Abstract
Introduction:
Limited data exist on intravenous thrombolysis (IVT) effect on fibrinogen depletion, and on possible associations between low fibrinogen levels and hemorrhagic complications, among acute ischemic stroke (AIS) patients.
Patients and methods:
We conducted a prospective cohort study evaluating fibrinogen levels prior to IVT and at two prespecified timepoints following IVT end (2 and 24 h). Safety and efficacy outcomes were assessed. Fibrinogen depletion was defined as reduction < 200 mg/dl, or as reduction ⩾ 50% of baseline fibrinogen levels after 2 and 24 h from IVT. Furthermore, we conducted a systematic review and meta-analysis, comparing fibrinogen level changes from baseline to a timeframe of 2–12 h, and to 24 h post-IVT between patients receiving alteplase versus tenecteplase.
Results:
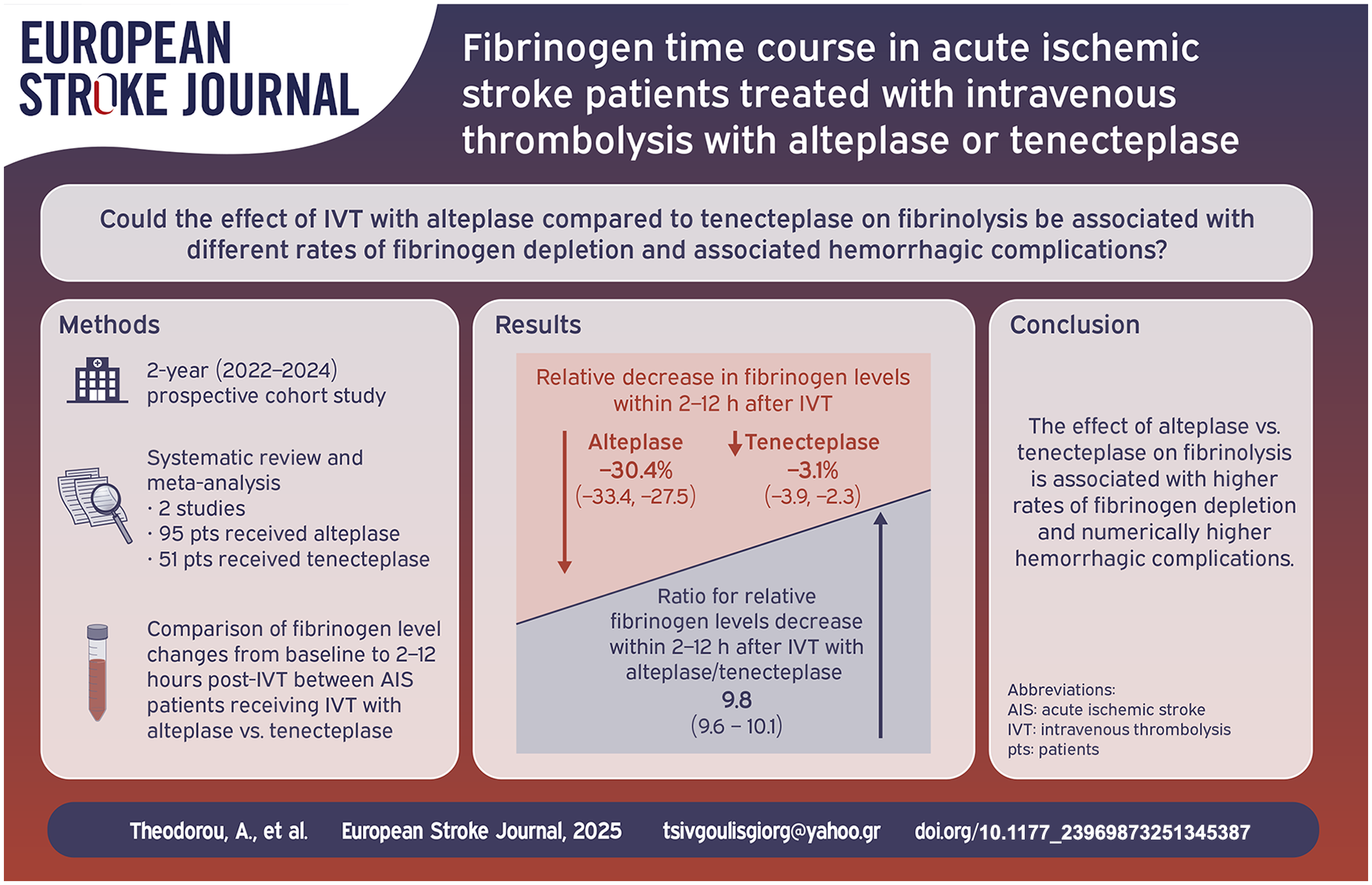
One hundred and sixteen AIS patients, treated with IVT (alteplase = 81, tenecteplase = 35) were included in this study. Alteplase was associated with higher rates of fibrinogen depletion, observed in 2 (38% vs 0%; p-value < 0.001) and 24 h (26% vs 0%; p-value: 0.002) post-IVT, compared to tenecteplase. Three symptomatic intracranial hemorrhages (sICH) and one major extracranial bleeding were observed in patients receiving alteplase (two out of four with fibrinogen depletion) and one sICH without associated fibrinogen depletion was documented in the tenecteplase subgroup. Major hemorrhagic complications were numerically higher in patients with fibrinogen depletion (6.5% vs 3.5%, p = 0.609). Fibrinogen depletion was documented in 50% of alteplase-treated patients complicated with sICH or major extracranial bleeding and in 0% of respective tenecteplase-treated patients. Two cohorts were included in the meta-analysis. Mean relative changes of fibrinogen levels from baseline to a timeframe of 2–12 h post-IVT (RoM: 9.84; 95% CI: 9.63–10.05) were significantly higher in patients receiving alteplase.
