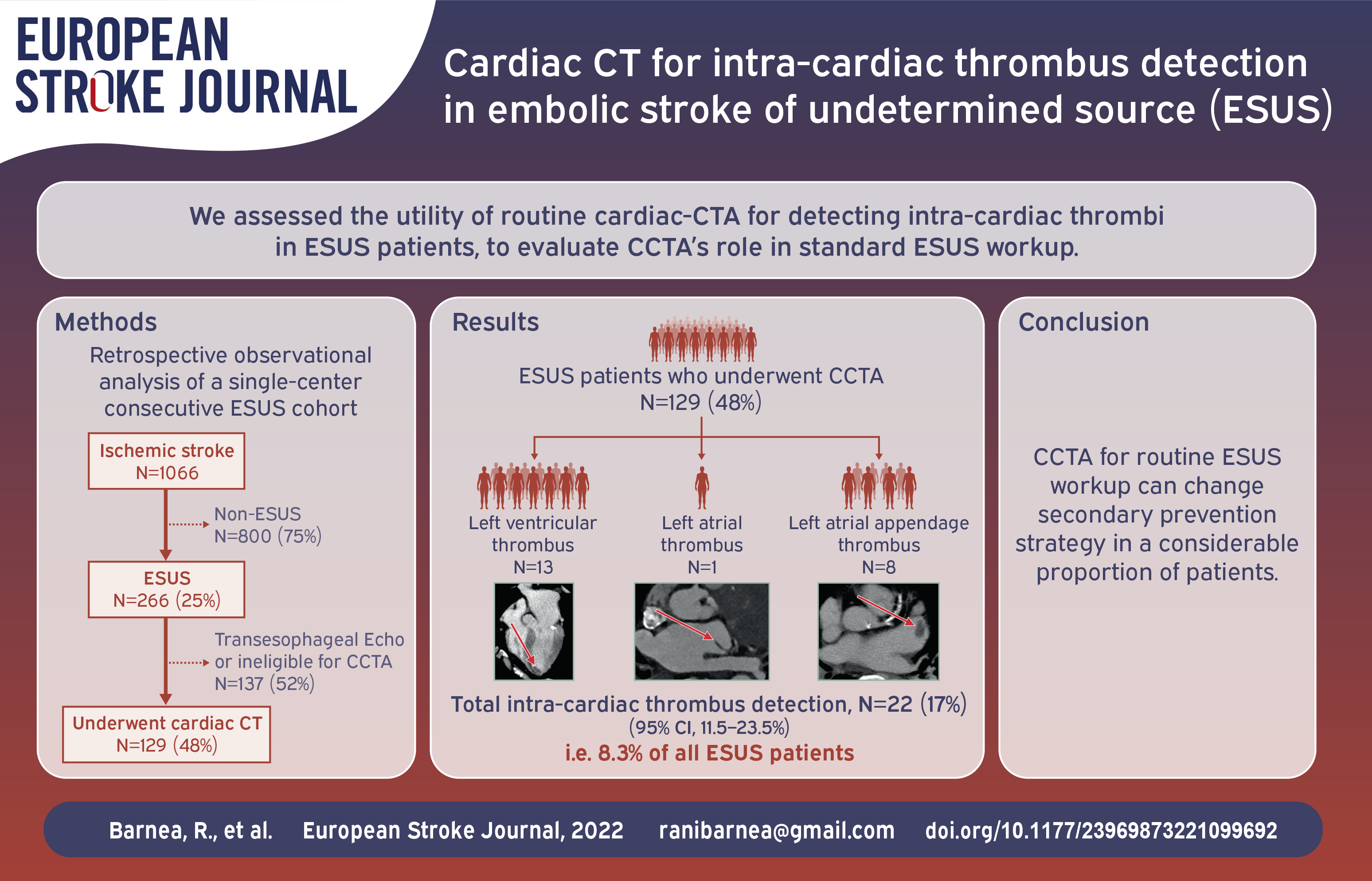
Abstract
Introduction:
Embolic stroke of undetermined source (ESUS) is a common medical challenge regarding secondary prevention strategy. Cardiac imaging is the cornerstone of embolic stroke workup, in an effort to diagnose high risk cardio-embolic sources. Cardiac computed tomography angiography (CCTA) is an emerging imaging modality with high diagnostic performance for intra-cardiac thrombus detection. The yield of CCTA implementation in addition to standard care in ESUS workup is unknown. Thus, the aim of this study was to assess the utility of CCTA in detecting intra-cardiac thrombi in the routine ESUS workup.
