Abstract
Introduction:
Prolonged cardiac monitoring after stroke increases the detection of device-detected atrial fibrillation (DDAF), leading to a clinical dilemma regarding anticoagulation for secondary stroke prevention. While anticoagulation reduces thromboembolic risk in clinical AF, its benefit-risk profile in DDAF remains uncertain.
Methods:
In this systematic review and meta-analysis, randomized-controlled clinical trials (RCTs) evaluating anticoagulation among patients with DDAF post stroke or transient ischemic attack (TIA) were pooled. The primary efficacy outcome was any stroke recurrence, while stroke or systemic embolism, ischemic stroke recurrence, myocardial infarction, and cardiovascular mortality were assessed as secondary efficacy outcomes. The primary safety outcome was major bleeding, while hemorrhagic stroke and all-cause mortality were assessed as secondary safety outcomes.
Results:
Two RCTs with 599 patients (294 anticoagulation, 305 no-anticoagulation) were included. Anticoagulation significantly reduced any stroke recurrence (RR: 0.47; 95% CI: 0.23–0.94;
Discussion:
Anticoagulation in DDAF patients with prior stroke/TIA reduces any stroke recurrence but increases major bleeding risk without raising hemorrhagic stroke incidence. This trade-off underscores the need for individualized risk stratification.
Conclusions:
Anticoagulation lowers any stroke recurrence in DDAF patients post-stroke/TIA but raises major bleeding risk.

Introduction
Prolonged cardiac monitoring after stroke has been associated with increased detection of atrial fibrillation (AF), often leading to the initiation of anticoagulation for secondary stroke prevention.1–3 However, the impact of prolonged monitoring on stroke recurrence remains uncertain.1–3 Subclinical AF detected post-stroke has been hypothesized to result from heart-brain interactions following ictus in some cases – particularly in the context of insular infarction – yet, early high-burden AF may be causally linked to the index event rather than being a secondary phenomenon. 4 The precise relationship between subclinical AF detected after stroke and future ischemic events remains an area of ongoing investigation.5,6
A key clinical dilemma is whether anticoagulation should be routinely offered to stroke or transient ischemic attack (TIA) patients with device-detected AF (DDAF). Current guidelines do not provide definitive recommendations for this population,7–9 underscoring the need for a comprehensive evaluation of potential benefits and risks. To address this uncertainty, we conducted a systematic review and meta-analysis to assess the efficacy and safety of anticoagulation in patients with a history of stroke or TIA and DDAF.
Methods
Standard protocol approvals, registrations, and patient consents
The pre-specified protocol of the present systematic review and meta-analysis has been registered in the International Prospective Register of Ongoing Systematic Reviews PROSPERO (registration ID: CRD42025646099). The meta-analysis is reported according to the updated Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 10 This study did not require an ethical board approval or written informed consent by the patients according to the study design (systematic review and meta-analysis).
Data sources, searches and study selection
A systematic literature search was conducted according to the patient, intervention, comparison, outcome (PICO) model 11 to identify available randomized-controlled clinical trials (RCTs) including adult patients with history of stroke or TIA with DDAF (P) receiving treatment with anticoagulation (I) versus no anticoagulation (C; defined as usual care, antiplatelets, or placebo) and investigating the outcomes of interest as outlined below (O). For the diagnosis of DDAF, use of insertable cardiac monitoring, pacemaker, implantable cardioverter defibrillator, or cardiac resynchronization therapy were all considered amenable for inclusion. The literature search was performed independently by four reviewers (LP, AHK, KM, MIS). The electronic databases MEDLINE and Scopus were searched, using search strings that included the terms “stroke,” “device-detected atrial fibrillation,” and “anticoagulants.” No language or other restrictions were applied. Our search spanned from inception of each database to February 2nd, 2025.
Observational studies, cohort studies, non-controlled studies, case series and case reports reporting on DDAF and anticoagulation with the aim of stroke prevention were excluded. Commentaries, editorials, and narrative reviews were also excluded. All retrieved studies were independently assessed by four reviewers (LP, AHK, KM, MIS) and any disagreements were resolved after discussion with a fifth tie-breaking evaluator (GT).
