Abstract
Introduction:
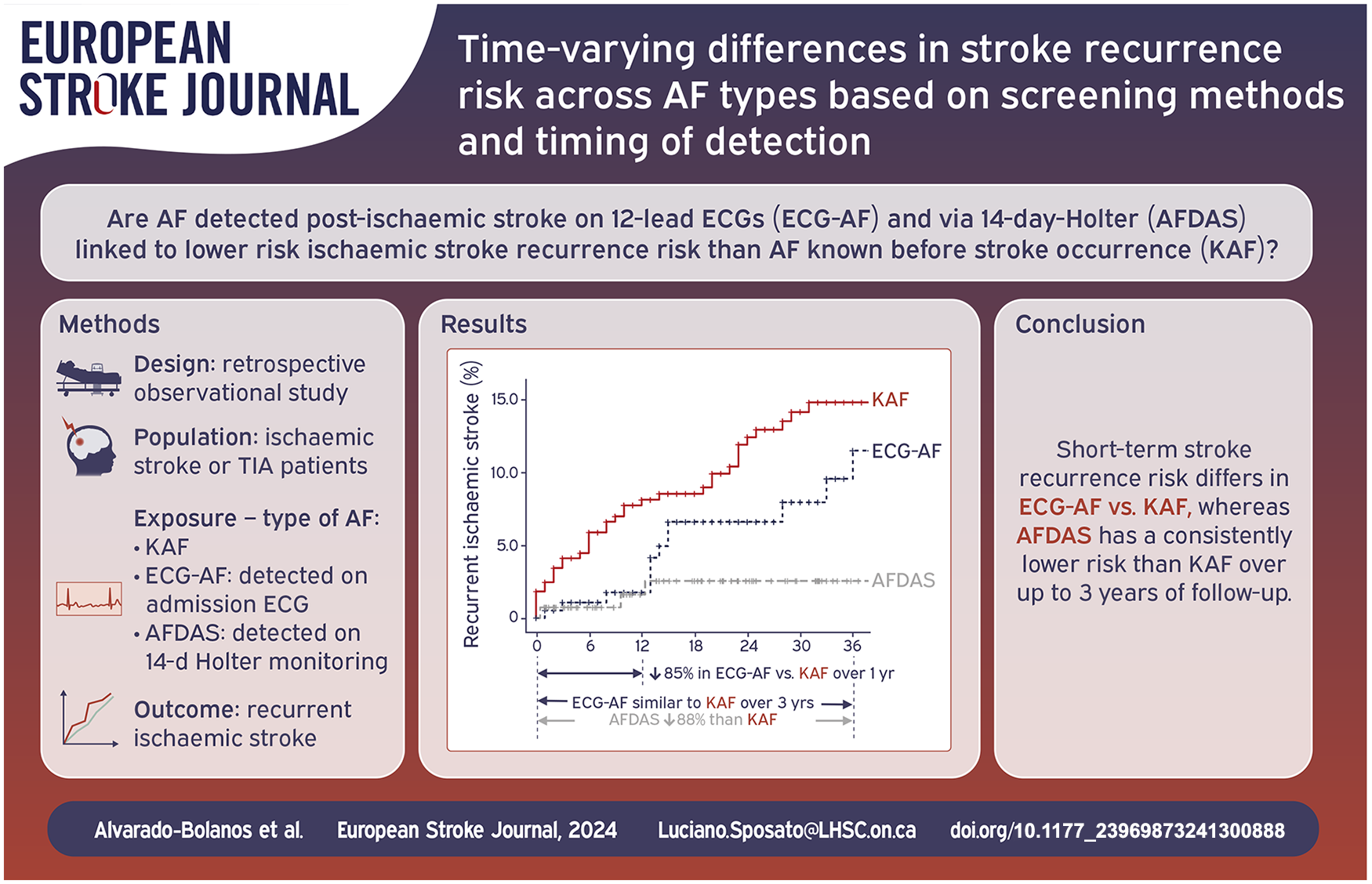
Atrial fibrillation (AF) burden progresses with time. Among ischemic stroke (IS) patients, AF can be detected at different burden progression stages based on the timing and screening method. We hypothesized that AF detected after IS on 12-lead ECGs (ECG-AF) and via 14-day-Holter prolonged cardiac monitoring (AFDAS) are linked to lower IS recurrence risk than AF known before stroke occurrence (KAF) because of being at an earlier progression stage than KAF. Additionally, we posited that IS recurrence risk differences between AF types vary over time due to their differential progression stages.
Patients and methods:
Retrospective observational cohort study including IS/TIA patients with KAF, ECG-AF, and AFDAS [2018–2021]. Adjusted hazard ratios (aHR) were estimated using multivariable cause-specific Cox proportional-hazard models to compare IS recurrence between ECG-AF versus KAF and AFDAS versus KAF. Proportional hazards assumptions were tested to assess whether IS recurrence risk differences were time-varying.
Results:
Of 758 AF patients (385 KAF, 236 ECG-AF, 137 AFDAS), 603 received anticoagulation and 59 experienced a recurrent IS after 1441 patient-years of follow-up. No IS recurrence risk differences were observed at the end of follow-up between ECG-AF and KAF (aHR 0.67, 95% CI 0.36–1.26), although ECG-AF showed lower risk only within the first year (aHR 0.15; 95% CI 0.04–0.56). AFDAS exhibited a lower IS recurrence risk than KAF (aHR 0.22, 95% CI 0.08–0.63), without time-varying differences.