Patients and methods:
This is a retrospective observational analysis of ESUS cases managed in vascular neurology unit between 2019 and 2021. Within this ESUS registry, consecutive patients undergoing CCTA were included and carefully analyzed.
Results:
During the study period 1066 Ischemic stroke (IS) cases were treated and evaluated. 266/1066 (25%) met ESUS criteria and 129/266 (48%) underwent CCTA. Intra-cardiac thrombus was detected by CCTA in 22/129 (17%; 95% CI, 11.5%−23.5%) patients: left ventricular thrombus (LVT) in 13 (10.1%) patients, left atrial appendage (LAA) thrombus in 8 (6.2%) patients, and left atrial (LA) thrombus in 1 (0.8%) patient. Only 5/22 (23%) of these thrombi were suspected, but could not be confirmed, in trans-thoracic echocardiogram (TTE). Among CCTA-undergoing patients, 27/129 (21%; 95% CI, 14%−28%) were found to have an indication (including pulmonary embolism) for commencing anticoagulation (AC) treatment, rather than anti-platelets. In favor of CCTA implementation, 22/266 (8.2%; 95% CI, 4.9%−11.5%) patients within the entire ESUS cohort were diagnosed with intra-cardiac thrombus, otherwise missed.
Conclusion:
CCTA improves the detection of intra-cardiac thrombi in addition to standard care in ESUS patients. The implementation of CCTA in routine ESUS workup can change secondary prevention strategy in a considerable proportion of patients.
Introduction
Embolic stroke of undetermined source (ESUS) is an evolving stroke subtype that poses great clinical challenge for secondary prevention strategy. 1 One main aim of ESUS workup is to detect cardiac sources of embolism, such as intra-cardiac thrombi that indicates commencing anti-coagulants (AC). 2 However, according to current stroke prevention guidelines, the use of advanced cardiac imaging is not obligatory and might be reasonable (class IIb recommendation) in ESUS cases, 1 as data supporting this strategy is scarce.
While echocardiography is still the mainstay of post-stroke cardiac imaging, 3 reports on cardiac computed tomography angiography (CCTA) in ischemic stroke (IS) of presumed cardiac source, have been increasingly published in recent years, 4 and CCTA has become a promising imaging modality for more reliable detection of intra-cardiac thrombi. 5 Relying on the preliminary published experience of implementing CCTA in IS workup,6–8 our hospital’s vascular neurology unit adopted this strategy as a supplemental imaging modality in ESUS workup, since 2019. According to this diagnostic strategy, ESUS cases that are not considered for transesophageal echocardiography (TEE) as standard of care, would undergo CCTA as advanced cardiac imaging for major cardio-embolic source detection.
In the present study, we evaluated the impact of CCTA in the diagnosis of intra-cardiac thrombus, in a consecutive group of patients hospitalized with the diagnosis of ESUS. In addition, we tested the impact of CCTA findings on medical management.
Methods
Intravenous injection of 60–85 ml of nonionic contrast agent (Iopromide 370; Bayer Schering, Berlin, Germany) was administered at a flow rate of 5 ml/s and was followed by a 40-ml saline chase bolus. Data acquisition consisted of two acquisitions. The first acquisition was automatically initiated using a bolus tracking with a region of interest placed in the descending aorta and a threshold level of 180 Hounsfield units, and the second acquisition was automatically initiated 60 s after administration of the contrast agent. Acquisition was performed during an inspiratory breath-hold while the electrocardiogram was recorded simultaneously to allow retrospective gating of data.
Results
During the study period, 1066 IS patients were hospitalized to the stroke unit and/or neurology department at our center, forming our consecutive IS cohort. Figure 1 provides a graphical representation of diagnostic flow-chart of all patients diagnosed with IS during the study period. 266/1066 (25%) of IS cases (69.7 ± 15.7 years, 120 [44%] females) met ESUS criteria. CCTA was performed in 129/266 (48%) of ESUS patients (73.6 ± 9.9 years, 58 [45%] females). Most of these patients had at least one comorbidity, the most common ones being HTN (76%), DM (41%) and IHD (33%). Moreover, 41/129 (32%) patients had a history of prior stroke. The theoretical CHADS VASC score was 3.9 ± 1.9. Patients’ baseline characteristics are presented in Table 1.
