Abstract
Background:
Early ischemic changes on baseline imaging are commonly evaluated for acute stroke decision-making and prognostication.
Aims:
We assess the association of early ischemic changes on clinical outcomes and whether it differs between intravenous tenecteplase and Alteplase.
Methods:
Data are from the phase 3, Alteplase compared to Tenecteplase (AcT) trial. Subjects with anterior circulation stroke were included. Early ischemic changes were assessed using the Alberta Stroke Program Early CT score (ASPECTS). Efficacy outcomes included modified Rankin scale (mRS) 0–1, mRS 0–2, and ordinal mRS at 90 days. Safety outcomes included 24-h symptomatic intracerebral hemorrhage (sICH), any hemorrhage on follow-up scan, and 90-day mortality rate. Mixed-effects logistic regression was used to assess the association of ASPECTS (continuous and categorical (0–4 vs 5–7 vs 8–10)) with outcomes and if these associations were modified by thrombolytic type after adjusting for age, sex, and baseline stroke severity.
Results:
Of the 1577 patients in the trial, 901 patients (56.3%; median age 75 years (IQR 65–84), 50.8% females, median National Institute of Health Stroke Scale (NIHSS) 14 (IQR 17–19)) with anterior circulation stroke were included. mRS 0–1 at 90 days was achieved in 1/14 (0.3%), 43/160 (14.7%), and 252/726 (85.1%) in the ASPECTS 0–4, 5–7, and 8–10 groups respectively. Every one-point decrease in ASPECTS was associated with 2.7% and 1.9% decrease in chances of mRS 0–1 and mRS 0–2 at 90 days, respectively, and 1.9% chances of increase in mortality at 90 days. Subgroup analysis in endovascular thrombectomy (EVT)-treated population showed similar results. Thrombolytic type did not modify this association between ASPECTS and 90-day mRS 0–1 (P-interaction 0.75). There was no significant interaction by thrombolytic type with any other outcomes.
Conclusion:
Similar to prior studies, we found that every one-point decrease in ASPECTS was associated with poorer clinical and safety outcomes. This effect did not differ between alteplase and tenecteplase.
Data access statement:
Data shall made available on reasonable request from the PI (BMM).
Introduction
The Alberta Stroke Program Early CT Score (ASPECTS) is one of the commonly used decision-making tools to evaluate early ischemic changes on baseline imaging prior to acute stroke treatment in patients with anterior circulation stroke. It has shown to be useful in prognostication of stroke outcomes, such as the functional outcome on modified Rankin scale (mRS) at 90 days, symptomatic intracerebral hemorrhage (sICH), and mortality after intravenous thrombolysis (IVT) treatment.1–5 An ASPECTS cut-off > 7 has shown to be associated with good functional outcome, while ASPECTS ⩽ 4 has been shown to be associated with higher rates of sICH after receiving IVT treatment, especially with ASPECTS ⩽ 2.3,4
IVT is one of the main treatment options for patients with acute ischemic stroke. Treatment with intravenous alteplase, and now tenecteplase, has shown to have a substantial benefit with higher odds of achieving excellent outcomes (mRS 0–1) when given within 4.5 h of symptom onset.6–8 Tenecteplase, a second-generation thrombolytic agent, is more fibrin-specific and has been shown to be non-inferior to alteplase in various clinical trials,9,10 leading to change in treatment guidelines across many countries.11–14 Some non-randomized studies have shown potentially lower rates of sICH with tenecteplase, but this has not been seen in randomized data. 15 It is unknown whether the relationship between tenecteplase and ASPECTS is similar to alteplase across all ASPECTS scores.
Thus, this study aimed to assess the influence of ASPECTS on stroke outcomes and the treatment effect and safety of tenecteplase as compared to alteplase across ASPECTS in patients with anterior circulation stroke who were eligible for standard IVT within AcT trial. We further aimed to demonstrate the contribution of ASPECTS on predicting outcomes after acute stroke treatment following IVT and endovascular thrombectomy (EVT).
Methods
The data supporting this study’s findings are available from the corresponding author on request and the local ethics committee.
Study protocol and patient population
The Alteplase compared to Tenecteplase (AcT) trial was a multicenter, open-labeled, randomized controlled trial to investigate the non-inferiority of tenecteplase 0.25 mg/kg as compared to alteplase 0.9 mg/kg acute ischemic stroke patients eligible for IVT presenting within 4.5 h of symptom onset (NCT 03889249). Patients were enrolled from 22 primary and comprehensive stroke centers across Canada. This study’s findings are based on data from the AcT trial, patients enrolled between 10 December 2019 and 25 January 2022. The detailed methodology, inclusion criteria, and results for the trial have been published elsewhere. 16 Briefly, the inclusion and exclusion criteria were informed by the Canadian Stroke Best Practice Recommendations (CSBPR 2018), 17 with imaging showing early signs of extensive infarction as a relative exclusion criterion and required clinical judgment based on the specific situation. The AcT trial (Clinical Trials Application number 231509) was approved by local ethics boards at all participating centers. The trial used deferred consent procedures wherever approved by local research ethics boards. Informed written or verbal consent was obtained from the patients or their surrogates as per local ethical approval. This secondary analysis followed the CONSORT guidelines (Supplemental material).
For the current analysis, we include patients with internal carotid artery (ICA) and middle cerebral artery (MCA) stroke using available interpretable imaging of baseline non-contrast computed tomography (NCCT), CT angiography (CTA), and follow-up scan. Patients with final diagnosis of anterior cerebral artery and vertebrobasilar occlusion were excluded. Baseline demographics (age, sex, and baseline National Institute of Health Stroke Scale (NIHSS)) were compared between patients included in the study versus those who were excluded. Baseline characteristics and outcomes were reported across three ASPECTS categories, namely 0–4, 5–7, and 8–10, as reported in prior studies. 18 We further reported a subgroup analysis in patients undergoing EVT within this patient population.
