Abstract
Background:
Secondary stroke prevention in patients with atrial fibrillation (AF) is one of the fastest growing areas in the field of cerebrovascular diseases. This scientific statement from the World Stroke Organization Brain & Heart Task Force provides a critical analysis of the strength of current evidence on this topic, highlights areas of current controversy, identifies knowledge gaps, and proposes priorities for future research.
Methods:
We select topics with the highest clinical relevance and perform a systematic search to answer specific practical questions. Based on the strength of available evidence and knowledge gaps, we identify topics that need to be prioritized in future research. For this purpose, we adopt a novel classification of evidence strength based on the availability of publications in which the primary population is patients with recent (<6 months) cerebrovascular events, the primary study endpoint is a recurrent ischemic stroke, and the quality of the studies (e.g. observational versus randomized controlled trial).
Summary:
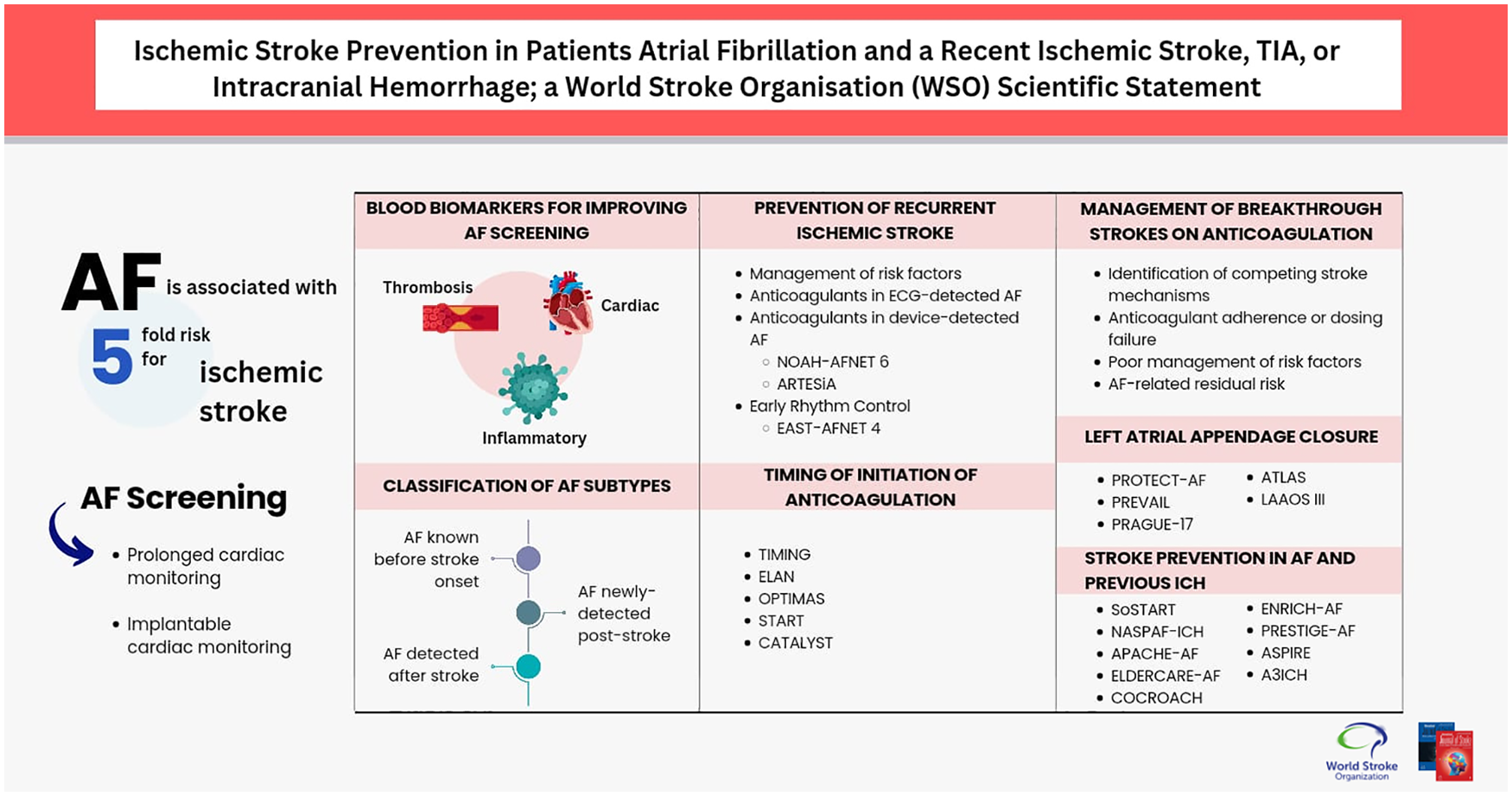
Priority areas include AF screening, molecular biomarkers, AF subtype classification, anticoagulation in device-detected AF, timing of anticoagulation initiation, effective management of breakthrough strokes on existing anticoagulant therapy, the role of left atrial appendage closure, novel approaches, and antithrombotic therapy post-intracranial hemorrhage. Strength of currently available evidence varies across the selected topics, with early anticoagulation being the one showing more consistent data.
Conclusion:
Several knowledge gaps persist in most areas related to secondary stroke prevention in AF. Prioritizing research in this field is crucial to advance current knowledge and improve clinical care.
Keywords
Introduction
Atrial fibrillation (AF) is a cardiac arrhythmia affecting approximately 59.7 million individuals globally as of 2019, which represents a 111% increase from 1990. 1 Population-based projections estimate a two- to threefold increase in the global prevalence of AF by 2050–2060 due to population growth, aging, and advanced AF detection methods. 2 AF is associated with a fivefold risk for ischemic stroke (IS), 3 is present in 18–30% of acute IS cases,4–6 and its prevalence in IS hospitalizations has increased to 22% in North America in recent decades.4,6 Several aspects of AF diagnosis and management have advanced significantly in the last decade. This position statement aims to review current evidence, classify its strength, and identify priority areas for future research.
Methods
The writing group selected relevant topics with clinical impact to be addressed in this document. We performed a systematic search for each topic (Supplemental Table S1). Statements were organized in sections focused on the diagnosis and management of AF patients with a recent IS, intracranial hemorrhage (ICH), or transient ischemic attack (TIA). Sections for which newer evidence was available or was more controversial were discussed more extensively than others. The aim was to evaluate the strength of current evidence and identify knowledge gaps for future research instead of providing clinical recommendations. We implemented a novel classification of evidence focused on clinical needs for physicians managing patients with acute IS. As such, we classified levels of evidence based on whether data addressed patients with a recent cerebrovascular event defined as ⩽6 months (as opposed to remote cerebrovascular events) before inclusion in randomized controlled trials (RCTs) or observational studies (Figure 1). The classification of strength of evidence also prioritized studies in which recurrent IS was the primary endpoint or a prespecified secondary endpoint. Members of the Writing Group and the World Stroke Organization Brain & Heart Task Force reviewed each statement and their level of evidence. If a co-author disagreed with a statement, the wording and level of evidence adjudication were revised until reaching a consensus. All authors approved the final version of each statement and level of evidence adjudication.