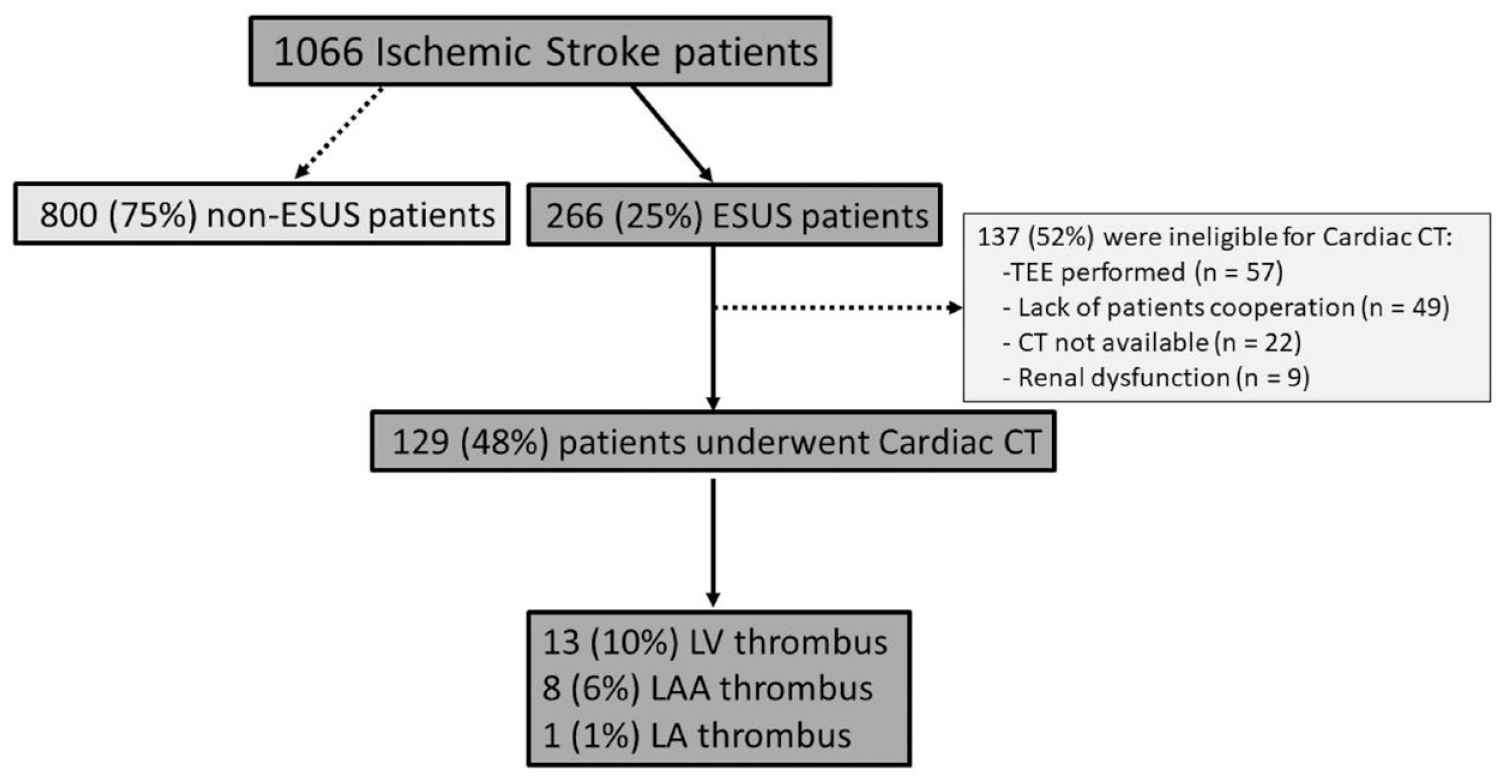
Diagnostic flow-chart of all patients diagnosed with ischemic stroke during the study period.
Patients’ characteristics.
