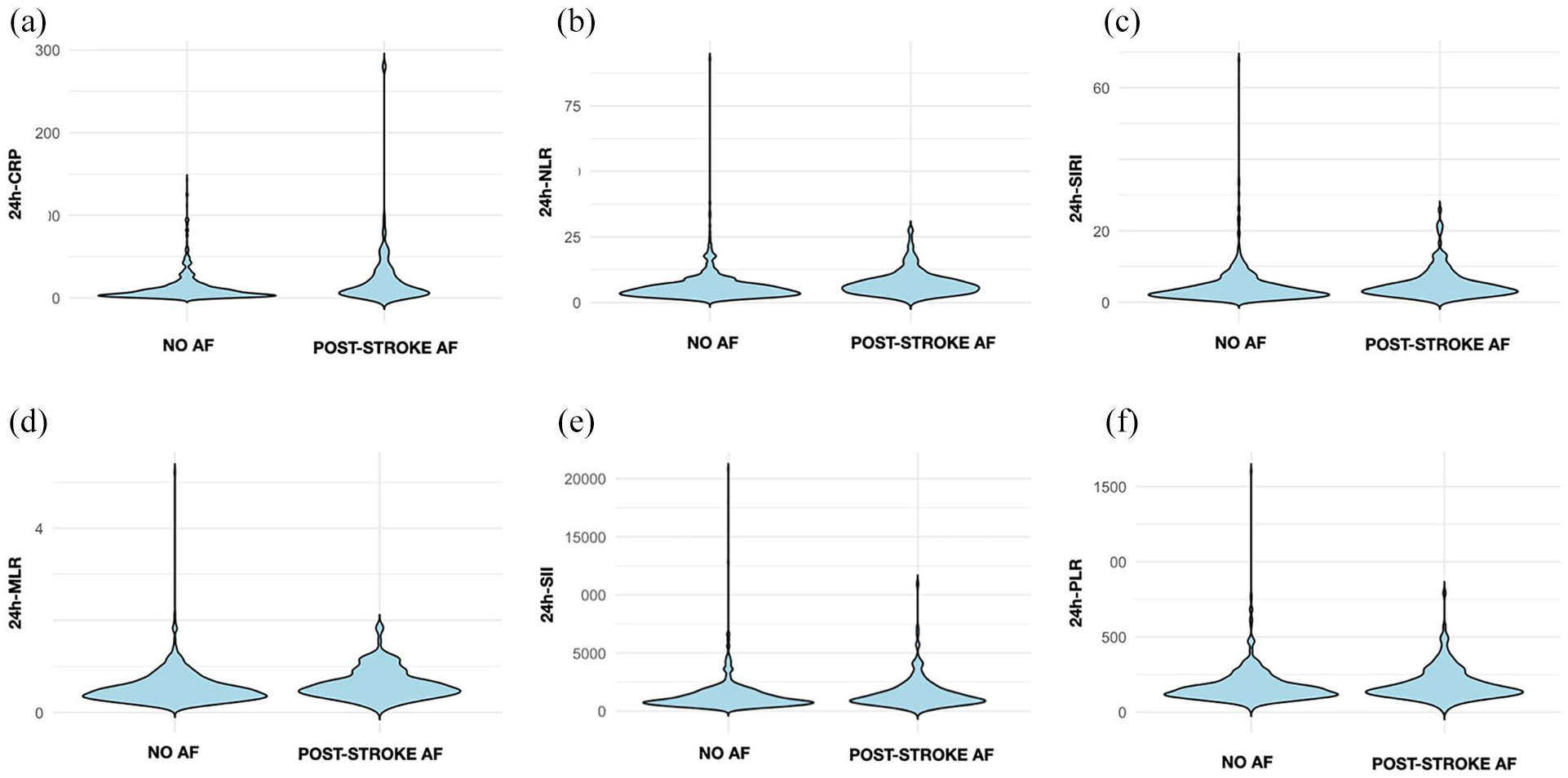
Violin plots showing the distribution of blood tests collected at 24 h from the index event in the weighted population. C-reactive protein (CRP), neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), monocyte-to-lymphocyte ratio (MLR), systemic inflammation response index (SIRI), and systemic immune-inflammation index (SII).
In the logistic regression analysis (Supplemental Table 4), admission CRP was independently associated with increased likelihood of post-stroke AF (odds ratio (OR) = 1.01 per unit increase, 95% confidence interval (CI) = 1.00–1.03, p < 0.001). A similar association was observed with 24-h CRP levels (OR = 1.02 per unit increase, 95% CI = 1.01–1.03, p < 0.001) (Figure 3). Other biomarkers did not remain significant in multivariate models.
