Abstract
Background
Covert brain infarcts are associated with important neurological morbidity. Their incidence in patients with embolic stroke of undetermined source (ESUS) is unknown.
Aims
To assess the incidence of covert brain infarcts and cerebral microbleeds using MRI in a prospective substudy of the NAVIGATE ESUS randomized trial and to evaluate the effects of antithrombotic therapies.
Methods
At 87 sites in 15 countries, substudy participants were randomly assigned to receive rivaroxaban 15 mg daily or aspirin 100 mg daily and underwent brain MRI near randomization and after study termination. The primary outcome was incident brain infarct (clinical ischemic stroke or covert brain infarct). Brain infarcts and microbleeds were ascertained centrally by readers unaware of treatment. Treatment effects were estimated using logistic regression.
Results
Among the 718 substudy participants with interpretable, paired MRIs, the mean age was 67 years and 61% were men with a median of 52 days between the qualifying ischemic stroke and randomization and a median of seven days between randomization and baseline MRI. During the median (IQR) 11 (12) month interval between scans, clinical ischemic strokes occurred in 27 (4%) participants, while 60 (9%) of the remaining participants had an incident covert brain infarct detected by MRI. Assignment to rivaroxaban was not associated with reduction in the incidence of brain infarct (OR 0.77, 95% CI 0.49, 1.2) or of covert brain infarct among those without clinical stroke (OR 0.85, 95% CI 0.50, 1.4). New microbleeds were observed in 7% and did not differ among those assigned rivaroxaban vs. aspirin (HR 0.95, 95% CI 0.52–1.7).
Conclusions
Incident covert brain infarcts occurred in twice as many ESUS patients as a clinical ischemic stroke. Treatment with rivaroxaban compared with aspirin did not significantly reduce the incidence of covert brain infarcts or increase the incidence of microbleeds, but the confidence intervals for treatment effects were wide.
Registration: https://www.clinicaltrials.gov. Unique identifier: NCT 02313909
Keywords
Introduction
Covert brain infarcts (CBI) detected by magnetic resonance imaging (MRI) are frequent in the elderly and are relevant to brain health, associated with impairment of cognition and motor function and with dementia.1,2 Interventions proven to reduce CBI are urgently needed, but none has been convincingly demonstrated to be efficacious by randomized clinical trials.
Embolic stroke of undetermined source (ESUS) is a subset of cryptogenic strokes with a presumed embolic mechanism and consists of non-lacunar ischemic stroke with no major cardiac source of embolism and no relevant large artery stenosis. 3 In the NAVIGATE ESUS MRI Substudy, incident CBI determined by repeat MRI was assessed in a subset of participants in an international, double-blind randomized trial comparing rivaroxaban, a selective factor Xa inhibitor anticoagulant, with aspirin for secondary stroke prevention in ESUS patients. We report the relative effects of these antithrombotic therapies on recurrent brain infarction (i.e. clinical recurrent ischemic stroke and incident CBI), on CBI alone in patients without clinical recurrent ischemic stroke, and on incident cerebral microbleeds.
Hypotheses
We hypothesized that recurrent brain infarction and incident CBI would be reduced by treatment with rivaroxaban compared with aspirin in patients with recent ESUS.
Methods
The New Approach riVaroxaban Inhibition of Factor Xa in a Global trial vs. ASA to prevent Embolism in Embolic Stroke of Undetermined Source (NAVIGATE ESUS) trial was an international, double-blinded, randomized phase III trial conducted at 459 centers in 31 countries that enrolled 7213 participants comparing rivaroxaban 15 mg once daily to aspirin 100 mg once daily for secondary stroke prevention in patients with recent ESUS.4,5 The trial design, participant features, and main results have been previously reported.4,5 In brief, patients with recent (three days to six months) ischemic stroke visualized by either computed tomography or MRI imaging were eligible if they met criteria for ESUS as proposed by the Cryptogenic Stroke/ESUS International Working Group, 3 but without the requirement for intracranial arterial imaging. 4 Participants with contraindications to brain MRI as determined by local standards did not participate in the MRI substudy. The trial was stopped early at an interim analysis after a median participant follow-up of 11 months because of lack of efficacy coupled with an increase in bleeding associated with assignment to rivaroxaban. 5
For the prospective MRI Substudy, the primary objective was to determine the effect of rivaroxaban compared with aspirin on MRI-defined incident CBI and clinical recurrent ischemic stroke (i.e. all incident brain infarcts) in individuals with a recent ESUS. Secondary objectives were to assess the effects of rivaroxaban compared with aspirin on incident CBI alone in participants without a clinical recurrent stroke and on incident cerebral microbleeds. The target sample size of 1000 patients would allow detection of a 35% reduction in CBI among those assigned to rivaroxaban with 80% power (two-sided alpha 0.05) assuming an annual CBI rate of 10% among aspirin-assigned patients and a mean follow-up of 24 months. Participation in the substudy required signed informed consent, and the substudy was approved by the local ethics boards at all participating sites.