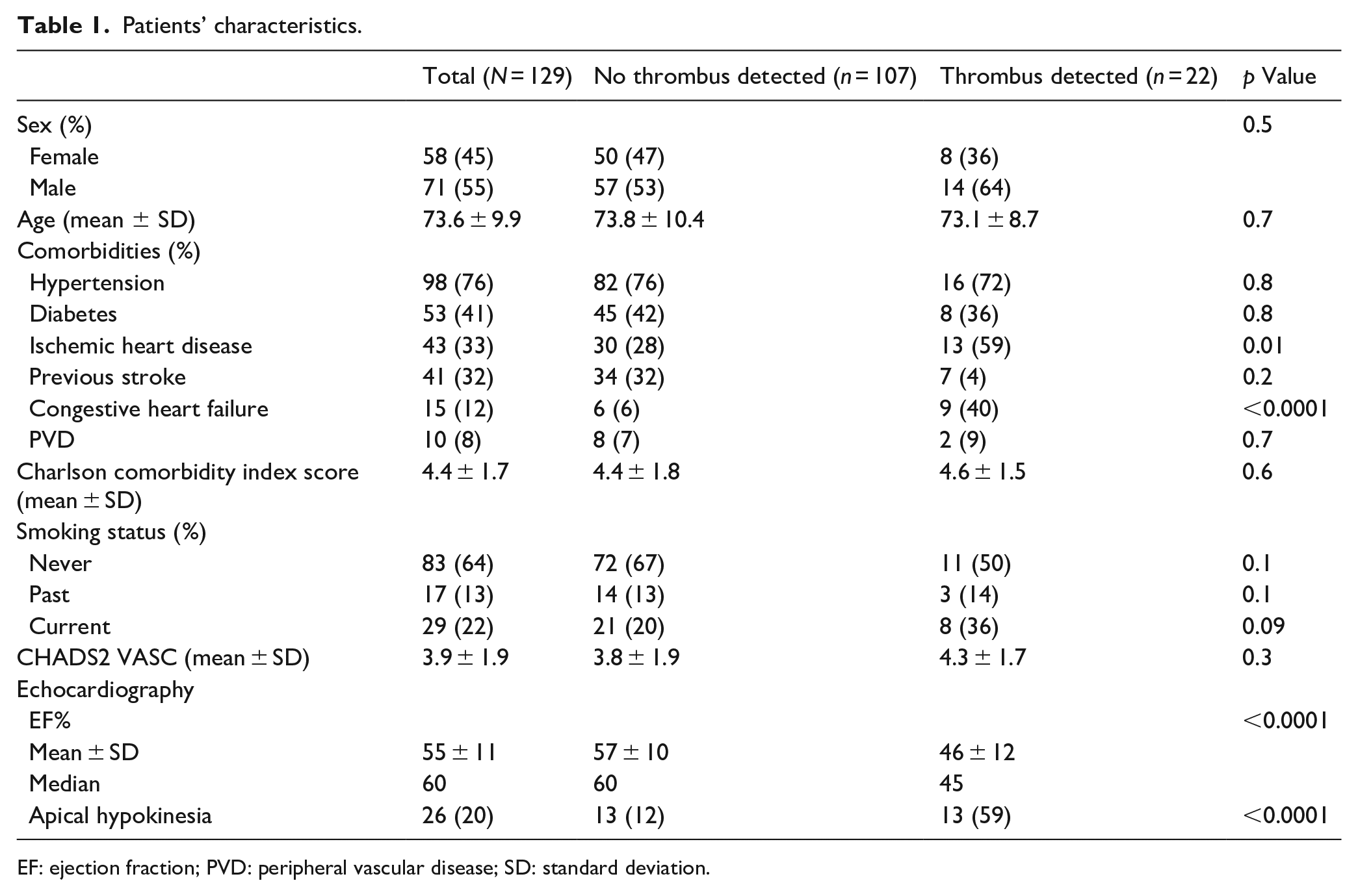
EF: ejection fraction; PVD: peripheral vascular disease; SD: standard deviation.
CCTA detected intra-cardiac thrombus in 22/129 (17%; 95% CI, 11.5%−23.5%) of ESUS patients. Left ventricular thrombus (LVT) was detected in 13 (10.1%) patients; LAA thrombus in 8 (6.2%) patients; and LA thrombus in 1 (0.8%) patient. As shown in Table 1, patients in whom intra-cardiac thrombus was detected by CCTA had higher prevalence of IHD and congestive heart failure (CHD) compared with patients in whom thrombus was not detected; this was reflected also by significantly lower ejection fraction (EF%) and higher prevalence of apical hypo-kinesia, demonstrated on TTE. Figure 2 provides selected images of intra-cardiac thrombi detected on cardiac CT scans.