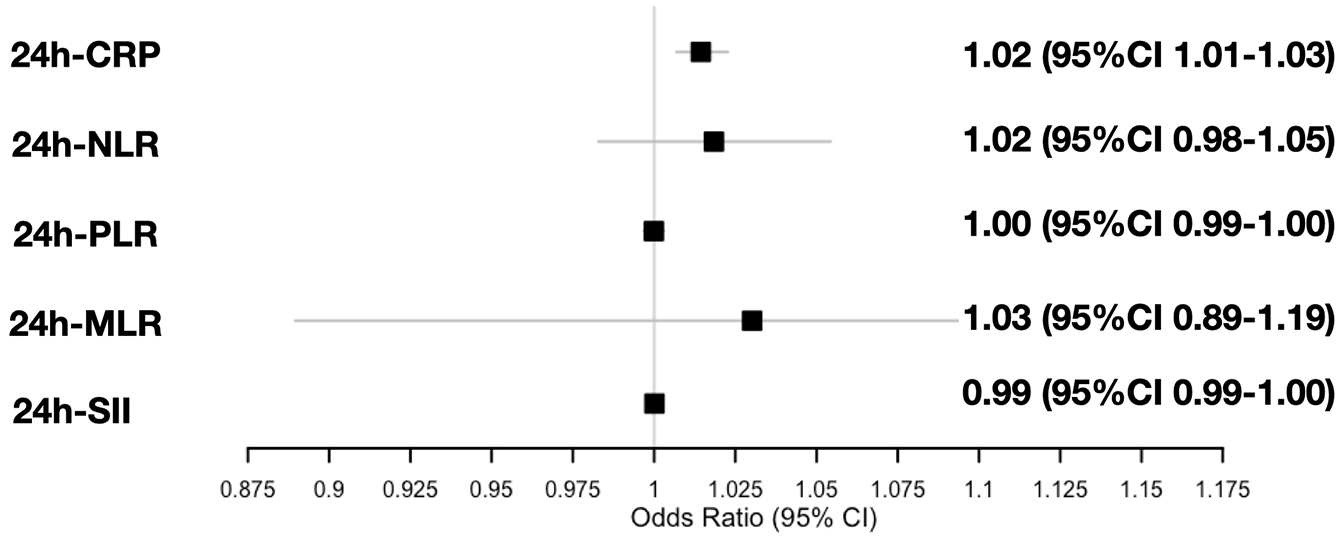
Forest plot for post-stroke AF. C-reactive protein (CRP), neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), monocyte-to-lymphocyte ratio (MLR), and systemic immune-inflammation index (SII). CI: confidence interval.
ROC analysis of CRP for predicting post-stroke AF in acute stroke patients
Using the ROC curves from the logistic regression analysis, we identified the predictive accuracy for post-stroke AF in acute stroke patients undergoing MT of two binomial logistic regression models (Figure 4). The first included age, sex, hypertension, heart failure, alcoholism, coronary artery disease, diabetes mellitus, smoking, previous transient ischemic attack (TIA), and ischemic stroke. The second model included the same variables as the model above, with the addition of CRP. The ROC curves showed an area under the curve (AUC) of 0.667 (95% CI = 0.64–0.69) for the model without CRP and 0.723 (95% CI = 0.71–0.74) for the model with CRP. DeLong’s test for comparing correlated ROC curves revealed a statistically significant difference between the two models (Z = −2.0943, p = 0.03623), with a 95% CI for the AUC difference of −0.109 to −0.004. To evaluate the independent predictive value of CRP for post-stroke AF in patients undergoing MT, we calculated the ROC curve using CRP as the sole predictor variable. The AUC for this analysis was 0.641 (95% CI = 0.62–0.66). In addition, the optimal cutoff for CRP to predict post-stroke AF was determined using Youden’s Index. The analysis identified an optimal cutoff value of 4.25 for CRP for post-stroke AF after MT in acute stroke patients.
