Abstract
Introduction:
Covert brain infarcts (CBIs) identified by imaging are more frequent than symptomatic ischemic strokes and are usually small and/or involve non-eloquent brain areas. The spectrum of incident CBIs in patients following acute ischemic stroke has not been well characterized.
Patients and methods:
Exploratory observational cohort study of the size, location, multiplicity and risk factors of incident CBIs identified using serial MRIs in patients with acute non-cardioembolic ischemic stroke participating in the PACIFIC-STROKE trial.
Results:
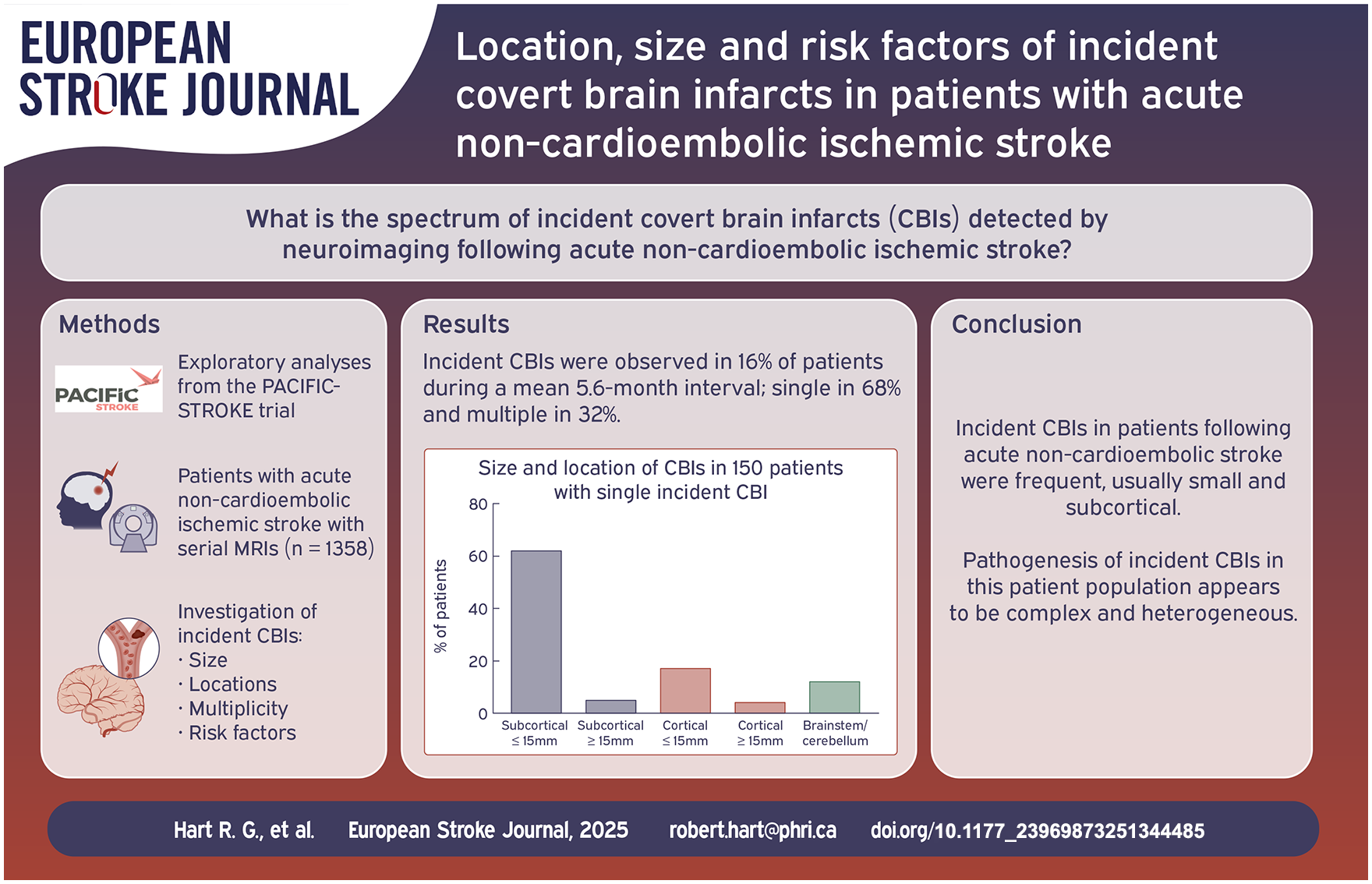
Incident CBIs were identified in 16% (220/1358) of participants during the mean 5.6-month interval between MRIs. Single incident CBIs occurred in 150 (68%) of incident CBI patients and multiple CBIs in 70 (32%); 42% (93/220) of incident CBI patients had a single, small, subcortical CBI. There was no clear relationship between the location of incident CBI and the location of index stroke. Significant independent features associated with incident CBI included diabetes (adjusted OR 1.52, 95%CI 1.09–2.12), current tobacco use (adjusted OR 1.50, 95%CI 1.07–2.09), prior stroke/TIA (adjusted OR 1.59, 95%CI 1.08–2.36), carotid artery atherosclerosis (adjusted OR 1.47, 95%CI 1.07–2.02), and diastolic blood pressure (adjusted OR 1.02 per 1 mmHg increase, 95%CI 1.01–1.04).