Quality control, bias assessment and data extraction
Eligible studies were subjected to quality control and bias assessment employing the Cochrane Collaboration tool (RoB 2) for RCTs. 12 Quality control and bias assessment was conducted independently by four reviewers (LP, AHK, AT, GP), and disagreements were settled by consensus after discussion with the corresponding author (GT).
Data extraction was performed in structured reports, including study name, country, recruitment period, intervention and comparison characteristics, included patients and their baseline characteristics.
Outcomes
The primary efficacy outcome of interest was any stroke recurrence at follow-up, defined as either ischemic stroke, hemorrhagic stroke, undefined stroke, or TIA. Secondary efficacy outcomes were (i) stroke recurrence or systemic embolism, (ii) ischemic stroke recurrence, (iii) myocardial infarction, and (iv) cardiovascular mortality. Cardiovascular mortality was defined as per each study definition. The primary safety outcome was major bleeding and the secondary safety outcomes were (i) hemorrhagic stroke and (ii) all-cause mortality at follow-up.
Statistical analysis
For the pairwise meta-analysis, we calculated for each dichotomous outcome of interest the corresponding risk ratios (RR) with 95% confidence interval (95% CI) for the comparison of outcome events among patients receiving anticoagulation versus no-anticoagulation. The number needed to treat (NNT) for the primary efficacy outcome was calculated using the formula:
Data availability statement
All data generated or analyzed during this study are included in this article and its Supplemental Information files.
Results
Literature search and included studies
The systematic database search yielded a total of 932 and 1109 records from the MEDLINE and SCOPUS databases, respectively (eFigure 1). After excluding duplicates and initial screening, we retrieved the full text of 10 records that were considered potentially eligible for inclusion. After reading the full-text articles, eight were further excluded (eTable 1). Finally, we identified two eligible studies17,18 for inclusion in the systematic review and meta-analysis (Table 1), comprising a total of 599 patients with a history of stroke or TIA and DDAF, receiving either anticoagulation (
Characteristics of studies included in the systematic review and meta-analysis.
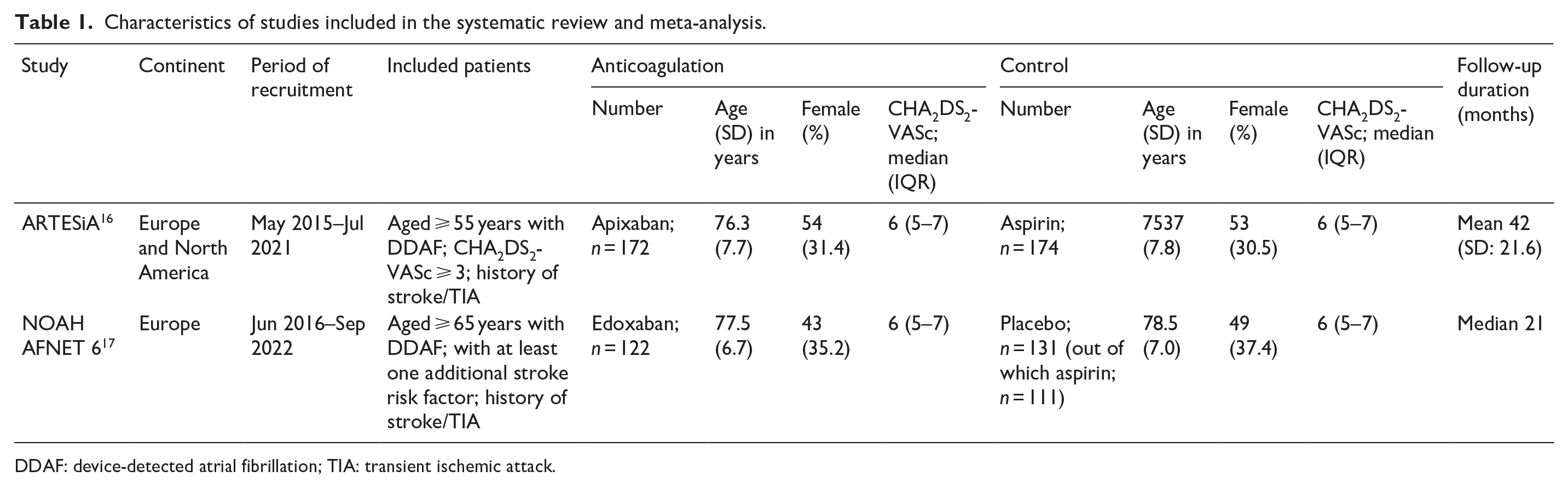
DDAF: device-detected atrial fibrillation; TIA: transient ischemic attack.