Characteristics of substudy participants
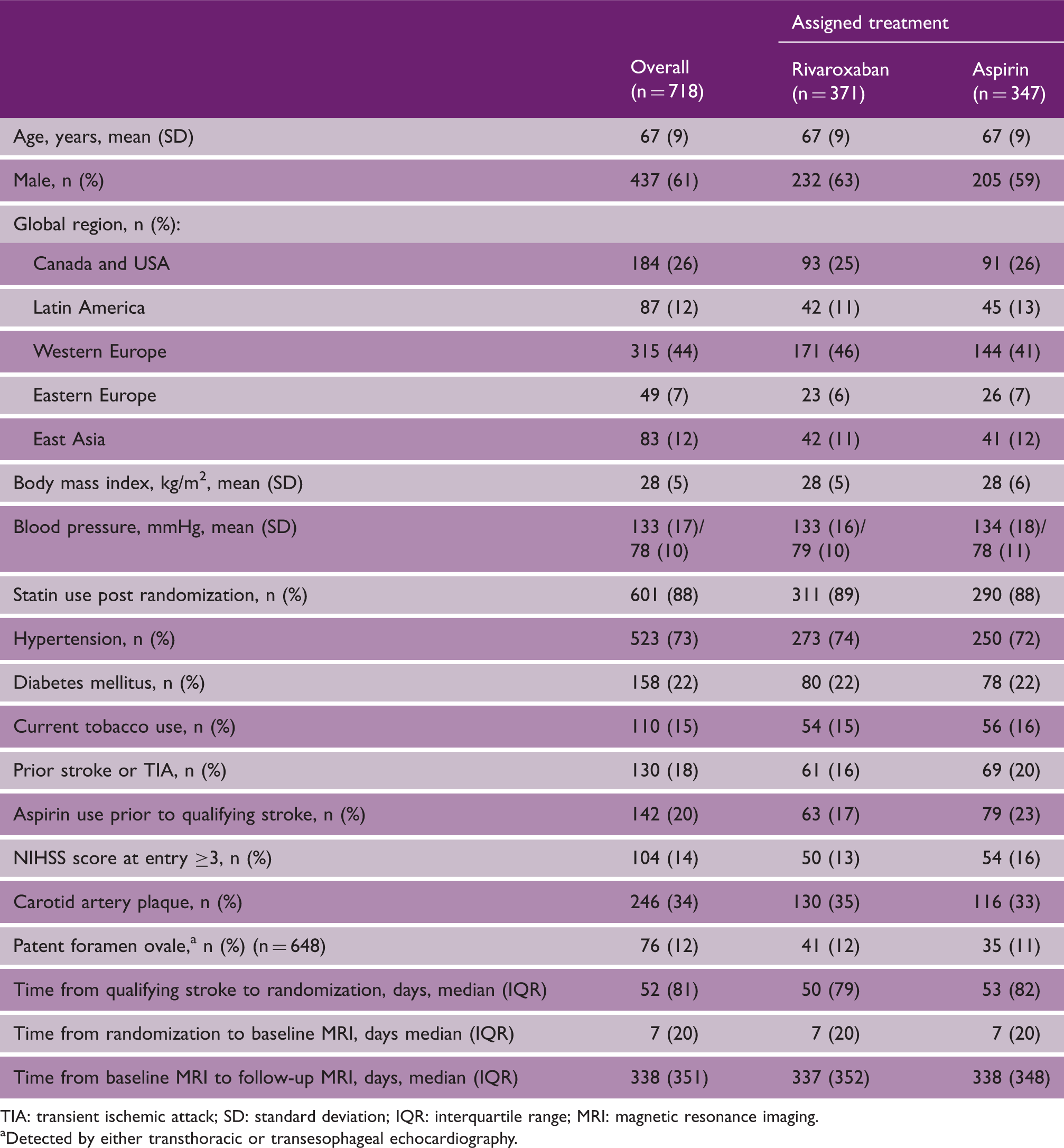
TIA: transient ischemic attack; SD: standard deviation; IQR: interquartile range; MRI: magnetic resonance imaging.