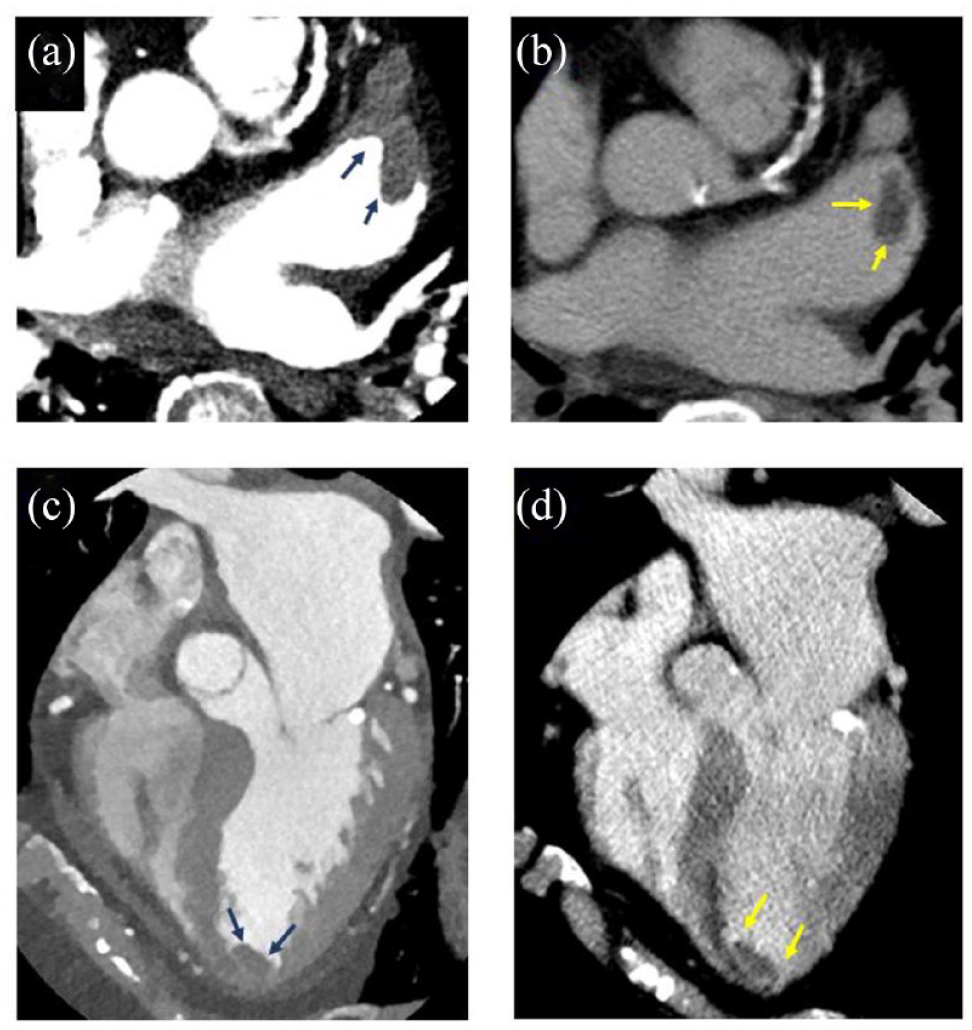
Selected images of intra-cardiac thrombi detected on cardiac CT scans. Representative left atrial appendage thrombus assessed on contrast enhanced CT images (a) and post-contrast CT scan (b). Representative left ventricular apical thrombus assessed on contrast enhanced CT images (c) and post-contrast CT scan (d).
Twenty-six (20%) patients in the ESUS-CCTA cohort had apical hypo/akinesia demonstrated on NCTTE. All the 13 LVT detected by CCTA were located in the apex with a concomitant apical wall motion abnormality demonstrated on NCCTE. NCTTE raised suspicion of LV thrombus in 5/13 (38%) of these cases, but could not confirm this diagnosis. Five of 13 (38%) of LVT were measured >20 mm on the longest plain on CCTA, 6/13 (46%) 10–20 mm, and 2/13 (15%) were <10 mm. All 13 patients with LVT had evidence of previous myocardial infarction on CCTA, however, the presence of IHD was previously unknown in two of them.
CCTA revealed pulmonary embolism (PE) in seven patients (in two cases, PE and LV thrombus were detected concurrently). Another important finding of CCTA was hyper-attenuated leaflet thickening (HALT), representing subclinical leaflet thrombosis (SLT), on aortic prosthetic valve in two patients (in one case, concurrent with LAA thrombus). Altogether, anticoagulation was found to be indicated in 27/129 (21%; 95% CI, 14%−28%) of ESUS patients undergoing CCTA, otherwise treated with anti-aggregation.
Considering the entire ESUS cohort, CCTA implementation strategy led to intra-cardiac thrombus detection in 22/266 (8.2%; 95% CI, 4.9%−11.5%) patients.
Within our ESUS registry, 137/266 (52%) patients did not undergo CCTA. As shown in Figure 1, the main reason for not performing CCTA was the preference of TEE in 57/137 (42%) of these patients. Table 2 compares baseline characteristics of this TEE group with CCTA group. TEE was implemented in 41/57 (72%) patients, who were younger than 60 years, for which patent foramen ovale (PFO) closure would be contemplated, if diagnosed. As shown in Table 2, PFO was detected in 17/57 (30%) of the TEE group versus 6/129 (5%) of the CCTA group (
Baseline characteristics of patients undergoing CCTA vs. TEE, and detection of intra-cardiac thrombus or PFO among these groups.