Neuroimaging analysis
Since AcT was a pragmatic clinical trial, baseline imaging including CT/CT angiogram and CT perfusion and follow-up imaging (CT and magnetic resonance imaging (MRI) brain) was done as part of the routine care in each center. These imaging were then anonymized and transferred to the imaging core laboratory hosted at the University of Calgary. An independent imaging core laboratory including authors F.B., N.S., B.M., and M.A. with 4–20 years of experience read all imaging data.
All imaging was interpreted on the standard high-resolution imaging workstation computers in a similar environment (for lighting and brightness). The ASPECTS was assessed for early ischemic change within the anterior circulation distributed over the MCA territory on the baseline imaging. Early ischemic changes were defined as any parenchymal hypoattenuation and loss of gray-white-matter differentiation compared to the same structure on the contralateral hemisphere with or without focal brain swelling. 1 Independent core laboratory members who scored the ASPECTS were blinded to the patient’s clinical outcomes and treatment allocation while reading the scans. The collection and assessment of the imaging data were reviewed in consensus readings. Disagreements between two readers were solved by an experienced independent third reader for consensus read. Collaterals on single-phase CT angiography was assessed using scoring by Tan et al. 19 Thus, leptomeningeal collateral status was graded with a scale of 0–3, with following definitions, poor collaterals (0—no collateral supply to the occluded MCA territory), intermediate collaterals (1—the collateral supply filling ⩽ 50% of the occluded MCA territory), and good collaterals (2—the collateral supply filling > 50% but < 100% of the occluded MCA territory or 3—for 100% collateral supply of the occluded MCA territory). Standard follow-up scans, including NCCT and MRI, are assessed at 24 h.
Outcomes and measures
Clinical outcome measures included excellent functional outcome (mRS 0–1), functional independence (mRS 0–2), ordinal mRS, and return to baseline functioning at 90–120 days. The mRS for functional outcome is an ordinal scale from 0 to 6, with 0 indicating no neurological symptoms and 6 indicating death. Return to pre-stroke level of function was a pragmatic patient-reported outcome collected in the AcT trial to assess if the patients felt that they were back to their pre-stroke functioning status (yes/no). All outcomes were collected by the blinded study personnel on the same centralized telephone interview. Safety outcomes include death, symptomatic intracranial hemorrhage (sICH), peripheral bleeding requiring blood transfusion, and angioedema. sICH was defined as intracranial hemorrhage that is temporally related to and directly responsible for the worsening of the patient’s neurological condition and, in the investigator’s opinion, was the most important factor for the neurological worsening that occur within 24 h of thrombolytic drug administration. Radiological outcome measures included any imaging identified intracranial hemorrhage using Heidelberg classification, 20 angiographic outcomes included successful reperfusion and recanalization after endovascular treatment defined as extended Thrombolysis in Cerebral Infarction (eTICI) ⩾ 2b/3 and revised arterial occlusive lesion score (rAOL) ⩾ 2b/3, respectively.21,22
Statistical analysis
The study population was divided into three ASPECTS categories, namely 0–4 (low), 5–7 (moderate), and 8–10 (good) to ensure comparability with prior published studies. 18 Baseline characteristics were compared across these three ASPECTS categories. Data were expressed as medians with interquartile ranges (IQRs) (non-normal distribution), mean with standard deviation (normal distribution) for continuous variables, and counts and percentages for categorical variables. These were compared by chi-square test, Fisher’s exact test, or Kruskal–Wallis test as appropriate. To assess effect modification by thrombolytic type (alteplase versus tenecteplase) on treatment outcomes, we used two-way multiplicative interaction terms (ASPECTS and treatment type) in an unadjusted model. If the interaction term was not significant, then we only reported main effects in the model mentioned below without stratifying by treatment type. To assess the association between ASPECTS (continuous) and functional outcomes, we used mixed-effects logistic regression with age, sex, baseline NIHSS, onset to thrombolysis start time, and interaction term (ASPECTS and thrombolytic type, if applicable) as fixed-effects variables and participating site as random-effects variable. Proportions of all outcomes were reported across different ASPECTS categories and adjusted analyses were done using mixed-effects linear regression model adjusted for with age, sex, and baseline NIHSS as fixed-effects variables and participating site as random-effects variable. Shapley values were calculated for all independent variables in the regression model to determine the marginal contribution of each variable to the overall pseudo R-squared in the functional outcome model including excellent functional outcome (mRS 0–1), good functional outcome (mRS 0–2), and mortality model at 90 days. Similar analysis was repeated in patients who underwent EVT. All statistical tests were two-sided, and p-values less than 0.05 were considered statistically significant. STATA 16 was used to perform the analysis mentioned above.
