Abstract
Background
Previous studies on stroke recurrence in younger adults often contain small sample size which makes it difficult to study trends in stroke recurrence over a long period of time.
Aims
The aim of the present study was to investigate temporal trends in the risk of recurrence in younger patients with a first ischemic stroke.
Methods
All men and women aged 18–54 years who had survived at least 28 days after a first ischemic stroke from 1987 to 2006 were identified in the Swedish Inpatient Register. The patients were stratified into four 5-year periods according to their admission period and were followed up for a total of four years after the index event with regard to recurrent ischemic stroke. A Cox regression model was used to analyze the risk of recurrent ischemic stroke.
Results
Of the 17,149 ischemic stroke patients who were identified, 2432 (14.2%) had a recurrent ischemic stroke event within four years. From the first to the last periods (1987–1991 versus 2002–2006), the four-year risk of recurrent ischemic stroke decreased by 55% (hazard ratio 0.45, 95% confidence interval 0.39–0.53) in men and 59% (hazard ratio 0.41, 95% confidence interval, 0.33–0.50) in women. The cumulative four-year risk was 11.8% (95% CI 10.55–13.25) in men and 9.8% (95% CI 8.40–11.46) in women during the last five-year period (2002–2006).
Conclusions
The risk of recurrence among younger ischemic stroke patients has decreased over the past 20 years. Despite these improvements, younger patients are still at a high risk for recurrent ischemic stroke.
Introduction
Stroke is a serious condition with a high risk of disability and long-lasting consequences, which increase not only the risk of premature death, but also of future vascular events.1–3 Over the past years, mortality of stroke in most Western countries has decreased and the number of patients surviving their first stroke is increasing.4–6 Vascular outcomes, such as recurrent stroke, are becoming increasingly more important because they may further increase the risk of disability and premature death.
The Global Burden of Disease Study reported that a significant amount of the incidence of stroke, prevalence of stroke, and death-related stroke, and over two-thirds of disability-adjusted life years lost occur in people younger than 75 years old. 7 However, most studies on outcome after stroke are based on elderly people with findings that may not be applicable to younger adults. Previous studies have reported that the risk of a recurrent stroke among younger people within the first year varies from 2 to 4%, decreasing thereafter to less than 3% yearly.8–11 Most prior studies on younger adults have been limited by small sample sizes. To the best of our knowledge, no studies have investigated trends and changes in risk over a long period of time in younger patients with ischemic stroke (IS). Information on prognosis and outcome is important for understanding the etiology of stroke and also in understanding how to improve outcome after a stroke.
Aims
This study aimed to examine temporal trends and the cumulative risk of recurrent IS over time in younger adults aged 18–54 years who were hospitalized with a first-time IS in Sweden from 1987 to 2006.
Methods
Swedish hospitals are required to report principal and contributory diagnoses of all patients to the national Inpatient Register (IPR), which started in 1964, and has been operating on a nationwide basis since 1987. In the current study, data from the IPR and Cause of Death Register were linked using the unique Swedish personal identifier number (a unique 10-digit number). All diagnoses are coded according to the International Classification of Disease (ICD) system, with ICD-8 from 1968 until 1987, ICD-9 from 1987 until 1996, and ICD-10 from 1996 and onward. The positive predictive value (PPV) for discharge diagnosis differs for diagnoses in the IPR but is generally 85–95%. 12 In 2004, the PPV for first-ever stroke was 94%. 13
Study population
All patients aged 18–54 years who survived at least 28 days after a first hospitalization with a principal discharge diagnosis of IS from 1987 to 2006 were identified using ICD-8 and ICD-9 codes of 434 and 436 and the ICD-10 codes I63 and I64. The use of ICD-9 code 433 was considered but not included in the final analysis due to uncertainty of the diagnosis in the IPR (625 men and 344 women). Because the IPR data were not complete until 1987, all IS cases were consistently regarded as a first-time event after a time frame of seven years using data from 1980 and onward. This was performed for each separate year from 1987 to 2006 to ensure that all data were treated as uniformly as possible. A total of 17,149 cases were identified as a first-time IS and were stratified into four time periods according to the admission period: 1987–1991, 1992–1996, 1997–2001, and 2002–2006. Patients were followed at 1–6 months, 6–12 months, 1–2 years, 2–3 years, 3–4 years, and 0–4 years for recurrent IS.