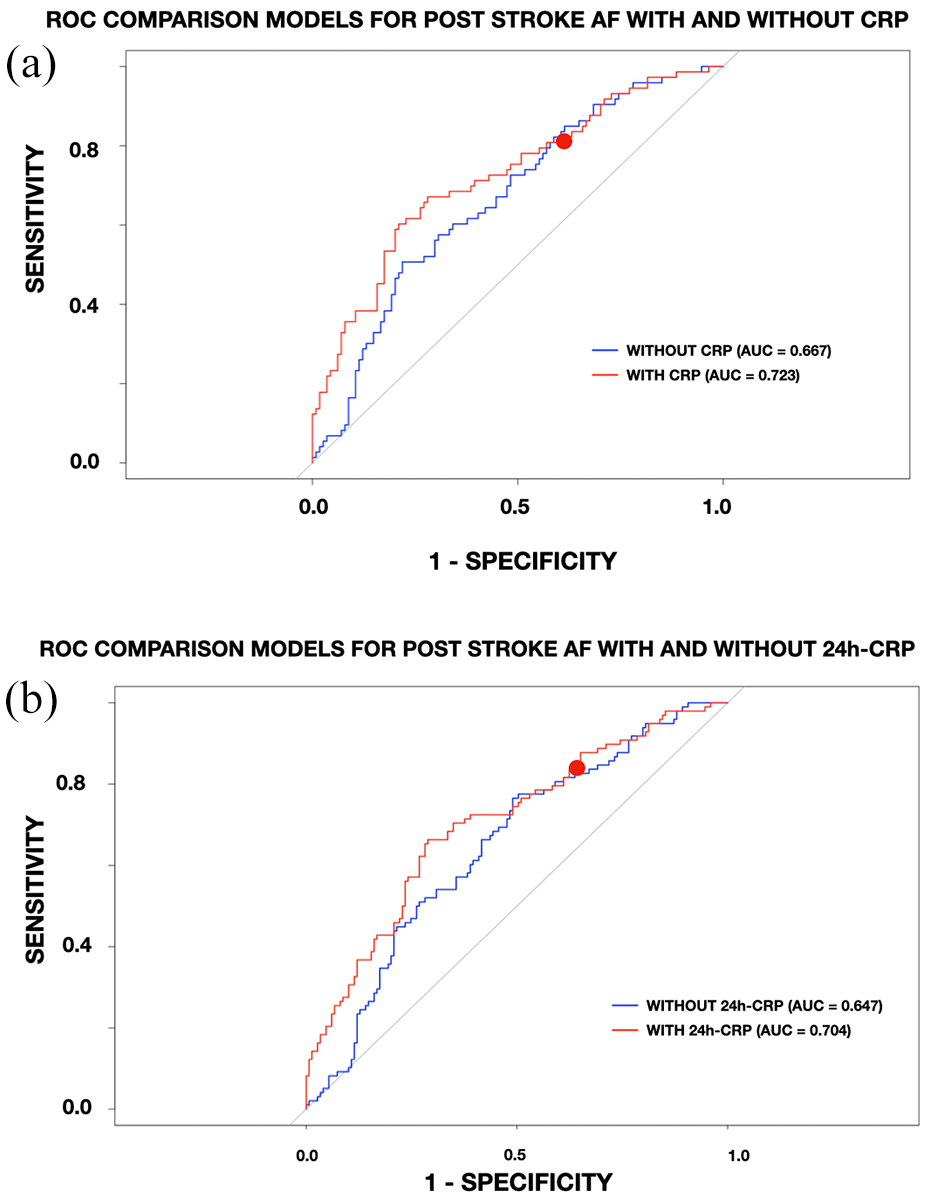
Receiver operating characteristics (ROC) curves for predicting post-stroke AF after mechanical thrombectomy. C-reactive protein (CRP).
ROC analysis of 24-h CRP for predicting post-stroke AF in acute stroke patients
We conducted a similar analysis for 24-h CRP, evaluating its predictive accuracy for post-stroke AF using ROC curves from logistic regression (Figure 4). The first model, excluding 24-h CRP, had an AUC of 0.647 (95% CI = 0.61–0.69), while the second model, which included 24-h CRP, achieved a higher AUC of 0.704 (95% CI = 0.69–0.72). ROC curves were generated for both models and visually compared. DeLong’s test for two correlated ROC curves revealed a statistically significant difference between the models’ AUCs (Z = −2.1629, p = 0.03055), with a 95% CI for the difference ranging from −0.108 to −0.005. To assess the predictive power of 24-h CRP alone for post-stroke AF, we calculated the ROC curve with CRP24 as the sole predictor variable. The AUC was 0.623 (95% CI = 0.60–0.64). Using Youden’s Index to maximize sensitivity and specificity, the optimal cutoff for CRP24 to predict post-stroke AF was determined to be 14.69 after MT in acute stroke patients.
Interaction analysis of CRP and 24-h CRP for predicting post-stroke AF
Figure 5 shows the margin plots showing the probability of achieving post-stroke AF in acute stroke patients after MT for CRP and 24-h CRP in different subgroups. CRP was associated with the incremental probability of post-stroke AF detection regardless of age (p-values for interaction = 0.129), presence of heart failure (p-values for interaction = 0.295), and coronary artery disease (p-values for interaction = 0.559). There is a significant interaction between CRP and history of hypertension in predicting post-stroke AF after MT in acute ischemic stroke (p-values for interaction = 0.009). Conversely, 24-h CRP was associated with the incremental probability of post-stroke AF detection regardless of the age (p-values for interaction = 0.127), presence of heart failure (p-values for interaction = 0.701), coronary artery disease (p-values for interaction = 0.743), and hypertension (p-values for interaction = 0.070).