Quality control of included studies
The risk of bias in included studies was assessed by the Cochrane risk-of-bias (RoB 2) tool 10 and is presented in eFigure 6. Both studies presented low risk of bias with the exception of some concerns due to performance bias owning to treatment discontinuations which were balanced between the two groups.
Quantitative analyses
An overview of analyses for primary and secondary outcomes is summarized in Table 2.
Overview of analyses for the outcomes of interest.

NNT: number needed to treat; NNH: number needed to harm; NA: not applicable.
Patients receiving anticoagulants for DDAF post stroke/TIA had a reduced risk of any stroke recurrence compared to controls (RR: 0.47; 95% CI: 0.23–0.94;
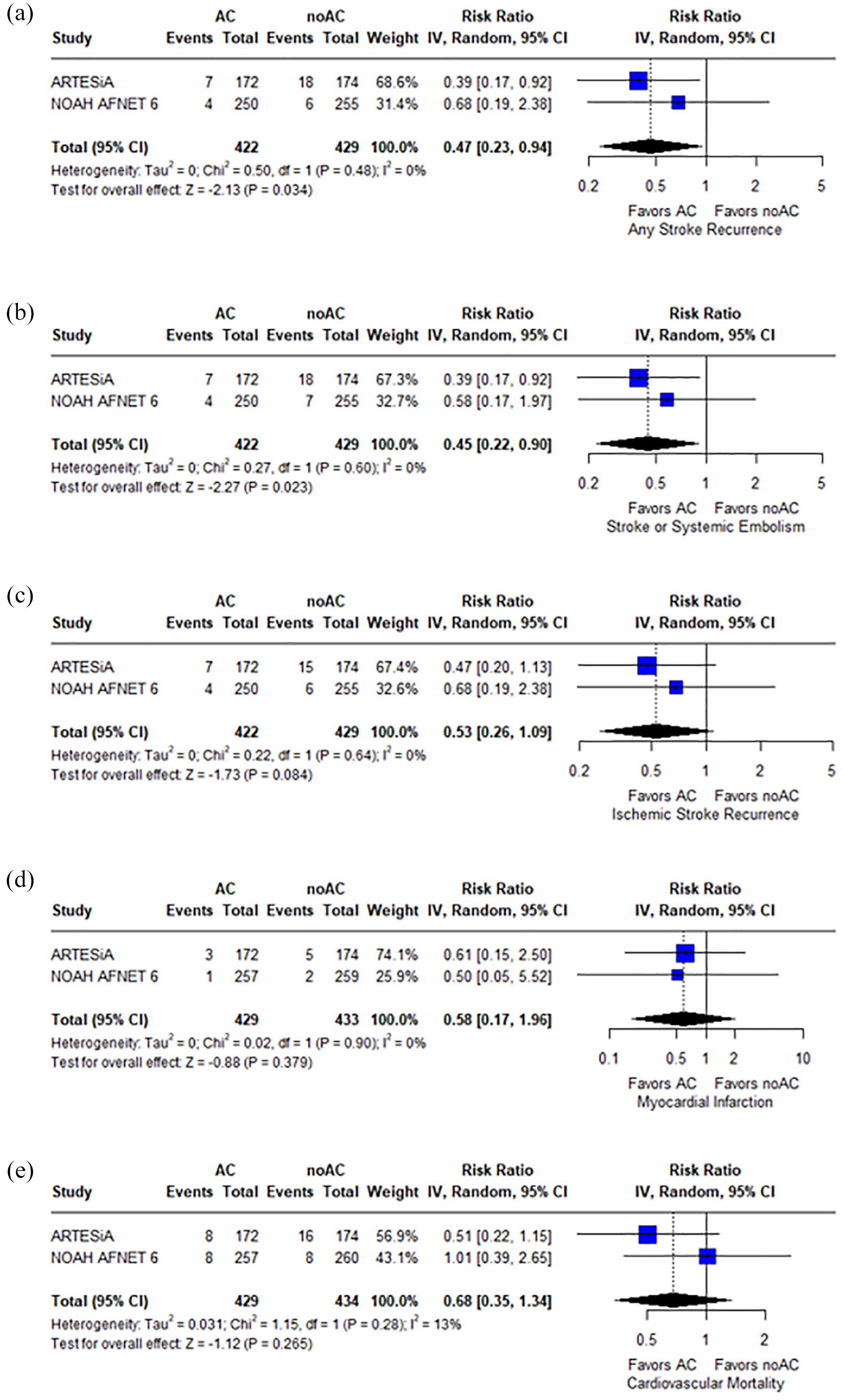
Forest plot presenting the risk ratio of any stroke recurrence (panel a), the composite of stroke recurrence and systemic embolism (panel b), ischemic stroke recurrence (panel c), myocardial infarction (panel d), and cardiovascular mortality (panel e) among patients treated with anticoagulation (AC) versus no-anticoagulation (noAC).
Regarding the secondary efficacy outcomes, anticoagulation was associated with lower rates of stroke recurrence or systemic embolism (RR: 0.45; 95% CI: 0.22–0.90;
Concerning safety outcomes, anticoagulation was associated with a higher risk of major bleeding (RR: 2.30; 95% CI: 1.06–4.98;