Recurrent IS was defined as either a fatal (death in hospital) or nonfatal event that occurred 28 days after index hospitalization and with at least two days between discharge from hospital and the next admission (Figure 1). Because a death outside hospital ascribed to IS could not be considered as a recurrent event with certainty, we excluded 20 cases where this had occurred. From 1987 to 2006, there was a total of 2432 recurrent IS (1579 men and 853 women) among 17,149 IS patients (Figure 1). A limited number of hemorrhagic stroke events were not included (179 men and 79 women). The main author (KWG) and a stroke physician (CHS) separately reviewed a subsample (100 cases) of all cases to ascertain that the selection process was appropriate, particularly with respect to readmission to a rehabilitation unit for the index stroke, which could be ruled out in all cases.
Flow chart of the selection process of recurrent IS.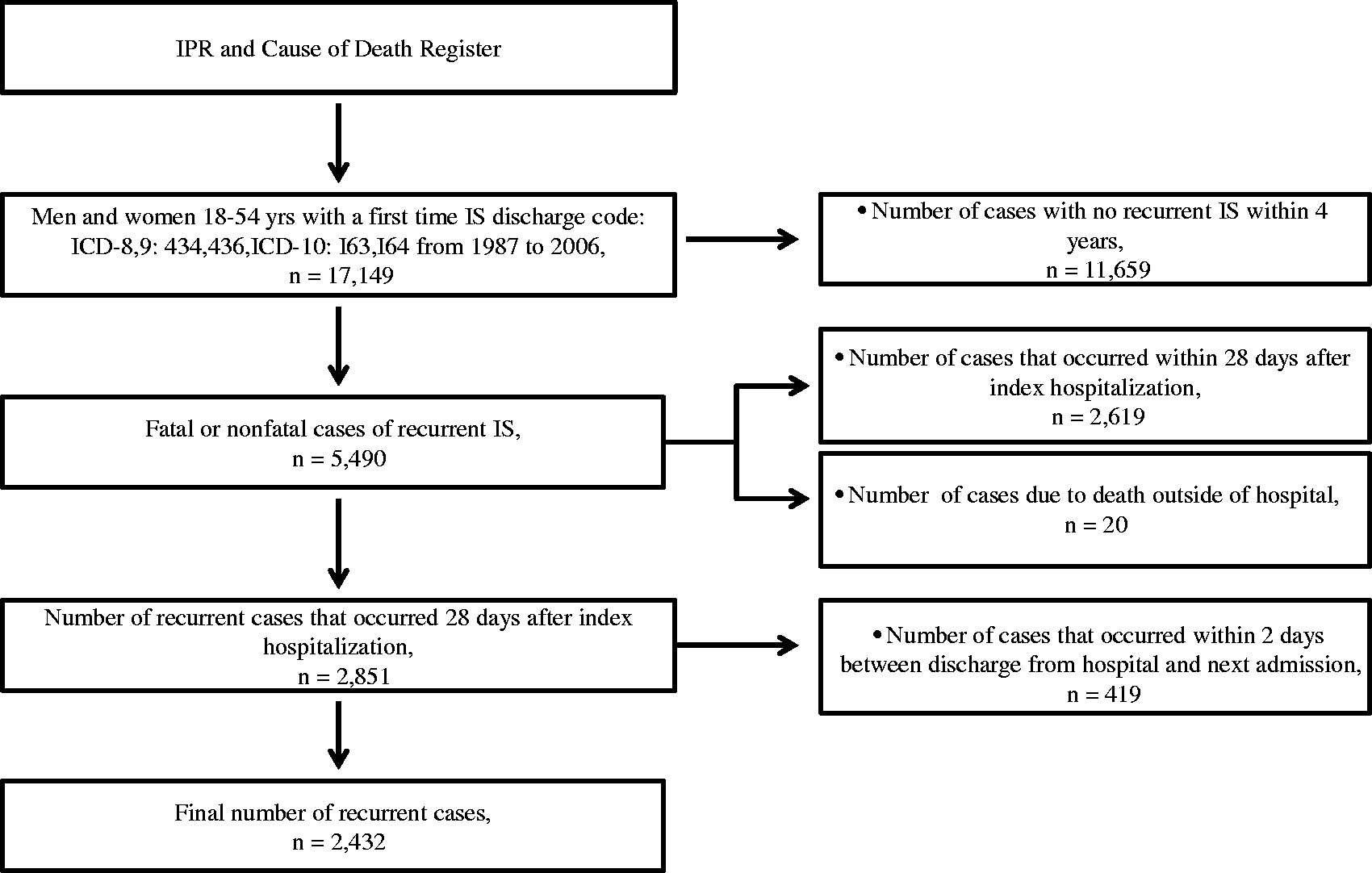
The IPR and outpatient register was used to identify all comorbidities. For surgery codes only IPR was used. Comorbidities over a period extending seven years before and including the index hospitalization for the first IS event were included. The following main or contributory discharge codes were used (ICD-10 (ICD-8; ICD-9)) atrial fibrillation, I48 (427.92; 427D); coronary heart disease, I20–I25 (410–414); heart failure, I50 (427.00, 427.10; 428); hypertension, I10–I15 (401–405); malignancy, C00–C97 (140–208); congenital heart disease, Q20–Q26 (746–747; 745–747); valvular disease, I05–I09 and I34–I35 (394–397; 424) diabetes, E10–E14 (250); and previous acute myocardial infarction I21 (410). Surgical treatment was defined by the following codes: coronary artery bypass grafting 3066, 3067, 3105, 3127, FNA, FNB, FNE, and FNC; and percutaneous coronary intervention: 3080, FNG 00, FNG 02, and FNG 05.
Ethics
For anonymity, all personal identifiers were removed and replaced with a code in the final data set. The present study was approved by the regional Ethics Board of Gothenburg.
Statistical analysis
All calculations were performed with SAS version 9.3 (SAS Institute, Cary, NC, USA) and graphs were produced using R version 2.15.1. Baseline characteristics are presented as proportions and percentages for each individual comorbidity by sex and time period. Incidence rate for recurrent IS was calculated as the number of individuals having recurrence divided by the total follow-up time for each time interval of 1–6 months, 6–12 months, 1–2 years, 3–4 years, and 0–4 years. This was done separately by sex and time period.
Cox proportional hazard regression with 95% confidence intervals (CIs) was used to calculate changes in the risk of recurrent IS over time. The first period (1987–1991) was used as the reference. All final models were adjusted for age with interaction of time to adjust for proportionality. The cumulative risk of recurrence and stroke-free survival (defined as free from recurrent IS and death) were estimated with the Kaplan–Meier method.