Results
Of the 1600 patients in the AcT trial, 901 (56.3%) were included in this study. Of these, 470 (52.2%) were in tenecteplase arm and 431 (47.8%) were in alteplase arm (Supplemental Figure S1, Study Flowchart). Overall, median age was 75 years (IQR 65–84); 458 patients (50.8%) were females, and median NIHSS was 14 (IQR 17–19). The baseline ASPECTS was related to stroke severity (Supplemental Figure S2). Overall, there were 727 (80.7%) participants with high ASPECTS (ASPECTS 8–10), 160 (17.8%) moderate ASPECTS (ASPECTS 5–7), and 14 (1.5%) low ASPECTS (ASPECTS 0–4). The median ASPECTS in the three groups were 9, 7, and 4, respectively. The intraclass correlation coefficient (ICC) between two readers for the total ASPECTS was 0.72, calculated based on the absolute agreement and a two-way mixed-effects model. Patients excluded from the study were younger with lower median NIHSS (median age 71 (IQR 60–82); median NIHSS 7 (IQR 5–10)) and 43.9% females. Further details of this patient population are reported in Supplemental Table S1. Patient’s baseline characteristics across the three ASPECTS categories are summarized in Table 1. When compared with moderate and low ASPECTS patients, those with high ASPECTS group had lower NIHSS scores (median 11 (IQR 6–19) vs median 18 (IQR 14–22) vs median 22 (IQR 19–27)), lower proportion of large vessel occlusion (36.5% vs 71.3% vs 92.9%), greater prevalence of good collateral grading on CTA (55.4% vs 24.8% vs 0%), and shorter time from stroke onset to hospital arrival (74 (51–124) vs 84 (58–145) vs 106 (66–169) min). Patients’ characteristics were similar between alteplase and tenecteplase across all three ASPECTS groups (Supplemental Table S2).
Baseline characteristics comparing patients stratified by ASPECTS categories.
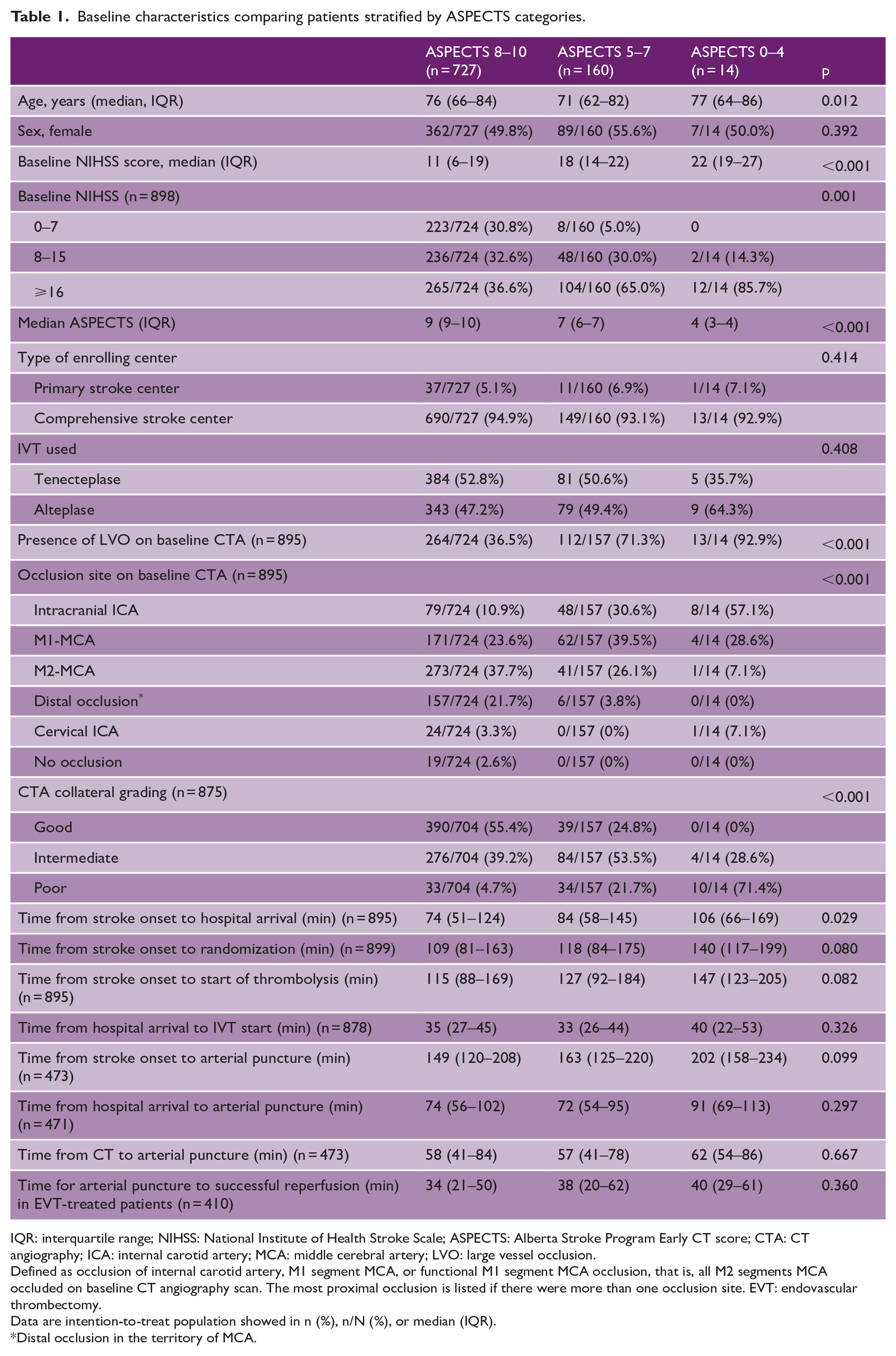
IQR: interquartile range; NIHSS: National Institute of Health Stroke Scale; ASPECTS: Alberta Stroke Program Early CT score; CTA: CT angiography; ICA: internal carotid artery; MCA: middle cerebral artery; LVO: large vessel occlusion.
Defined as occlusion of internal carotid artery, M1 segment MCA, or functional M1 segment MCA occlusion, that is, all M2 segments MCA occluded on baseline CT angiography scan. The most proximal occlusion is listed if there were more than one occlusion site. EVT: endovascular thrombectomy.