Detected by either transthoracic or transesophageal echocardiography.
Incident CBI was defined as a new brain infarct visible on MRI that did not occur in a patient with recurrent clinical stroke. MRI definition of brain infarction was based on the Standards for Reporting Vascular Changes on Neuroimaging consensus criteria. 6 An incident infarct was defined as a new lesion on the follow-up MRI which was hyperintense on T2-weighted or FLAIR images, hypointense on T1-weighted images, or if positive on diffusion-weighted images. If subcortical, the lesion had to be at least 3 mm in size. To increase precision, incident infarct(s) were identified by simultaneous comparison of the baseline with the follow-up scan. Cerebral microbleeds were defined as hypointense lesions on T2* images that were ≤10 mm in diameter. A single radiologist interpreted all scans; the reader was blinded to clinical characteristics and to treatment assignment, but not to the sequences of images (i.e. baseline and follow-up). Topographically, MRI-detected infarcts were divided into two groups: small subcortical infarcts were defined as entirely subcortical and ≤15 mm axial diameter on FLAIR sequences or ≤20 mm axial diameter on diffusion-weighted imaging; large and/or cortical infarcts were either larger and subcortical or were of any size and involving the cortex.
The primary analysis assessed the effect of rivaroxaban compared with aspirin on the outcome of clinical recurrent ischemic stroke plus MRI-defined incident CBI (i.e. recurrent brain infarction). Clinical recurrent ischemic stroke was defined and adjudicated as previously reported. 5 Secondary analyses included assessing treatment effects on all incident CBI excluding patients with clinical recurrent strokes and on incident cerebral microbleeds.
Analyses were based on the intent-to-treat population that had readable study MRIs at baseline and at follow-up. Patient characteristics were described using proportions for discrete variables and means with standard deviations (SD) or medians with interquartile ranges (IQR) for continuous variables. The odds ratio (OR) and 95% confidence interval (CI) from a logistic regression model were computed to describe odds of brain infarct associated with each characteristic. Continuous variables were empirically checked for “linearity” by evaluating if the changes in the beta (natural logarithm of the OR) between quartiles were similar with ORs for clinically important groups alternatively reported. Characteristics statistically different (p < 0.10) between those with vs. without incident brain infarcts by univariable analyses were included in forward stepwise multivariable analyses using to identify characteristics independently associated with brain infarct. Fit of the final multivariable model was estimated by computing the C-statistic. No adjustment was made for multiple comparisons, and all reported p-values were two-sided. Statistical analysis was done using SPSS for Windows version 25.0.0 (IBM Corp., Armonk, NY) and MedCalc Statistical Software version 19 (MedCalc Software bvba, Ostend, Belgium; https://www.medcalc.org; 2019).
Results
Between 2014 and 2017, 718 patients from 87 sites in 15 countries (listed in the Supplement) consented to participate in the substudy and had interpretable baseline and follow-up MRIs obtained within the time restrictions (Figure 1). Trial recruitment ended six weeks before the unexpected early termination of the main trial, and substudy participants who underwent initial MRI but who had fewer than three months of follow-up before study termination did not undergo a repeat MRI. Clinical features of the 718 patients were similar to those of other participants with the exception of geographic region of enrollment, i.e. a higher frequency of Canada/USA participants (26% vs. 11%), fewer Eastern Europeans (12 vs. 20%), and fewer East Asians (12% vs. 20%) (Supplement Table 2).