Discussion:
Differences in IS recurrence risk between ECG-AF and KAF varied over time. However, AFDAS showed a consistently lower IS risk than KAF throughout the entire study period.
Introduction
Atrial fibrillation (AF) is currently understood as a disease continuum that progresses from an early “at risk” phase determined by the presence of modifiable and nonmodifiable risk factors to a “permanent” stage in which it becomes irreversible. 1 At earlier stages, AF burden is modest and its detection is thus more elusive unless prolonged cardiac monitoring (PCM) is pursued for weeks to years. In later AF stages, left atrial substrate is more severe, AF burden is greater, and patients become symptomatic, making AF detection more likely, even with a 12-lead ECG. AF identified on 12-lead ECGs is considered high-risk as it has been associated with a fivefold higher risk of stroke than the absence of AF. 2 However, ischemic strokes can occur in patients at a wide range AF stages, going from asymptomatic, paroxysmal, early-onset to symptomatic, persistent or permanent. 3 Furthermore, the intensity of cardiac monitoring and timing of AF diagnosis (before vs after stroke onset) determines the stage at which AF is detected in stroke patients.4,5 AF in stroke patients has been classified as known before stroke occurrence (KAF), newly detected on 12-lead ECG (ECG-AF), or AF detected after stroke on PCM (AFDAS).3,4 No studies have yet compared the risks of ischemic stroke recurrence of ECG-AF and AFDAS relative to KAF, nor have they examined how these risks change over time after stroke onset. The latter is a relevant question, given the progressive nature of AF burden. We therefore conducted a retrospective observational study to determine if the risk of ischemic stroke recurrence in patients with KAF is different from that of ECG-AF, and AFDAS. We hypothesized that KAF would show the highest stroke recurrence risk, with ECG-AF yielding an intermediate hazard, and AFDAS displaying the lowest stroke recurrence rates. 4 We further hypothesized that the risk of stroke recurrence in patients with ECG-AF and AFDAS relative to KAF would vary over time, with differences between groups being larger during the first months and progressively attenuating toward the end of the follow-up.
Methods
Study design and patient selection
Data was derived from the London Ontario Stroke Registry, which includes patients from London Health Sciences Centre (LHSC) with ischemic stroke or transient ischemic attack (TIA) assessed as inpatients and at the TIA and stroke prevention clinic. LHSC serves a catchment area of 2 million population and functions as the primary referral stroke centre for acute stroke therapy for 13 hospitals in the region of Southwestern Ontario, Canada. 6 Patients are assessed by certified stroke neurologists and data are collected consecutively and prospectively. All patients in the registry have at least a 12-lead ECG as part of their basic workup. We included patients with ischemic stroke or TIA with KAF, ECG-AF, and AFDAS. We defined KAF as AF known before stroke or TIA occurrence. ECG-AF was defined as newly diagnosed AF on a 12-lead ECG during the initial assessment or stroke admission. AFDAS was defined as AF lasting ⩾30 s on 14-day Holter monitoring. PCM was conducted with a 14-day single-lead CardioSTAT™ Holter monitor (Icentia Inc, Quebec, Canada), which has been previously validated for arrhythmia detection in adult patients. 7 We excluded patients with AF associated with recent cardiac interventions (e.g. cardiac surgery or patent foramen ovale closure).
Data collection and outcomes
All outcomes were ascertained by a stroke neurologist or a nurse practitioner at the last post-stroke follow-up visit. The primary outcome was recurrent ischemic stroke, defined as a new focal neurological deficit not attributable to a nonischemic cause with imaging evidence of a new brain infarction. 8 As part of the prespecified hypothesis, recurrent ischemic stroke risk was compared in patients with KAF versus ECG-AF, and KAF versus AFDAS. The secondary outcome was major adverse cardiovascular events (MACE), defined as the composite of acute coronary syndrome, decompensated chronic heart failure exacerbation requiring admission, recurrent ischemic stroke, or cardiovascular death. Tertiary outcomes were major bleeding as per the International Society on Thrombosis and Hemostasis (ISTH) criteria, 9 intracranial bleeding, and all-cause death. All outcomes were ascertained until the end of the available follow-up. Clinical and demographic data included the type of cerebrovascular event (ischemic stroke or TIA), age, sex, comorbidities, echocardiographic findings (left ventricular ejection fraction and left atrial volume index), and high-sensitivity troponin T. AF burden on PCM was quantified as the total duration of all detected AF paroxysms and the proportion of total time on AF relative to the net duration of monitoring. AF burden was also stratified into 4 categories: <2 min, 2–6 min, 6 min to 24 h, and >24 h. 10 As an exploratory analysis, we evaluated the clinical characteristics and AF burden of patients with AFDAS experiencing recurrent ischemic strokes and intracranial hemorrhages.