Data are intention-to-treat population showed in n (%), n/N (%), or median (IQR).
Distal occlusion in the territory of MCA.
Outcomes across ASPECTS categories
In an unadjusted analysis, the absolute rates of mRS 0–1, mRS 0–2, and return to pre-stroke function were highest in ASPECTS 8–10 category and were lowest in ASPECTS 0–4 category. Conversely, rates of symptomatic hemorrhage were highest in ASPECTS 5–7 and death were highest in ASPECTS 0–4 category. Patients with high ASPECTS have the lowest rate of sICH and death. (Supplemental Table S3). In an adjusted analysis with ASPECTS 8–10 as the reference category, there were no significant differences for functional outcomes across the three ASPECTS categories except that ASPECTS 0–4 were significantly associated with lower rates of mRS 0–2 at 90 days as compared to ASPECTS 8–10 (Table 2). Patients with ASPECTS 0–4 had significantly higher mortality rates as compared to moderate and high ASPECTS (57.1% vs 22.5% vs 16.8%, Supplemental Table S3) (adjRR 2.14 (1.17–3.89) for ASPECTS 0–4, adjRR 1.25 (0.92–1.69) for ASPECTS 5–7, reference ASPECTS 8–10, Table 2). Compared to high ASPECTS group, patients with moderate ASPECTS had higher rate of sICH (14/160 (8.75%) vs 18/727 (2.48%); adjRR 2.85 (1.15–7.07) and had higher rate of any ICH on follow-up scans (69/157 (43.9%) vs 167/718 (23.3%); adjRR 1.66 (1.23–2.25) (Supplemental Tables S2 and S3). There were no significant differences in other safety and angiographic outcomes (Supplemental Tables S2 and S3). The distribution of mRS by category is shown in Figure 1(a).
Adjusted analysis reporting outcomes stratified by ASPECTS categories with ASPECTS 8–10 as the reference.
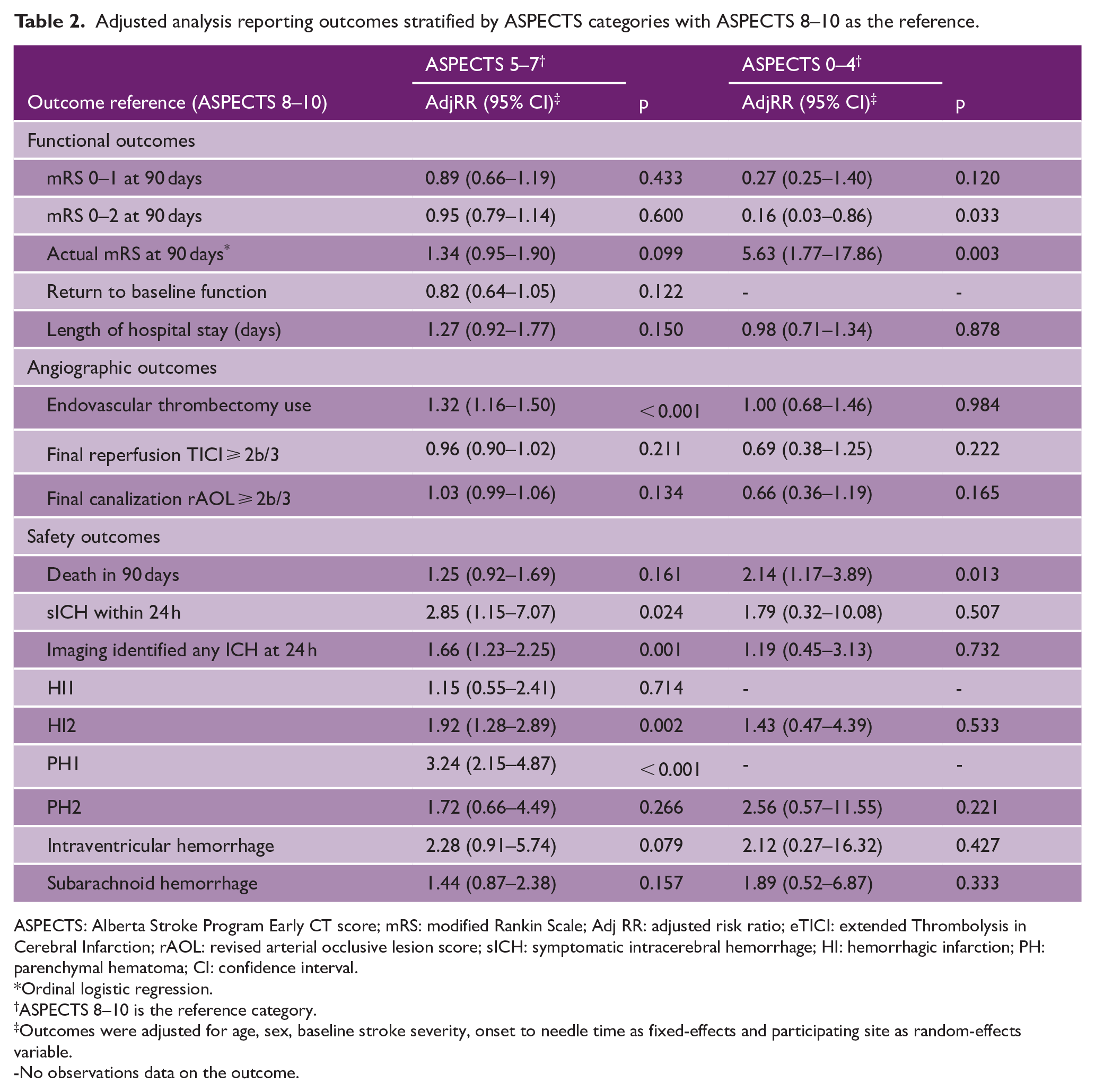
ASPECTS: Alberta Stroke Program Early CT score; mRS: modified Rankin Scale; Adj RR: adjusted risk ratio; eTICI: extended Thrombolysis in Cerebral Infarction; rAOL: revised arterial occlusive lesion score; sICH: symptomatic intracerebral hemorrhage; HI: hemorrhagic infarction; PH: parenchymal hematoma; CI: confidence interval.
