Abstract
Background
Embolic stroke of undetermined source (ESUS) accounts for up to 25% of strokes. Understanding risk factors associated with ESUS is important in reducing stroke burden worldwide. However, ESUS patients are younger and present with fewer traditional risk factors. Significant global variation in ESUS populations also exists making the clinical picture of this type of stroke unclear.
Methods and results
ESUS patients were pair matched for age, sex, and ethnicity with a group of all other strokes (both n = 331). Exploratory factor analysis was applied in both groups to 14 risk and clinical factors to identify latent factors. In ESUS patients, two latent factors emerged consisting primarily of heart-related variables such as left ventricular wall motion abnormalities, reduced ejection fraction, and increased left atrial volume index, as well as aortic arch atherosclerosis. This is in comparison to the all other strokes group, which was dominated by traditional stroke risk factors.
Conclusions
Our findings support the existence of a unique pattern of risk factors specific to ESUS. We show that LVWMA and corresponding changes in left heart function are a potential source of emboli in these patients. In addition, the clustering of aortic arch atherosclerosis with left heart factors suggests a causal link. Through the application of exploratory factor analysis, this work contributes to a further understanding of stroke mechanisms in ESUS.
Introduction
Since its formal characterization, 1 embolic stroke of undetermined source (ESUS) has generated much interest. Several risk factors have been associated with an ESUS diagnosis, such as aortic arch atherosclerosis, 2 hypertension, 3 and left ventricular wall motion abnormalities (LVWMA). 4 However, the clinical picture of ESUS remains unclear with covert atrial fibrillation the most widely speculated upon cause.5,6
One of the difficulties in identifying risk profiles for ESUS patients is the consistent finding that they are younger and carry fewer traditional risk factors than other forms of stroke diagnosis. 7 Another difficulty is the global variation in ESUS populations, likely stemming from genetic and culturally determined factors.8,9 In addition, the majority of work on this topic uses multivariate statistics to show how risk factors differ from other forms of stroke diagnoses (or normal populations) but not how they manifest in ESUS only populations.
To address these issues, this study applies exploratory factor analysis to investigate the interactions between risk factors for ESUS while controlling for age, sex, and ethnicity. Exploratory factor analysis assesses underlying interactions between variables and specifies latent factors. 10 Latent (meaning “hidden”) variables are unobserved but identified through shared features of observed variables. The usefulness of understanding risk factor interactions in this way is highlighted by metabolic syndrome, whereby vascular risk and metabolic abnormalities combine to confer increased cerebro- and cardiovascular hazard. 11 To our knowledge, no studies have explored similar clustering of risk factors in ESUS patients and this work is the first to do so.
Methods
Stroke data were prospectively collected at a tertiary referral center between 1 January 2017 and 31 March 2020. Stroke subtypes were classified according to TOAST criteria by stroke neurologists. 12 ESUS diagnosis was confirmed using criteria described by Hart et al. 1 Each patient underwent vascular imaging using cervico–cranial magnetic resonance angiography, computed tomography angiography, digital subtraction angiography, and/or carotid Doppler studies. Transthoracic echocardiogram and Holter for at least 24–48 h were also performed. Patients with large PFO were excluded.
Seventeen risk factors were studied as determined by the literature and stroke neurologists. These were; age, sex, and ethnicity (used for pair matching), hypertension (HTN), diabetes mellitus (DM), dyslipidemia (DYSLIP), smoking, coronary artery disease (CAD), left atrial volume index (LAVI), ejection fraction (EF), left ventricular diastolic dysfunction (LVDD), mitral calcification (MITCAL), aortic arch atherosclerosis (AAA), left ventricular wall motion abnormalities (LVWMA), aortic root diameter (ART), left ventricular mass index (LVMI), and regional wall thickness (RWT).
Data analysis
Where appropriate, variables were converted to binary or categorical outcomes. Two data sets were created; one composed of ESUS patients and the second, all other strokes. All other strokes consisted of non-cardioembolic, multiple etiologies, and cardioembolic strokes. All other strokes served as a comparator to validate the underlying pattern of clinical and risk features identified in ESUS were unique to that diagnosis. Pair matching was based first on sex, then country of origin, followed by age (within two years). Unless a match for all three was found, the patient was excluded.