Statistical analysis
Continuous variables were reported as median and interquartile range (IQR). Discrete variables were described as total numbers and proportions. Baseline characteristics between groups were compared using standardized mean differences (SMD). Crude incidences and Kaplan-Meier curves for the primary and secondary outcomes were calculated. Cause-specific Cox-proportional hazard models accounting for the competing risk of death were applied to estimate adjusted hazard ratios (aHRs) to compare the association of ECG-AF versus KAF and PCM-AF versus KAF for all clinical endpoints. Models were adjusted by including variables yielding SMD > 0.2 on bivariate analysis and those judged a priori to impact outcomes regardless of the SMD. Schoenfeld residual plots and Pearson correlation of ranked times and residuals were generated to assess if the ratio of the hazards remained constant over time. If the proportional hazards assumption was violated, specific aHR for different periods were estimated. Variables with ⩽20% of missing data were resolved by using multiple imputations. Multicollinearity was assessed using variation inflation factors. Based on prior evidence suggesting that anticoagulation at the time of the qualifying cerebrovascular event is associated with ischemic stroke recurrence risk, we performed a post-hoc analysis by including anticoagulation at baseline in both models.
Results
Cohort characteristics
This study included 758 patients, of whom 385 had KAF, 236 ECG-AF and 137 AFDAS followed for 1441 patient-years. Overall, 584 (77.0%) patients experienced an ischemic stroke as the qualifying event, and 603 (79.6%) were anticoagulated a median follow-up time of 23.4 (IQR 4.8–38.9) months. No patient received a percutaneous left atrial appendage occlusion procedure. There were no significant differences in the rate of anticoagulation between groups (Table 1). A total of 59 (7.8%) patients experienced a recurrent ischemic stroke at the end of follow-up, and 50 (84.7%) were receiving anticoagulants at the time of the recurrence. There were no variables with >20% of missing data.
Baseline characteristics.
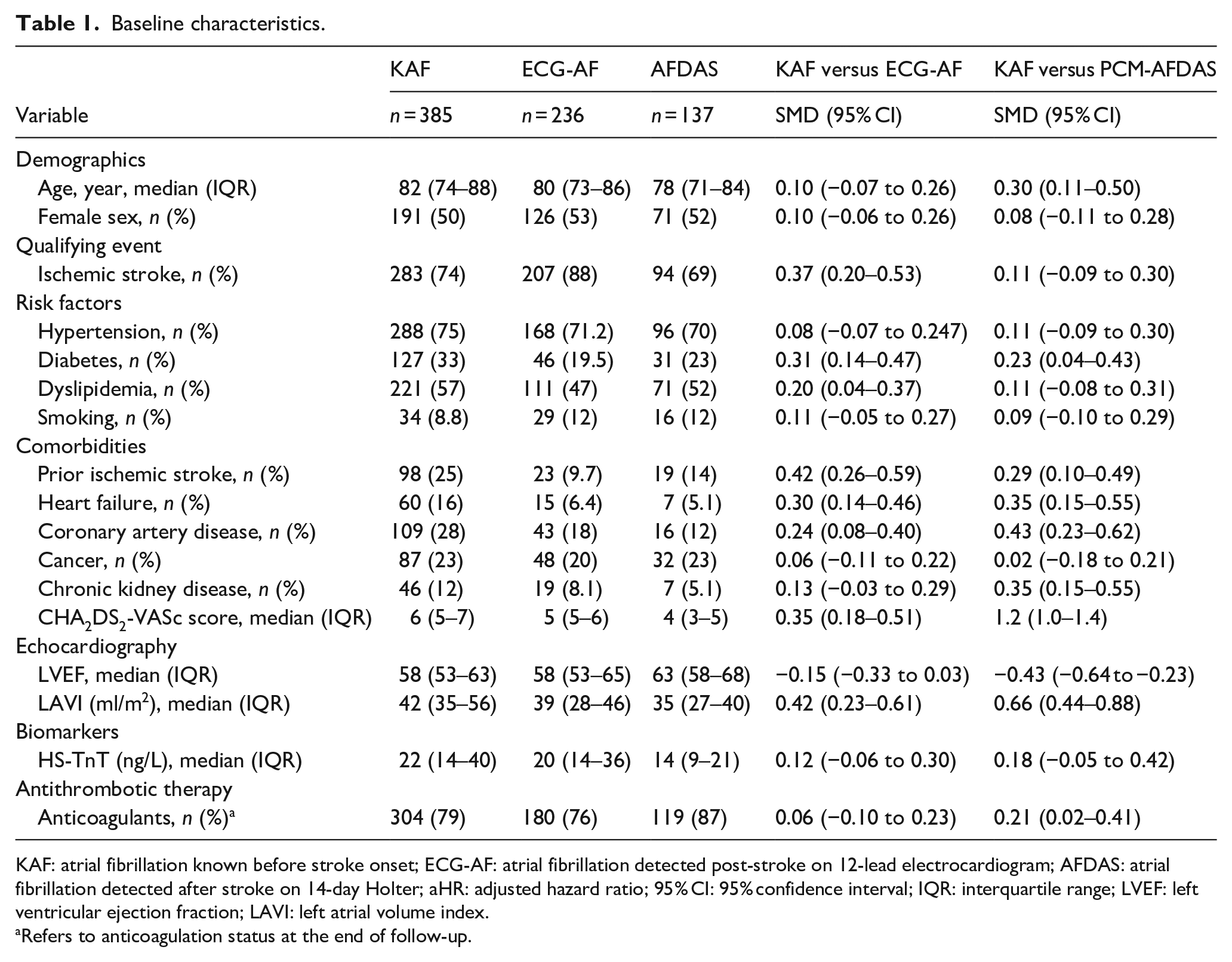
KAF: atrial fibrillation known before stroke onset; ECG-AF: atrial fibrillation detected post-stroke on 12-lead electrocardiogram; AFDAS: atrial fibrillation detected after stroke on 14-day Holter; aHR: adjusted hazard ratio; 95% CI: 95% confidence interval; IQR: interquartile range; LVEF: left ventricular ejection fraction; LAVI: left atrial volume index.