Results
Baseline characteristics in 17,149 men and women with a first IS
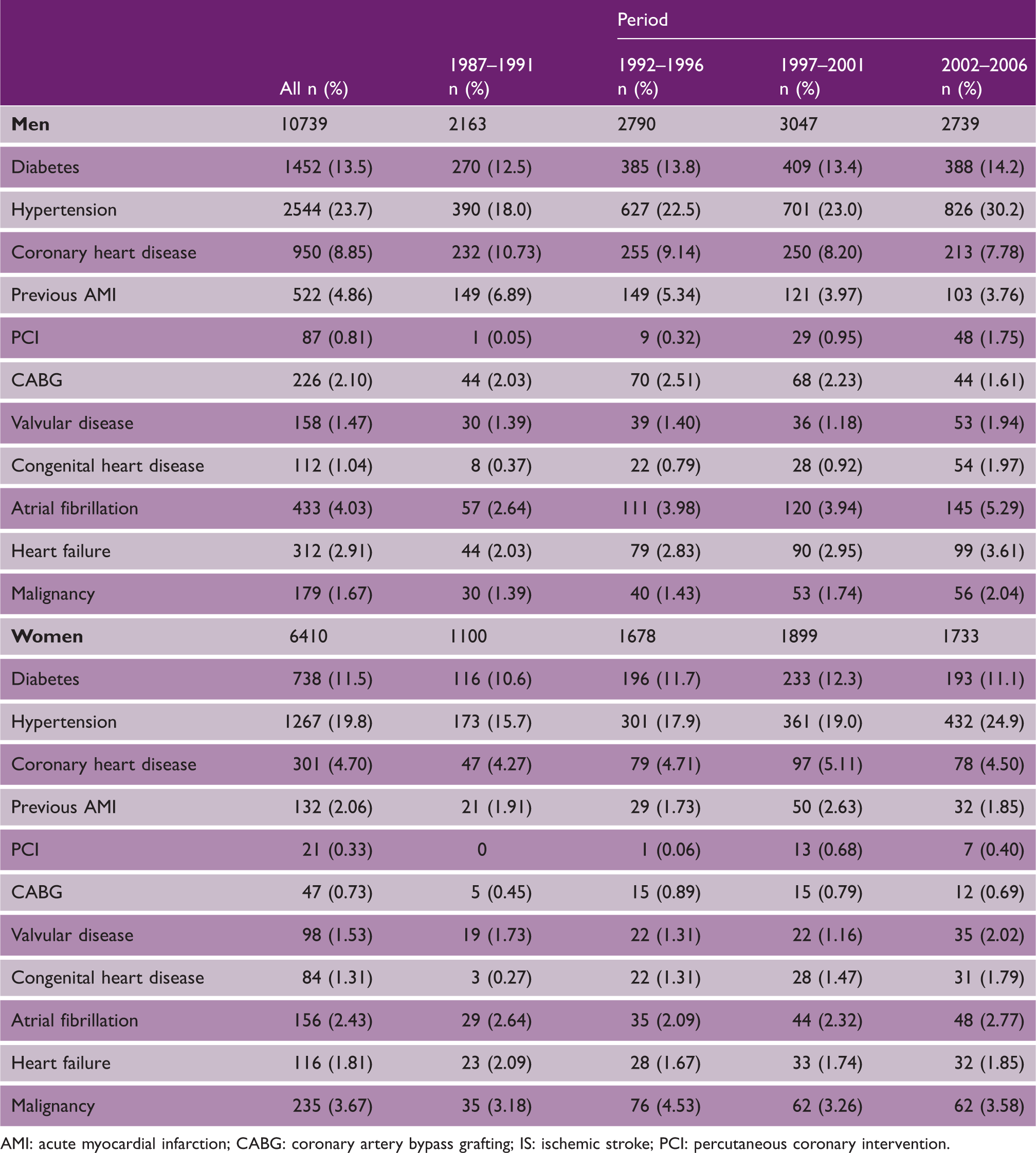
AMI: acute myocardial infarction; CABG: coronary artery bypass grafting; IS: ischemic stroke; PCI: percutaneous coronary intervention.