Ordinal logistic regression.
ASPECTS 8–10 is the reference category.
Outcomes were adjusted for age, sex, baseline stroke severity, onset to needle time as fixed-effects and participating site as random-effects variable.
-No observations data on the outcome.
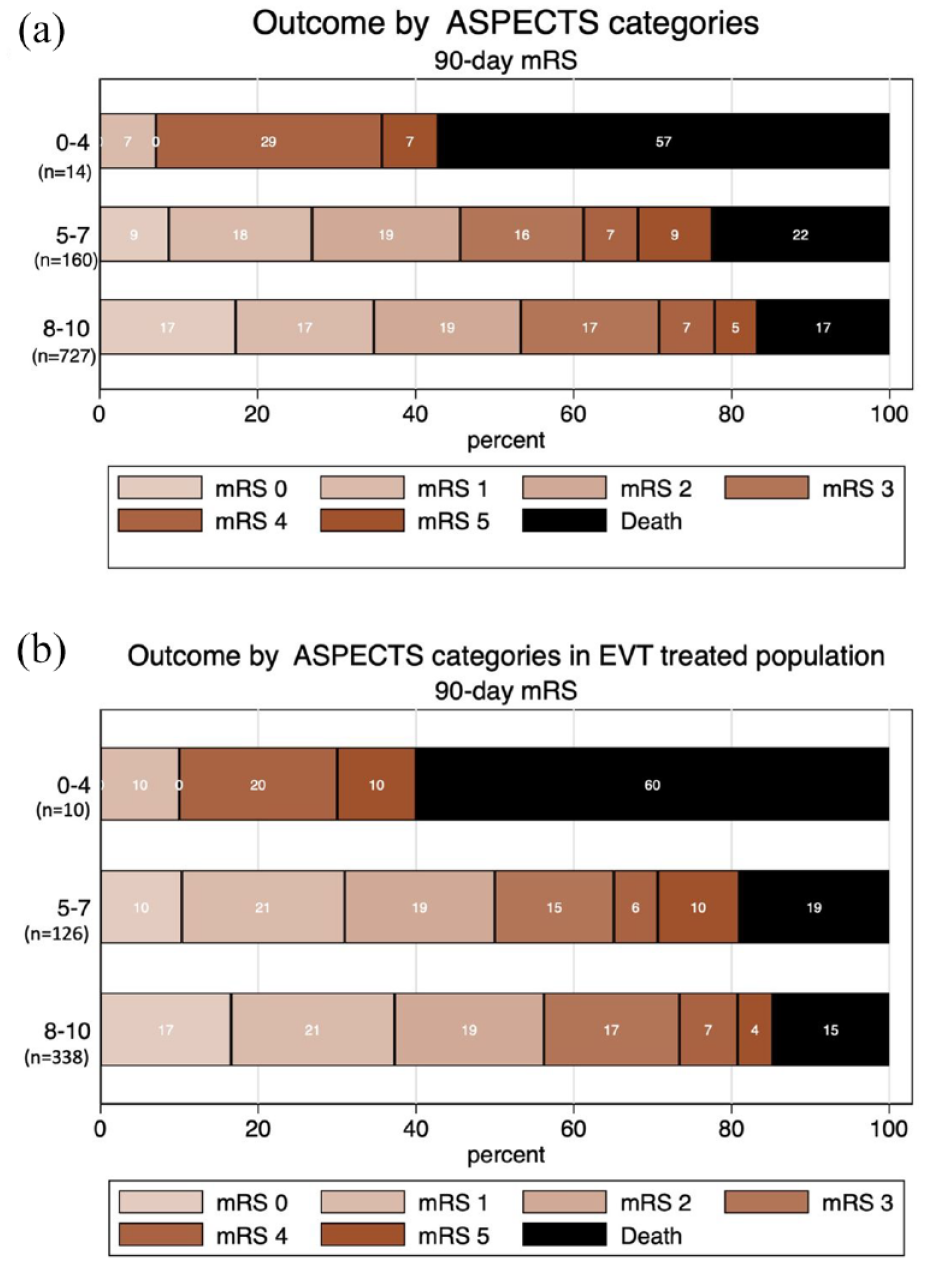
Distribution of mRS outcomes by ASPECTS categories. Horizontal bar graph showing mRS outcomes by ASPECTS categories. The distribution of ordinal mRS in (a) the total study population (n = 901) and (b) EVT-treated population (n = 474).