Discussion and conclusions:
Fibrinogen depletion was observed only in AIS patients receiving alteplase and was associated with numerically higher rates of major hemorrhagic complications. These findings require independent confirmation in larger, international cohort studies.
Introduction
Tenecteplase represents a genetic modified variant of the tissue plasminogen activator (TPA) alteplase, with higher fibrin specificity (approximately ~15-fold higher) and lower rates of systemic hemorrhage, compared to alteplase.1,2 Currently, tenecteplase at a dose of 0.25 mg/kg has been recommended as alternative to alteplase for unselected patients with acute ischemic stroke (AIS) within the first 4.5 h of symptoms onset. 3 However, there is scarce data regarding the effect of alteplase compared to tenecteplase on the fibrinolytic activity, leading to early fibrinogen depletion and hemorrhagic outcomes among patients with AIS treated with intravenous thrombolysis (IVT).
In view of the former considerations, we performed an observational prospective cohort study aiming to evaluate serial fibrinogen levels in patients with AIS, treated with alteplase (standard-dose of 0.9 mg/kg) compared to tenecteplase (recommended treatment dose of 0.25 mg/kg) in two tertiary stroke centers. We measured fibrinogen levels prior to IVT and in predefined timepoints (2 and 24 h) after the end of IVT and we compared them between patients who received alteplase versus tenecteplase. Furthermore, we conducted a systematic review and meta-analysis to investigate and compare fibrinogen level changes from baseline to a timeframe of 2–12 h, and to 24 h after the end of IVT between AIS patients receiving alteplase versus tenecteplase.
To the best of our knowledge, this is the first prospective observational cohort study, and the first systematic review and meta-analysis with the aim to robustly address all available data evaluating the effect of IVT with alteplase versus tenecteplase on fibrinogen depletion and on possible associations between decreased fibrinogen levels and hemorrhagic complications, among AIS patients receiving IVT.
Methods
Prospective cohort study
Study design
We prospectively evaluated consecutive AIS patients, treated with alteplase or tenecteplase during a 2-year period (December 2022–November 2024) in two tertiary stroke centers in Greece (Department of Neurology, Attikon University Hospital, and Stroke Unit, Metropolitan Hospital). 4 This study was performed in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for reporting observational research. 5
Ethical approval and patient consent
The study was approved by the local Ethics Committees of Attikon University Hospital (protocol number: ΕΒD526/12-9-2022) and Metropolitan Hospital (protocol number: 3423/30-09-2022). All participants or their legal representatives provided signed informed consent before enrollment.
The data that support the findings of the present study are available from the corresponding author upon reasonable request.
Study population and selection criteria
Consecutive AIS patients treated with either alteplase or tenecteplase were prospectively evaluated during a 2-year period in two tertiary stroke centers in Greece. Patients who underwent endovascular treatment (EVT) following IVT, were also included in the current study. Both centers participate in SITS (Safe Implementation of Thrombolysis in Stroke) Registry. 4 Patients who did not provide written informed consent to participate and patients with unavailable serial fibrinogen measurements were excluded from the current study.
Data collection
The following data were collected for each patient: age, sex, risk factors, medical comorbidities, stroke clinical severity assessed by National Institutes of Health Stroke Scale (NIHSS), fibrinogen levels at admission, 2 and 24 h after the end of IVT, and different laboratory parameters on admission, 2 and 24 h after the end of IVT. Fibrinogen depletion was defined as a reduction below 200 mg/dl, or as a reduction ⩾ 50% of baseline fibrinogen levels after 2 and 24 h post IVT. These thresholds were based on previous studies, reporting a statistically significant association between hemorrhagic complications and a more pronounced decrease in fibrinogen concentrations, following IVT with alteplase.6 –8 All patients included in this cohort had a 3-month follow-up. The NIHSS score was assessed on admission, at 2 and 24 h following IVT and the modified Rankin scale (mRS) score was assessed on discharge and at 3 months after stroke onset. 9
Peripheral blood samples were drawn from each patient prior to IVT, and at prespecified timepoints after the end of IVT (2 and 24 h). Fibrinogen levels were determined on fresh plasma obtained from blood collected in 5 ml vacuum tubes containing 0.5 ml sodium citrate (0.129 mol/l). Moreover, we measured hemoglobin levels, prothrombin time (PT), activated partial thromboplastin time (APTT), d-dimers and international normalized ratio (INR).