Discussion and conclusion:
Incident CBIs in patients following acute non-cardioembolic stroke were frequent, usually small and subcortical, and associated with traditional stroke risk factors. Observations from PACIFIC-STROKE, particularly the absence of correlation between the location of incident CBI and the index infarct and the high frequency of incident small, subcortical CBIs, suggest that incident CBI pathogenesis is complex and heterogeneous in this population.
Trial registration:
https://clinicaltrials.gov (NCT04304508).
Introduction
Focal brain ischemia includes both symptomatic ischemic strokes and clinically covert brain infarcts (CBIs). CBIs detected by neuroimaging tend to be small and located either in the subcortex or in clinically silent areas of the cortex. Even so they are associated with cognitive and motor dysfunction and an increased risk of symptomatic stroke. 1 In the PACIFIC-STROKE randomized trial that enrolled patients with acute, non-cardioembolic ischemic stroke, incident CBIs were detected using standardized MRIs performed at study entry and repeated after 26 weeks or when assigned study drug was discontinued. 2 The frequency of incident CBIs and the effect of asundexian according to CBI location have been previously reported. 3 In these exploratory analyses, we report additional details about the locations of incident CBIs relative to the initial qualifying stroke and features associated with location and multiplicity.
Methods
This is an observational cohort study involving a subgroup participants in the PACIFIC-STROKE international, randomized, placebo-controlled, double-blind, phase-2 trial involving patients ⩾45 years of age with non-cardioembolic ischemic stroke within 48 h of symptom onset. 2 Eligibility required intention to treat with standard-of-care antiplatelet therapy chosen by local investigators and being willing/able to undergo baseline and follow-up study MRIs. The study design, protocol, and main results have previously been published. 2 Patients from 196 clinical sites in 23 countries were randomly assigned in a 1:1:1:1 ratio to receive one of three asundexian dosages (10, 20, and 50 mg once daily) or matching placebo. Local investigators classified ischemic stroke etiology using the TOAST (Trial of ORG in Acute Stroke Treatment) criteria. 4
All participants were required to undergo at least two MRIs that met study requirements. In addition to the baseline MRI (done prior to or within 72 h after randomization), participants underwent a final study MRI at 26 weeks or as soon as possible if there was early termination of assigned study treatment. Required MRI sequences were diffusion-weighted imaging (DWI), apparent diffusion coefficient, fluid-attenuated inversion recovery, and a hemorrhage-sensitive sequence which could be either T2*weighted gradient-recalled echo or susceptibility-weighted imaging according to local site protocols. Acquisition requirements included slice thickness ⩽5 mm, pixel sizes no larger than 0.94 mm by 0.94 mm, and slice gap ⩽2 mm.
MRI data were transmitted to the central MRI Core Laboratory at the University of Calgary for independent interpretation by two (of three) neuroradiologists blinded to participant data and to treatment assignment, with differences resolved by consensus. Incident brain infarcts were identified by direct comparison of the baseline MRI with the follow-up study MRI. Kappa values for the identification of new infarcts for each pair of readers, based on a set of 50 PACIFIC-STROKE cases, were 0.74, 0.77, and 0.90, indicating good to excellent agreement. Infarcts were defined according to Standards for Reporting Vascular Changes on Neuroimaging (STRIVE). 5 For the analysis of CBI subtypes, incident CBIs were dichotomized by location (entirely subcortical vs cortical or with cortical involvement; brainstem or cerebellar CBI were not categorized as cortical or subcortical), size (small = ⩽15 mm in largest axial diameter vs large = >15 mm), and single versus multiple (see Supplemental Table 1 for categorization of CBI locations).
Of 1808 total randomized participants, 1428 had baseline and follow-up MRIs deemed adequate to assess for new brain infarcts (Figure 1). Incident CBIs were defined as new infarcts that were detected on follow-up MRI in participants without a history of symptomatic recurrent ischemic stroke between the study MRIs; the 1358 participants without recurrent symptomatic stroke between study MRIs comprise the subgroup cohort for these exploratory analyses and include participants pooled from all treatment arms. Expanded areas of brain infarction on follow-up MRI that were contiguous with the acute baseline infarction and in the same vascular territory were defined as extension of previous infarction and are not considered as incident CBI. Correlation of the location of incident CBIs with index ischemic stroke was limited to the participant subgroup with a single DWI-positive infarct on the baseline MRI identifying the location of the index stroke.

Study flow diagram.
Statistical analysis
Continuous variables are presented as mean ± standardized deviation and compared using either Student’s t-test or Analysis of Variance. Categorical variables are reported as count (percentage) and compared using Fisher’s exact test if the expected cell count in one of the two arms is less than 5, otherwise, chi-square test was used. A multivariable logistic regression model was used to test independent baseline patient features that are associated with all incident CBIs. Covariates included in the full model were those that reached a significance level of 0.2 in the univariable analysis. The final reduced model was developed from the full model by using backward stepwise selection procedure, forcing current tobacco use, diabetes, prior stroke or TIA, carotid artery atherosclerosis and diastolic blood pressure (i.e. as traditional stroke risk factors) into the model. The model for single versus multiple covert brain infarcts and for single, small subcortical covert brain infarcts was constructed in a similar fashion. A p-value of <0.05 was considered statistically significant. All analyses were performed using SAS version 9.4 (SAS Institute, Cary, USA).