Refers to anticoagulation status at the end of follow-up.
AFDAS burden
The median AF duration in patients with AFDAS was 5.25 (IQR 0.35–33) h. The proportion of AFDAS patients distributed across categories of AF burden <2 min, 2–6 min, 6 min to 24 h, and >24 h were 14.6%, 5.1%, 51.1%, and 29.2%, respectively. Median AF duration relative to the length of PCM (AF duration/total PCM time) was 2.23% (IQR 0.14–12.25). The median time from the qualifying ischemic stroke or TIA to AF detection was 36.6 (24.5–94.5) days.
ECG-AF versus KAF
Patients with ECG-AF more often had an ischemic stroke as a qualifying event, had lower median CHA2DS2-VASc scores, and a lower prevalence of diabetes, dyslipidemia, chronic heart failure, coronary artery disease, and prior stroke than those with KAF. Likewise, patients with ECG-AF had a smaller left atrium (Table 1).
Based on the Schoenfeld residuals and Pearson correlation analysis, differences in ischemic stroke recurrence risk between ECG-AF and KAF violated the proportional hazards assumption. As such, they were deemed time-varying, with an 85% lower risk for ECG-AF relative to KAF within the first 12 months of follow-up (aHR 0.15; 95% CI 0.04–0.56; p = 0.005), but no significant differences thereafter (Figure 1).
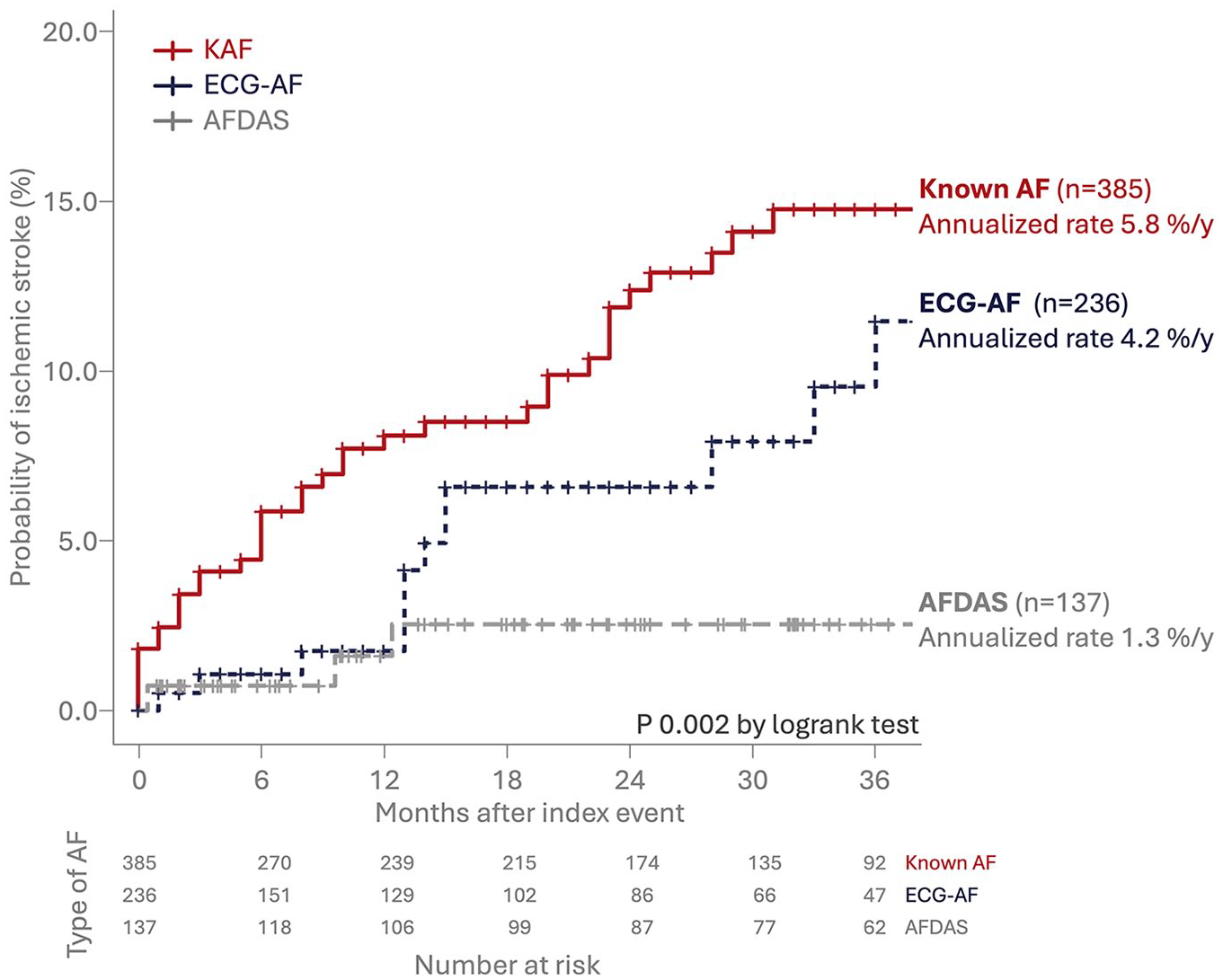
Kaplan Meier curves for recurrent ischemic stroke stratified by AF type.