Statistical analysis
Descriptive analysis was performed using Statistical Package for Social Sciences Version 24.0. 13 Continuous data are presented as mean and standard deviation (M, ±SD) with absolute numbers and percentage reported for categorical variables. To explore differences in clinical and risk factors between ESUS and all other strokes, N-1 Chi square test of proportions was used. 14
Exploratory factor analysis was applied to each data set using Mplus version 6.12, with mean- and variance-corrected-weighted least squares (WLSMV) and Geomin rotation. 15 Model fit cut-off values were ≥.90 for comparative fit index (CFI) and Tucker Lewis index (TFI) with root mean square error of approximation (RMSEA) ≤.08. Loadings ≥.3 indicated satisfactory contribution to a latent variable. 10 See Supplementary Material 1 for full description of exploratory factor analysis.
Results
Demographic, clinical, and radiological/echocardiographic characteristics of ESUS and all other stroke groups.
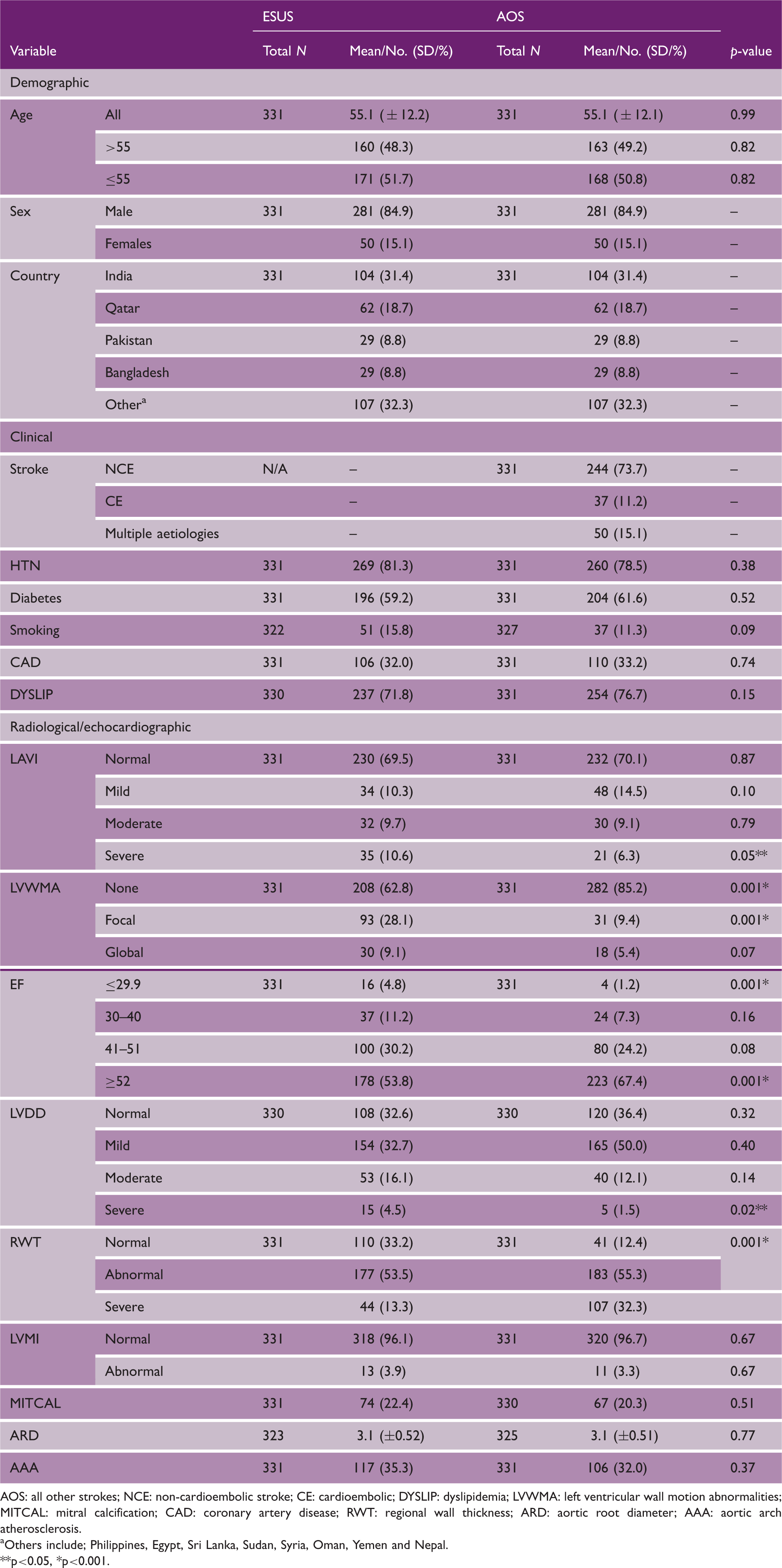
AOS: all other strokes; NCE: non-cardioembolic stroke; CE: cardioembolic; DYSLIP: dyslipidemia; LVWMA: left ventricular wall motion abnormalities; MITCAL: mitral calcification; CAD: coronary artery disease; RWT: regional wall thickness; ARD: aortic root diameter; AAA: aortic arch atherosclerosis.