Results
Among the 1358 participants comprising the CBI-eligible cohort (Figure 1), the mean age was 66 years, 32% were women, 84% were recruited from Europe or Australia, and the mean NIH Stroke Scale score at randomization was 2.7 (Table 1). Based on TOAST criteria applied by local investigators, small vessel disease was the etiology of the qualifying stroke in 47%, stroke was of unknown cause in 34%, and large-artery atherosclerosis was deemed etiologic in 16%. On the baseline study MRI, one or more DWI positive lesions were present in 92% (1255/1358) of patients.
Features of patients with versus without incident covert brain infarcts.
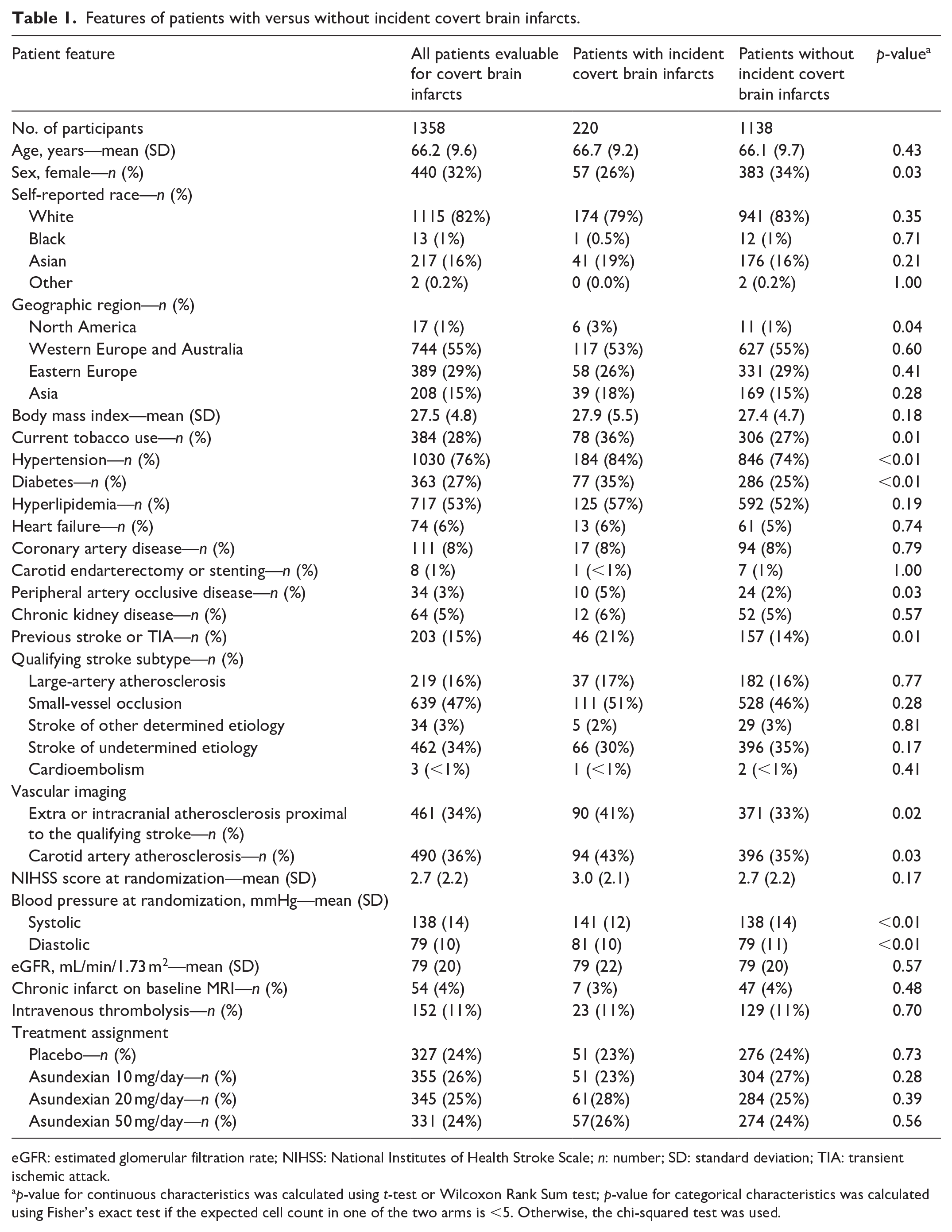
eGFR: estimated glomerular filtration rate; NIHSS: National Institutes of Health Stroke Scale; n: number; SD: standard deviation; TIA: transient ischemic attack.
p-value for continuous characteristics was calculated using t-test or Wilcoxon Rank Sum test; p-value for categorical characteristics was calculated using Fisher’s exact test if the expected cell count in one of the two arms is <5. Otherwise, the chi-squared test was used.