At the end of the follow-up period, there were no differences in the risk of ischemic stroke recurrence (aHR 0.67, 95% CI 0.36–1.26; p = 0.22) between KAF (n = 38/385; annualized incidence rate of 5.8% per patient-year) and ECG-AF (n = 16/236; annualized incidence rate of 4.2% per patient-year; Figure 2) or any other secondary or safety outcome (Table 2).
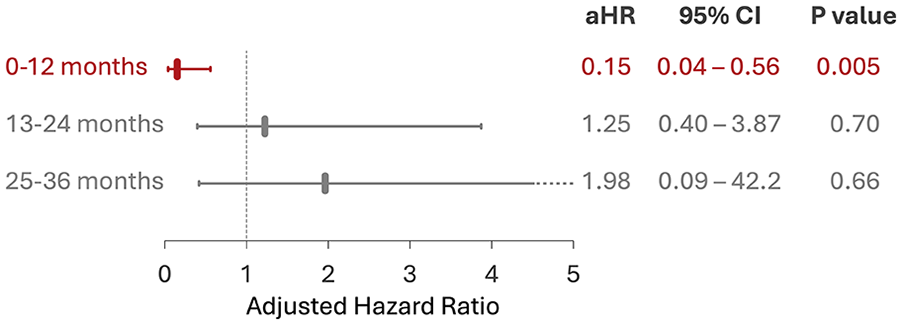
Time-varying risk of recurrent ischemic stroke in ECG-AF versus KAF.
Adjusted hazard ratios for study outcomes.
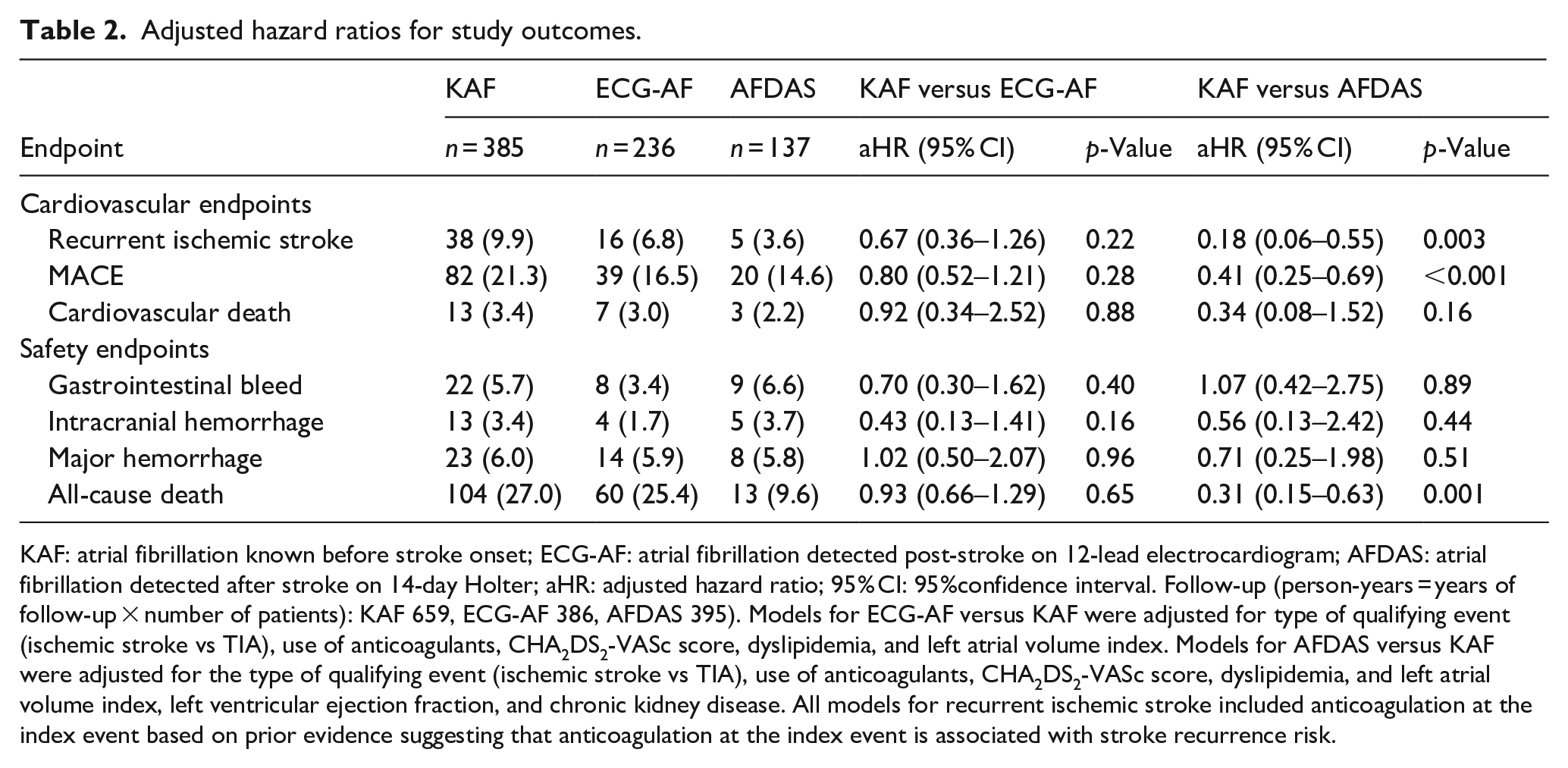
KAF: atrial fibrillation known before stroke onset; ECG-AF: atrial fibrillation detected post-stroke on 12-lead electrocardiogram; AFDAS: atrial fibrillation detected after stroke on 14-day Holter; aHR: adjusted hazard ratio; 95% CI: 95%confidence interval. Follow-up (person-years = years of follow-up × number of patients): KAF 659, ECG-AF 386, AFDAS 395). Models for ECG-AF versus KAF were adjusted for type of qualifying event (ischemic stroke vs TIA), use of anticoagulants, CHA2DS2-VASc score, dyslipidemia, and left atrial volume index. Models for AFDAS versus KAF were adjusted for the type of qualifying event (ischemic stroke vs TIA), use of anticoagulants, CHA2DS2-VASc score, dyslipidemia, and left atrial volume index, left ventricular ejection fraction, and chronic kidney disease. All models for recurrent ischemic stroke included anticoagulation at the index event based on prior evidence suggesting that anticoagulation at the index event is associated with stroke recurrence risk.