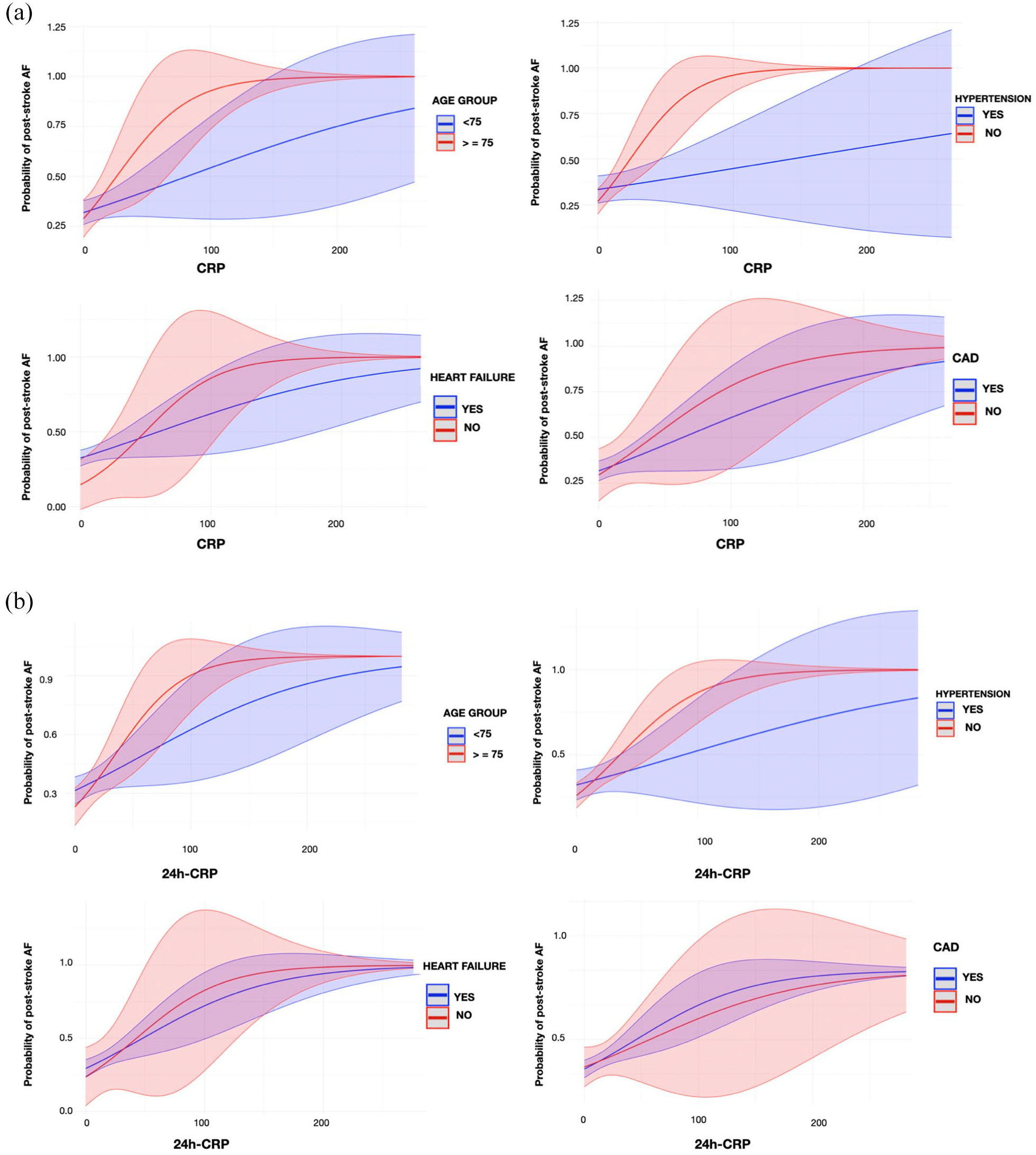
Margin plots of CRP (A) and 24-h CRP (B) with probability of post-stroke AF by age subgroups, presence of hypertension, heart failure, and coronary artery disease (CAD). Each graph shows the predicted probability (solid line) and 95% confidence interval (shade) of different outcomes.
ROC analysis of the percentage change in CRP over 24 h for predicting post-stroke AF in acute stroke patients is presented in Supplemental Result and Supplemental Figure 4.