Others include; Philippines, Egypt, Sri Lanka, Sudan, Syria, Oman, Yemen and Nepal. **p<0.05, *p<0.001.
In the ESUS group, a final two-factor model was identified (χ2 = 56.4, df, 53, RMSEA, 0.01, CFI, 1.00, TFI, 1.00). In the first latent factor “Cardiac 1”, the highest loading variable was LVWMA (− 0.98), which was negatively correlated (LVWMA were coded 0 –2 for none, focal, and global) followed by EF (0.71) and LAVI (0.48) (see Figure 1, Table 2). The second latent factor “Cardiac 2” also included heart-related features, LVDD (0.36), and LVMI (0.32) clustered with aortic arch atherosclerosis (0.50), and dyslipidemia (−0.43). Aortic arch atherosclerosis was the dominant Cardiac 2 contributor and the only positively loading non-heart feature found in ESUS patients. The correlation between Cardiac 1 and Cardiac 2 was moderate (0.34).
Schematic representation of latent variables in the final ESUS (left) and all other strokes (right) models. Indicator variables appear on the left of each diagram and latent variables on the right. Arrows point to indicators that cluster with latent variables and each loading is displayed in the boxes. Between latent variables, double-headed arrows indicate correlation with r-values shown in the box. AAA: aortic arch atherosclerosis; DYSLIP: dyslipidemia; WMA: wall motion abnormalities; AOS: all other strokes. Geomin rotated factor loadings for final ESUS and all other strokes models. AOS: all other strokes; MITCAL: mitral calcification; AAA: aortic arch atherosclerosis; RWT: regional wall thickness; ARD: aortic root diameter; DYSLIP: dyslipidemia; LVWMA: left ventricular wall motion abnormalities. Note: Loadings ≥.3 are in bold. Deleted in second ESUS analysis. Deleted in second analysis of all other strokes.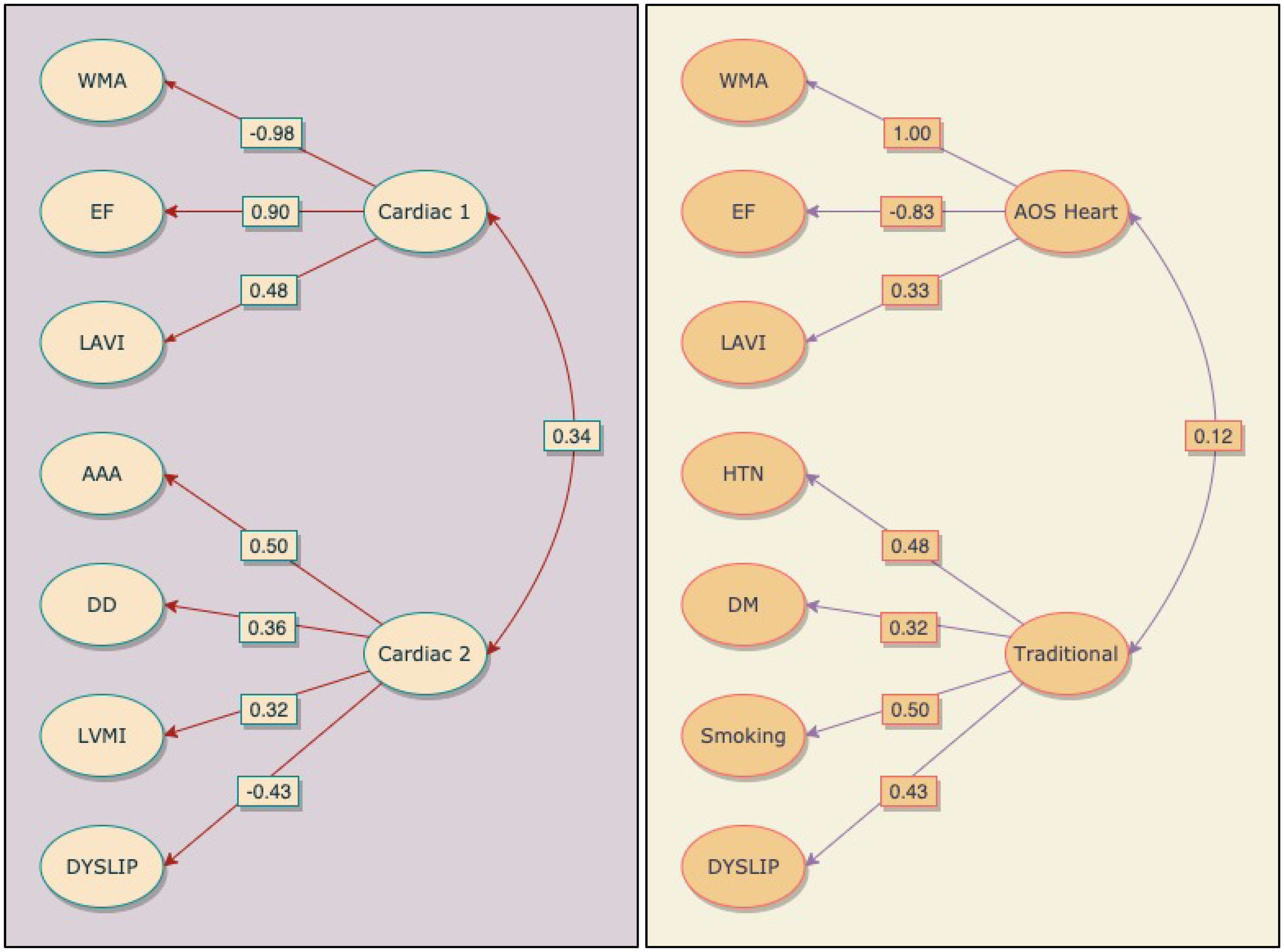
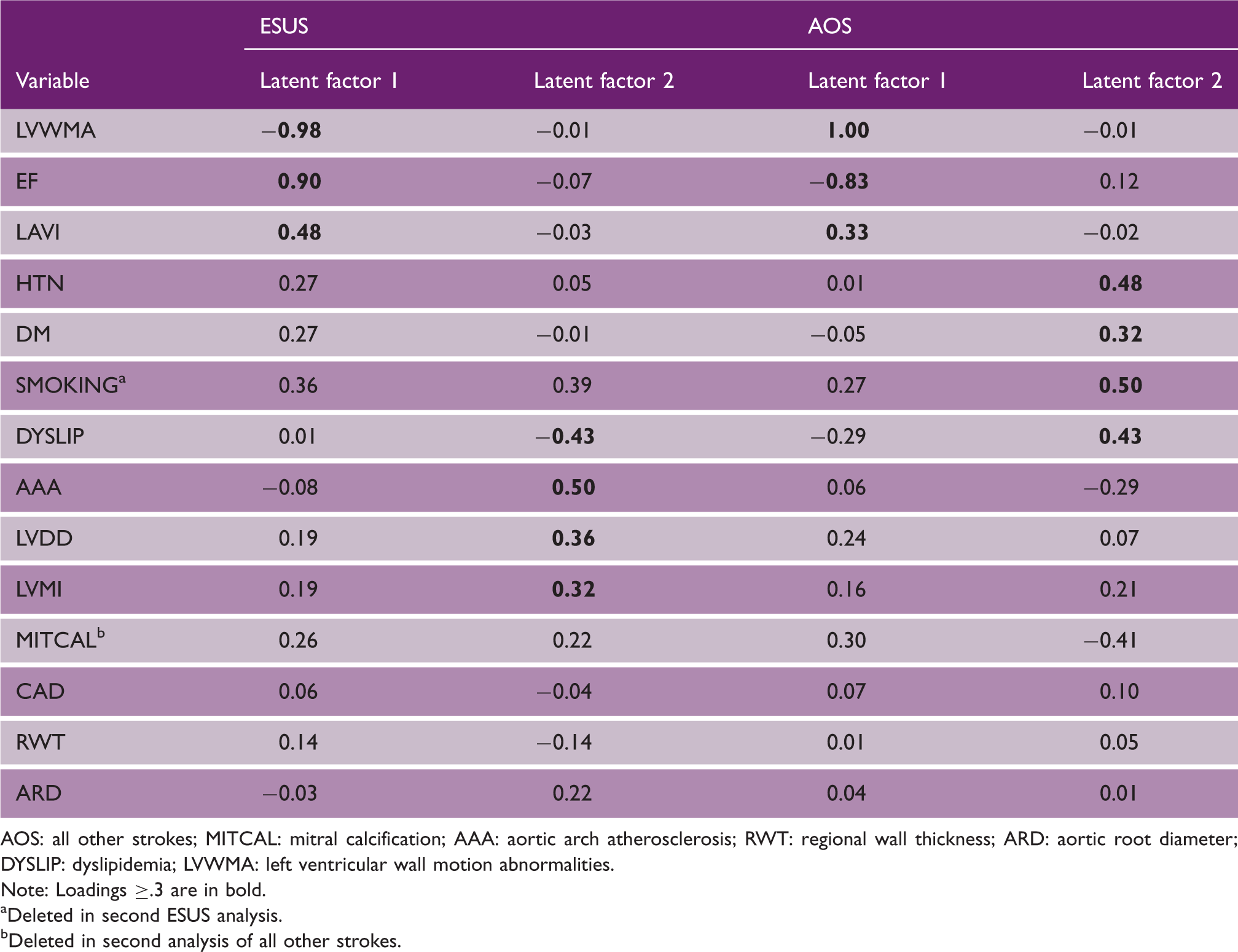
In the all other strokes data, a two-factor model also emerged (χ2 = 64.7, df, 53, RMSEA, 0.03, CFI, 0.98, TFI, 0.97). The first latent factor “AOS Heart” mirrored ESUS findings and included LVWMA (1.0) and EF (−0.83) but loaded in reverse meaning that normal or focal/ LVWMA and normal or mild/EF, clustered with the third contributor, LAVI (0.48), (see Figure 1, Table 2). A second latent factor “Traditional” consisted entirely of tradition risk factors. The highest loading variable was smoking (0.50), followed by HTN (0.48), dyslipidemia (0.43), and DM (0.32). The correlation between AOS Heart and Traditional latent factors was small at 0.12. A full description of results can be found in Supplementary Material 1.
Discussion