Outcomes across ASPECTS categories in the EVT-treated subgroup
Among the patients included in the study, 474 (52.6%) were treated with EVT. In this subgroup, there were 338 (71.3%) participants with high ASPECTS, 126 (26.6%) moderate ASPECTS, and 10 (2.1%) low ASPECTS. The median age was higher in the low ASPECTS as compared to the moderate and high ASPECTS (80 (IQR 64–88) vs 70 (IQR 61–79) vs 74 (IQR 66–82) years, respectively). When compared with moderate and low ASPECTS patients, those with high ASPECTS had lower NIHSS scores (median 16 (IQR 10–21) vs median 18 (IQR 14–22) vs median 23 (IQR 19–27)) and a higher proportion of good collateral grading on CTA (40.7% vs 24.8% vs 0%, Supplemental Table S3). Outcomes across the ASPECTS categories were similar to the overall study population. The ASPECTS 8–10 category showed the highest absolute rates of mRS 0–1, mRS 0–2, and return to pre-stroke function, while the rates of symptomatic hemorrhage and death were highest in ASPECTS 0–4 category (Figure 1(b), Supplemental Table S4). The adjusted analysis with ASPECTS 8–10 as the reference category showed no significant differences in functional outcomes across the three ASPECTS categories. The ASPECTS 0–4 category had significantly higher rate of death as compared to moderate and high ASPECTS (60.0% vs 19.1% vs 14.8%, Supplemental Table S5) (adjRR 2.52 (1.21–5.22) for ASPECTS 0–4, adjRR 1.38 (0.91–2.08) for ASPECTS 5–7, reference ASPECTS 8–10, Supplemental Table S6). Similar to the overall population, patients with moderate ASPECTS had higher rate of sICH and any ICH on follow-up scans as compared to high ASPECTS (9.5% vs 2.7%, adjRR 3.25 (1.29–8.23) for sICH and 44.8% vs 30.1%, adjRR 1.44 (1.04–2.01) for any ICH).
Continuous ASPECTS and the influence on outcomes
There was no significant interaction between continuous ASPECTS and thrombolytic type for any functional outcomes (Supplemental Table S7). Thus, we only reported the main effects of the influence of ASPECTS on functional outcomes. The influences of ASPECTS on functional outcomes at 90 days are shown in Figure 2. In the total study population, every one-point decrease in ASPECTS was associated with 2.7% and 1.9% decrease in chances of mRS 0–1 and mRS 0–2, respectively (Figure 2(a) and (c)). Every one-point decrease in ASPECTS was associated with 1.9% chance of an increase in mortality (Figure 2(e)). Among the patients treated with EVT, every one-point decrease in ASPECTS was associated with 3% and 3.1% decrease in chances of mRS 0–1 and mRS 0–2, respectively (Figure 2(b) and (d)). The mortality increases 2.4% for every one-point decrease in ASPECTS (Figure 2(f)).
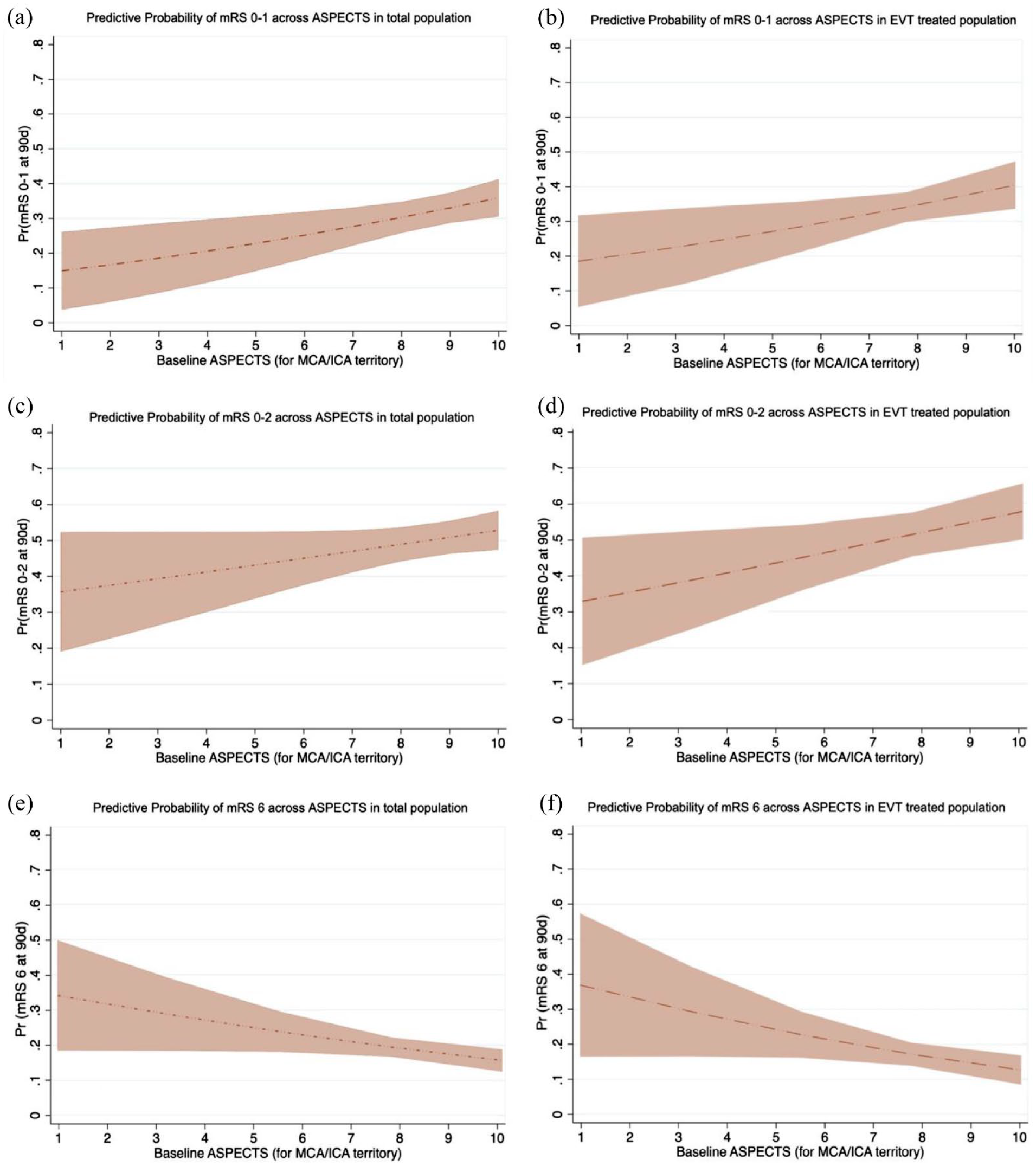
The influence of ASPECTS on functional outcomes at 90 days. The association between ASPECTS and functional outcomes using mixed-effects logistic regression with age, sex, baseline NIHSS as fixed-effects variables. Figures demonstrated the total study population (a, c, and e) and EVT-treated population (b, d, and f). For every one-point decreased in ASPECTS, the probability at 90 days of mRS 0–1 decreased by (a) 2.7% and (b) 3%, the probability of mRS 0–2 decreased by (c) 1.9% and (d) 3.1%. The probability of mortality at 90 days increased by (e) 1.9% and (f) 2.4% for every one-point decreased in ASPECTS.