Discussion
Our study based on an IPW analysis provides evidence supporting the predictive value of CRP for detecting post-stroke AF in acute ischemic stroke patients following MT. By measuring CRP levels at both admission and 24-h post-procedure, we demonstrated that early inflammatory indicators could offer predictive insights into post-stroke AF risk in stroke patients due to LVO. An effective and rapid post-stroke AF detection is crucial in the acute stroke setting, as timely identification of post-stroke AF allows for earlier initiation of oral anticoagulation therapy, significantly reducing the risk of subsequent ischemic events, particularly in the acute phase when recurrence risk is high.24–26 Our study findings are consistent with the previously published literature that suggested an association between CRP and post-stroke AF risk. Previous research suggested that increased serum CRP levels were associated with a high risk of post-operative AF and recurrence of AF after electrical cardioversion.27,28 Pang et al. 29 developed a predictive model for assessing the incidence of post-stroke AF after acute ischemic stroke. The authors showed that their prediction model based on age, CRP on admission, presence of heart failure, history of stroke/TIA, type B natriuretic peptide NIHSS, and neuroimaging features had good calibration and discrimination to predict post-stroke AF after an acute ischemic stroke. In contrast to Pang et al., our analysis included two measurements of CRP levels, on admission and after 24 h from the index event. This is because a single measurement of serum CRP would not be an ideal candidate for determining a chronic inflammatory state since CRP levels are prone to fluctuate over time. Moreover, it is noteworthy to mention that we used an IPW analysis to balance the baseline characteristics of the two groups (post-stroke AF vs. NO-AF group), aiming to reduce the confounding effect of a potential bias by indication given the retrospective and non-randomized nature of the study.
Inflammation is considered to play an important role in the pathogenesis of post-stroke AF.4,11 Indeed, neurogenic mechanisms contributing to post-stroke AF are increasingly recognized within the framework of the recently described Stroke-Heart Syndrome, where inflammation plays a central role.30,31 Indeed, our findings are consistent with previous research that highlights the association between elevated CRP levels and the risk of post-stroke AF. A large cohort study involving over 100,000 ischemic stroke patients demonstrated that higher CRP levels at admission, particularly those classified as moderate or high by the American Heart Association’s thresholds, 13 were associated with an increased likelihood of developing AF. 32 These results align with our observations and underscore the importance of systemic inflammation as a key mechanism contributing to the development of AF after stroke. Inflammatory and autonomic pathways are activated following stroke, triggering focal ectopic firing at the ganglionated plexi.12,33 Over time, these inflammatory processes can lead to re-entry mechanisms linked to stroke-induced cardiac injury, such as subendocardial hemorrhage, further promoting arrhythmogenic activity.12,33 CRP, a representative biomarker of systemic inflammation, is thought to be involved in the immunological process that triggers cardiac and vascular remodeling and is associated with various cardiovascular risks. 34 Our findings support the hypothesis that CRP may be a mechanistic link between systemic inflammation and post-stroke AF. Moreover, the optimal CRP thresholds identified in our study—4.25 on admission and 14.69 at 24 h—may serve as clinically relevant cutoffs to stratify patients by post-stroke AF risk. These thresholds could allow clinicians to identify high-risk patients early, facilitating targeted monitoring and potentially enabling earlier therapeutic interventions.
Another important finding of our analysis was the association between CRP and post-stroke AF risk is particularly pronounced in hypertensive patients, indicating that CRP may reflect compounded inflammatory risk in this subgroup. Hypertension is already a recognized risk factor for AF and is associated with chronic vascular inflammation, which may contribute to the atrial remodeling that predisposes patients to AF. 35 The interaction between elevated CRP and hypertension highlights the need for further investigation into the synergistic role of inflammation and blood pressure control in managing AF risk after stroke. This finding also suggests that clinicians could prioritize CRP monitoring and follow-up specifically in hypertensive stroke patients treated with MT, potentially offering a more personalized approach to post-stroke AF risk assessment.