One or more incident CBIs were reported in 220 patients (16%) during the mean interval of 5.6 months between MRIs (Table 2). Among those with incident CBI, a single incident CBI was reported in 150 patients (68%), while multiple incident CBIs were identified in 70 patients (32%) with 218 total CBIs among those with multiple incident CBIs, range 2–11, median of 2, mean of 3.1 (SD 1.9) per patient. Among the 150 patients with single incident CBIs, a single, small, subcortical CBIs was observed in 62% (93/150 patients), a single large subcortical CBI in 5% (7/150), a single CBI involving the cortex in 21% (32/150), and a single brainstem or cerebellar CBI in 12% (18/150) involved the brainstem or cerebellum (Table 2, Figure 2). Restricting analysis to placebo-assigned participants yielded similar distributions of location and size (Supplemental Table 2). Of 368 total incident CBIs, 348 (95%) were small (⩽15 mm axial diameter). Most (83%, 183/220) patients with one or more incident CBIs had at least one small, subcortical hemispheric CBI, while 25% (55/220) of patients had an incident CBI with hemispheric cortical involvement (Table 2). The frequency of one or more small, subcortical CBI was 13% (183/1358) during the 5.6 month mean interval between MRIs.
Location and size of incident covert brain infarcts.
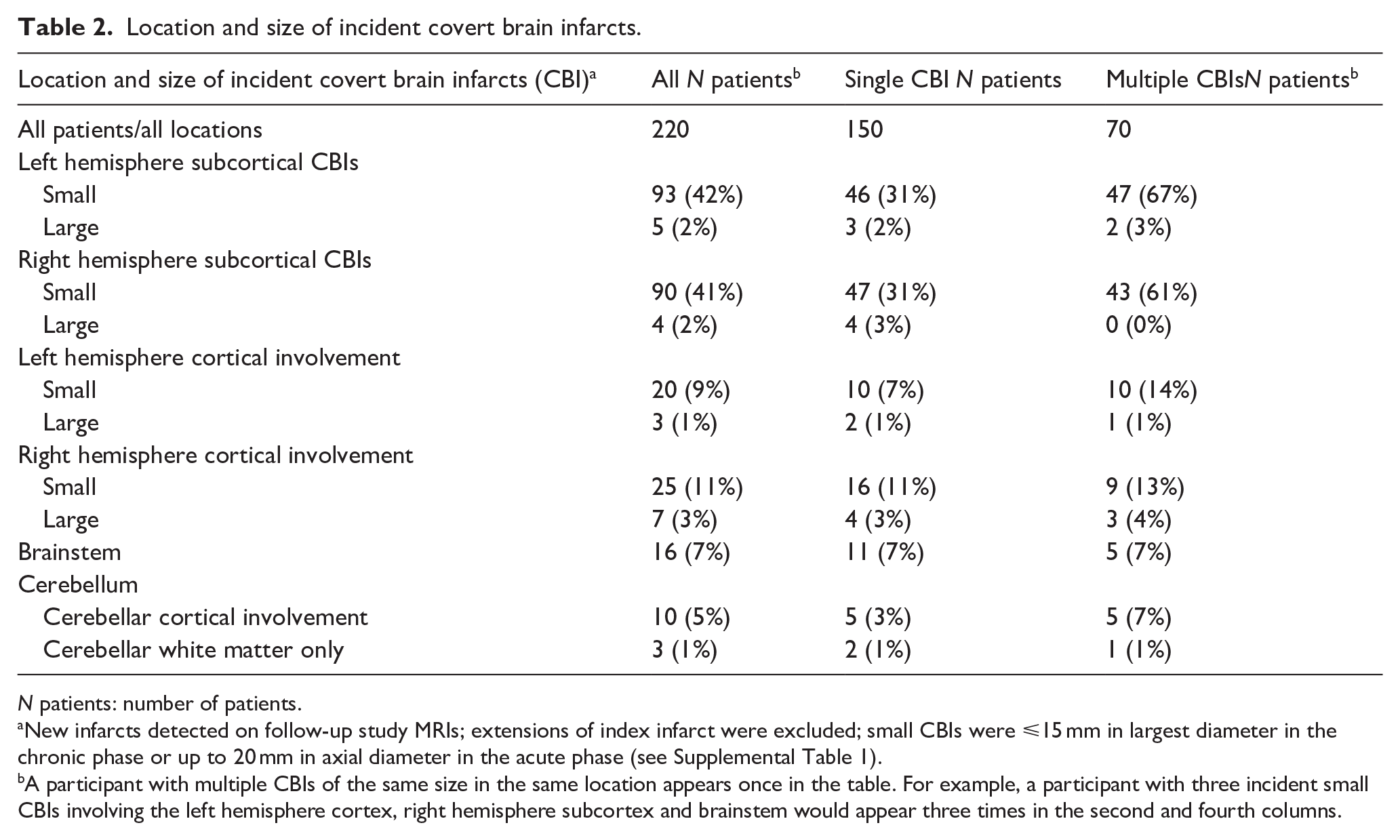
N patients: number of patients.
New infarcts detected on follow-up study MRIs; extensions of index infarct were excluded; small CBIs were ⩽15 mm in largest diameter in the chronic phase or up to 20 mm in axial diameter in the acute phase (see Supplemental Table 1).