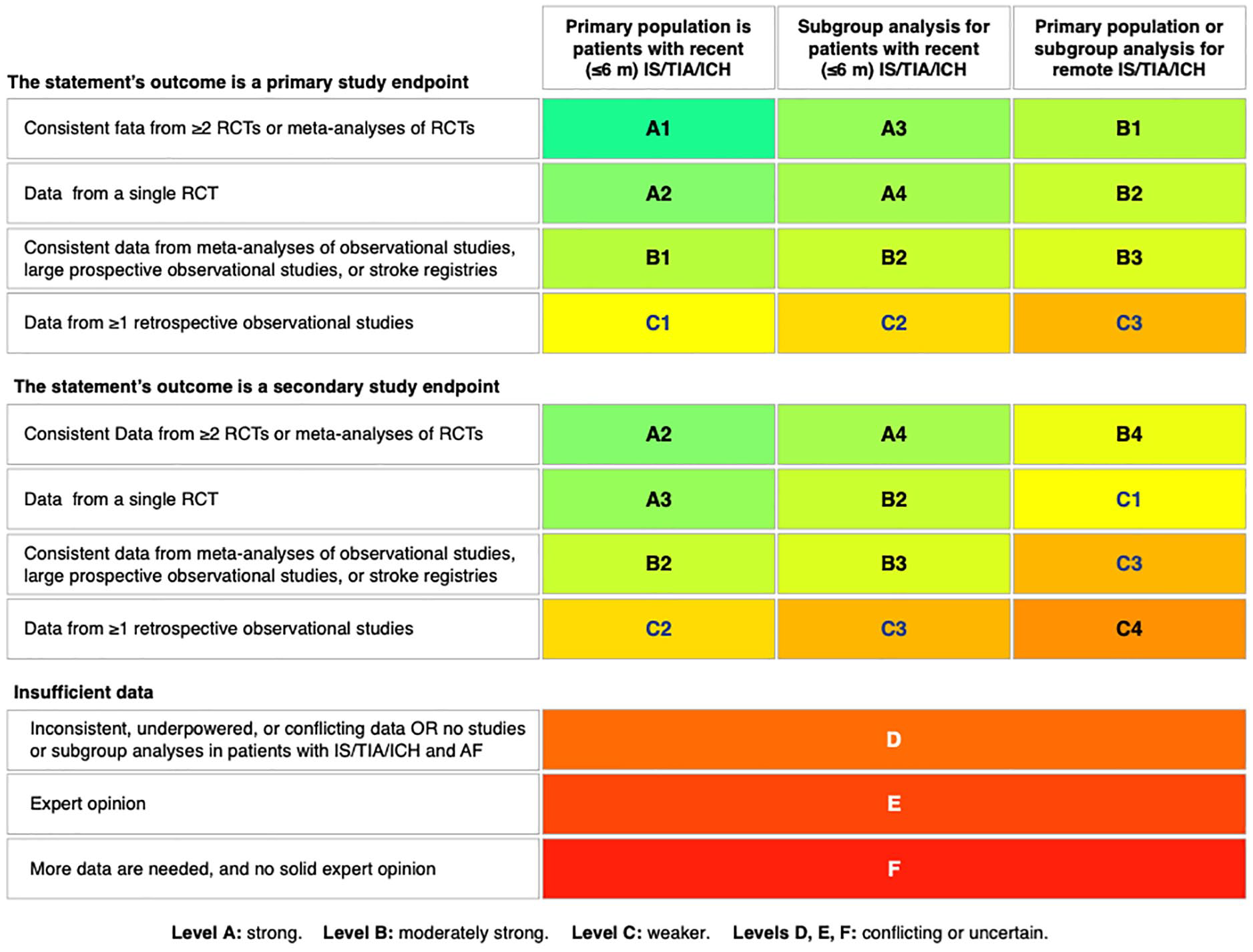
Classification of levels of evidence. RCT: randomized controlled trial; IS/TIA/ICH: ischemic stroke, transient ischemic attack or intracranial hemorrhage.
AF screening
AF is associated with AF recurrence and IS risk, and thus, prolonged cardiac monitoring (PCM) is used to screen for subclinical AF. In patients with IS and TIA, RCTs have shown significantly increased AF detection using external devices and implantable cardiac monitors (ICM) (Supplemental Table S2) than standard-of-care diagnostics. None of the RCTs on PCM was designed to test whether PCM reduces IS recurrence, and all were underpowered to show a significant effect. A study-level meta-analysis of six clinical trials with 68,556 patient-months of follow-up showed no association between PCM use and IS recurrence (incidence rate ratio (IRR): 0.90; 95% confidence interval (CI): 0.71–1.15), recurrent IS or TIA (IRR: 0.97; 95% CI: 0.80–1.18), or recurrent IS/ICH/TIA (IRR: 0.99; 95% CI: 0.80–1.20). 7 It must be noted that the type of cardiac monitoring (e.g. external vs implantable), duration (7 days to ≅3 years), and timing of initiation (3 days to 6 months) were heterogeneous across studies. Two RCTs are currently evaluating whether different intensities of PCM reduce stroke risk in patients with a recent IS or TIA (NCT04371055, NCT05134454).
Blood biomarkers for improving AF screening
Measuring blood biomarkers capable of identifying patients more likely to have PCM-detected AF could potentially streamline AF screening. Blood biomarkers can be classified into cardiac, thrombotic, and inflammatory. 8
Cardiac biomarkers
Elevated cardiac troponin has been associated with increased AF detection (area under the curve (AUC): 0.660–0.697) in several observational studies (Supplemental Table S3). 9 Natriuretic peptides are released from the cardiac atria or ventricles under strain.8,10 Although both N-terminal pro b-type natriuretic peptide (NT-proBNP) and midregional pro-atrial natriuretic peptide (MR-proANP) are associated with AF diagnosis post-stroke,11–15 NT-proBNP is less atrial-specific than MR-proANP. In the BIOSIGNAL (Biomarker Signature of Stroke Aetiology) study, which prospectively measured MR-proANP in 1759 patients within 24 h of acute IS onset, Log10MR-proANP levels were strongly associated with new AF diagnosis (adjusted odds ratio (aOR): 35.3, 95% CI: 17.6–71.0). 15 A simple model with age and MR-proANP showed good discrimination (AUC: 0.810) and higher net benefit than existing clinical AF risk scores.