Our analysis has the following strengths: (1) large cohort of MT patients; (2) multicenter study; (3) data ascertainment undertaken systematically; and (4) the use of an IPW analysis to adjust for the indication bias. Nevertheless, our study also has several limitations. The retrospective observational study design limits the ability to establish a causal relationship between CRP levels and post-stroke AF onset. Although we used IPW to balance baseline covariates and reduce confounding, unmeasured factors could still influence CRP levels and post-stroke AF outcomes. Future prospective studies or randomized trials would be essential to confirm the causal relationship and further elucidate the role of CRP role in post-stroke AF. Second, while CRP is a widely used biomarker of inflammation, it is a non-specific marker and can be influenced by various conditions unrelated to AF or stroke, including infections, autoimmune diseases, and recent surgeries. To mitigate this issue, we excluded patients with active infections or inflammatory conditions known to elevate CRP. Nonetheless, subtle or undiagnosed inflammatory processes might have affected CRP levels in some patients. Another limitation is the lack of longitudinal CRP data beyond the initial 24-h period. Our analysis focused on CRP levels at two specific time points, which may not fully capture the inflammation trajectory and its relationship to post-stroke AF. Serial measurements of CRP or other inflammatory markers over a longer follow-up period might provide a clearer picture of how inflammation evolves in relation to post-stroke AF development. The study relies on standard CRP rather than including both conventional CRP and high-sensitivity CRP (hs-CRP), which could have provided additional insights into low-grade inflammation. Our study’s inclusion criteria may limit the generalizability of our findings. By focusing on patients undergoing MT for LVO in the anterior circulation, our results may not apply to patients with other types of strokes, such as patients with minor strokes, small vessel disease, or strokes treated with medical management alone or those not eligible for MT. In addition, patients with differing pathophysiological mechanisms of stroke may exhibit distinct inflammatory profiles, potentially altering the applicability of our model. Further research in more diverse stroke populations could determine whether CRP retains predictive value for post-stroke AF across different patient groups and treatment modalities. Another limitation of our study is the absence of measurements for B-type natriuretic peptide (BNP) and troponin levels, which are well-documented in the literature as biomarkers associated with AF detection. These biomarkers were not included as they are not part of routine testing in our participating centers. Another limitation of our study is the exclusion of patients with chronic inflammatory conditions during the screening process. While this ensured a homogeneous cohort for evaluating acute CRP dynamics, it limits the applicability of our findings to patients with chronic inflammation. Future studies should assess the model’s performance in broader populations, including those with chronic inflammatory states, to evaluate its generalizability.
We did not collect data on other outcomes (e.g. acute coronary syndromes, heart failure, ventricular arrhythmias, and Takotsubo cardiomyopathy) as they were beyond the scope of this analysis. Although we utilized robust methodologies to exclude patients with a documented history of AF, the possibility remains that individuals with undiagnosed subclinical AF prior to stroke onset were included in the post-stroke AF group. Moreover, conducting a competing risk analysis was not feasible due to the difficulty in precisely aligning the timelines of competing events (death and post-stroke AF) given the retrospective nature of our data collection. However, since AF was exclusively detected during the hospital stay, it is unlikely that mortality significantly influenced its detection in our cohort. Future studies employing competing risk regression models could provide deeper insights into the interplay between systemic inflammation, post-stroke AF, and long-term survival outcomes. Finally, echocardiographic measurements were not available for the entire cohort. Finally, echocardiographic measurements were not recorded for the entire cohort.
In conclusion, our findings indicate that CRP is a promising predictive biomarker for post-stroke AF in acute ischemic stroke patients treated with MT. The observed associations between elevated CRP levels and post-stroke AF risk suggest an inflammatory component in AF pathogenesis post-stroke. CRP measurements on admission and 24-h post-procedure could be incorporated into clinical pathways to support early identification of high-risk patients, potentially informing individualized monitoring and therapeutic strategies. Given the limitations of our study, future research should focus on prospective validation of the predictive utility of CRP, as well as the exploration of additional biomarkers and the potential synergistic role of inflammation and hypertension in post-stroke AF development. These insights may ultimately lead to more targeted approaches in the management of AF in stroke patients, improving outcomes in this vulnerable population.