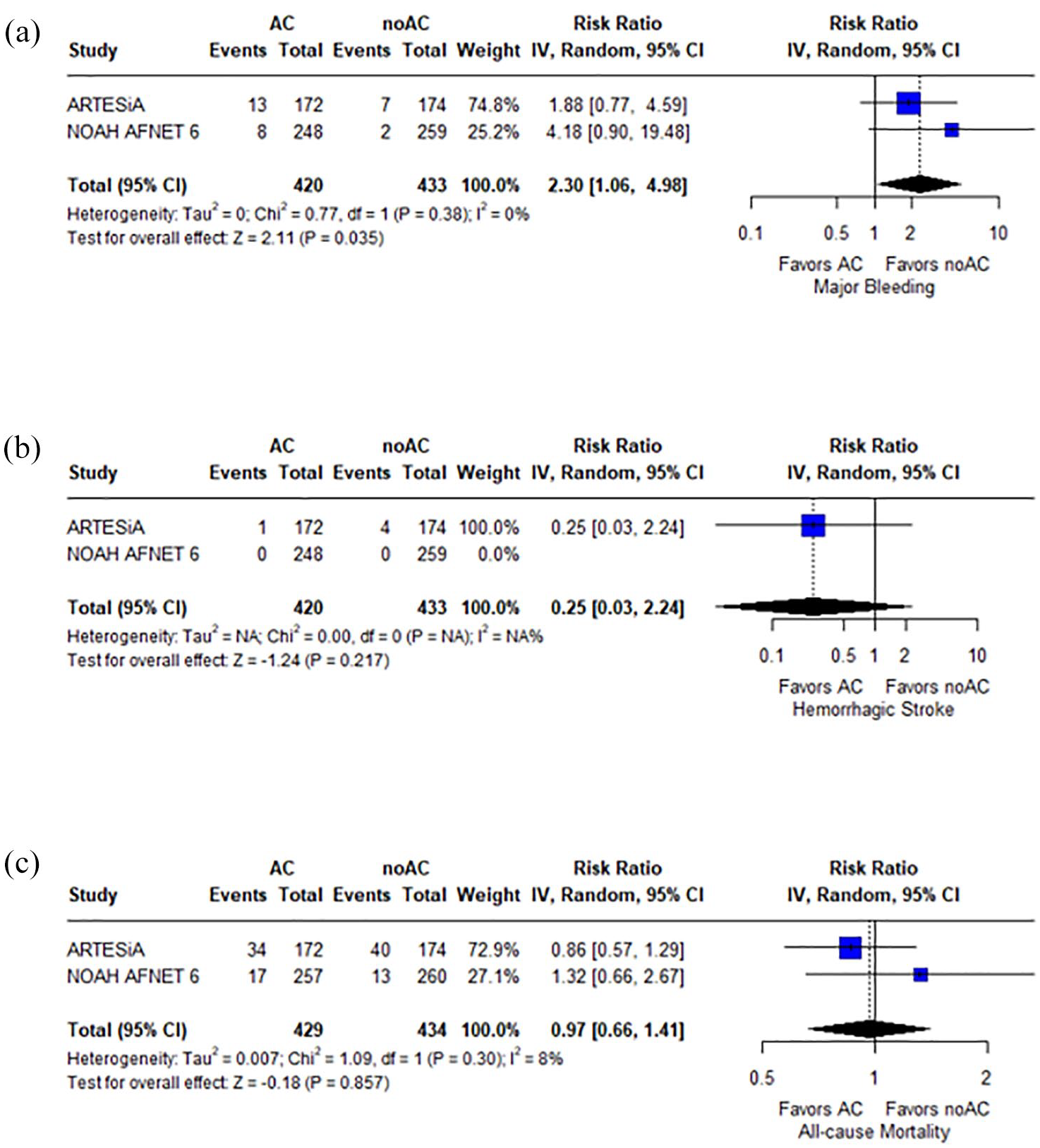
Forest plot presenting the risk ratio of major bleeding (panel a), hemorrhagic stroke (panel b), and all-cause mortality (panel c) among patients treated with anticoagulation (AC) versus no-anticoagulation (noAC).
The pooled proportions per arm for each outcome of interest are presented in eTable 2.
Discussion
The present meta-analysis demonstrates that anticoagulation in patients with a history of stroke or TIA and DDAF is associated with a significant reduction in any stroke recurrence, with a NNT of 34. Anticoagulation also reduced the composite outcome of stroke recurrence and systemic embolism with a NNT of 31. However, these benefits come at the cost of an increased risk of major bleeding (NNH = 37), highlighting the delicate balance between efficacy and safety in this population. Notably, there was no increase in hemorrhagic stroke with anticoagulation in this population.
Previous studies have suggested that AF detected after stroke is associated with a relatively low rate of stroke recurrence.4,19–21 Our meta-analysis supports this observation, as the pooled recurrence rates for stroke (4%; 95% CI: 1–11; eFigure 7), stroke & systemic embolism (4%; 95% CI: 1–11; eFigure 8) and ischemic stroke (4%; 95% CI: 1–9; eFigure 9) in patients with DDAF were low and similar to prior evidence. It should be noted that the follow-up duration was a mean of 42 months in ARTESiA and a median of 21 months in NOAH-AFNET 6.17,22 However, despite these low event rates, anticoagulation still led to a significant reduction in recurrent stroke, in recurrent stroke or systemic embolism as well as a trend toward lower ischemic stroke recurrence. Importantly, the rates of disabling or fatal stroke were also lower in the ARTESiA trial (3 in the anticoagulated vs 12 in the non-anticoagulated group) with a hazard ratio of 0.26 (95% CI: 0.07–0.93). 17
One significant consideration in interpreting these results is the uncertainty regarding the etiopathogenic mechanism of stroke in patients with DDAF. In the ARTESiA trial, 17 for example, eight patients with a history of hemorrhagic stroke were included and randomized, yet the optimal anticoagulation strategy for such patients remains unclear. Even in cases of clinical AF, the decision to anticoagulate after an intracerebral hemorrhage remains highly debated, 23 making it even more complex in the context of DDAF. It is possible that different results might have been observed if the analysis had exclusively included patients with truly cryptogenic ischemic stroke. Additionally, in both the ARTESiA and NOAH-AFNET 6 trials, DDAF was detected on average quite distant from the index stroke event, raising further questions about its causal role in stroke recurrence and the appropriateness of anticoagulation in this setting.