In a post-hoc analysis, adding anticoagulation at the time of the qualifying cerebrovascular event to the regression model did not modify the association between ECG-AF and ischemic stroke recurrence risk relative to KAF at 12 months (0.20; 95% CI 0.05–0.86; p = 0.033) or at the end of follow-up (aHR 0.82; 95% CI 0.38–1.75; p = 0.60). In this model, anticoagulation at baseline was not associated with ischemic stroke recurrence risk at 12 months (aHR 1.75; 95% CI 0.57–5.41; p = 0.31) or at the end of follow-up (aHR 1.41; 95% CI 0.70–2.85; p = 0.33).
There were no differences in any of the secondary or tertiary outcomes between KAF and ECG-AF or KAF and AFDAS (Table 2).
AFDAS versus KAF
Participants with AFDAS were younger, 11 had a lower prevalence of diabetes, chronic heart failure, coronary artery disease, chronic kidney disease, prior ischemic stroke, a lower median CHA2DS2-VASc score, a smaller left atrial volume index, and a higher left ventricular ejection fraction than those with KAF (Table 1).
At the end of the study follow-up, the risk of ischemic stroke recurrence was lower in patients with AFDAS (n = 5/137; annualized incidence rate of 1.3% per patient-year) than in those with KAF (aHR 0.22, 95% CI 0.08–0.63; p = 0.003; Table 2 and Figure 1). Differences in stroke recurrence risk between AFDAS and KAF did not violate the proportional hazards assumption.
In a post-hoc analysis, adding anticoagulation at the time of the qualifying cerebrovascular event to the regression model did not modify the association between AFDAS and ischemic stroke recurrence risk relative to KAF at the end of follow-up (aHR 0.23; 95% CI 0.07–0.79; p = 0.020). In this model, anticoagulation at baseline was not associated with ischemic stroke recurrence risk at the end of follow-up (aHR 1.52; 95% CI 0.70–3.32; p = 0.28).
The risks of MACE (aHR 0.41, 95% CI 0.25–0.69; p < 0.001) and all-cause death (aHR 0.31, 95% CI 0.15–0.63; p = 0.001) were lower in the AFDAS group than in those with KAF.
Recurrent ischemic stroke and intracranial hemorrhages in patients with AFDAs
Patients with AFDAS experienced an equal number of ischemic stroke recurrences (n = 5) and intracranial hemorrhages (n = 5; Tables 2 and 3). Three of five patients with recurrent ischemic stroke were receiving anticoagulants at the time of the recurrence (Table 3). High-burden AFDAS was considered a possible cause of recurrent ischemic stroke in only one of three patients receiving anticoagulants. This patient had evidence of severely impaired left atrial appendage flow on a cardiac computed tomography. All intracranial hemorrhages occurred in patients receiving oral anticoagulants and two were fatal. Four of them occurred in patients with low-burden AFDAS (<2.5 h) and AF never recurred clinically or in subsequent ECGs in three of these patients.
Characteristics of recurrent ischemic strokes and intracranial hemorrhages in patients with AFDAS.