The aims of this work were to explore the underlying relationships between different risk factors for ESUS. After the exploratory factor analysis, two latent factors emerged in both ESUS and all other strokes groups with unique patterns of risk observed despite being matched for age, sex, and ethnicity. As the pattern unique to ESUS was predominantly heart related, it indicates a cardioembolic source of emboli in these patients.
As mentioned, ESUS patients are younger and consequently have fewer traditional risk factors.7,9 In this work, 48.3% of participants were over 55 years of age and yet traditional risk factors were absent in the ESUS latent factors (dyslipidemia loaded negatively in Cardiac 2 suggesting abnormal blood lipids are not involved in ESUS pathology). This finding cannot be attributed to differences in sex or ethnicity as these were controlled for and contrasts starkly with the all other strokes group where traditional risk factors clustered as an independent latent factor. This is significant as it is evidence of a pattern of risk features that occur in ESUS patients of any age. This pattern of risk may extend to ethnicity, which was similarly controlled. A question of interest is whether Caucasians would produce comparable findings? Data presented here suggest this may be so as wide ethnic disparities exist between the South East and Western Asians in this sample. In addition, the risk factors found in this work have been reported in Caucasians elsewhere and further studies are warrented.2,8
Of the two latent factors identified in ESUS patients, Cardiac 1 was the largest contributor and included the solely heart-related factors, LVWMA, LAVI, and EF. LVWMA are both associated and predictive of EF and increased LAVI is reported in patients with wall motion problems.16-18 ESUS Cardiac 1 differed from all other strokes where LVWMA were absent or minimally involved in stroke as was reduced EF. There is growing evidence of a role for LVWMA in ESUS pathology.4,19,20
The finding of predominantly heart-related clustering extended to Cardiac 2 where reduced LVDD and increased LVMI featured. The moderate correlation between Cardiac 1 and 2 reflects an association between these variables and Cardiac 1 items. For instance, LVDD and LVWMA are associated with ventricular remodelling after myocardial infarction and increased atrial volume occurs in response to the additional burden in regulating ventricular filling LVDD causes.21,22 LVWMA have also been linked to diastolic dysfunction and ventricular enlargement independently.4,9,23 A final risk factor in Cardiac 2 was aortic arch atherosclerosis, which has previously been identified in ESUS patients and is discussed further below.2,24