Thrombosis biomarkers
Anti-thrombin III, D-dimer, and the MOCHA profile (markers of coagulation and hemostatic activation, including serum d-dimer, prothrombin fragment 1.2, thrombin-antithrombin complex, and fibrin monomer) have been associated with new AF detection, underlying malignancy, and stroke recurrence, with a good predictive ability when associated with left atrial volume index (AUC: 0.800).14,16 The AUC of thrombotic markers for AF detection was 0.700 in another study and appeared to be a stronger association with underlying malignancy and venous thromboembolism. 17
Inflammatory and novel biomarkers
In a larger systematic review and meta-analysis, there was only a non-significant trend toward association with AF detection among people with higher levels of C-reactive protein. 10 Novel biomarkers, including Bone morphogenic protein 10, 18 symmetric dimethylarginine, 19 and soluble suppression of tumorigenicity-2, 20 have been associated with AF detection in stroke patients but more evidence is needed. Cytokines (IL-4, IL-6, IL-10, tumor necrosis factor, interferon-gamma, etc.) have been associated with AF relative to sinus rhythm. IL-17 has been implicated in the pathogenesis of AF, 21 and IL-6 is associated with increased AF incidence in patients undergoing cardiac surgery22,23 and with AF recurrence after electrical cardioversion. 24 We did not identify any studies evaluating the role of cytokines for predicting AF detection in patients with a recent IS or TIA.
Classification of AF subtypes
Stroke recurrence rates according to the timing of AF diagnosis
The timing of AF detection relative to stroke onset and the intensity of monitoring determine the characteristics of the detected AF, with a gradient of stroke risk ranging from very high in patients with AF known before stroke onset to significantly lower risk in PCM-detected AF. 25 AF known before stroke occurrence is detected incidentally on 12-lead electrocardiograms (ECGs) performed during routine physical examination or when patients become symptomatic before they experience a stroke. Therefore, by the time it is diagnosed on an ECG, it has matured long enough to become a symptomatic high-burden arrhythmia. In contrast, AF detected on opportunistic PCM pursued post-stroke is generally an earlier and lower-burden arrhythmia. 25
Based on meta-analyses of RCTs and observational studies, AF in patients with a recent IS or TIA has been categorized into three main subtypes based on the timing of AF diagnosis: AF known before stroke onset or “Known AF” (KAF), AF newly-detected post-stroke on 12-lead ECG, and AF detected after stroke (AFDAS) on PCM, ranging from short (24 h or 48 h Holter) to long term (⩾7 days). 26 The rationale behind this categorization is that KAF has a higher prevalence of risk factors and vascular comorbidities, more severe left atrial substrate, greater AF burden, and higher risk of stroke recurrence than AFDAS.7,27 AF newly detected on 12-ECGs post-stroke has a fivefold higher risk of stroke recurrence than PCM-detected AF and is considered a pre-existing AF that remained undiagnosed until stroke occurrence despite being high-burden, with a risk profile similar to KAF. 28 Therefore, newly 12-lead ECG-detected AF at any time-point post-stroke should not be considered AFDAS and has a similar long-term risk of stroke as KAF (Figure 2). 26 AFDAS is always PCM-detected.
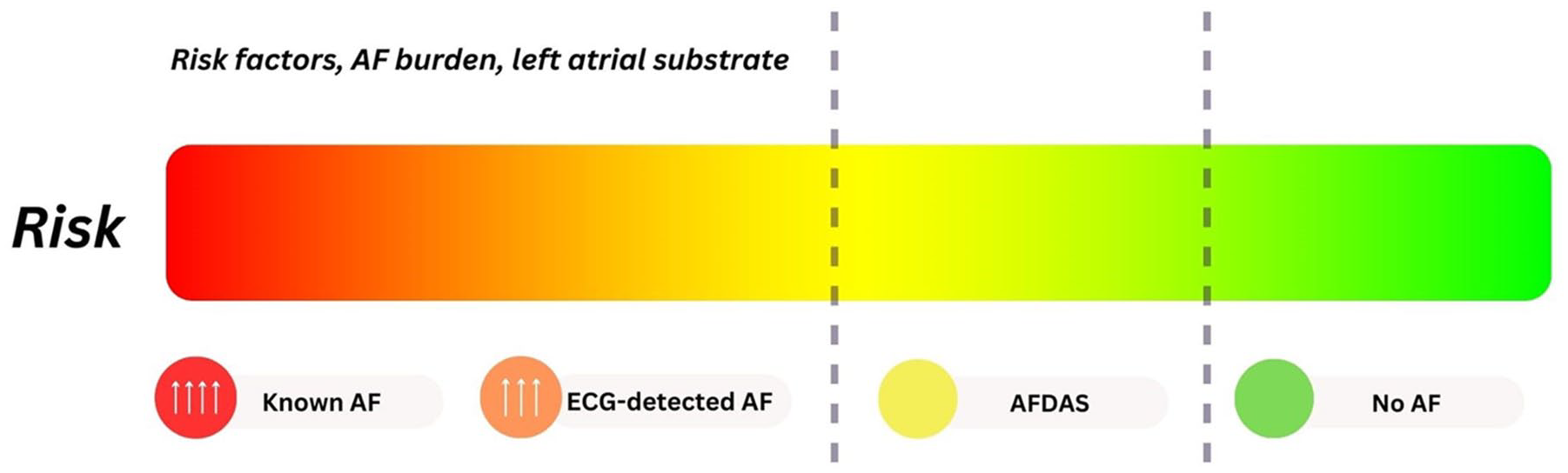
AF risk based on the timing of detection and intensity of cardiac monitoring. AF: atrial fibrillation; ECG: 12-lead electrocardiogram; AFDAS: atrial fibrillation detected after stroke.
Prevention of recurrent ischemic stroke
The pillars of IS prevention in AF are the management of risk factors, anticoagulation, rate/rhythm control, and minimizing the risk of bleeding. Patients with a recent IS or TIA usually undergo PCM, which adds a layer of complexity due to the wide range of AF burden found in this population.
Management of risk factors
Strong evidence from RCTs supports that optimizing the control of risk factors is crucial for IS prevention, regardless of the presence of AF. 29 In patients with AF, the strongest evidence from RCTs has shown that physical activity, reducing alcohol intake, and treating hypertension, sleep-disordered breathing, obesity, and diabetes can reduce AF incidence and recurrence.30,31 No specific RCT has assessed the effect of risk factor management on recurrent IS in AF patients with a recent IS or TIA.
RCTs of anticoagulants in ECG-detected AF
Robust evidence from multiple large RCTs and meta-analyses of RCTs demonstrates that vitamin K antagonists (VKA) reduce IS risk by approximately 67% compared to placebo or no therapy, and the risk of stroke (ischemic and hemorrhagic) by 38% relative to Aspirin. 32 In more recent RCTs, direct oral anticoagulants (DOACs) were at least as effective as VKAs for the prevention of IS (relative risk (RR): 0.92, 95% CI: 0.83–1.02), resulting in a 52% lower risk of ICH (RR: 0.48; 95% CI: 0.39–0.59) and 19% lower risk of stroke/SE (RR: 0.81; 95% CI: 0.73–0.91) in patients with and without a remote stroke/TIA. 33
Secondary analyses of RCTs of anticoagulants in device-detected AF
NOAH-AFNET 6 (Non-vitamin K Antagonist Oral Anticoagulants in Patients With Atrial High Rate Episodes) reported neutral findings in a randomized trial comparing edoxaban 60 mg daily versus placebo or aspirin for the prevention of stroke, systemic embolism (SE), or cardiovascular death in patients 65 years of age or older with subclinical device-detected AF lasting ⩾6 min and at least one risk factor. 34 It was stopped early due to excess major bleeding with edoxaban and had a low number of stroke events, potentially limiting the trial’s power to detect differences in the primary efficacy outcome. In contrast, the ARTESiA (Apixaban for Stroke Prevention in Subclinical Atrial Fibrillation) trial reported superior prevention of stroke or SE with random assignment to apixaban 5 mg twice daily compared with aspirin 81 mg daily in patients 55 years of age or older with subclinical device-detected AF lasting 6 min to 24 h. 35 An aggregate meta-analysis of the two trials demonstrated that oral anticoagulation with these agents reduced IS risk (RR: 0.68, 95% CI: 0.50–0.92) and reported consistent estimates of treatment effect between the two trials (I2 = 0%). 36 However, less than 10% of participants in these trials had a history of IS or TIA. Subanalyses from NOAH-AFNET 6 and ARTESiA comparing the effect of DOACs versus aspirin or placebo on IS recurrence risk in patients with remote IS or TIA were conflicting (Supplemental Table S4). In both trials, DOACs significantly increased major bleeding risk.