Outcome measures
Safety outcomes included symptomatic intracranial hemorrhage (sICH), according to the SITS-MOST criteria (presence of local or remote parenchymatous hemorrhage type 2 within 22–36 h post-IVT, combined with NIHSS score increase of 4 or more points or leading to death), any intracranial hemorrhage, and 3-month mortality. 10 Efficacy outcomes included the 3-month excellent functional outcome, defined as mRS scores: 0-1 and the 3-month good functional outcome, defined as mRS scores: 0–2. 9 Asymptomatic ICH, documented within 48 h post-IVT were classified according to Heidelberg Bleeding Classification. 11
Patients who received bridging therapy (IVT coupled with EVT) were also included in the current analysis. A monthly center-based check of collected data was implemented to avoid missing data.
Statistical analysis
Categorical variables are presented as number and percentages, whereas continuous variables are presented with their corresponding mean values and standard deviations (SDs) in cases of normal distribution and as medians with interquartile ranges (IQRs) in cases of skewed distributions. Comparisons of demographics, clinical characteristics and laboratory findings (before IVT, 2 and 24 h after the end of IVT) were performed with χ2-test (Fisher exact test), unpaired t-test and Mann-Whitney U test.
Meta-analysis
Standard protocol approvals, registrations, and patient consents
Moreover, an aggregate data meta-analysis was performed including all the eligible cohort studies and the current study as well. The meta-analysis is reported according to the updated Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines 12 and was written according to the Meta Analysis of Observational Studies in Epidemiology (MOOSE) proposal. 13 The study protocol, comprising pre-determined PICOS (Population, Intervention, Comparison, Outcome and Study) framework, was a priori designed and registered in the International Prospective Register of Ongoing Systematic Reviews PROSPERO database (CRD42024591829).
Data sources and database searches
In this systematic review and meta-analysis three independent reviewers (AT, KM, KP) searched for published eligible studies reporting fibrinogen levels prior and after IVT with either alteplase or tenecteplase for AIS. We searched PUBMED and Scopus, using search strings that included the following terms: “acute ischemic stroke,” “intravenous thrombolysis,” “alteplase,” “tenecteplase”, “fibrinogen,” “fibrinolysis.” The complete search algorithms used in PUBMED and Scopus are provided in the Supplemental Material.
No language or other restrictions were applied. The search spanned from electronic database’s inception to March 10, 2024. An additional manual search of bibliographies of articles meeting study inclusion criteria was conducted to ensure the comprehensiveness of the literature.
Study selection
Registry-based studies, observational cohort studies, and case series reporting on adult patients (⩾18 years old) with AIS, who received IVT with either alteplase or tenecteplase and the fibrinogen levels prior and after the end of IVT were available, were considered eligible. Included patients received IVT either with alteplase or tenecteplase, followed in eligible cases by mechanical thrombectomy (MT). Exclusion criteria comprised: (1) studies that did not report data with regard to fibrinogen levels prior and following IVT; (2) studies with overlapping data; (3) study population of <10 patients; (4) reported outcomes not aligned with our inclusion criteria; (5) narrative and systematic reviews, commentaries, pre-prints or non-paper reviewed studies, and conference abstracts.
All retrieved studies were independently assessed by three reviewers (AT, KM, and KP), and any disagreements were resolved after discussion with a fourth tie-breaking evaluator (GT).
Data extraction
All retrieved studies were assessed by three reviewers (AT, KM, KP) independently, and disagreements were settled by consensus after discussion with the corresponding author (GT).
The following information were extracted: name of the study, first author and year of publication, study design, and data collection interval, total number of study participants, mean age, sex distribution, relative fibrinogen levels change in 2–12 h after the end of IVT and in 24 h after the end of IVT compared to baseline levels.
Study quality and assessment of publication bias
Quality control and bias assessment among eligible studies were performed employing the Risk of Bias in Non-randomized Studies of Interventions (ROBINS-I) tool for the observational studies, independently by two authors (AT and KM). 14 Any disagreements were settled by consensus after discussion with the corresponding author (GT).
The publication bias across individual studies was evaluated graphically using funnel plots, when at least four studies were included in the analysis of the outcomes of interest. Funnel plot asymmetry was assessed using Egger et al.’s linear regression test, with a two-sided p-value < 0.1 being considered statistically significant. 15
Statistical analysis
Meta-analysis was performed using the R–software version 2023.06.0+421 (packages: base, meta and metafor). 16 Continuous outcomes were assessed by raw means. With regard to fibrinogen level changes, we calculated the Ratio of Means (RoM) fibrinogen levels change from baseline to a timeframe of 2–12 h, and to 24 h after the end of IVT, meta-analyzing patients who received alteplase to tenecteplase.
RoM exhibits comparable performance characteristics to other methods, including mean difference, controls for the variability in concentration ranges between studies and simplifies the clinical interpretation of the results. The variance of this ratio was assessed using the delta method and we used inverse variance weighting to pool ratios. RoM < 1 and RoM > 1 indicates lower and higher mean fibrinogen level changes from baseline to 2–12 h and 24 h after the end of IVT among patients receiving alteplase compared to those receiving tenecteplase, respectively.