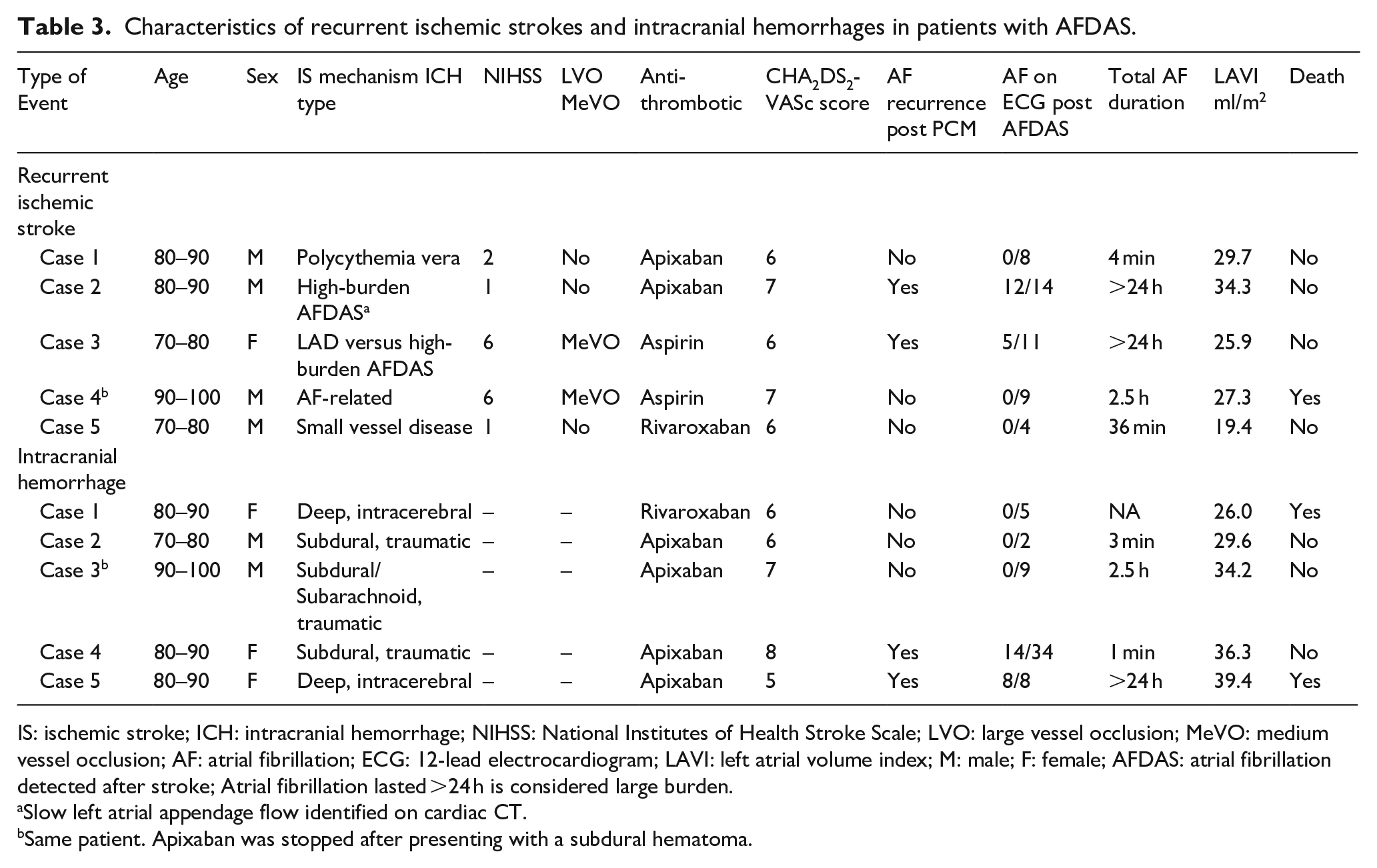
IS: ischemic stroke; ICH: intracranial hemorrhage; NIHSS: National Institutes of Health Stroke Scale; LVO: large vessel occlusion; MeVO: medium vessel occlusion; AF: atrial fibrillation; ECG: 12-lead electrocardiogram; LAVI: left atrial volume index; M: male; F: female; AFDAS: atrial fibrillation detected after stroke; Atrial fibrillation lasted >24 h is considered large burden.
Slow left atrial appendage flow identified on cardiac CT.
Same patient. Apixaban was stopped after presenting with a subdural hematoma.
Discussion
This observational cohort study, including 758 ischemic stroke and TIA patients with three different types of AF classified by the timing of diagnosis and screening method, revealed several novel findings. First, there were no differences in stroke recurrence at the end of the follow-up period between KAF and ECG-AF. However, differences in stroke risk between both types of AF were time-varying and patients with KAF had a 6.7-fold higher relative risk of ischemic stroke recurrence within the first 12 months of follow-up compared to ECG-AF (HR 0.15 for ECG-AF compared to KAF). Second, stroke recurrence risk in patients with AFDAS was substantially and consistently lower than KAF throughout the entire duration of follow-up, and each of the three prespecified study periods, supporting the idea that AFDAS is at an earlier-stage AF than KAF. Third, patients with AFDAS experienced an equal number of ischemic stroke recurrences and anticoagulant-associated intracranial hemorrhages.
Time-varying differences in stroke recurrence risk between KAF and ECG-AF have potential explanations. ECG-AF may represent a slightly earlier stage AF than KAF, requiring approximately a year to match the embolic risk of the latter. Supporting this concept, patients with ECG-AF had a lower prevalence of risk factors and CHA2DS2-VASc score, and less severe left atrial substrate changes. Clinically, the rapid ECG-AF matching KAF’s stroke risk suggests that ECG-AF and KAF should be grouped under the KAF category despite ECG-AF being diagnosed post-stroke. We hypothesize that ECG-AF is KAF that remained undiagnosed because of being less symptomatic or occurring in patients with more limited access to the health care system, or less prone to seek medical attention for non-severe symptoms. These hypotheses should be tested in future studies.