Supplemental Material
sj-docx-1-wso-10.1177_17474930251332489 – Supplemental material for C-reactive protein elevation and subsequent atrial fibrillation detection after ischemic stroke treated with mechanical thrombectomy: An inverse probability weighting analysis
Supplemental material, sj-docx-1-wso-10.1177_17474930251332489 for C-reactive protein elevation and subsequent atrial fibrillation detection after ischemic stroke treated with mechanical thrombectomy: An inverse probability weighting analysis by Lucio D’Anna, Gabriele Prandin, Mariarosaria Valente, Liqun Zhang, Paresh Malhotra, Simona Sacco, Matteo Foschi, Raffaele Ornello, Viva Levee, Katherine Chulack, Fahad Sheikh, Feras Fayez, Francesco Toraldo, Domenico Maisano, Caterina Del Regno, Filippo Komauli, Adelaida Gartner Jarmillo, Hakam AL-Karadsheh, Hamza Zahid, Piers Klein, Mohamad Abdalkader, Edoardo Pirera, Paolo Manganotti, Kyriakos Lobotesis, Thanh N Nguyen, Gian Luigi Gigli, Soma Banerjee and Giovanni Merlino in International Journal of Stroke
Supplemental Material
sj-docx-2-wso-10.1177_17474930251332489 – Supplemental material for C-reactive protein elevation and subsequent atrial fibrillation detection after ischemic stroke treated with mechanical thrombectomy: An inverse probability weighting analysis
Supplemental material, sj-docx-2-wso-10.1177_17474930251332489 for C-reactive protein elevation and subsequent atrial fibrillation detection after ischemic stroke treated with mechanical thrombectomy: An inverse probability weighting analysis by Lucio D’Anna, Gabriele Prandin, Mariarosaria Valente, Liqun Zhang, Paresh Malhotra, Simona Sacco, Matteo Foschi, Raffaele Ornello, Viva Levee, Katherine Chulack, Fahad Sheikh, Feras Fayez, Francesco Toraldo, Domenico Maisano, Caterina Del Regno, Filippo Komauli, Adelaida Gartner Jarmillo, Hakam AL-Karadsheh, Hamza Zahid, Piers Klein, Mohamad Abdalkader, Edoardo Pirera, Paolo Manganotti, Kyriakos Lobotesis, Thanh N Nguyen, Gian Luigi Gigli, Soma Banerjee and Giovanni Merlino in International Journal of Stroke
Supplemental Material
sj-docx-6-wso-10.1177_17474930251332489 – Supplemental material for C-reactive protein elevation and subsequent atrial fibrillation detection after ischemic stroke treated with mechanical thrombectomy: An inverse probability weighting analysis
Supplemental material, sj-docx-6-wso-10.1177_17474930251332489 for C-reactive protein elevation and subsequent atrial fibrillation detection after ischemic stroke treated with mechanical thrombectomy: An inverse probability weighting analysis by Lucio D’Anna, Gabriele Prandin, Mariarosaria Valente, Liqun Zhang, Paresh Malhotra, Simona Sacco, Matteo Foschi, Raffaele Ornello, Viva Levee, Katherine Chulack, Fahad Sheikh, Feras Fayez, Francesco Toraldo, Domenico Maisano, Caterina Del Regno, Filippo Komauli, Adelaida Gartner Jarmillo, Hakam AL-Karadsheh, Hamza Zahid, Piers Klein, Mohamad Abdalkader, Edoardo Pirera, Paolo Manganotti, Kyriakos Lobotesis, Thanh N Nguyen, Gian Luigi Gigli, Soma Banerjee and Giovanni Merlino in International Journal of Stroke
Supplemental Material
sj-docx-7-wso-10.1177_17474930251332489 – Supplemental material for C-reactive protein elevation and subsequent atrial fibrillation detection after ischemic stroke treated with mechanical thrombectomy: An inverse probability weighting analysis
Supplemental material, sj-docx-7-wso-10.1177_17474930251332489 for C-reactive protein elevation and subsequent atrial fibrillation detection after ischemic stroke treated with mechanical thrombectomy: An inverse probability weighting analysis by Lucio D’Anna, Gabriele Prandin, Mariarosaria Valente, Liqun Zhang, Paresh Malhotra, Simona Sacco, Matteo Foschi, Raffaele Ornello, Viva Levee, Katherine Chulack, Fahad Sheikh, Feras Fayez, Francesco Toraldo, Domenico Maisano, Caterina Del Regno, Filippo Komauli, Adelaida Gartner Jarmillo, Hakam AL-Karadsheh, Hamza Zahid, Piers Klein, Mohamad Abdalkader, Edoardo Pirera, Paolo Manganotti, Kyriakos Lobotesis, Thanh N Nguyen, Gian Luigi Gigli, Soma Banerjee and Giovanni Merlino in International Journal of Stroke
Supplemental Material
sj-docx-8-wso-10.1177_17474930251332489 – Supplemental material for C-reactive protein elevation and subsequent atrial fibrillation detection after ischemic stroke treated with mechanical thrombectomy: An inverse probability weighting analysis