Heterogeneity between studies was assessed with the Cochran Q and I2 statistics. A p-value, resulting from the Cochran’s Q test, and less than 0.1 indicated heterogeneity. Moreover, and for the qualitative interpretation of the heterogeneity, I2 > 50% and I2 > 75% indicated substantial and considerable heterogeneity, respectively. 17 Random-effects models (DerSimonian and Laird) were used, incorporating between-study heterogeneity. 18 A p-value of 0.05 or less was considered significant.
Results
Patient characteristics
Our cohort comprised 116 AIS patients [mean age: 73 ± 13, 59% men, median National Institutes of Health Stroke Scale (NIHSS)-score: 11 (6–18)] treated with IVT; 81 received alteplase (mean age: 73 ± 13; 64% men) and 35 received tenecteplase (mean age: 75 ± 14; 54% men). The risk factors, medical comorbidities, clinical and laboratory findings on admission are summarized in Supplemental Table S1. Patients who received tenecteplase had statistically significant higher NIHSS-scores on admission [15 (12–19) vs 9 (5–18); p-value: 0.003] and higher rates of large vessel occlusion [23 (66%) vs 27 (33%); p-value: 0.005], compared to those receiving alteplase. Otherwise, key baseline characteristics, cardiovascular risk factors, laboratory findings, including results from coagulation tests, were similar between the two groups.
At 2 h after the end of IVT, patients who received alteplase had significantly lower fibrinogen levels (220.3 ± 114.7 vs 326.3 ± 74.6; p-value: <0.001) and higher rates of fibrinogen depletion [31 (38.3%) vs 0 (0%); p-value: <0.001] compared to those receiving tenecteplase (Table 1). At 24 h after the end of IVT, differences in the absolute decrease of fibrinogen levels [82.7 ± 143.3 vs 3.1 ± 62.1; p-value: 0.002] and in the rate of fibrinogen depletion [21 (25.9%) vs 0 (0%)] remained statistically significant between alteplase and tenecteplase groups.
Temporal course of fibrinogen levels prior and at predefined timepoints following the end of intravenous thrombolysis.

IQR: interquartile range; IVT: intravenous thrombolysis; SD: standard deviation.
Similar rates of asymptomatic ICH, functional outcome at 3 months and mortality, were observed between patients who received alteplase or tenecteplase (Table 2). In detail, 12 asymptomatic ICH were reported in the alteplase and eight in the tenecteplase subgroup. Among patients who received alteplase, eight patients had a parenchymal hematoma type 1 (PH1; defined as a hematoma within infarcted tissue, occupying <30%, without substantive mass effect), two patients has a parenchymal hematoma type 2 (PH2; defined as a hematoma occupying 30% or more of the infarcted tissue, with obvious mass effect) and two patients had a parenchymal hematoma remote from the infarcted tissue. In the tenecteplase subgroup, six patients had a PH1, one patient had a PH2 hemorrhagic transformation and one patient a remote parenchymal hematoma. 11
Temporal course of clinical parameters and laboratory findings prior and after the end of intravenous thrombolysis.
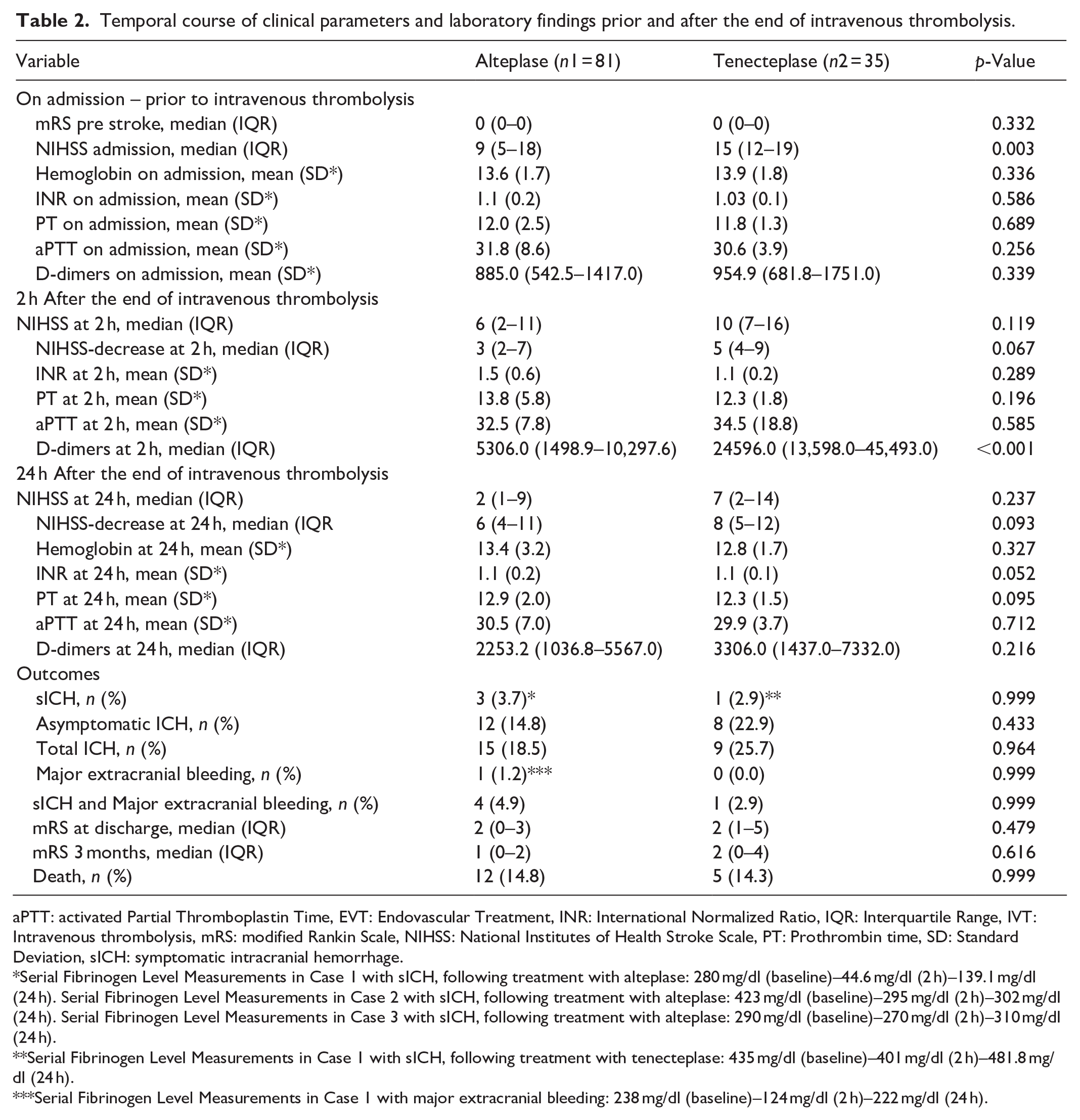
aPTT: activated Partial Thromboplastin Time, EVT: Endovascular Treatment, INR: International Normalized Ratio, IQR: Interquartile Range, IVT: Intravenous thrombolysis, mRS: modified Rankin Scale, NIHSS: National Institutes of Health Stroke Scale, PT: Prothrombin time, SD: Standard Deviation, sICH: symptomatic intracranial hemorrhage.