Regarding safety, while the included studies reported low overall rates of major bleeding (4%; 95% CI: 1–8; eFigure 10), the difference between anticoagulated and non-anticoagulated patients was statistically significant. Importantly, the majority of major bleeding events were gastrointestinal hemorrhages rather than intracerebral hemorrhages. Nonetheless, these findings do not support a blanket recommendation for anticoagulation in all patients with DDAF for secondary stroke prevention. Instead, treatment decisions should be guided by a more nuanced risk stratification approach, integrating key patient characteristics such as DDAF burden (duration and frequency), 24 prior bleeding history, stroke mechanism (cryptogenic vs other etiologies), and overall frailty.
It is important to note that the comparator arms in NOAH-AFNET 6 and ARTESiA differed in their design, which may have influenced the observed outcomes. NOAH-AFNET 6 was a double-blind study in which patients were randomized to receive edoxaban or placebo. However, aspirin use was permitted in the placebo group as part of standard care for patients with an independent indication, such as prior myocardial infarction, percutaneous coronary intervention, coronary artery bypass grafting, or secondary stroke prevention. 18 In contrast, ARTESiA employed a double-blind, double-dummy design in which patients were explicitly randomized to receive either apixaban or aspirin. 17 Unlike NOAH-AFNET 6, aspirin was the designated comparator in ARTESiA rather than an adjunct therapy. Although these differences in study design may have influenced the overall rates of stroke recurrence and bleeding events, aspirin use was highly prevalent in the control arm of NOAH-AFNET 6, with almost 85% of patients receiving this treatment. 18 This suggests that despite differences in trial methodology, the majority of patients in both studies’ non-anticoagulated arms received an antithrombotic agent, which should be considered when interpreting the results of our meta-analysis.
Another important consideration in interpreting our findings is the definition of cardiovascular mortality across the included trials. In ARTESiA, cardiovascular mortality encompassed deaths due to cardiac causes and other vascular causes, including stroke, pulmonary embolism, and ruptured aortic aneurysm or dissection. 25 Notably, all hemorrhagic deaths were classified as cardiovascular deaths in ARTESiA. 25 In contrast, NOAH-AFNET 6 defined cardiovascular mortality as death resulting from myocardial infarction, sudden cardiac death, heart failure, stroke, cardiovascular procedures, and cardiovascular hemorrhage specifically excluding non-cardiovascular hemorrhagic deaths. 22 Given these differences, fatal bleeding events were included under cardiovascular mortality in ARTESiA but only in cases of cardiovascular hemorrhage in NOAH-AFNET 6. This discrepancy in classification may have influenced the observed rates of cardiovascular mortality in our meta-analysis and should be considered when interpreting the overall balance between thrombotic and hemorrhagic risks associated with anticoagulation in patients with DDAF.
Treatment adherence and crossover between study arms are important factors that may have influenced the findings of the included trials. In ARTESiA, permanent discontinuation of the assigned treatment occurred in 24.3% of patients in the apixaban group and 23.8% in the aspirin group, primarily due to the development of subclinical AF lasting more than 24 h or clinical AF. 17 Similarly, in NOAH-AFNET 6, 21 patients in the edoxaban arm and 24 in the placebo arm developed AF during follow-up, which may have led to changes in treatment allocation. 18 Additionally, 9 patients (7.4%) in the edoxaban group and 12 patients (9.2%) in the placebo group withdrew consent, potentially resulting in treatment discontinuation or loss to follow-up. 18 These treatment modifications underscore the challenges of long-term anticoagulation management in patients with DDAF and may have influenced the observed event rates in the original trials and, consequently, our meta-analysis. Future studies should further examine the impact of treatment persistence and crossover on anticoagulation outcomes in this population.
Given the challenges in individualizing anticoagulation therapy for patients with DDAF, future research should focus on refining risk stratification models that incorporate clinical, imaging, and rhythm-monitoring parameters to better predict the net benefit of anticoagulation. Specifically, upcoming trials could evaluate: (i) the role of AF burden (e.g. daily duration, frequency of episodes) in modulating stroke risk and whether a threshold exists for initiating anticoagulation; (ii) biomarker-based approaches, such as elevated NT-proBNP or mid-regional proANP, to enhance thromboembolic risk prediction; (iii) patient-centered strategies integrating shared decision-making and frailty assessments to guide treatment in elderly or multimorbid patients; and (iv) alternative anticoagulation strategies to reduce bleeding risk while preserving stroke prevention benefits.