Supplemental material, sj-docx-8-wso-10.1177_17474930251332489 for C-reactive protein elevation and subsequent atrial fibrillation detection after ischemic stroke treated with mechanical thrombectomy: An inverse probability weighting analysis by Lucio D’Anna, Gabriele Prandin, Mariarosaria Valente, Liqun Zhang, Paresh Malhotra, Simona Sacco, Matteo Foschi, Raffaele Ornello, Viva Levee, Katherine Chulack, Fahad Sheikh, Feras Fayez, Francesco Toraldo, Domenico Maisano, Caterina Del Regno, Filippo Komauli, Adelaida Gartner Jarmillo, Hakam AL-Karadsheh, Hamza Zahid, Piers Klein, Mohamad Abdalkader, Edoardo Pirera, Paolo Manganotti, Kyriakos Lobotesis, Thanh N Nguyen, Gian Luigi Gigli, Soma Banerjee and Giovanni Merlino in International Journal of Stroke
Supplemental Material
sj-jpeg-3-wso-10.1177_17474930251332489 – Supplemental material for C-reactive protein elevation and subsequent atrial fibrillation detection after ischemic stroke treated with mechanical thrombectomy: An inverse probability weighting analysis
Supplemental material, sj-jpeg-3-wso-10.1177_17474930251332489 for C-reactive protein elevation and subsequent atrial fibrillation detection after ischemic stroke treated with mechanical thrombectomy: An inverse probability weighting analysis by Lucio D’Anna, Gabriele Prandin, Mariarosaria Valente, Liqun Zhang, Paresh Malhotra, Simona Sacco, Matteo Foschi, Raffaele Ornello, Viva Levee, Katherine Chulack, Fahad Sheikh, Feras Fayez, Francesco Toraldo, Domenico Maisano, Caterina Del Regno, Filippo Komauli, Adelaida Gartner Jarmillo, Hakam AL-Karadsheh, Hamza Zahid, Piers Klein, Mohamad Abdalkader, Edoardo Pirera, Paolo Manganotti, Kyriakos Lobotesis, Thanh N Nguyen, Gian Luigi Gigli, Soma Banerjee and Giovanni Merlino in International Journal of Stroke
Supplemental Material
sj-jpeg-4-wso-10.1177_17474930251332489 – Supplemental material for C-reactive protein elevation and subsequent atrial fibrillation detection after ischemic stroke treated with mechanical thrombectomy: An inverse probability weighting analysis
Supplemental material, sj-jpeg-4-wso-10.1177_17474930251332489 for C-reactive protein elevation and subsequent atrial fibrillation detection after ischemic stroke treated with mechanical thrombectomy: An inverse probability weighting analysis by Lucio D’Anna, Gabriele Prandin, Mariarosaria Valente, Liqun Zhang, Paresh Malhotra, Simona Sacco, Matteo Foschi, Raffaele Ornello, Viva Levee, Katherine Chulack, Fahad Sheikh, Feras Fayez, Francesco Toraldo, Domenico Maisano, Caterina Del Regno, Filippo Komauli, Adelaida Gartner Jarmillo, Hakam AL-Karadsheh, Hamza Zahid, Piers Klein, Mohamad Abdalkader, Edoardo Pirera, Paolo Manganotti, Kyriakos Lobotesis, Thanh N Nguyen, Gian Luigi Gigli, Soma Banerjee and Giovanni Merlino in International Journal of Stroke
Supplemental Material
sj-jpeg-5-wso-10.1177_17474930251332489 – Supplemental material for C-reactive protein elevation and subsequent atrial fibrillation detection after ischemic stroke treated with mechanical thrombectomy: An inverse probability weighting analysis
Supplemental material, sj-jpeg-5-wso-10.1177_17474930251332489 for C-reactive protein elevation and subsequent atrial fibrillation detection after ischemic stroke treated with mechanical thrombectomy: An inverse probability weighting analysis by Lucio D’Anna, Gabriele Prandin, Mariarosaria Valente, Liqun Zhang, Paresh Malhotra, Simona Sacco, Matteo Foschi, Raffaele Ornello, Viva Levee, Katherine Chulack, Fahad Sheikh, Feras Fayez, Francesco Toraldo, Domenico Maisano, Caterina Del Regno, Filippo Komauli, Adelaida Gartner Jarmillo, Hakam AL-Karadsheh, Hamza Zahid, Piers Klein, Mohamad Abdalkader, Edoardo Pirera, Paolo Manganotti, Kyriakos Lobotesis, Thanh N Nguyen, Gian Luigi Gigli, Soma Banerjee and Giovanni Merlino in International Journal of Stroke
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.B. is a key opinion leader for RAPIDAI. T.N.N. is Associate Editor of Stroke; advisory board for Brainomix and Aruna Bio; and speaker for Genentech and Kaneka. All other authors have no conflicts of interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially funded by the Graham-Dixon charity.
Ethical approval
This study has obtained approval from the UK Health Regulator Authority (HRA) (HRA Reference No. 275260). The study has also received confirmation of capacity and capability from the Imperial College Healthcare NHS Trust.
Informed consent
Informed consent was not a legal requirement, as the research was carried out using data collected as part of routine care, and any researchers outside the direct care team only had access to anonymized data.
Data availability
Data available upon reasonable request.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