A participant with multiple CBIs of the same size in the same location appears once in the table. For example, a participant with three incident small CBIs involving the left hemisphere cortex, right hemisphere subcortex and brainstem would appear three times in the second and fourth columns.
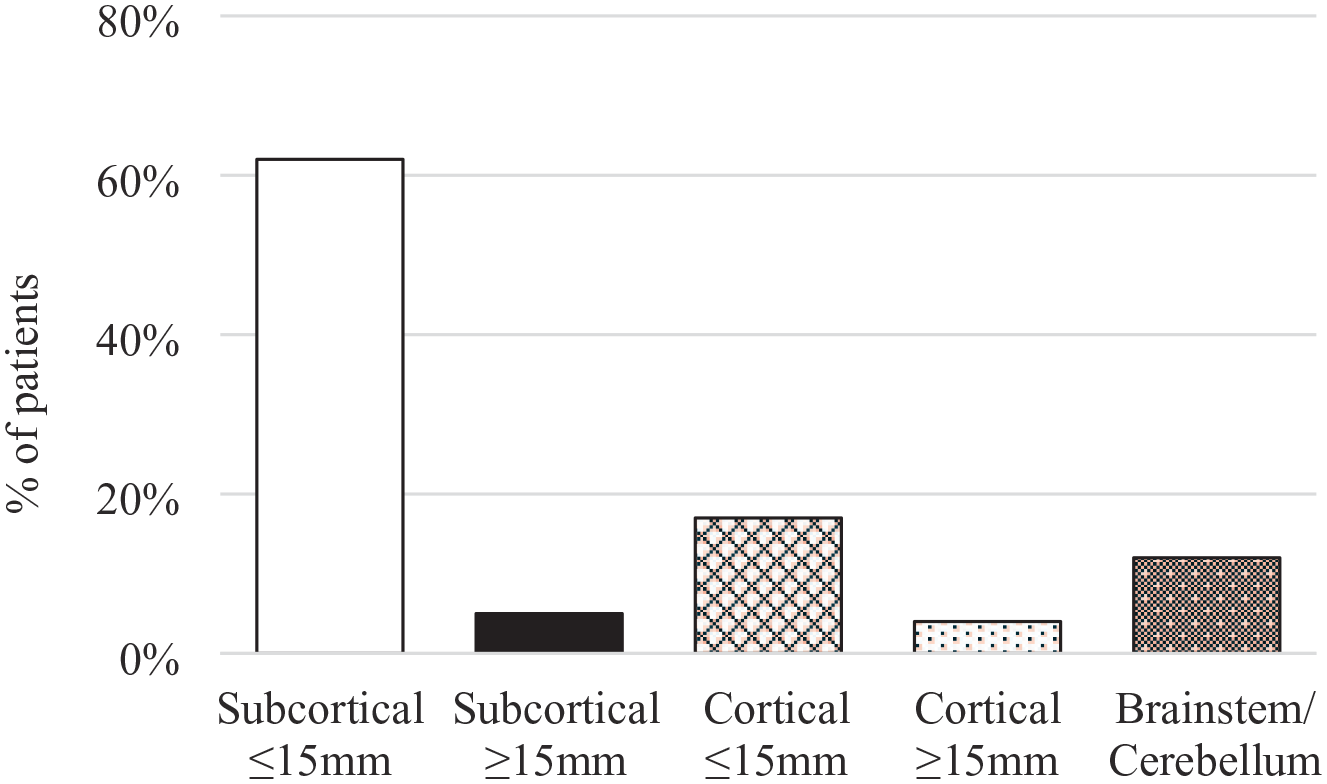
Size and location of covert brain infarcts in patients with single incident covert brain infarcts.
Location of incident CBI relative to the index stroke
These analyses were restricted to 891 participants with a single DWI-positive lesion on the baseline study MRI permitting the location of the index infarct to be reliably determined. Among 53 patients whose index ischemic stroke was hemispheric, 45% (24/53) of incident single CBIs were located in the same hemisphere, 47% (25/53) in the contralateral hemisphere, and 8% (4/53) in the brainstem or cerebellum (Table 3). Of 35 patients with a single DWI-positive index infarct involving the left hemisphere (33 subcortical, two cortical), single incident covert CBI were located in the left hemisphere in 40% (14/35), the right hemisphere in 54% (19/35), and the brain or cerebellum in 6% (2/35; Table 3). Of 18 patients with a single DWI-positive index infarct involving the right hemisphere (16 subcortical, two cortical), single incident covert CBI were located in the right hemisphere in 56% (10/18), the left hemisphere in 33% (6/18), and the brain or cerebellum in 11% (2/18; Table 3). There was no clear relationship between the location of incident CBI to the index stroke when the presumed etiology of the index stroke was considered, albeit based on small numbers (Supplemental Tables 3a and 3b).
Location of incident covert brain infarcts relative to the index infarct.