Concerns have been raised by experts37–39 and recent guidelines 31 regarding a one-size-fits-all approach for anticoagulation in IS or TIA patients with device-detected subclinical AF lasting <24 h. A more comprehensive and personalized approach considering the interplay of AF burden, atrial substrate, and time between stroke occurrence and AF diagnosis has been proposed for patients with AFDAS. For instance, the B 2 AD-RISK scheme, which comprises the longitudinal measurement of biomarkers (B), AF burden (B), atrial substrate (A), age and sex demographics (D), and risk factors (R), is currently being tested in a pilot study (NCT0658970). 26
Early rhythm control
The EAST-AFNET 4 (Early Treatment of Atrial Fibrillation for Stroke Prevention Trial) trial randomized 2789 patients with AF diagnosed within the previous 12 months to early rhythm control (ERC) with antiarrhythmic drugs or ablation versus standard of care. The primary composite efficacy outcome of cardiovascular death, stroke, or hospitalization with worsening of heart failure or acute coronary syndrome was less frequent in the ERC group (hazard ratio: HR: 0.79, 95% CI: 0.66–0.94). Patients receiving ERC had a lower risk of stroke than the control group (HR: 0.65, 95% CI: 0.44–0.97). Several observational studies and a subanalysis of EAST-AFNET 4 in patients with prior IS or TIA have shown similar results. 40 A small open-label, randomized, multicenter RCT including 300 patients with acute IS and AF within 2 months of stroke onset found lower recurrent IS rates in patients undergoing ERC than in those receiving usual care (HR: 0.251; 95% CI: 0.063–1.003). 41 EAST-STROKE (Early Treatment of Atrial Fibrillation for Stroke Prevention Trial in Acute STROKE) will test a similar approach in patients with recent ischemic cerebrovascular events (NCT05293080).
Timing of initiation of anticoagulation
Clinicians considering early initiation of anticoagulation therapy must balance the potential benefit of improved recurrent stroke prevention on the one hand and the potential harm of symptomatic ICH on the other hand. Observational studies found that DOAC therapy is initiated early (median within 4 days) after a recent stroke in clinical routine, even in the absence of RCT data and despite more conservative historical guideline recommendations.42–47 Several registry-based observational studies were conducted to answer the “timing question” in various populations (Supplemental Table S5).42–47 These studies found no strong evidence of a heightened ICH risk in patients with early initiation of anticoagulation. However, most studies artificially split timing to make a comparison. The most relevant limitations are that early and late start were defined differently across studies, with thresholds ranging from ⩽2 days to ⩽7 days; they had retrospective designs with risk of confounding by indication, and had no standardized procedures for early or later treatment selection.
Three RCTs specifically addressed the topic of early or late initiation of DOACs. The TIMING (Timing of Oral Anticoagulant Therapy in Acute IS With Atrial Fibrillation) was a registry-based noninferiority RCT that randomized 888 IS patients (median NIHSS 4) with AF admitted within 72 h of symptom onset to either early (⩽4 days) or delayed (5–10 days) start of DOAC treatment. 48 Early DOAC initiation was non-inferior to delayed start (IS rates: 3.1% vs 4.6%), and no patient in either group experienced a symptomatic ICH. ELAN (Early versus Late Initiation of Direct Oral Anticoagulants in Post-IS Patients with Atrial Fibrillation) is the largest RCT comparing early versus later initiation of DOAC treatment in AF-related IS. 49 The time frame for early or late start of DOAC treatment was defined according to the infarct size on neuroimaging. 49 Patients with minor or moderate stroke randomized to early initiation of DOACs were started within 48 h, and patients with major stroke on day 6 or 7 (n = 1006). The primary outcome, a composite of recurrent IS, SE, major extracranial bleeding, symptomatic ICH, or vascular death within 30 days, occurred in 2.9% versus 4.1% in the early and late groups, respectively. Numerically, fewer patients in the early group had recurrent IS within 30 days (1.4% vs 2.5%). Two patients in each group had symptomatic ICH. The OPTIMAS (Optimal Timing of Anticoagulation After Acute Ischaemic Stroke) trial was a phase 4, multicenter, parallel-group, RCT applying an open-label intervention and blinded endpoint adjudication. 50 It used a hierarchical non-inferiority-superiority gatekeeper design (sequentially assessing a non-inferiority margin of 2 percentage points and then proceeding to test for superiority) to compare early initiation of DOACs (within 4 days after stroke onset) versus delayed initiation (7–14 days following stroke onset) in 3621 patients with AF and IS. 50 The primary endpoint was a composite of recurrent IS, symptomatic ICH, stroke of unknown type, or systemic embolism at 90 days in a modified intention-to-treat analysis. Early DOAC initiation within 4 days post-IS was noninferior to delayed initiation for the composite primary endpoint. Early initiation was not superior to late initiation.
CATALYST (CollAboration on the optimal Timing of anticoagulation after ischaemic stroke and Atrial fibrillation: prospective individuaL participant data meta-analYsiS of randomized controlled Trials) is an individual participant data meta-analysis of RCTs investigating the optimal timing of DOAC initiation after acute IS in patients with AF. CATALYST included data from 5411 patients from TIMING, ELAN, OPTIMAS, and START. 51 The primary endpoint was a composite of recurrent IS, symptomatic intracerebral hemorrhage, or unclassified stroke at 30 days. Early DOAC initiation (within 4 days) was superior to later initiation (⩾5 days) for the primary endpoint at 30 days (2.12% vs 3.02%, odds ratio (OR): 0.70, 95% CI: 0.50–0.98). Symptomatic intracerebral hemorrhage rates were low in both the early and late groups: 0.45% and 0.40%, respectively. At 90 days, primary endpoint events were numerically lower in the earlier than the later group, but without reaching statistical significance. The CATALYST meta-analysis supports the initiation of DOACs early after acute IS in patients with atrial fibrillation.