The contribution of ASPECTS to the functional outcomes prediction
For predicting functional outcome at 90 days in a model including baseline clinical, imaging, and treatment variables, we found ASPECTS alone contributes 5.6% in predicting excellent functional outcome (mRS 0–1), 2.8% for good functional outcome (mRS 0–2), and 6.8% for mortality. The average of the absolute Shapley values of each variable contributing toward the functional outcome at 90 days is illustrated in Figure 3.
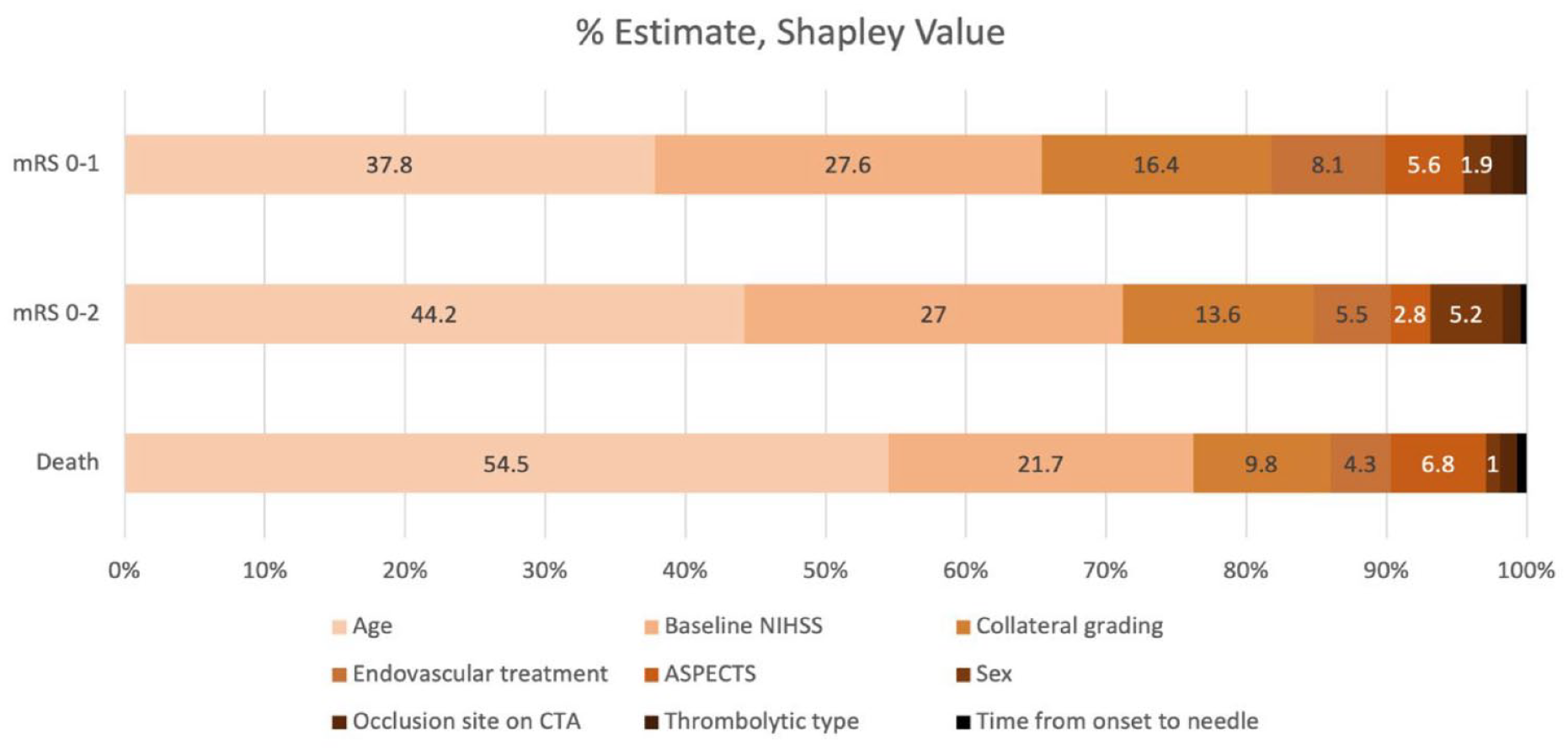
The contribution of factors on the functional outcome and death in patients with IVT treatment (Shapley value; percentage estimate).