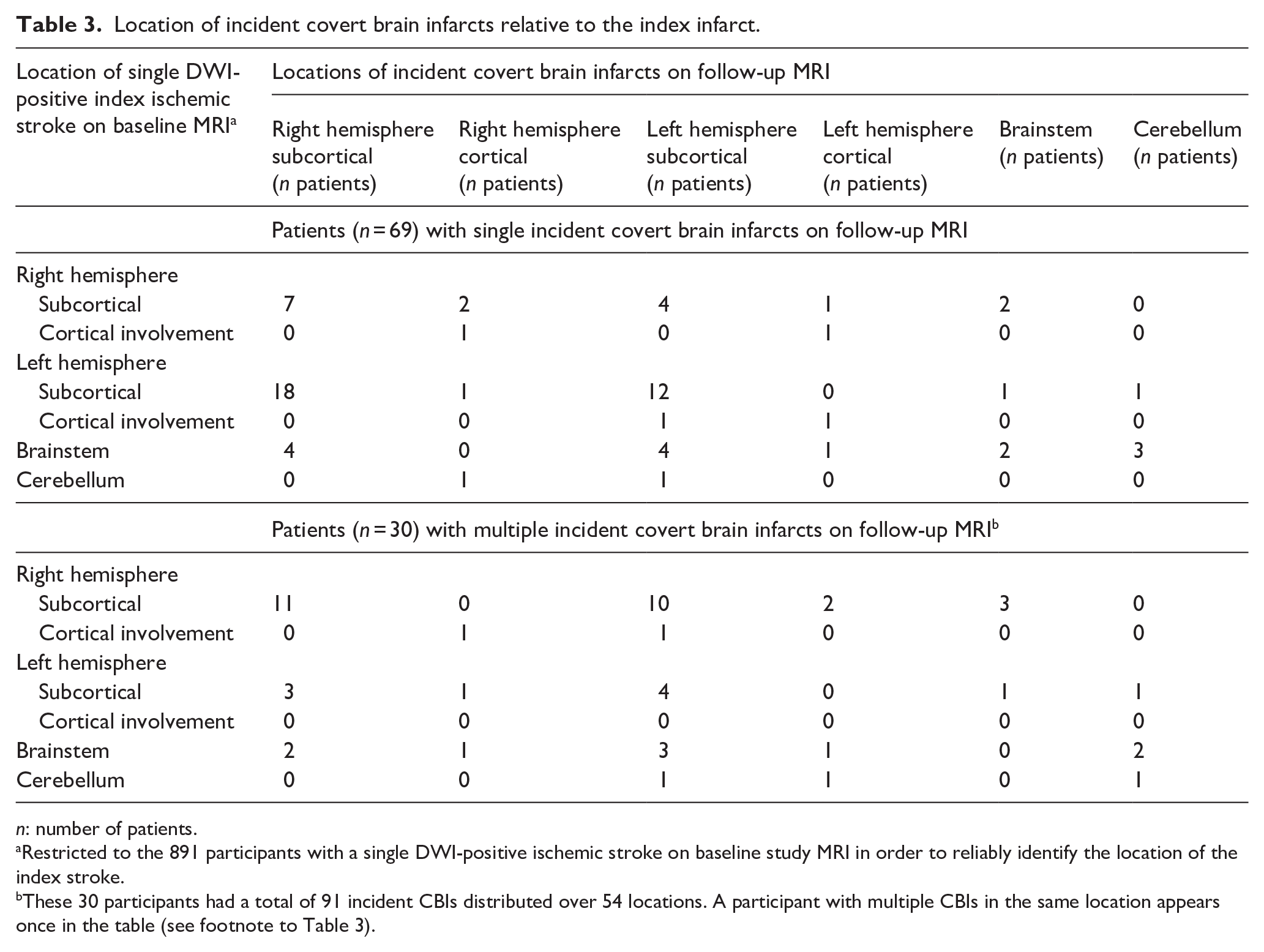
n: number of patients.
Restricted to the 891 participants with a single DWI-positive ischemic stroke on baseline study MRI in order to reliably identify the location of the index stroke.
These 30 participants had a total of 91 incident CBIs distributed over 54 locations. A participant with multiple CBIs in the same location appears once in the table (see footnote to Table 3).
Participant features associated with incident CBI
By multivariable analysis, significant independent risk factors for incident CBI were diabetes (adjusted OR 1.52, 95%CI 1.09–2.12), current tobacco use (adjusted OR 1.50, 95%CI 1.07–2.09), prior stroke/TIA (adjusted OR 1.59, 95%CI 1.08–2.36), carotid artery atherosclerosis (adjusted OR 1.47, 95%CI 1.07–2.02), and diastolic blood pressure (adjusted OR 1.02, 95%CI 1.01–1.04 per 1 mmHg increase; Table 4). A sensitivity analysis that removed prior stroke/TIA resulted in two additional variables becoming significantly associated with incident CBI: hypertension (adjusted OR 1.53, 95%CI 1.02–2.29) and peripheral vascular disease (adjusted OR 2.20, 95%CI 1.00–4.84; Table 4, footnote).
Patient features significantly associated with incident covert brain infarcts by multivariate analysis a .