MRI substudy profile.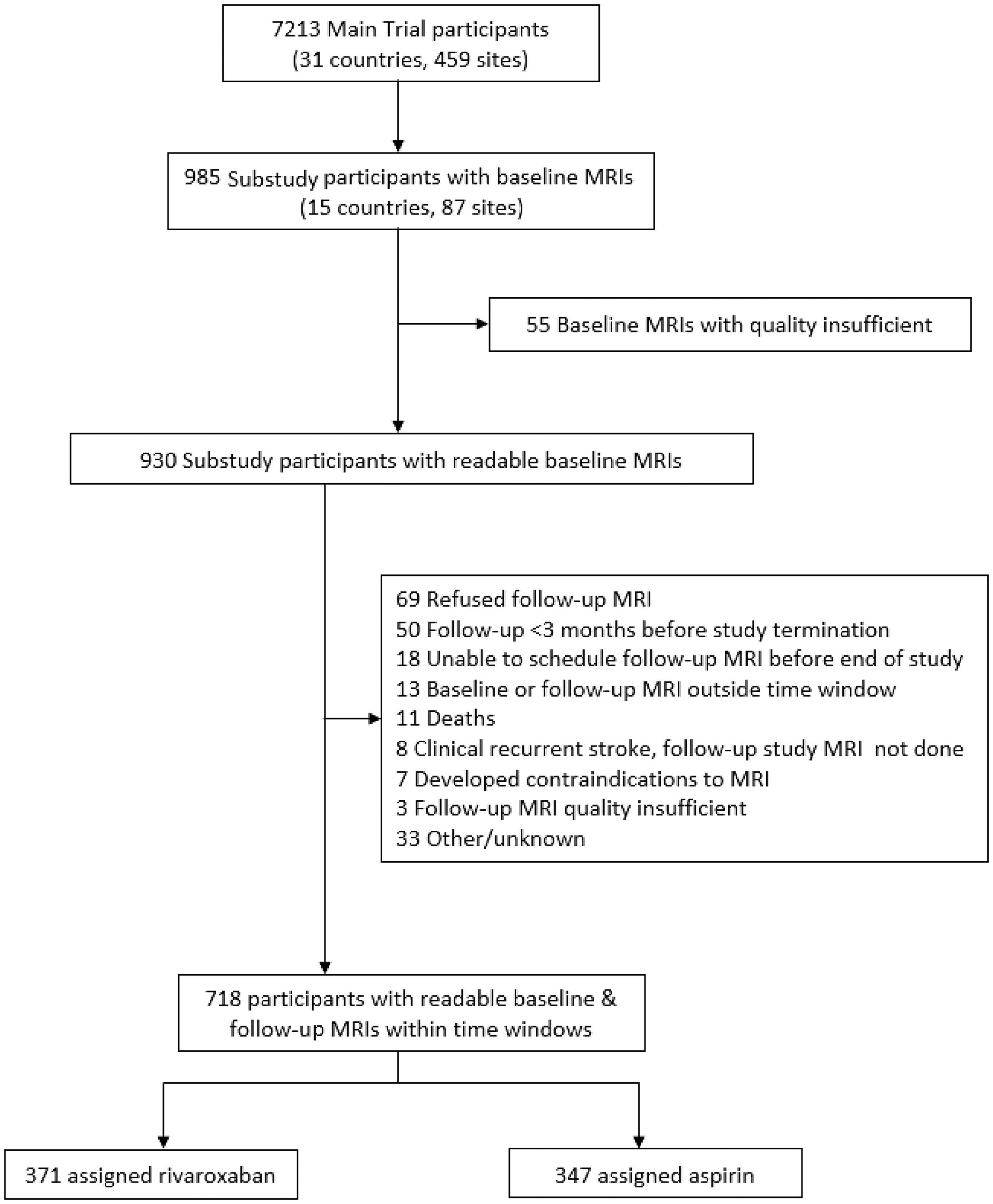
The mean age (SD) of the 718 substudy participants was 67 (9) years, and 61% were men with a median (IQR) of 52 (81) days between the index stroke and randomization and a median (IQR) of 7 (20) days between randomization and the baseline MRI. Patient features were similar for substudy participants assigned rivaroxaban vs. aspirin (Table 1).
Incident brain infarcts during follow-up and effects of treatment
During the median (IQR) 11 (12) months between MRI scans, an incident brain infarct (i.e. clinical recurrent ischemic stroke or CBI) occurred in 87 (12%) of the 718 participants: a clinical recurrent ischemic stroke occurred in 27 of the 718 (4%) participants, and CBI without recurrent clinical stroke occurred in 60 of the remaining 691 (9%). Of the 27 patients with a clinical recurrent ischemic stroke, 17 (63%) had a new brain infarct on the follow-up substudy MRI; nine of the 10 without a new brain infarct on the follow-up substudy MRI were reported to have evidence of acute brain infarction on a clinically-obtained MRI done at the time of recurrent stroke. The median (IQR) interval between the clinical recurrent stroke and follow-up substudy MRI was 7 (20) days for the 17 with a new brain infarct and 63 (439) days for the 10 without visualization of a new brain infarct on the substudy follow-up MRI (p = 0.01, Mann Whitney U test). In addition, one substudy participant had a clinical intraparenchymal hemorrhage.