Serial Fibrinogen Level Measurements in Case 1 with sICH, following treatment with alteplase: 280 mg/dl (baseline)–44.6 mg/dl (2 h)–139.1 mg/dl (24 h). Serial Fibrinogen Level Measurements in Case 2 with sICH, following treatment with alteplase: 423 mg/dl (baseline)–295 mg/dl (2 h)–302 mg/dl (24 h). Serial Fibrinogen Level Measurements in Case 3 with sICH, following treatment with alteplase: 290 mg/dl (baseline)–270 mg/dl (2 h)–310 mg/dl (24 h).
Serial Fibrinogen Level Measurements in Case 1 with sICH, following treatment with tenecteplase: 435 mg/dl (baseline)–401 mg/dl (2 h)–481.8 mg/dl (24 h).
Serial Fibrinogen Level Measurements in Case 1 with major extracranial bleeding: 238 mg/dl (baseline)–124 mg/dl (2 h)–222 mg/dl (24 h).
Notably, sICH and major extracranial bleeding were numerically higher among patients who received alteplase (4.9% vs 2.9%, p = 0.999, Fisher’s exact test). In addition, major hemorrhagic complications were also numerically higher in patients with fibrinogen depletion (6.5% vs 3.5%, p = 0.609, Fisher’s exact test). Fibrinogen depletion was documented in 50% (2 out of 4) of alteplase-treated patients complicated with sICH or major extracranial bleeding and in 0% (0 out of 1) of respective tenecteplase-treated patients (p = 0.999, Fisher’s exact test). Supplemental Figure S1 shows an extensive delayed chest wall hematoma following IVT in a patient with AIS, whose laboratory findings revealed fibrinogen depletion 2 h after the end of IVT.
Meta-analysis
Study selection
The systematic database search yielded a total of 289 and 233 records from the MEDLINE and SCOPUS databases, respectively (PRISMA; Supplemental Figure S2). After excluding duplicates and initial screening, we retrieved the full text of 11 records, which were considered eligible for full text evaluation. After careful evaluation of the available literature, nine studies were excluded (Supplemental Table S2 and Figure S2) resulting in selection of two studies that met the inclusion criteria for our meta-analysis. 19 Our prospective cohort study was also included in the quantitative synthesis of the abovementioned studies (Supplemental Table S3).
Study quality and publication bias
The risk of bias in the included cohort studies was assessed by the ROBINS-I tool and is presented in the Supplemental Figures S3 and S4. Significant selection bias was observed, whereas control for confounders was restricted due to limited number of participants. Bias in classification of intervention, and bias due to deviations from intended interventions was low. However bias due to missing data and information/reporting biases were moderate.
Overall analysis
Mean relative changes of fibrinogen levels from baseline to a timeframe of 2–12 h, and to 24 h post IVT with alteplase or tenecteplase are summarized in Figures 1(a), 1(b), 2(a) and 2(b), respectively. In brief, the relative reduction in fibrinogen levels from baseline to 2–12 h after the end of IVT (RoM: 9.84; 95%CI: 9.63-10.05; p < 0.001) was significantly higher in patients receiving alteplase compared to those receiving tenecteplase (Figure 1(c)). However, the mean relative reduction in fibrinogen levels from baseline to 24 h post IVT did not differ between the two groups (RoM: 3.52; 95% CI: 0.00–8877.52; p = 0.289; Figure 2(c)).