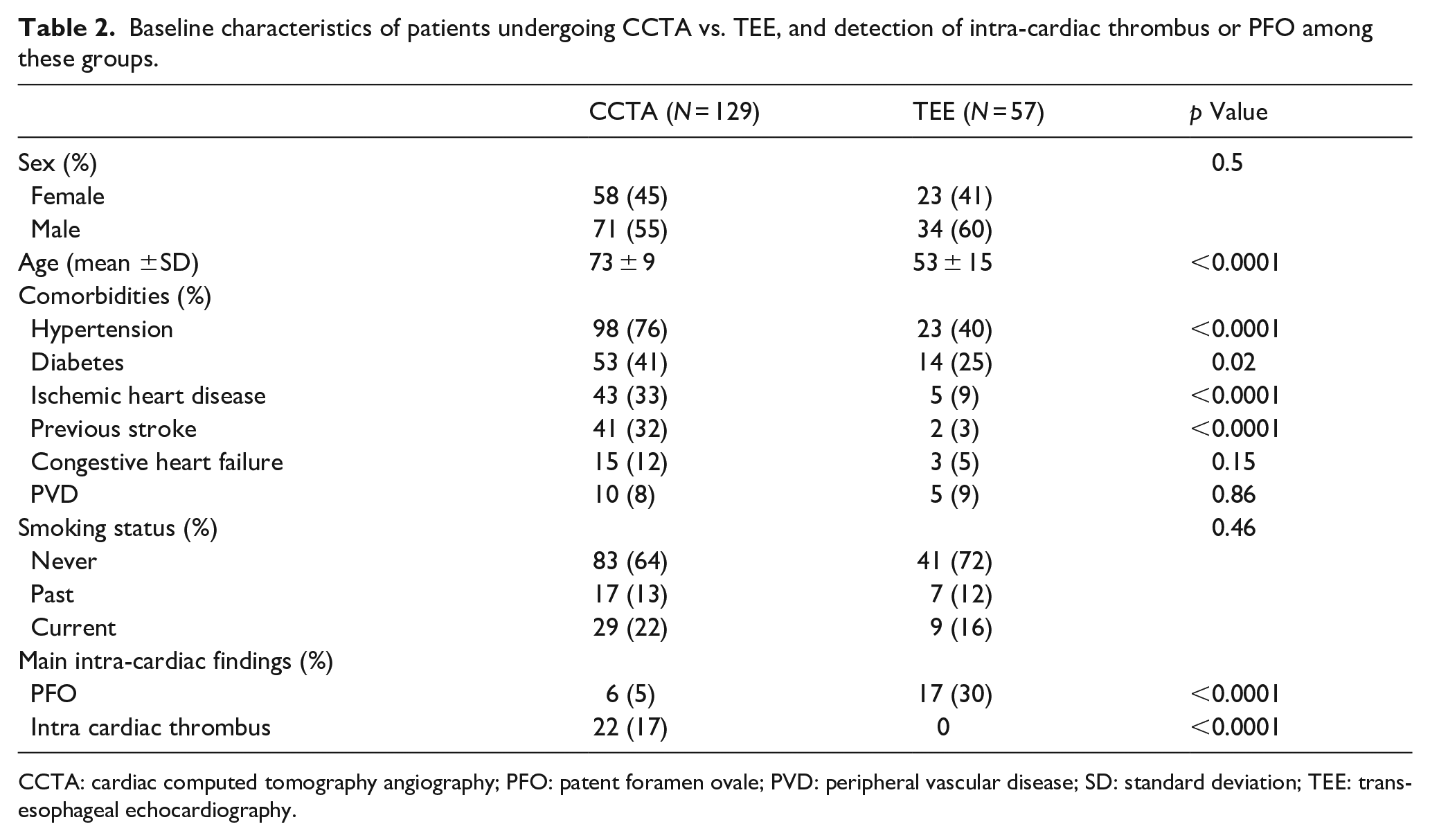
CCTA: cardiac computed tomography angiography; PFO: patent foramen ovale; PVD: peripheral vascular disease; SD: standard deviation; TEE: transesophageal echocardiography.
Of the entire ESUS cohort, 186/266 (70%) patients underwent advanced cardiac imaging (CCTA or TEE) as part of their ESUS workup.