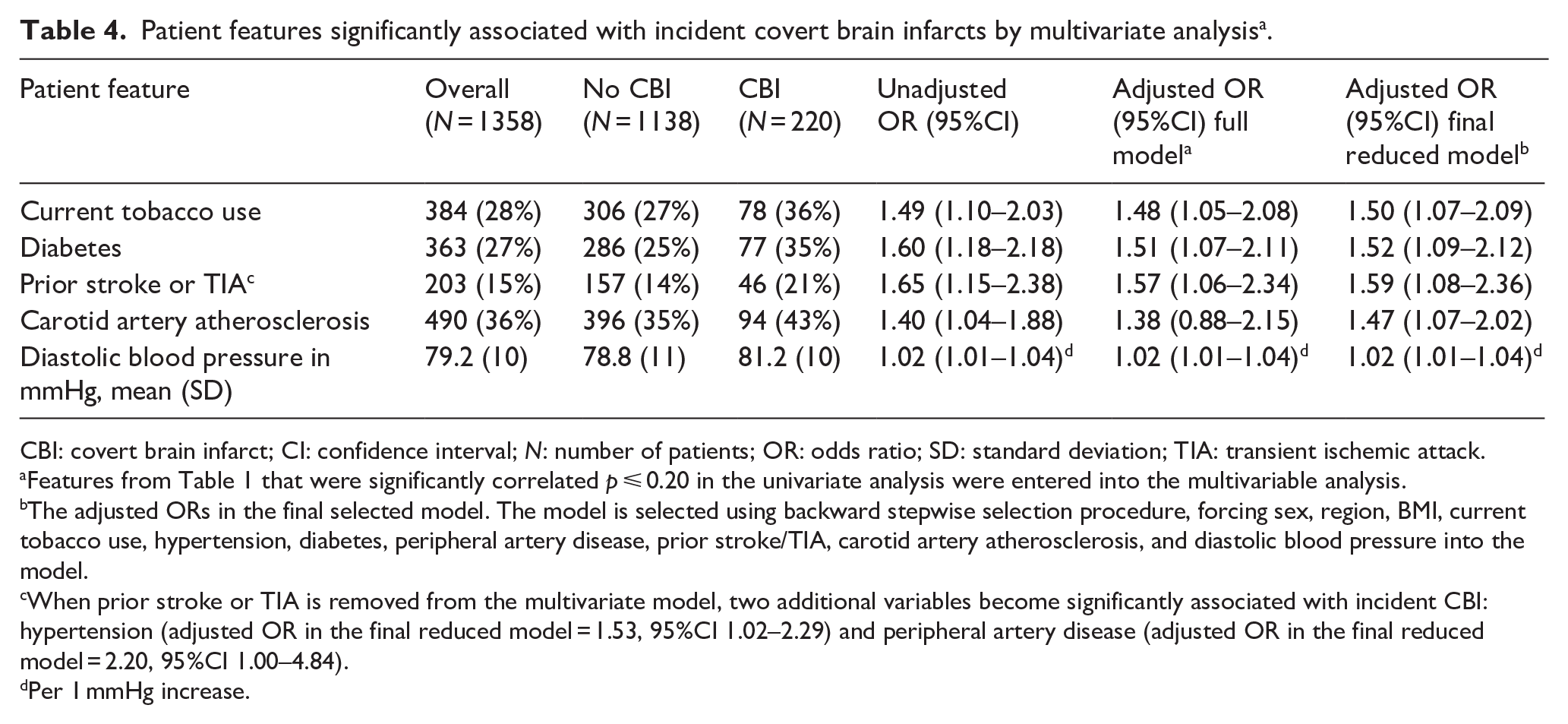
CBI: covert brain infarct; CI: confidence interval; N: number of patients; OR: odds ratio; SD: standard deviation; TIA: transient ischemic attack.
Features from Table 1 that were significantly correlated p ⩽ 0.20 in the univariate analysis were entered into the multivariable analysis.
The adjusted ORs in the final selected model. The model is selected using backward stepwise selection procedure, forcing sex, region, BMI, current tobacco use, hypertension, diabetes, peripheral artery disease, prior stroke/TIA, carotid artery atherosclerosis, and diastolic blood pressure into the model.
When prior stroke or TIA is removed from the multivariate model, two additional variables become significantly associated with incident CBI: hypertension (adjusted OR in the final reduced model = 1.53, 95%CI 1.02–2.29) and peripheral artery disease (adjusted OR in the final reduced model = 2.20, 95%CI 1.00–4.84).
Per 1 mmHg increase.
Features associated with multiple CBIs versus single incident CBIs
Features independently associated with multiple versus single incident CBIs were carotid artery atherosclerosis (adjusted OR 0.44, 95%CI 0.23–0.87), systolic blood pressure (adjusted OR 1.03, 95%CI 1.01–1.06 per 1 mmHg increase), intravenous thrombolysis given for the qualifying stroke (adjusted OR 2.73, 95%CI 1.02–7.32) and Eastern Europe region (reference: North America/Western Europe; adjusted OR 2.13, 95%CI 1.04–4.34; Supplemental Table 4).
Discussion
In the PACIFIC-STROKE trial assessing secondary stroke prevention involving participants with acute non-cardioembolic ischemic stroke, the frequency of incident CBI during the 5.6-month mean interval between MRIs was 16%, more than twice the frequency of recurrent symptomatic ischemic stroke. 2 Most (83%) patients with one or more incident CBIs had at least one small, subcortical hemispheric CBI (Table 2).