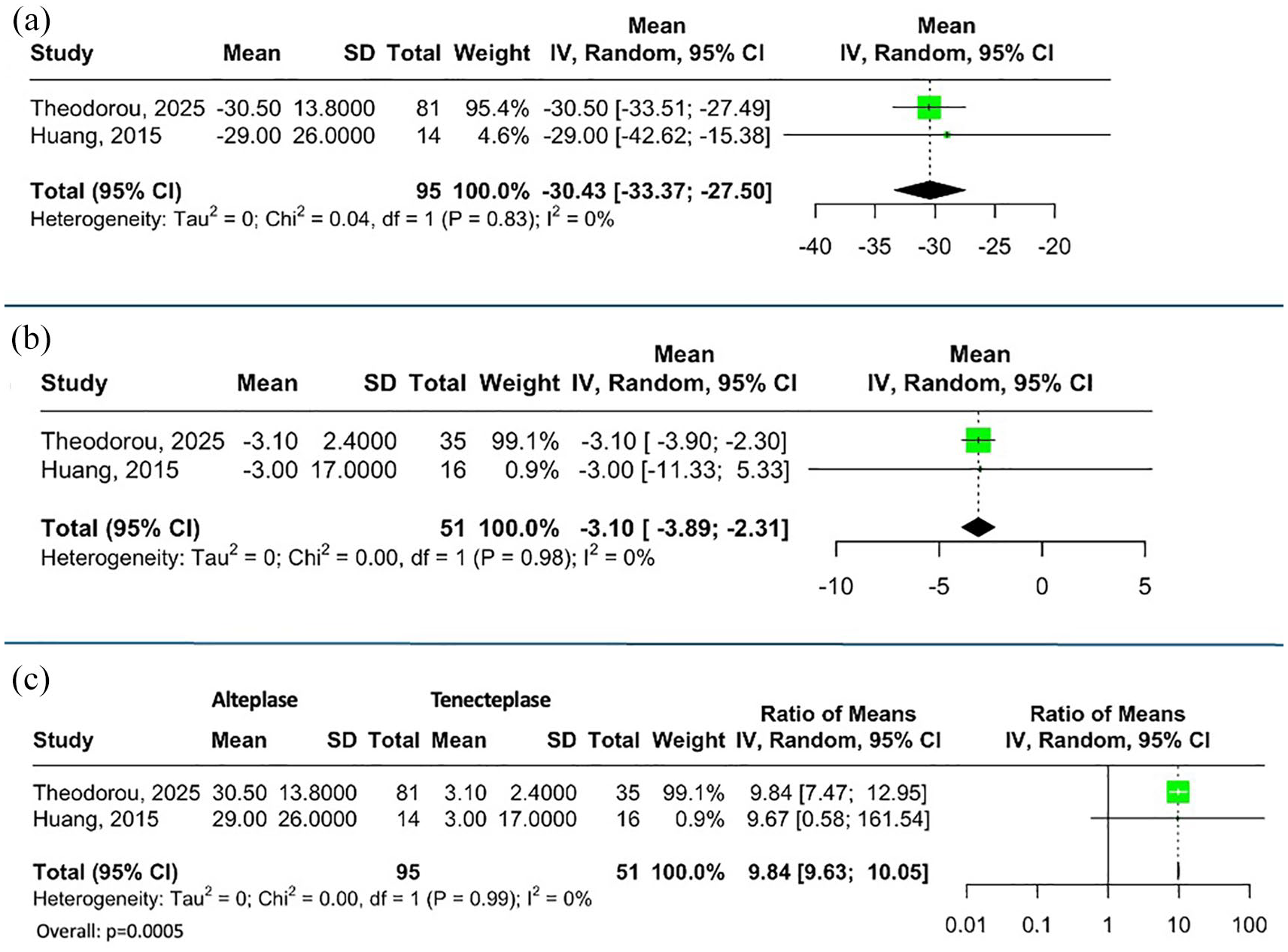
Relative Fibrinogen levels decrease at a timeframe of 2–12 h after the end of (a) alteplase and (b) tenecteplase administration. Ratio for relative fibrinogen levels change at a timeframe of 2–12 h after the end of alteplase to tenecteplase administration (c).