Fewer patients assigned rivaroxaban (40/371 = 11%) vs. aspirin (47/347 = 14%) had an incident brain infarct (i.e. recurrent ischemic stroke or CBI) (OR 0.77; 95% CI 0.49, 1.2), but this difference was not statistically significant (Figure 2). Among the 77 patients with new brain infarcts visualized on the follow-up study MRI, hemorrhagic transformation was observed in 20% (7/35) of patients assigned rivaroxaban vs. 25% (10/40) of those assigned aspirin (OR 0.75 (95% CI 0.25, 2.2) (in two, it could not be determined due to unavailability of the required sequences); all hemorrhagic infarcts were petechial.
Odds Ratios of Outcomes.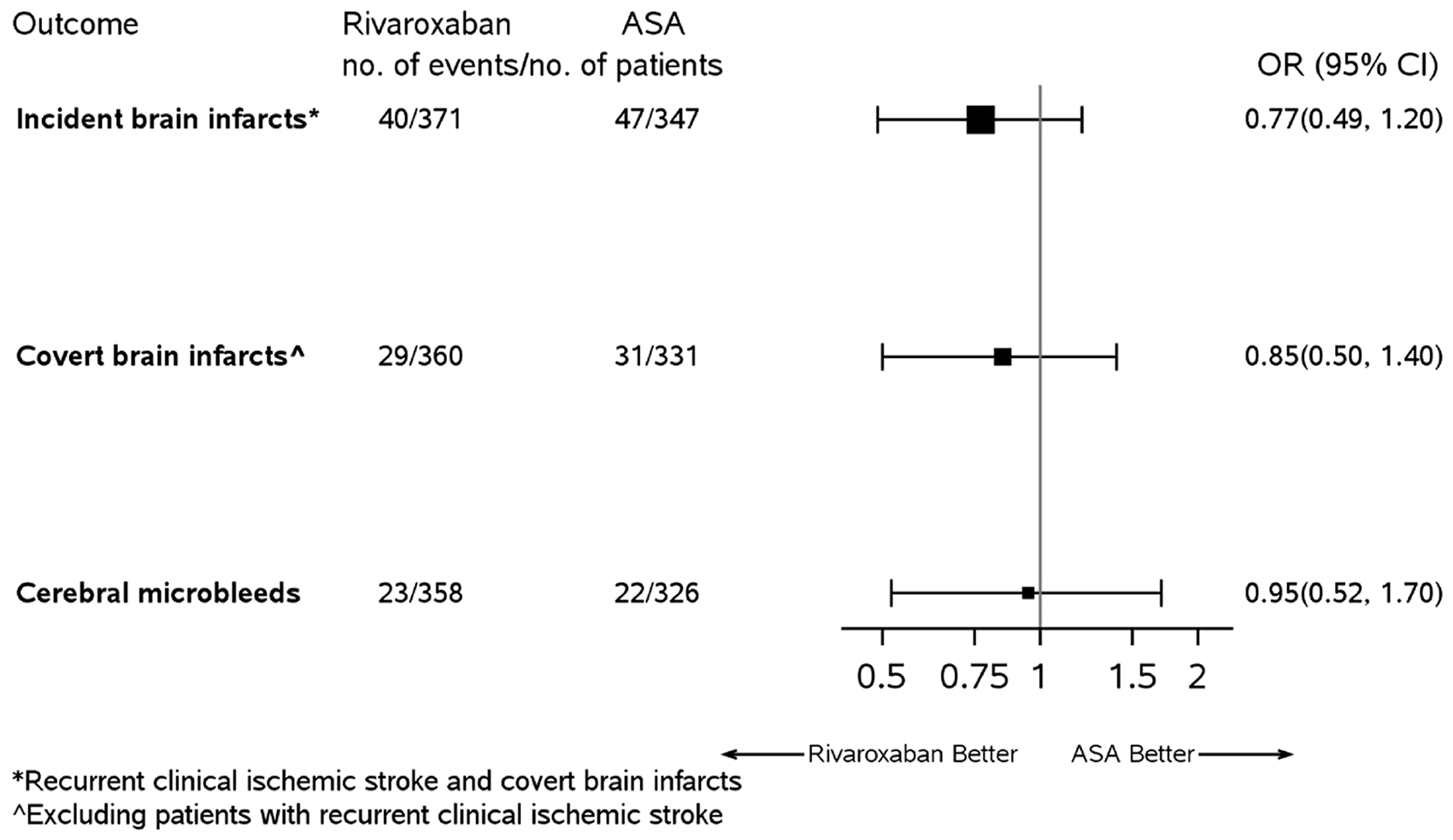
The 87 patients with an incident brain infarct were more frequently male (72% vs. 59%) and with coronary artery disease (13% vs. 6%), a history of cancer (17% vs. 10%), an NIHSS score ≥3 at entry (23% vs. 13%), and not aged 60–74 years compared with other substudy patients (Supplement Table 3). Each of these characteristics except for coronary artery disease was independently associated with incident brain infarct (Supplement Table 4). When the 27 patients with incident clinical recurrent stroke were excluded, results for the model were similar for CBI (Supplement Table 4).
Excluding the 27 patients with a clinical recurrent ischemic stroke, the proportion of participants with incident CBI was 8% (29/360) if assigned rivaroxaban vs. 9% (31/331) if assigned aspirin (OR 0.85, 95% CI 0.50, 1.4) during the median of 11 months between MRIs (Figure 2). Of these 60 patients with an incident CBI, 39 (65%) had a single incident CBI, 10 (17%) had two (7/10 in the same vascular territory), and 11 (18%) had 3–6. Including patients with multiple incident CBI, hemispheric cortical CBI occurred in 26 patients (43%), hemispheric subcortical in 39 (65%), and brainstem and/or cerebellum in seven (12%). Radiographically, 34 (57%) patients (16 assigned rivaroxaban, 18 assigned aspirin) had one or more large and/or cortical incident CBI.
Cerebral microbleeds
New cerebral microbleeds were observed in 45 (7%) of 684 substudy participants during follow-up, equally among those assigned to rivaroxaban (23/358) vs. aspirin (22/326) (OR 0.95, 95%CI 0.52, 1.7) (Figure 2). Two-thirds (30/45) of patients with a new cerebral microbleed had a single new microbleed, 16% (7/45) had two, and the remaining 18% (8/45) had 3–7. Including patients with multiple new microbleeds, 29 patients had cortical, 21 subcortical, one brainstem, and six cerebellar.