AFDAS showed a consistently lower stroke recurrence risk than KAF throughout the entire follow-up period, without significant differences at each of the three prespecified study periods. This finding, in association with our previous report showing that AFDAS has a lower risk of stroke recurrence than ECG-AF, 12 supports the notion that AFDAS is a relatively lower-risk AF. The lack of variation of difference in stroke recurrence between AFDAS and KAF across study periods suggests that AFDAS constitutes an earlier-stage AF with an overall low burden (median duration of 5.25 h, 2.23% of total recording time, and 70.8% lasting <24 h) or perhaps, that it is less likely to progress to a higher burden AF within the first few years post-stroke. A longer follow-up time would be needed to determine if AFDAS is also a progressive arrhythmia, with worsening of left atrial substrate and AF burden, ultimately resulting in a similar embolic risk as ECG-AF and KAF. The Evolution of Burden of AF, Biomarkers, Left Atrial Characteristics, Demographics, and Risk Factors in AF Detected After Stroke (B 2 AD-RISK AFDAS) pilot prospective observational study will evaluate this hypothesis (NCT06589700).
The risk of bleeding events was not significantly different between groups. However, it is worth noting that patients with AFDAS experienced an equal number of recurrent ischemic strokes and intracranial hemorrhages. A detailed analysis of these 10 events in the context of the overall AF burden and clinical characteristics of patients with AFDAS suggests that this group entails a widely heterogeneous embolic risk (Table 3). Importantly, all ischemic stroke recurrences occurred in patients with high-risk AFDAS or competing mechanisms (Table 3). Conversely, a proportion of intracranial hemorrhages occurred in patients with low-risk AFDAS, (low AF burden, no left atrial substrate, and no AF recurrence during follow-up), in whom anticoagulation may have not been entirely warranted and resulted in considerable harm (Table 3). These findings are concerning and should be further investigated in other cohorts.
Of note, 155 (20.4%) patients in this cohort were not on anticoagulants at a median of nearly 2 years of follow-up. This adherence rate is higher than that reported in a meta-analysis of 48 studies of direct oral anticoagulation (approximately 72%–81% at 12 months). 13 Reasons for not using anticoagulants in all types of AF are shown in the Supplemental File.
Our study has limitations. First, given that this is an observational study and we did not actively follow patients to ascertain their outcomes, we may have missed some events if patients were not assessed within the Southwestern Ontario healthcare system. However, this is unlikely given that our centre is the regional stroke centre for 13 hospitals with linked electronic medical records. 6 Second, the analysis of recurrent ischemic stroke and hemorrhagic events is limited by their low number and should therefore be considered only exploratory and hypothesis-generating. Third, the results of this study cannot be generalized to patients with cryptogenic ischemic stroke, as this study cohort included individuals with all ischemic stroke subtypes.
In summary, this study suggests that both KAF and ECG-AF constitute high-risk AFs, with differences between their embolic risk being time-varying in the short term. These findings support the concept that KAF and ECG-AF should be grouped under a single category of risk. AFDAS comprises a heterogeneous group of patients with an overall relatively lower risk of stroke recurrence than KAF. The difference in risk profiles between both types of AF does not change with time, at least in the first years post-stroke, suggesting that some AFDAS is detected at a very early stage, in which its embolic risk may be lower.
Supplemental Material
sj-docx-1-eso-10.1177_23969873241300888 – Supplemental material for Time-varying differences in stroke recurrence risk between types of atrial fibrillation based on screening methods and timing of detection
Supplemental material, sj-docx-1-eso-10.1177_23969873241300888 for Time-varying differences in stroke recurrence risk between types of atrial fibrillation based on screening methods and timing of detection by Alonso Alvarado-Bolanos, Diana Ayan, Facundo Lodol, Alexander V. Khaw, Lauren M. Mai, Jennifer L. Mandzia, Marko Mrkobrada, Maria Bres-Bullrich, Lorraine Fleming, Corbin Lippert, Meredith Cecile, Rodrigo Bagur, Sebastian Fridman and Luciano A. Sposato in European Stroke Journal
Footnotes
Acknowledgements
We thank the patients involved in this study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LAS: Speaker/consulting honoraria from Boehringer Ingelheim, Pfizer, Bayer, and Gore; member of the Editorial Board of Neurology, Stroke, and JAHA; Editor, Neurocardiology section of Stroke. Associate Editor, JAHA. AAB: no relevant conflicts to disclose. DA: no relevant conflicts to disclose. FL: no relevant conflicts to disclose. AVK: no relevant conflicts to disclose. LM: no relevant conflicts to disclose. JM: no relevant conflicts to disclose. MM: no relevant conflicts to disclose. MBB: no relevant conflicts to disclose. LF: no relevant conflicts to disclose. CL: no relevant conflicts to disclose. MC: no relevant conflicts to disclose. RB: no relevant conflicts to disclose. SF: no relevant conflicts to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: LAS was supported by the Kathleen and Dr. Henry Barnett Chair in Stroke Research.
Ethical approval
This study has been approved by Western University Ethics Review Board (2023-122969-78477).
Informed consent
To guarantee that all cases are included consecutively and given that all cases are gathered retrospectively, the Western University Health Sciences Research Ethics Board, Lawson Health Research Institute, and the Privacy office at London Health Sciences Centre granted a waiver of consent.
Guarantor
LAS
Contributorship
AAB, SF, and LAS conceived and were involved in the study design. AAB and SF performed the statistical analysis. DA, AAB, FL, and LF were involved in patient recruitment and dataset preparation. AAB and LAS drafted the manuscript. All authors contributed to the interpretation of the data, revised the manuscript for important intellectual content, approved the final version to be published, and participated sufficiently in the work to take public responsibility for the appropriateness of the content.
Study registration
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