Discussion
The current exploratory analysis from the AcT trial comprising patients with anterior circulation strokes shows that ASPECTS, as a continuous measurement, is an important predictor of poor outcomes in patients treated with IVT and EVT treatment within 4.5 h of symptom onset. The findings of this study contribute to the understanding that the use of ASPECTS is instrumental in identifying who stand to gain benefits from IVT and EVT treatment, with positive treatment effect consistent at higher ASPECTS. This study also provides new insights on the similar treatment effect of tenecteplase and alteplase in predicting functional outcomes across all ASPECTS values.
The findings of this study also support that duration of ischemia and collaterals are critical factors in determining the extent of early ischemic changes, especially in the early time window. We saw that patients with more extensive early ischemic changes had more prolonged onset to hospital arrival time and higher proportion of poor collaterals. These results correspond with the data from The Canadian Alteplase for Stroke Effectiveness Study (CASES), which demonstrated a linear association for better outcomes in ASPECTS value of 6–10.2,5 However, none of the previous IVT trials have showed the effect modification of ASPECTS on functional outcome.3–5 Regarding thrombolysis treatment, the ASPECTS > 7 was shown to be an independent factor for good outcome (mRS 0–2) in PROACT-II trial; however, the study used intraarterial thrombolytic for anterior circulation stroke who had proximal arterial occlusion. 23 Similar to our study, lower ASPECTS correlated with significantly lower rate of good functional outcomes (mRS 0–2) and more frequent rate of intracerebral hemorrhage.23,24 The notably higher incidence of sICH and any ICH in the moderate ASPECTS group in our study is possibly attributed to a higher number of patients with higher NIHSS and poor collateral status in this category. In addition, follow-up MRI is used in 21%, 36%, and 42% in the ASPECTS 0–4, 5–7, and 8–10, respectively. Caution is advised in interpreting results for the low ASPECTS category due to the limited number of patients in this subgroup. Nevertheless, it is noteworthy that the absolute rates of death within 90 days were high in this subgroup, consistent with prior studies demonstrating the inferior prognosis of IVT with alteplase compared to EVT alone in patients with low ASPECTS.25,26 The results of this study, although limited by small number, are relevant in context of recent large randomized trials on large core infarcts showing benefit of EVT as compared to the standard treatment.27–30 The rates of IVT in these large core trials ranged from 20% to 39% and a stratified analysis based on thrombolysis treatment is yet to be published. ASPECTS alone cannot negate the benefit of treating this subgroup of patients. In addition, data on treatment effect of tenecteplase across ASPECTS have not been studied until now.
Overall, the study results align with the previous studies highlighting the impact of ischemic change on treatment efficacy. ASPECTS has limitations. Its semiquantitative nature and reliance on CT imaging may limit precision and sensitivity, and the score’s interpretation may be subjective and operator-dependent. ASPECTS does not provide information on collateral circulation, timing of changes and is not applicable to non-MCA strokes. Despite its limitations, ASPECTS offers a semiquantitative estimate of volume combined with localization, stands out as a good scoring tool, allowing efficient assessment of CT scan interpretation for non-experts.
This study has some limitations. This is a post hoc exploratory analysis. Therefore, the results need to be interpreted in that context. The interpretation of ASPECTS is relevant to the early ischemic changes from ICA and MCA strokes, not in the context of all anterior circulation infarction. The small sample size in low ASPECTS subgroup led to wide confidence intervals and the results should be interpreted with caution. The effect of ASPECTS in our study can be applied to only IVT patients treated within 4.5 h, and the result may not be generalizable for stroke patients in the extended time window.
Conclusion
In thrombolysis-treated patients presenting within 4.5 h of symptom onset, ASPECTS is an important prognostic tool for functional and safety outcomes. These effects are similar for both alteplase and tenecteplase.
Supplemental Material
sj-pdf-1-wso-10.1177_17474930241273561 – Supplemental material for The treatment effect across ASPECTS in acute ischemic stroke: Analysis from the AcT trial
Supplemental material, sj-pdf-1-wso-10.1177_17474930241273561 for The treatment effect across ASPECTS in acute ischemic stroke: Analysis from the AcT trial by Chitapa Kaveeta, Ibrahim Alhabli, Fouzi Bala, MacKenzie Horn, Faysal Benali, Shelagh B Coutts, Atif Zafar, Olena Bereznyakova, Alexander Khaw, Houman Khosravani, Gary Hunter, Aleksander Tkach, Dar Dowlatshahi, Luciana Catanese, Chrysi Bogiatzi, Ramana Appireddy, Brian H Buck, Richard H Swartz, Tolulope T Sajobi, Mohammed Almekhlafi, Andrew M Demchuk, Aravind Ganesh, Bijoy Menon and Nishita Singh in International Journal of Stroke
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: L.C. received payments from Servier, consulting fees from iSchemaView RAPID, Circle Neurovascular Imaging (NVI), and Canadian Medical Protective Association. T.T.S. has received consulting fees from Circle NVI. R.H.S. has stock options in FollowMD and gets salary support for research from the Heart and Stroke Foundation of Canada, the Sandra Black Center for Brain Resilience & Recovery, and Ontario Brain Institute. B.M. has stock options in Circle NVI and has consulted for Biogen and Boehringer Ingelheim. M.A. reports grants from Canadian Institute of Health Research during the conduct of the study. No other disclosures are reported. N.S. gets salary support for research from the Heart and Stroke Foundation of Canada, Research Manitoba and University of Manitoba.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Alteplase compared to Tenecteplase (AcT) Trial was funded by Canadian Institutes of Health Research, by Alberta Innovates, and by AbSPORU (Alberta Strategy for Patient-Oriented Research Support Unit).
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