Discussion
In this study, a consecutive cohort of patients with ESUS underwent CCTA in addition to standard of care in order to better evaluate the prevalence of intra-cardiac thrombus as a possible cardio-embolic source. The major findings of this study are as follows: (1) performing CCTA in addition to screening NCTTE improves the detection of intra-cardiac thrombi, and (2) the use of CCTA in routine ESUS workup can change secondary prevention strategy in a considerable proportion of patients.
Initial diagnostic work-up of non-lacunar stroke includes imaging of the cervical and intracranial arteries, screening NCTTE, 12 lead ECG and at least 24 h of continuous heart-rhythm monitoring. 16 After this evaluation, approximately 17% of all IS (which are approximately 22% of non-lacunar strokes) are considered as ESUS according to the accepted definition in stroke literature.1,9 Once an ESUS diagnosis is made, further investigation is considered on a case-by-case basis and should include long-term cardiac monitoring, TEE, cervical vessel wall and plaque imaging, hypercoagulable testing and malignancy screening. 17 In this setting, post stroke intra-cardiac thrombus detection, is of considerable clinical importance, as it indicates commencing AC treatment.1,2,18
NCTTE is used as the basic screening cardiac imaging for IS and is therefore a pre-requisite for ESUS definition. TEE is considered as a more advanced and sensitive modality for post stroke cardiac imaging, particularly for PFO or valvular sources of embolism, but the general yield of both modalities in stroke patients is uncertain.19–21 TTE is preferred for detecting LV thrombi, especially if contrast enhanced echocardiography is performed, 22 while LA/LAA thrombi are best detected by TEE. 4 In recent years, studies reporting the use of CCTA for IS workup, as a supplementary imaging modality for detection of intra-cardiac thrombi, have been increasingly published. 5 However, in the majority of these studies, IS patients who underwent CCTA, were those with suspected cardio-embolic source, especially known AF.23,24 The clinical considerations and rational of implementing CCTA in AF patients (for which anticoagulation is indicated regardless of CCTA findings) in these studies, was not explicitly specified, but rather expressed as a general aim to define predictors of stroke recurrence. To our knowledge, this analysis of consecutive cohort of ESUS patients, is the first to assess the widespread implementation of CCTA in routine ESUS workup. One retrospective analysis, revealed 5/74 (6.7%) intra-cardiac thrombi detection rate among those who met ESUS criteria and underwent CCTA in a Chinese cohort of re-perfused IS patients. 25 In the consecutive ESUS cohort of our study, a significantly higher rate of intra-cardiac thrombi was detected, emphasizing the importance of CCTA use in ESUS workup. The significant difference between intra-cardiac thrombi detection rates in these two cohorts may be attributed to the different prevalence of atherosclerosis, being the most common IS subtype in China.26,27
TTE is an operator dependent technique, also influenced by individual patient’s characteristics such as chest wall anatomy. LVT can be detected by TTE with relatively low sensitivity of 21%–33%, that can be increased to 61% when contrast-enhanced TTE (CTTE) is performed. 29 CTTE allows significantly improved assessment of the presence or absence of LV thrombus.22,39,40 Among LV dysfunction patients, CTTE has the greatest impact over NCTTE, for LVT detection. 41 However, in common practice, CTTE is not included in routine IS/ESUS workup,17,42 and therefore is not considered standard of care. TTE has difficulty in visualizing LV thrombi, with lowest sensitivity (10%) for thrombi smaller than 10 mm, and highest sensitivity (46%) for thrombi larger than 20mm. This relatively low sensitivity for LV thrombi detection is related to the limited near field resolution, wherein adherent mural thrombi resemble the adjacent myocardium. 43 Among the 13 LVT detected in our cohort by CCTA, NCTTE raised suspicion of LVT in 5/13 (38%) of them, but could not confirm this diagnosis. In these cases, TTE demonstrated a substantial apical a-kinetic area, but the thrombus was indistinguishable from the adjacent myocardium so a confirmed diagnosis of LVT could not be reached.
TEE is considered the least sensitive modality for LVT detection.4,20
Cardiac MRI (CMRI) is the most sensitive and specific imaging modality for LVT detection in IS patients, and thus considered as the gold standard for this purpose.4,29,43 Unfortunately, CMRI has not been routinely adopted in ESUS workup due to its current cost and availability.