An advantage of exploratory factor analysis is that after isolating risk factors we may speculate on how variables influence each other and importantly, on the sequence of interactions that may cause pathology. In the ESUS group, the highest loading variable, and the one that underpins other conspicuous risk and clinical factors, was LVWMA. LVWMA are sensitive indicators of myocardial ischemia suggesting that many of these patients had previous or unrecognized myocardial infarction. 25
An interesting question from Cardiac 2 is why aortic arch atherosclerosis and LVDD were coupled? We speculate that it relates to altered hemodynamics from poor left heart function with increased likelihood of blood stagnation and disturbed flow.26,27 Changes in hemodynamics are a well-described cause of atherosclerotic plaque through pathways such as; increased exposure of atherosclerotic initiators with the vessel wall, changes in wall shear stress and activation of flow-sensitive coding and non-coding genes.28–30 Although flow was not directly measured in this work, it is a plausible explanation for the aortic arch atherosclerosis-LVDD clustering presented here and future studies of flow dynamics in ESUS patients are of interest. Furthermore, the location of plaque formation is not random, including in the aorta.31,32 This implies that a distinct pattern of aortic atheroma may be present if altered flow dynamics are a cause of plaque in ESUS. In our model, approximately one-third of ESUS patients had aortic arch atherosclerosis suggesting a role as a source of emboli. However, the detection and categorization of aortic arch atherosclerosis require transesophageal echocardiography (TEE), which is limited in ESUS studies as shown by the NAVIGATE ESUS trial where only 19% of patients underwent TEE.2,33 Our work also highlights the importance of TEE in ESUS.
Left heart dysfunction and resultant hemodynamic changes are also associated with hypercoagulability and increased likelihood of thrombus formation.34,35 Ventricular thrombi are common after myocardial infarction and almost always appear at the site of LVWMA. 36 However, thrombi are sometimes missed in patients with ESUS for example, Takasugi et al. report a LV thrombus detection rate of 20-1 using cardiac magnetic resonance imaging versus TEE. 37