Discussion
In addition to patients experiencing recurrent clinical ischemic stroke, twice as many patients developed incident CBI during follow-up of the substudy cohort, indicating a substantial burden of clinically undiagnosed ischemic brain injury in patients with ESUS. The observed rate of incident CBI of 9% per year may have been higher if patients underwent baseline study MRI sooner after the index stroke in order to capture the early post-stroke high-risk period for recurrent brain ischemia (the median interval between qualifying stroke and baseline MRI was 62 days). The absence of significant reductions in all incident brain infarct and in CBI alone by rivaroxaban over aspirin in the substudy cohort is consistent with the neutral treatment effect on clinical recurrent ischemic stroke in the much larger main trial (hazard ratio 1.01, 95% CI 0.81, 1.26 comparing rivaroxaban vs. aspirin). 5 The treatment effects, or lack thereof, were consistent whether considering CBI or clinical stroke. Randomization to rivaroxaban, an oral anticoagulant factor Xa inhibitor, did not increase the incidence of new cerebral microbleeds over treatment with aspirin. Similarly, we did not observe an increase in the occurrence of hemorrhagic transformation of incident infarcts among those assigned rivaroxaban, but numbers were small and the resulting confidence interval was wide.
Our observations are consistent with other recent multicenter randomized trials, demonstrating that the rate of incident CBI exceeds that of clinical ischemic strokes.7–9 The effects of antithrombotic therapies on incident CBI paralleled the treatment effects on recurrent clinical ischemic stroke in our study and in the COMPASS trial. 7 Consequently, incorporating CBI into the efficacy outcome of future randomized trials aimed at prevention of brain infarction (in contrast to restricting to clinical recurrent ischemic strokes) could potentially reduce by half or more the trial sample sizes. CBI are not benign, representing brain infarction that goes unrecognized mainly because of location and size.