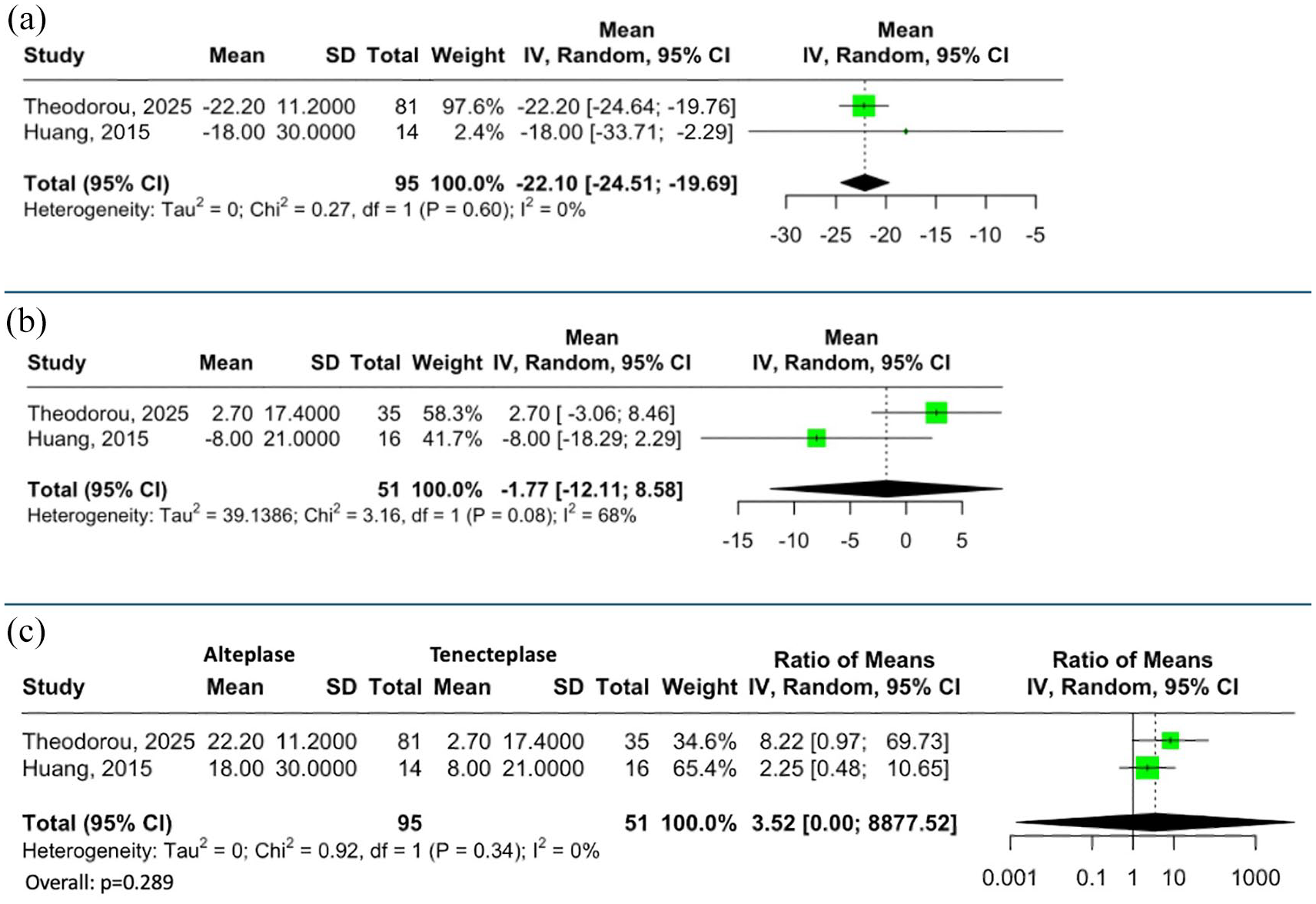
Relative Fibrinogen levels decrease at 24 h after the end of (a) alteplase and (b) tenecteplase administration. Ratio for relative fibrinogen levels change at 24 h after the end of alteplase to tenecteplase administration (c).
Discussion
In this prospective cohort study, we sought to evaluate fibrinogen level changes prior and at predefined timepoints post IVT among AIS patients treated with alteplase or tenecteplase and to assess the effect of these changes on prespecified safety and efficacy outcomes. Our study showed that alteplase administration resulted in significantly higher rates of fibrinogen depletion, with an associated greater absolute and relative mean decrease in fibrinogen levels compared to tenecteplase. To the best of our knowledge this is the largest observational cohort study, evaluating the effect of IVT with alteplase versus tenecteplase on fibrinogen depletion. Although the rates of hemorrhagic complications were comparable between the two groups, sICH and major extracranial bleeding events were observed predominantly in AIS patients treated with alteplase and were associated with fibrinogen depletion. In addition, there were numerically higher rates of major hemorrhagic complications among patients with fibrinogen depletion. The results of our systematic review and meta-analysis confirmed the significant decrease of fibrinogen levels following alteplase administration compared to tenecteplase.
Tenecteplase has previously shown significantly lower rates of fibrinogen degradation, due to the higher clot fibrin affinity compared with alteplase. 20 This has been also highlighted in a substudy, of the Alteplase–Tenecteplase Trial Evaluation for Stroke Thrombolysis (ATTEST) study, published in 2015 by Huang et al., aiming to compare the effect of alteplase versus tenecteplase on coagulation system and specifically on fibrinolysis, and to assess possible associations with ICH. 19 This study showed that in the acute ischemic stroke setting, tenecteplase caused significantly less disruption to the coagulation and fibrinolytic system compared to alteplase. This observation was also confirmed in our systematic review and meta-analysis, showing a statistically significant relative fibrinogen levels decrease in a timeframe of 2–12 h following IVT and a marginally higher decrease of relative fibrinogen levels from baseline, between patients receiving alteplase versus tenecteplase. Notably, in our observational cohort study, fibrinogen depletion was only reported in the subgroup of alteplase. More specifically, the rate of fibrinogen depletion in our study was estimated at 38%, which was quite similar to the rate (32%) reported in an Italian study but higher than the rate (20%) documented in an Austrian study.6,21 These disparities could be attributed to the different study populations and settings.