Management of breakthrough strokes on anticoagulation
Data from RCTs and population-based studies show that approximately 1% of patients on DOACs experience a breakthrough IS annually. 52 Recurrent IS risk is particularly high in patients with breakthrough stroke ranging between 5% and 9% annually. 53 While suboptimal adherence to anticoagulants is still common, 54 breakthrough strokes can occur even with the best medication compliance and prescribing practices. 53 Several aspects must be considered before labeling a breakthrough event as DOAC failure-related. The specific cause of breakthrough strokes can be identified by applying a comprehensive and systematic investigation, which in turn can help tailor secondary prevention strategies (Figure 3). 55
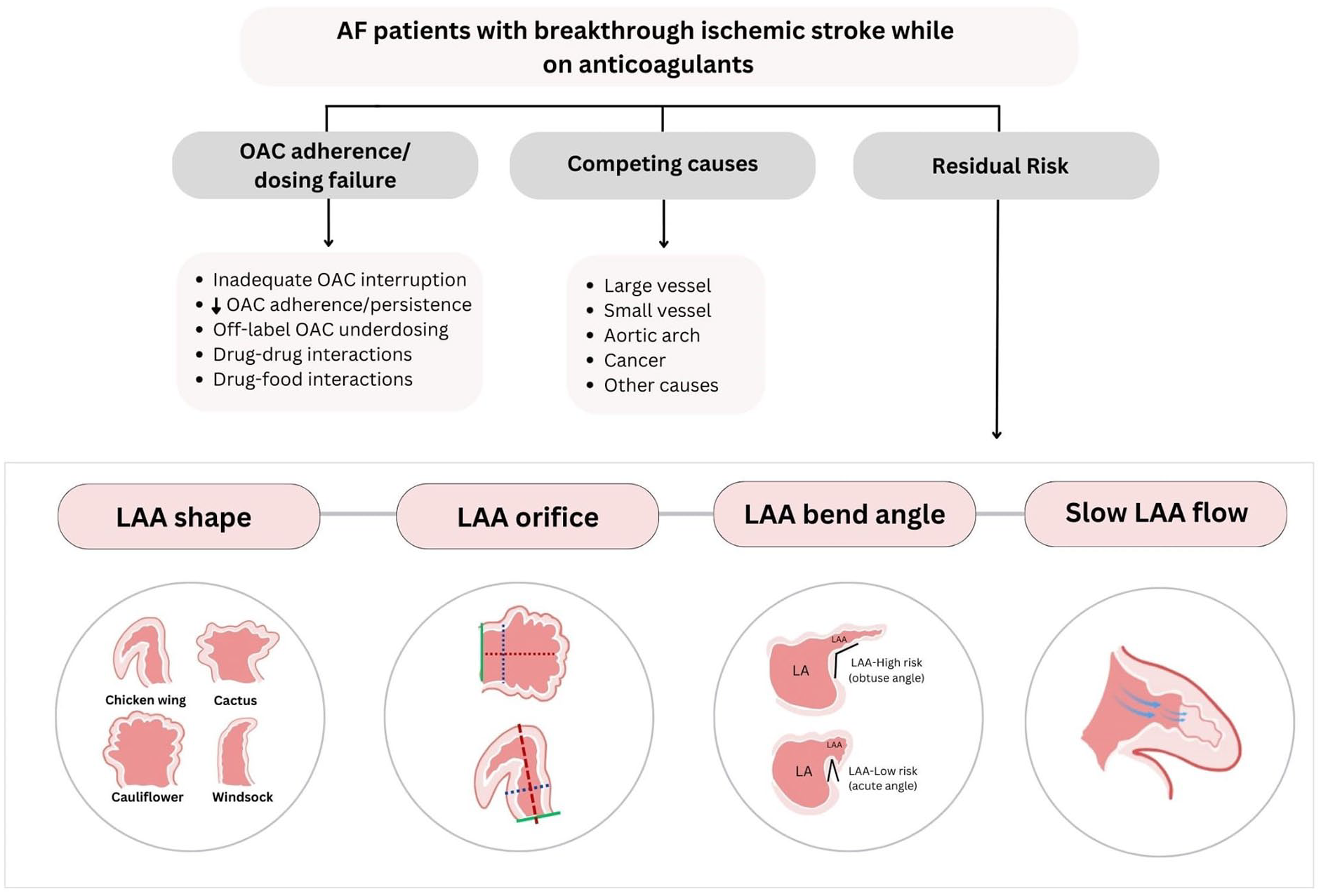
Etiological investigation of breakthrough strokes. OAC: oral anticoagulant; AF: atrial fibrillation; LAA: left atrial appendage.
Identification of competing stroke mechanisms
The proportion of breakthrough strokes explained by competing mechanisms other than AF ranges between 24% and 35%.56,57 While some series have shown that competing mechanisms are more frequent in AF patients on anticoagulants at the time of the event than among those off anticoagulation, 57 others have shown a similar prevalence on and off anticoagulation. 58 Among competing causes, the most frequently reported are large (18–61%) and small (25–26%) artery disease.56–58 Cancer-related coagulopathy is a potential competing mechanism. Approximately 7% of patients with AF have cancer and this is associated with an increased risk of IS (e.g. breast). 59 If cancer-related coagulopathy is suspected, further targeted investigations should be undertaken if the results are likely to change treatment.
Anticoagulant adherence or dosing failure
Adherence or dosing issues represent 32% of all breakthrough strokes. 56 Poor adherence and persistence are the leading causes of inefficient anticoagulation in patients with AF. In a meta-analysis of 48 observational studies including 594,784 AF patients, the pooled proportion of good adherence to oral anticoagulants at 12 months, defined as >80% of days covered or medication possession ratio, was only 68%. 54 Similarly, the pooled proportion of persistence on anticoagulation at 12 months was 62%. 54 Both non-persistence (HR: 4.6; 95% CI: 2.8–7.4) and poor adherence (HR: 1.4; 95% CI: 1.06–1.8) were associated with increased stroke risk. 54 Other causes of inefficient anticoagulation beyond the scope of this work but still of clinical importance include poor absorption, underdosing, drug–drug interactions, and inappropriate interruption surrounding surgical procedures. 60
Poor management of risk factors
As discussed previously, the management of risk factors is an essential component of stroke prevention, which is sometimes suboptimal, and explains a proportion of stroke recurrences. 29 Although not a stroke mechanism, part of the risk of stroke recurrence can be explained by poor control of risk factors.
AF-related residual risk
The most frequent cause of breakthrough strokes in patients on optimal anticoagulation and no competing mechanisms is cardioembolism from AF-related residual risk (44%). 56 This risk is explained by AF-specific structural and functional factors, including left atrial appendage (LAA) morphology (e.g. LAA shape, 61 bend angle, 62 and orifice size) 63 and flow. 64 Approximately 90% of cardiac thrombi in patients with AF originate in the LAA. 65 The prevalence of LAA thrombus among individuals receiving DOACs is approximately 2.3%. 66 Among AF patients on DOACs, LAA thrombi seem to be more frequent in those with a prior stroke than among those without 67 and the general population. 66 These LAA features can be investigated with transesophageal echocardiography, cardiac computed tomography, and cardiac magnetic resonance imaging.