CCTA demonstrates LVT as a hypodense mass within enhanced LV cavity, with significantly lower attenuation in comparison with the myocardium. CCTA diagnose LVT and distinguishes it from myocardial wall with sensitivity, specificity and positive and negative predictive values of 94%, 97%, 94%, and 97%, respectively. 44 Currently, there are few validated data on the role of cardiac CT in the detection of LVT in comparison with MRI. 29 However, in the setting of IS, CCTA may be superior to TTE regarding LV thrombi detection. 23 LVT is associated with increased risk of stroke and systemic embolism and indicates commencing anti-coagulation treatment for at least 3 months. Furthermore, embolic events appear to occur even after the resolution of LVT, suggesting that this therapy should be considered for a longer period in some cases. 45
LV thrombus detection in our ESUS cohort (10%) suggests that this diagnostic strategy is justified, as it changed the medical management in these cases. According to our findings, NCTTE is not sensitive enough and therefore insufficient for comprehensive ESUS workup. One possible conclusion from our study is that the presence of global or regional LV dysfunction in the setting of ESUS, particularly apical wall motion abnormality, may justify advanced cardiac imaging (CCTA or CMRI) or at least CTTE, in order to improve LVT detection. Further studies are needed to assess this hypothesis.
Overall, while CTTE is LV sensitive and TEE is LA/LAA sensitive, CCTA serves as a more comprehensive cardiac imaging for ESUS, addressing both diagnostic challenges at the same time.
Another finding of CCTA in our cohort was hyper-attenuated leaflet thickening (HALT), representing subclinical leaflet thrombosis (SLT),
47
on aortic prosthetic valve in two patients (in one case, concurrent with LAA thrombus). While SLT is relatively frequent after TAVR (occurring in 15%−30% of patients),
48
and its clinical significance in means of valve functionality is debatable, this pathology was shown to be associated with increased stroke risk.
49
Oral anticoagulation (OAC) treatment after SLT diagnosis is associated with significantly higher odds of SLT resolution compared with no OAC treatment (OR: 0.01; 95% CI: 0.00–0.06;
Limitations
The retrospective design of this single-center cohort study is the main limitation of this study. Accordingly, the decision of whether or not a patient would undergo advanced cardiac imaging, either CCTA or TEE, was not randomized nor blinded, and therefore introduces selection bias.
Of the entire ESUS cohort, 80/266 (30%) did not undergo advanced cardiac imaging (neither CCTA nor TEE). The main reason for not performing advanced cardiac imaging, in 49/80 (61%) of these patients, was lack of patient cooperation due to neurological or medical condition (aphasia, altered mental status, reduced consciousness, respiratory or hemodynamic instability, or general fragility). Lack of patients’ cooperation limited the possibility of implementing CCTA, as this may cause significant artifacts in the exam. Moreover, altered mental status or aphasia impaired patients’ capability to provide informed consent for TEE, as an invasive procedure. In 9/80 (11%) patients, another factor limiting the use of CCTA was the relative contra-indication of acute or chronic kidney injury or single functioning kidney. Finally, as CCTA is a valuable and sometimes scarce resource for coronary artery disease workup (cardiology, internal medicine, emergency medicine etc.), it was not available for 22/80 (28%) patients during their hospitalization in the stroke unit or vascular neurology ward, and therefore was not performed. Accordingly, only 70% of the ESUS cohort underwent advanced cardiac imaging, and this impairs the generalization of our findings and moderates their impact.
Our study is unable to perform a direct comparison between CCTA and TEE as none of the patients included in our cohort were examined by both modalities. The comparison between CCTA and TEE undergoing patients (Table 2) does not lead to any comparative conclusions, as the baseline characteristics of these two groups are distinctly and significantly different, and retrospectively reflect the workup strategy in the study period.
Since CCTA was not compared to another imaging modality for similar patient group, we could not define the incremental intra-cardiac thrombus detection rate of CCTA, but rather the net clinical impact of implementing it in ESUS workup, expressed as added AC indications and intra-cardiac thrombus detections.
Furthermore, as contrast-enhanced TTE is not routinely used in IS workup, all patients in our cohort underwent NCTTE, impairing LVT detection by echocardiography.
Conclusion
The implementation of CCTA in routine ESUS workup can change secondary prevention pharmacologic strategy in a considerable proportion of patients, by providing an indication for anti-coagulation rather than conventional antiplatelet regimen. Future studies are needed to evaluate the impact of changes in medical management on the outcome of ESUS patients.
Footnotes
Author contributions
RB conceived the study and wrote the first draft of the manuscript, INA was involved in protocol development and equally contributed to this study. AH, RK, and EA were involved in protocol development. RM, SP and JN were involved in data acquisition and analysis, GS, AS, and AH were involved in imaging analysis. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was not sought for the present study because of its retrospective observational nature.
Ethical approval
Ethical approval for this study was obtained from the Ethics Committee of Rabin Medical Center.