The effect of tenecteplase on fibrinogen, due to its higher affinity for clot fibrin, further supports the growing evidence related to the multiple advantages of tenecteplase compared to alteplase.1,2 Beyond the emerging data with regard to the effectiveness and safety of tenecteplase, and the practical advantages of its bolus administration, the lower rates of fibrinogen depletion following IVT with tenecteplase, further underscores the safety of this thrombolytic agent. 19 Moreover, the documented maintenance of fibrinogen levels following tenecteplase administration could be used in clinical practice as an independent risk factor, identifying patients with lower risk of developing major bleedings and could be associated with reduced need for fibrinogen replacement therapy in patients, experiencing hemorrhagic complications post-IVT. This represents a notable consideration in patient monitoring and therapeutic management.
In addition, the results of this prospective cohort study and aggregate data meta-analysis are in accordance with previous studies, showing an increased risk of sICH and major extracranial bleeding following IVT with alteplase among AIS patients, manifesting fibrinogen depletion.6,7,21–23 However, the vast majority of these studies assessed only the effect of IVT with alteplase on fibrinolysis among AIS patients. The comparative effect of alteplase versus tenecteplase on fibrinolysis was evaluated only in the substudy by ATTEST trial, by Huang et al. 19
Remarkably, the largest to date real-world study documented a lower sICH rate with tenecteplase versus alteplase among AIS patients receiving IVT. 24 However, a more recent systematic review and meta-analysis of randomized controlled trials (RCTs) reported comparable rates of sICH between patients treated with alteplase versus tenecteplase. 25 Nevertheless, heterogeneity in reported rates among studies is anticipated and may arise from heterogenous study populations, and differences in inclusion criteria and methodologies.
There are several limitations to our study, including first the small sample size of our cohort and the very limited number of available studies comparing the effect of alteplase versus tenecteplase on fibrinolysis in AIS patients. Moreover, the discrepancy between the number of participants receiving alteplase versus tenecteplase in the prospective cohort study should be acknowledged. Tenecteplase was offered only in patients with large vessel occlusion and after obtaining written informed consent (off-label use).3,26 Additional several risk factors, including arterial hypertension or blood pressure variability, abnormal renal function, coagulopathies, etc, could also contribute to an increased risk of bleeding complications following IVT among AIS patients. These implications could be further assessed in future larger international prospective cohort studies.
In conclusion, the findings of the present cohort study, coupled with the results of a systematic review and meta-analysis, highlight the potential detrimental effect of alteplase compared to tenecteplase on fibrinolysis, which is associated with higher rates of fibrinogen depletion and numerically higher hemorrhagic complications.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251345387 – Supplemental material for Fibrinogen time course in acute ischemic stroke patients treated with intravenous thrombolysis with alteplase or tenecteplase
Supplemental material, sj-docx-1-eso-10.1177_23969873251345387 for Fibrinogen time course in acute ischemic stroke patients treated with intravenous thrombolysis with alteplase or tenecteplase by Aikaterini Theodorou, Klearchos Psychogios, Odysseas Kargiotis, Apostolos Safouris, Maria Chondrogianni, Eleni Bakola, Konstantinos Melanis, Stella Fanouraki, Frantzeska Frantzeskaki, Eftihia Polyzogopoulou, Georgios Magoufis, Marios Themistocleous, Lina Palaiodimou, Maria-Ioanna Stefanou, Guillaume Turc, Andrei V Alexandrov and Georgios Tsivgoulis in European Stroke Journal
Footnotes
Acknowledgements
None
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
The study was approved by the local Ethics Committees of Attikon University Hospital (decision number: ΕΒD526/12-9-2022) and Metropolitan Hospital (decision number: 3423/30-09-2022). All participants provided signed informed consent.
Ethical approval
The systematic review and meta-analysis did not require an ethical board approval or written informed consent by the patients.
Guarantor
GT.
Contributorship
Dr Theodorou participated in study concept and design, acquisition of data, analysis and interpretation, writing original draft. Drs Psychogios, Kargiotis and Safouris participated in acquisition of data, critical revision of the article for important intellectual content. Drs Chondrogianni, Bakola, Melanis, Fanouraki, and Frantzeskaki participated in acquisition of data, critical revision of the article for important intellectual content. Drs Polyzogopoulou, Magoufis, Themistocleous, Palaiodimou, and Stefanou participated in acquisition of data, critical revision of the article for important intellectual content. Drs Turc and Alexandrov were responsible for the study design, analysis and interpretation, and performed critical revision of the article for important intellectual content. Dr. Tsivgoulis was responsible for study concept, acquisition of data, analysis and interpretation, critical revision of the article for important intellectual content.
Data availability
All data needed to evaluate the conclusions in the paper are present in the main manuscript and in the supplemental material. Additional data related to this paper may be requested from the corresponding author, upon reasonable request.
Group authorship
No. There is no study group involved in our research.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