Secondary stroke prevention in patients with breakthrough strokes
Given the high risk of early recurrence, secondary stroke prevention in patients with breakthrough strokes is essential. There are no data from RCTs evaluating whether switching a DOAC to a different DOAC or a VKA at the time of experiencing a breakthrough stroke reduces recurrent stroke risk. A study-level meta-analysis of six retrospective observational studies comprising 12,159 patients suggests that remaining on a DOAC instead of switching from DOACs to VKAs is associated with lower risk of IS recurrence (RR: 0.55; 95% CI: 0.43 -0.70) and ICH (RR: 0.37, 95% CI: 0.25 -0.55). Additionally, switching from a DOAC to a VKA was associated with an 85% increased risk of death compared to remaining on a DOAC (RR: 1.85; 95% CI: 1.06 -3.24). 68 This analysis is subject to the limitations of retrospective observational studies. In observational studies, adding an antiplatelet agent to anticoagulants was not associated with lower IS risk reduction. 56 In a subanalysis of RCTs, adding an antiplatelet agent was linked to increased ICH risk, 69 and a meta-analysis of RCTs and observational studies showed overall increased bleeding risk. 70 Evidence is missing for short-term addition of antiplatelet agents in patients with a competing large-artery stroke mechanism. The Frail Atrial Fibrillation (FRAIL AF) trial randomized frail individuals (⩾75 years of age and a Groningen Frailty Indicator score ⩾3) with AF who were receiving VKAs to continue VKA therapy versus switching to a DOAC. 71 The primary outcome of major or clinically relevant nonmajor bleeding complication was more frequent in the DOAC group (HR: 1.69; 95% CI: 1.23-2.32), without differences in the risk of thromboembolic events at 12 months of follow-up. The proportion of patients with a previous stroke and the risk of IS were not reported.
A potential novel option for patients with breakthrough stroke is LAA occlusion as a matched observational cohort study found a lower risk of recurrent stroke compared to standard–of-care DOAC therapy alone in patients with breakthrough stroke (HR: 0.33, 95% CI: 0.19–0.58). 72 Although promising, a major limitation of these data is that for the LAA occlusion patients, follow-up started from the moment of the LAA occlusion procedure bypassing the high-risk early post-stroke time period, while for the non-LAA occlusion cohort, follow-up started immediately after the index event (inclusive of the high-risk early post-stroke time period) introducing substantial bias in favor of LAA occlusion. As such, uncertainty remains and this approach is currently being investigated in RCTs (NCT05976685; NCT05963698).
Left atrial appendage closure and other interventions
The left atrial appendage (LAA) is the primary cardioembolic structural source in AF patients. 65 As such, LAA closure (LAAC) has been tested in several RCTs as a potential strategy for stroke prevention in patients with AF.
Studies of percutaneous LAAC versus VKAs
The PROTECT-AF (Watchman Left Atrial Appendage System for Embolic Protection in Patients with Atrial Fibrillation) trial compared VKAs versus percutaneous LAAC in 707 anticoagulant-naïve AF with a CHADS2 score ⩾1. 73 LAAC met prespecified criteria for noninferiority and superiority (rate ratio (RR): 0.60, 95% CI: 0.41–1.05) for its primary efficacy endpoint (composite of stroke, SE, and cardiovascular/unexplained death). 73 The PREVAIL (Prospective Randomized Evaluation of the Watchman LAA Closure Device In Patients With Atrial Fibrillation Versus Long Term Warfarin Therapy) trial compared VKAs versus LAAC in 407 anticoagulant-naïve AF patients with a CHADS2 score ⩾2 or 1 and another risk factor. 74 Percutaneous LAAC was non-inferior to warfarin for IS prevention or SE >7 days post-closure but did not achieve the prespecified noninferiority threshold for the composite endpoint of stroke, SE, and cardiovascular or unexplained death. 74 A prospective registry found no differences in outcomes in patients with and without a prior stroke. 75
Studies of percutaneous LAAC versus DOACs
The PRAGUE-17 (Left Atrial Appendage Closure vs Novel Anticoagulation Agents in Atrial Fibrillation) trial included 402 AF patients with at least one of the following: bleeding requiring intervention or hospitalization; breakthrough stroke while on anticoagulants or CHA2DS2-VASc score ⩾3 + HAS-BLED score ⩾2. Patients were randomized to percutaneous LAAC versus DOACs. 76 LAAC was non-inferior to DOAC in preventing the composite outcome of stroke, TIA, SE, cardiovascular death, major or nonmajor clinically relevant bleeding, or procedure-/device-related complications. There were no significant differences between groups in the risk of IS or TIA (HR: 1.13, 95% CI: 0.44–2.93) or major/non-major bleeding (HR: 0.81, 95% CI: 0.44–1.52). A propensity-matched analysis comparing percutaneous LAAC versus DOACs in 587 patients with AF and a prior stroke (median time between stroke and LAAC of 7.6 months) showed no differences in the rates of IS and ICH, but a lower risk of the primary composite outcome of IS, major bleeding, and all-cause death. 77
Trials of surgical LAAC
The ATLAS (AtriClip Left Atrial Appendage Exclusion Concomitant to Structural Heart Procedures) pilot trial included patients undergoing nonmechanical valve and/or coronary artery bypass grafting without preoperative AF or needing anticoagulation, CHA2DS2-VASc score ⩾2, and HAS-BLED score ⩾2. 78 A total of 562 patients were randomized to surgical LAA exclusion (LAAE) versus no exclusion. 78 The proportion of patients with postoperative AF was 44.3%. The proportion of thromboembolic events was 3.4% in LAAE patients and 5.6% in the no-LAAE group. 78 LAAOS III (Left Atrial Appendage Occlusion Study) randomized 4770 AF patients with a CHA2DS2-VASc score ⩾2 undergoing cardiac surgery to surgical LAAE versus no-LAAE. 79 Surgical LAAE reduced the risk of stroke or SE compared to no-LAAE (HR: 0.67; 95% CI: 0.53–0.85) in a population where 80% of patients continued to receive oral anticoagulation. 79 Results were consistent in interaction analysis for patients with and without prior IS, TIA or SE. 79 The results from LAAOS III have catalyzed several ongoing trials testing the combination of mechanical therapy (percutaneous LAAC or carotid filter devices) combined with oral anticoagulation for improved stroke prevention in AF patients who remain at high risk of stroke despite anticoagulation. 53
Percutaneous carotid filters
VineTM is a novel permanent common carotid filter system that is implanted percutaneously under ultrasound guidance. It has been designed to prevent emboli >1.4 mm that result in large vessel occlusions from reaching the anterior circulation, which is affected by the majority of AF-related ischemic strokes.80,81 On the basis of a promising phase 2 program establishing the feasibility and safety of the device, in which there were no strokes due to large vessel occlusion following carotid filter implantation in over 268 patient-years of follow-up (106 participants), the INTERCEPT (Carotid Implants for PreveNtion of STrokE ReCurrEnce From Large Vessel Occlusion in Atrial Fibrillation Patients Treated With Oral Anticoagulation) RCT (NCT05723926) will be testing the superiority of bilateral carotid filter implantation + DOAC versus DOAC alone in patients with AF and stroke within the past year.82,83
Secondary prevention in AF and previous ICH
Patients with a previous ICH, particularly intracerebral hemorrhage, have an inherently high risk of recurrent ICH. Therefore, the decision to start or reinstate antithrombotic therapy in these patients is challenging.