Small CBI may not result in cavitated lesions or persisting focal white matter abnormalities and hence be undetected by a single repeat MRI done months later,10,11 Of note, 37% (10/27) of clinical recurrent ischemic strokes in this cohort did not have an incident brain infarct detected on the follow-up substudy MRI (interpreted without awareness of the interval clinical stroke), although nine of these 10 had brain infarction reported on clinical MRIs done acutely. The likelihood of detection of the clinical recurrent infarct on the follow-up study MRI was related to the time from the stroke to the follow-up MRI, as expected. In contrast to the subcortical location of CBI from population-based series, 2 in most substudy patients (57%), the incident CBI were large and/or cortical (i.e. potentially due to recurrent ESUS). A predominantly cortical location of incident CBI was reported in patients with systemic atherosclerosis participating in the COMPASS trial. 7 Taken together, these trials suggest that the size and location of CBI in patients with ESUS or with manifest systemic atherosclerosis differs from that in population-based cohorts and is more likely to be cortical in location. This distribution has implications for the pattern and burden of cognitive and functional deficits associated with accumulating CBI in these populations.
Those assigned rivaroxaban were no more likely to develop incident cerebral microbleeds than those assigned aspirin. Cerebral microbleeds are microscopic deposits of hemosiderin most commonly related to small vessel disease from hypertension or cerebral amyloid angiopathy. These lesions are common in cohorts of community-dwelling elderly patients with prevalences ranging from 6 to 25% and are associated with incident ischemic stroke, fatal brain hemorrhage, and dementia. Our findings in patients with a recent ESUS are similar to that observed in patients with atrial fibrillation and support a lack of influence of oral factor Xa inhibitors on incident cerebral microbleeds compared with aspirin. 12
Limitations of this study include that only 10% of NAVIGATE ESUS trial participants were enrolled in the substudy. However, participants appear generally representative of the overall trial cohort (Supplement Table 2). Further, this clinical trial-derived cohort may not be representative of ESUS patients in the general population. As well, the confidence intervals surrounding the point estimates of treatment effects were relatively wide, in part due to the early stopping of the main trial at an interim analysis.
Conclusions
The NAVIGATE ESUS MRI Substudy demonstrates the feasibility of assessment of incident CBI and cerebral microbleeds in an international trial of secondary stroke prevention. CBI were more than twice as frequent as clinical recurrent ischemic strokes. The concordance between treatment effects on recurrent clinical stroke and incident CBI in studies to date supports the potential incorporation of CBI as a trial outcome of future randomized trials aimed at the prevention of brain infarction.
Supplemental Material
sj-pdf-1-wso-10.1177_17474930211058012 - Supplemental material for Rivaroxaban versus aspirin for prevention of covert brain infarcts in patients with embolic stroke of undetermined source: NAVIGATE ESUS MRI substudy
Supplemental material, sj-pdf-1-wso-10.1177_17474930211058012 for Rivaroxaban versus aspirin for prevention of covert brain infarcts in patients with embolic stroke of undetermined source: NAVIGATE ESUS MRI substudy by Mukul Sharma, Eric E Smith, Lesly A Pearce, Kanjana S Perera, Scott E Kasner, Byung-Woo Yoon, Sebastian F Ameriso, Josep Puig, Dorte Damgaard, Jochen B Fiebach, Keith W Muir, Roland C Veltkamp, Roland C Veltkamp, Danilo S Toni, Nikolay Shamalov, Rubens J Gagliardi, Robert Mikulik, Stefan T Engelter, Daniel Bereczki, Martin J O’Donnell, Feryal Saad, Ashkan Shoamanesh, Scott D Berkowitz, Hardi Mundl, Robert G Hart and On behalf of the NAVIGATE ESUS MRI Substudy Investigators in International Journal of Stroke
Footnotes
Data availability
Additional aggregated data are available from the corresponding author upon request and pending the approval of the trial publications committee.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All co-authors or their institutions received payments from Bayer AG for participation in the NAVIGATE ESUS trial except for HM and SDB who are employed by Bayer AG.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The NAVIGATE ESUS trial and the MRI Substudy were sponsored by Bayer AG.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