LV dysfunction influences atrial hemodynamics and function as indicated in our model by the clustering of LAVI with LVWMA and other measures of left heart dysfunction.38,39 Atrial thrombi are a source of embolic stroke and a topic of great interest to ESUS research due to the often-seen atrial dysfunction (atrial fibrosis, left atrial size, and increased LAVI) observed in these patients.40,41 Currently, patients with atrial cardiomyopathy are being investigated for anticoagulants versus aspirin (the ARCADIA trial) and left atrial appendage flow velocity ≤0.2 m/sg (the ATTICUS trial) which will shed further light on the role of the left atrium in ESUS patients.42,43 An intriguing outcome regarding the NAVIGATE and RE-SPECT ESUS trials was that both failed to find a difference in anticoagulants versus antiplatelets in ESUS, which could be explained by some patients presenting with either atherosclerotic aetiologies versus thrombosis.33,44 Support for this proposition comes from the COMPASS trial where a combined low dose of rivaroxaban and aspirin was found to reduce stroke compared to either medication as monopharmacy. 45
Limitations
This study has several limitations. It is a retrospective analysis of prospectively collected data, therefore collection and registration bias may be present. As this study used a paired match design, the number of excluded participants was high. Only recorded indicator variables are included, and unregistered significant variables may have been omitted. Future work may need to explore other variables as ESUS data accumulates. Females were less than males due to expatriate population in Qatar. Despite these limitations, our study is the first to our knowledge to use exploratory factor analysis to investigate risk and clinical factors in patients with ESUS and provides the only model of how these factors interact in this group.
Supplemental Material
sj-pdf-1-wso-10.1177_17474930211009847 - Supplemental material for Partitioning risk factors for embolic stroke of undetermined source using exploratory factor analysis
Supplemental material, sj-pdf-1-wso-10.1177_17474930211009847 for Partitioning risk factors for embolic stroke of undetermined source using exploratory factor analysis by Jon D Perkins, Naveed Akhtar, Rajvir Singh, Asad Kamran and Saadat Ilyas in International Journal of Stroke
Footnotes
Acknowledgements
The authors acknowledge the valuable support of; Reny Francis, Deborah Morgan, Sujatha Joseph, Pooja George, Blessy Babu, Lamxi Ojhi, and Mark Santos, with data management.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical standards
The study adhered to the tenets of the declaration of Helsinki and was approved by the IRB of Hamad Medical Corporation, Qatar.
Informed consent
None required due to retrospective study design.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