Anticoagulation
Observational studies suggest that resumption of anticoagulation after ICH may be associated with reduced thromboembolic events without an offsetting increase in the risk of ICH recurrence. 84 In an individual patient-level meta-analysis combining information from three small early-phase RCTs and subgroup data of a single-phase III RCT totalling 412 patients with an ICH and AF, oral anticoagulation did not lead to a significant reduction in the primary outcome of any stroke or cardiovascular death. 85 Patients who reinitiated anticoagulation had a lower frequency of recurrent IS (4% vs 19%) and major ischemic cardiovascular events, including IS, SE, pulmonary embolism, and myocardial infarction (4% vs 19%). However, anticoagulation had numerically higher ICH recurrence events (6% vs 3%). 85 Ongoing phase 3 clinical trials testing the safety and efficacy of anticoagulation in ICH survivors with AF will provide more evidence to inform clinical decision-making in the future (Supplemental Table S6).
The ENRICH-AF (Edoxaban for Intracranial Haemorrhage Survivors with Atrial Fibrillation trial) trial is comparing standard dosing edoxaban with non-anticoagulant medical treatment for stroke prevention in intracranial hemorrhage survivors with atrial fibrillation. 86 Following an initial safety review of the first 699 patients—where 174 (25%) presented with lobar intracranial hemorrhage and 34 (5%) with isolated convexity subarachnoid hemorrhage—the trial’s Data Safety Monitoring Board advised halting the enrollment of those with these two hemorrhage subtypes, which are typically caused by underlying cerebral amyloid angiopathy. They additionally recommended discontinuing the study drug immediately in this subgroup of patients. 86 The data leading to this recommendation have yet to be published, and no treatment interactions were identified in patients with lobar or isolated convexity subarachnoid hemorrhage in the abovementioned meta-analysis. 85 Further data from ongoing RCTs where patients with CAA-related intracranial hemorrhage remain eligible, and repeated meta-analyses will be important to clarify the net-benefit in these high-risk patients.
Antiplatelet therapy
Antiplatelet monotherapy, while inferior to anticoagulation, offers a modest 23% reduction in the risk of thromboembolic events in patients with AF relative to placebo. 32 RESTART (REstart or STop Antithrombotics Randomised Trial) included 537 participants with ICH and a prior history of ischemic vascular disease. 87 Antiplatelet therapy did not increase the risk of recurrent ICH at a median follow-up of 2 years. Still, it significantly reduced the risk of major ischemic vascular disease, although it must be noted that only 25% (134/537) of the patients had AF. 87 The effect of Aspirin compared to no treatment or placebo in a population exclusively comprising AF patients with a prior ICH has not been tested in an RCT.
LAAC in patients with ICH
Meta-analyses have indicated that LAAC may have similar efficacy to warfarin in lowering the risk of IS, but the risk of ICH may be significantly lower with LAA closure. 88 Data on head-to-head comparisons between LAAC and DOACs in patients with prior ICH are lacking. In addition, given that studies evaluating LAAC excluded patients with ICH, it is unclear if these results can be extrapolated to patients with ICH.
Timing of antithrombotic therapy post-ICH
Clinical equipoise exists on the optimal timing of antithrombotic therapy after ICH. Literature-based estimates on the optimal timing of OAC following intracranial hemorrhage range broadly from 3 days to 30 weeks.89,90 While antiplatelet medications were started at a median of 76 days (interquartile range (IQR): 29–146) in the RESTART trial, 87 observational data are inconclusive. 91
Knowledge gaps and future directions
Despite significant advances in stroke prevention in patients with AF, we identified several knowledge gaps in this position statement. Relevant ongoing RCTs and observational studies addressing these gaps are listed in the supplementary file (Supplemental Table S6). This group has identified several research questions of clinical relevance that should be addressed in future studies (classified with a level of evidence C, D, E, or F in Table 1).
WSO Brain & Heart Task Force Statements on AF in Patients With Recent IS, TIA and ICH.
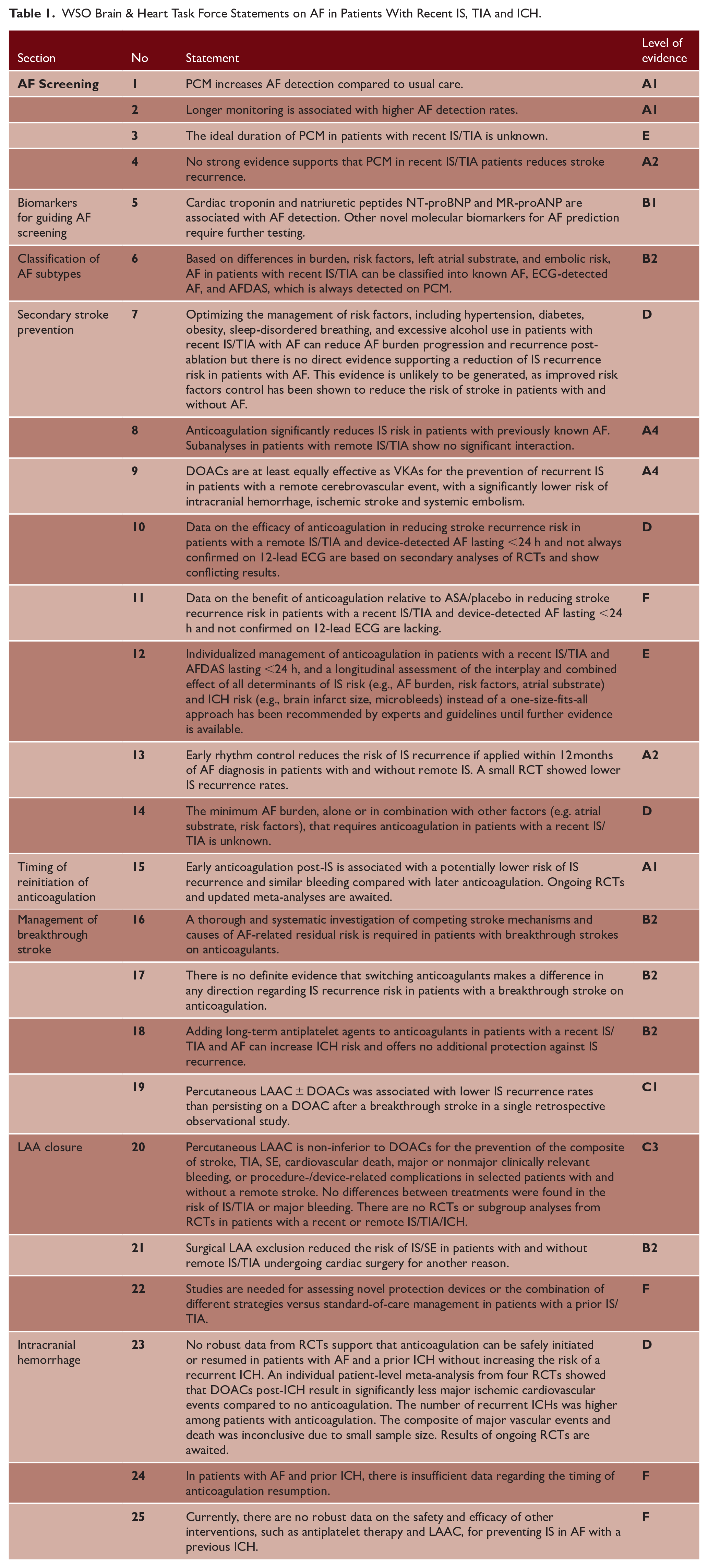
AF screening and classification of AF subtypes
Differences between subtypes of AF and their specific risk of IS outcomes are well-established. However, the clinical impact of this classification has not yet been demonstrated. Future studies are required to assess how the interplay between timing of AF detection, intensity of monitoring, AF burden, risk factors, and blood biomarkers affects stroke recurrence risk. It is also crucial to understand how AF burden and left atrial substrate progress over time. Given the increased risk of ICH resulting from the addition of antiplatelet agents to DOACs, there are concerns about potential harm when screening for AF in patients with an established competing cause (e.g. severe carotid artery stenosis). Therefore, the clinical implications of detecting AF in patients with a defined cause of stroke remains to be determined and adding oral anticoagulants to antiplatelet therapy in this population should be investigated in RCTs. One of the most pressing uncertainties, due to its potential impact on health care costs and clinical outcomes, is the ideal duration of monitoring for AF detection in patients with a recent ischemic cerebrovascular event. Whether a single device approach or is better than stepwise combination of short-term followed by longer-term cardiac monitoring in selected patients is unknown.