The present meta-analysis synthesizes data from the NOAH-AFNET 6 and ARTESiA trials, providing a comprehensive assessment of anticoagulation’s efficacy and safety in stroke/TIA patients with DDAF. While previous publications have examined stroke subgroups within each trial,17,18 our study enhances these findings by systematically pooling data to improve precision and generalizability. By integrating results across trials, we offer a more robust estimation of treatment effects, particularly for outcomes such as stroke recurrence, systemic embolism, and major bleeding. Moreover, our focus on patients with a history of stroke or TIA – who face a particularly high risk of recurrent cerebrovascular events – adds critical clinical relevance. Unlike the meta-analysis by McIntyre et al., 26 which included all DDAF patients regardless of prior stroke/TIA, our study specifically addresses the secondary prevention population. Given the ongoing debate surrounding anticoagulation in this group, our findings provide a unified estimate that may help refine clinical decision-making.
Certain shortcomings of this systematic review and meta-analysis should be acknowledged. First, this study is a study-level meta-analysis, and its conclusions are inherently limited by the available data. Second, only two RCTs were included, yet both presented prespecified secondary analyses and were of high quality. However, both RCTs were terminated early,22,25 limiting statistical power. Additionally, while our study provides valuable insights into anticoagulation for secondary stroke prevention in patients with DDAF, further research is needed to determine whether specific high-risk subgroups – such as elderly patients, those with multimorbidity, or individuals with high CHA2DS2-VASc scores – derive a greater net benefit from anticoagulation while minimizing bleeding risk. Future studies should aim to refine risk stratification strategies to better balance the risks and benefits of treatment in these vulnerable populations.
Conclusion
In conclusion, the current meta-analysis showed that anticoagulation following DDAF in patients with history of stroke or TIA is associated with a reduced risk of any stroke recurrence and the composite of stroke recurrence and systemic embolism, but part of this benefit is offset by an increased risk of major bleeding without a higher risk of hemorrhagic stroke. Further randomized data are needed to refine anticoagulation strategies and individualize treatment decisions in DDAF patients with prior stroke/TIA.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251334278 – Supplemental material for Recurrent stroke in patients with history of stroke/transient ischemic attack and device-detected atrial fibrillation: A systematic review and meta-analysis
Supplemental material, sj-docx-1-eso-10.1177_23969873251334278 for Recurrent stroke in patients with history of stroke/transient ischemic attack and device-detected atrial fibrillation: A systematic review and meta-analysis by Lina Palaiodimou, Aristeidis H Katsanos, Konstantinos Melanis, Maria-Ioanna Stefanou, Michele Romoli, Georgia Papagiannopoulou, Aikaterini Theodorou, Martin Köhrmann, Polychronis Dilaveris, Konstantinos Tsioufis, Gkikas Magiorkinis, Christos Krogias, Marios Themistocleous, Simona Sacco, Mira Katan, Gerasimos Filippatos and Georgios Tsivgoulis in European Stroke Journal
Footnotes
Acknowledgements
Dr. Katsanos is supported by the Heart and Stroke Foundation Canada and the Canadian Institutes of Health Research.
Correction (December 2025):
Article updated to change the Article Type from Original Research Article to Review
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Informed consent
Not applicable.
Trial registration
The pre-specified protocol of the present systematic review and meta-analysis has been registered in the International Prospective Register of Ongoing Systematic Reviews PROSPERO (registration ID: CRD42025646099).
Guarantor
GT
Contributorship
Conceptualization: LP, and GT; Data curation: LP, AHK, KM, MIS, MR, GP, AT, MKo, PD, KT, GM, CK, MT, SS, MKa, GF, and GT; Formal analysis: LP, and AHK; Investigation: LP, AHK, KM, MIS, GP, AK, and GT; Methodology: LP, AHK, KM, MIS, GP, AT, and GT; Project administration: GT; Supervision: GT; Visualization: LP; Writing – original draft: LP, and GT; Writing – review & editing: AHK, KM, MIS, MR, GP, AT, MKo, PD, KT, GM, CK, MT, SS, MKa, and GF.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