The location of incident CBIs was not associated with the location of the qualifying infarct (Table 3). For example, among the subgroup of patients with a single DWI-positive infarct on the baseline MRI, allowing the location of index stroke to be reliably determined, of 35 patients with the index infarct involving the left hemisphere, the single covert incident CBI was located in the left hemisphere in 40% while located in the right hemisphere in 54% (Table 3). The observed frequency of small, subcortical incident CBIs (13% during the 5.6 months interval between MRIs) exceeds that expected in patients 6 and populations 7 without a recent stroke.
Patient factors independently associated with incident CBIs included many of the traditional risk factors for atherosclerosis (Table 4) and overlapped with risk factors associated with CBI in population-based studies with the exception of patient age.7,8 Ischemic stroke subtype of the qualifying stroke as classified by local investigators did not emerge as independent predictors of incident CBI. Of note, carotid artery atherosclerosis by imaging was independently associated with incident CBI (adjusted OR 1.47, 95%CI 1.07–2.02; Table 4) as well as with a reduced likelihood of multiple (vs single) incident CBI (adjusted OR 0.44, 95%CI 0.23–0.87; Supplemental Table 3).
The use of intravenous thrombolysis for the index stroke was not independently associated with incident CBI but was significantly associated with an increased likelihood that incident CBI would be multiple rather than single (adjusted OR 2.73, 95%CI 1.02–7.32; Supplemental Table 4). It is unclear why thrombolysis given for the index stroke should alter the spectrum of subsequent incident CBI.
Five other randomized trials involving patients with recent non-cardioembolic ischemic stroke reported incident CBI detected on serial MRI, but with few details about the spectrum or risk factors of CBI.9 –13 In the two trials enrolling patients with acute ischemic stroke, the frequencies of incident CBIs were comparable (6% at 30 days, 10 11% at 3 months 12 ) to that observed in PACIFIC-STROKE. In a multicenter cohort study involving serial MRIs of 172 patients with ischemic stroke attributed to cerebral small vessel disease recruited with 15 days, 15% of patients had one or more covert brain infarct at 6 months. 14
Limitations of these exploratory observations include that statistical significance was unadjusted for multiple post-hoc comparisons. These analyses were based on a subgroup of participants undergoing serial MRIs in a clinical trial made up of patients with mainly minor ischemic strokes dominated by small vessel disease as the presumed etiology. It is possible that small incident CBIs were overcalled due to subtle MRI acquisition differences (i.e. not detected on the baseline MRI but were interpreted as incident on follow-up MRI). Control of vascular risk factors was not recorded during follow-up, and consequently whether such control influenced the incidence of CBI could not be assessed.
It was previously reported from PACIFIC-STROKE that the effect of the novel oral anticoagulant asundexian appeared to differ on incident cortical CBIs versus incident small subcortical CBIs, 3 and this was supported by an analysis of CBI subtypes from the ARCADIA trial. 13 This supports the hypothesis that different locations and size of incident CBIs may reflect different pathogeneses and potentially have different responses to antithrombotic and other preventive therapies. Assessing subtypes of incident CBIs could potentially be a useful surrogate for assessing stroke prevention therapies, but its value in this regard has not been adequately established.
These observations from PACIFIC-STROKE, particularly the absence of association between the location of incident CBI and the index infarct and the remarkably high frequency of small, subcortical CBIs, suggest that pathogenesis of incident CBI in this population may be heterogeneous and complex.
Supplemental Material
sj-docx-1-eso-10.1177_23969873251344485 – Supplemental material for Location, size, and risk factors of incident covert brain infarcts in patients with acute non-cardioembolic ischemic stroke: PACIFIC-STROKE trial
Supplemental material, sj-docx-1-eso-10.1177_23969873251344485 for Location, size, and risk factors of incident covert brain infarcts in patients with acute non-cardioembolic ischemic stroke: PACIFIC-STROKE trial by Robert G Hart, Eric E Smith, Angela Wang, Hardi Mundl, Pablo Colorado, Raed A Joundi, Aristeidis H Katsanos, Mukul Sharma and Ashkan Shoamanesh in European Stroke Journal
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All coauthors or their institutions received financial support from Bayer for participation in the PACIFIC-STROKE trial except HM and PC who were employed by Bayer.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The trial was funded by Bayer AG.
Ethical considerations
The study was conducted in accordance with the World Medical Association Declaration of Helsinki and was approved by the relevant health authorities and the institutional review boards at each of the 196 clinical sites.
Consent to participate
All participants provided written informed consent prior to enrollment in the study.
Consent for publication
Not applicable.
Guarantor
Robert Hart.
Contributorship
The study was conceived by RGH and AS and designed by RGH, AS, EES, and AW. Data collection was done EES, HM, and PC. Analysis was done and statistical support provided by AW and interpretated by RGH, EES RAJ, MS, and AHK. The manuscript was drafted by RGH and AW with revisions by EES, AS, RAJ, PC, HM, MS, and AHK. All authors have reviewed the results and approved the final version of the manuscript.
Data availability
The individual participant-level data are not available.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