Blood biomarkers
Evidence supporting the role of blood biomarkers, mainly natriuretic peptides, suggests they could be incorporated into clinical practice to select patients who may benefit from PCM. In addition, due to the association with AF-related outcomes, using natriuretic peptides may also help identify patients who could benefit from anticoagulation if AF is detected. RCTs specifically addressing these questions are needed. The results of the MOSES (MidregiOnal Proatrial Natriuretic Peptide to Guide SEcondary Stroke Prevention) trial are awaited (NCT03961334). Other blood biomarkers are at earlier stages of investigation or are less specific for AF detection.
Secondary stroke prevention in AF and management of breakthrough strokes
Given the increasing use of PCM, one of the main uncertainties is whether oral anticoagulation can reduce stroke recurrence risk in patients with a recent IS or TIA and device-detected AF lasting <24 h and not confirmed on 12-lead ECG are lacking, relative to antiplatelet therapy. Early rhythm control therapy has proven to be effective in patients with remote cerebrovascular events. However, no large, multicenter RCT has demonstrated its benefit in patients with a recent IS or TIA. EAST-STROKE study will address this question. There is no clear strategy for the management of patients with breakthrough strokes. Several strategies are being tested, including switching from a DOAC to a VKA versus staying on a DOAC, carotid filter implantation + DOAC, and LAAC versus LAAC + DOAC therapy.
Resuming or starting anticoagulation post-ICH
Whether resuming or starting anticoagulation in patients with a previous ICH is safe, improves survival, or effectively provides net benefit remains unknown. The results of PRESTIGE-AF (PREvention of STroke in Intracerebral haemorrhaGE Survivors With Atrial Fibrillation, NCT03996772) and ENRICH-AF, which have completed recruitment, are awaited, and the ASPIRE (Anticoagulation in ICH Survivors for Stroke Prevention and Recovery, NCT03907046) and A3ICH (Avoiding Anticoagulation After IntraCerebral Haemorrhage, NCT03243175) trials are ongoing. The role of other approaches such as LAAC versus antiplatelet therapy or anticoagulation requires further investigation.
In summary, many uncertainties remain on how to screen for AF and how to prevent AF-related strokes in varying scenarios. Funding agencies should prioritize research in these fields. Academic–industry partnerships are also strongly encouraged to advance knowledge.
Supplemental Material
sj-docx-1-wso-10.1177_17474930241312649 – Supplemental material for Ischemic stroke prevention in patients with atrial fibrillation and a recent ischemic stroke, TIA, or intracranial hemorrhage: A World Stroke Organization (WSO) scientific statement
Supplemental material, sj-docx-1-wso-10.1177_17474930241312649 for Ischemic stroke prevention in patients with atrial fibrillation and a recent ischemic stroke, TIA, or intracranial hemorrhage: A World Stroke Organization (WSO) scientific statement by Luciano A Sposato, Alan C Cameron, Michelle C Johansen, Mira Katan, Santosh B Murthy, Micaela Schachter, Nicole B Sur, Shadi Yaghi, Sara Aspberg, Valeria Caso, Cheng-Yang Hsieh, Max J Hilz, Antonia Nucera, David J Seiffge, Mary N Sheppard, Sheila CO Martins, M Cecilia Bahit, Jan F Scheitz and Ashkan Shoamanesh in International Journal of Stroke
Footnotes
Acknowledgements
We thank Dr. Alba Hernandez Pinilla and Ioannis Farah for their contribution to the systematic search of current evidence and ongoing studies.
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LAS: Speaker/consulting honoraria from Boehringer Ingelheim, Pfizer, Bayer, AstraZeneca, Medtronic; ACC: Speaker honoraria from BMS, Pfizer, AstraZeneca, and Boehringer Ingelheim; MK: Speaker/Consulting honoraria from Astra Zeneca, BMS, Medtronic; NBS: Consulting honoraria from Medtronic; VC: Speaker/consulting honoraria from Boehringer Ingelheim, Pfizer, Bayer, EVER PHARMA, Daiichi Sankyo; C-YH: Speaker honoraria from Boehringer Ingelheim, Daiichi Sankyo, Pfizer, Bayer, Medtronic; MJH: Speaker honoraria from Sanofi; DJS: Speaker/consulting honoraria from Pfizer and AstraZeneca; MCB: Speaker/consulting honoraria from J&J, Anthos Therapeutics, Merck; JFS: Speaker honoraria from BMS, Pfizer, AstraZeneca; AS: Speaker/consulting honoraria from AstraZeneca, Bayer AG, Daiichi Sankyo Ltd, Javelin Inc; other authors: no disclosures relevant to this work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: LAS: Kathleen and Dr Henry Barnett Chair in Stroke Research; ACC: David Cargill Trust Clinical Senior Research Fellow; JFS: Support from Deutsche Herzstiftung e.V; MK: Swiss National Science Foundation (NR 182267, NR 213471) and Swiss Heart Foundation; other authors: no funding relevant to this work.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