Temporal trends in the incidence rate of IS and risk of recurrent IS
Four-year hazard ratios for recurrent IS in men and women
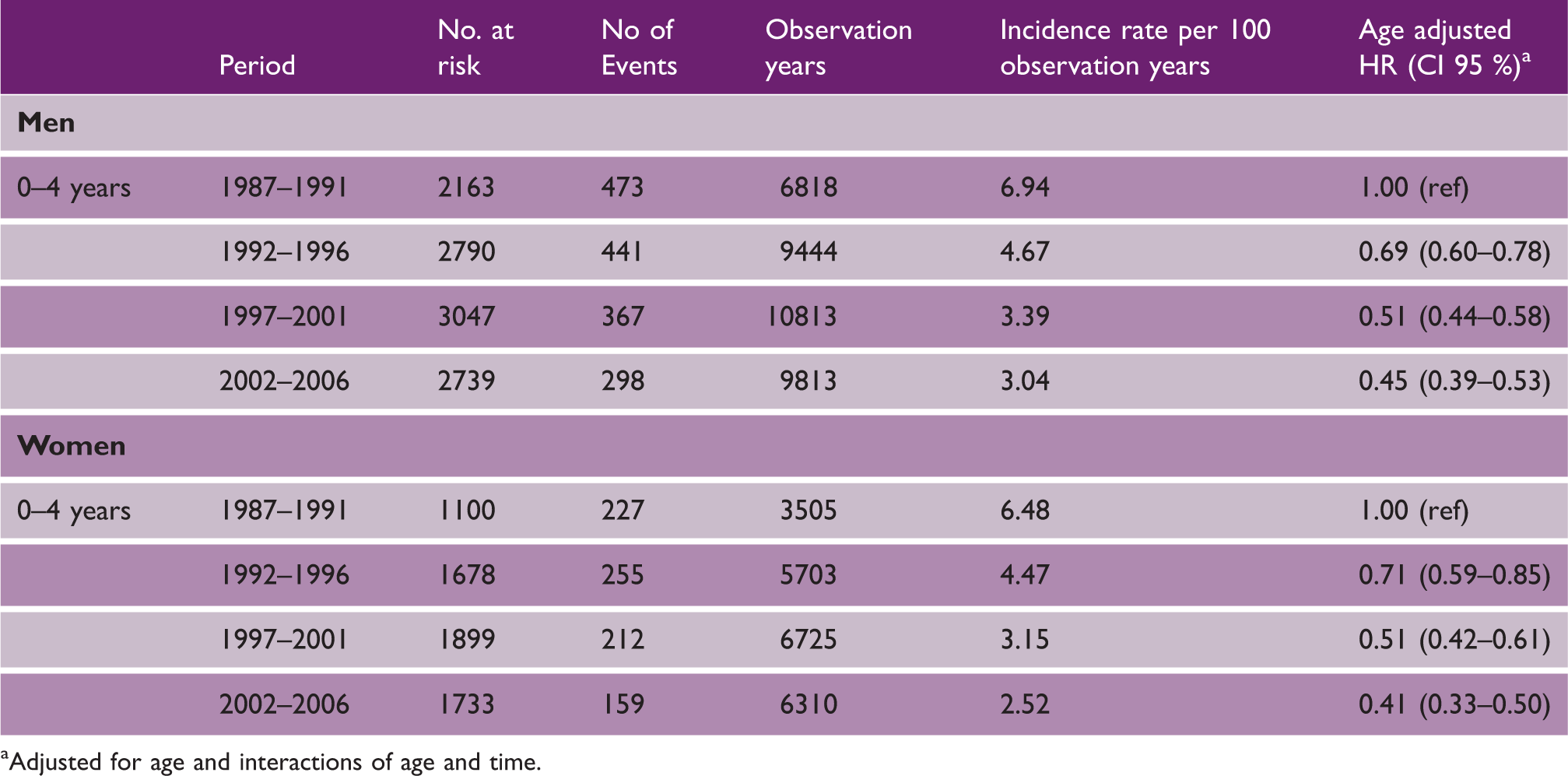
Adjusted for age and interactions of age and time.
Incidence rate and hazard ratios over time of recurrent IS in men and women

Death from other causes is not presented.
Adjusted for age and interactions of age and time.
IS: ischemic stroke.
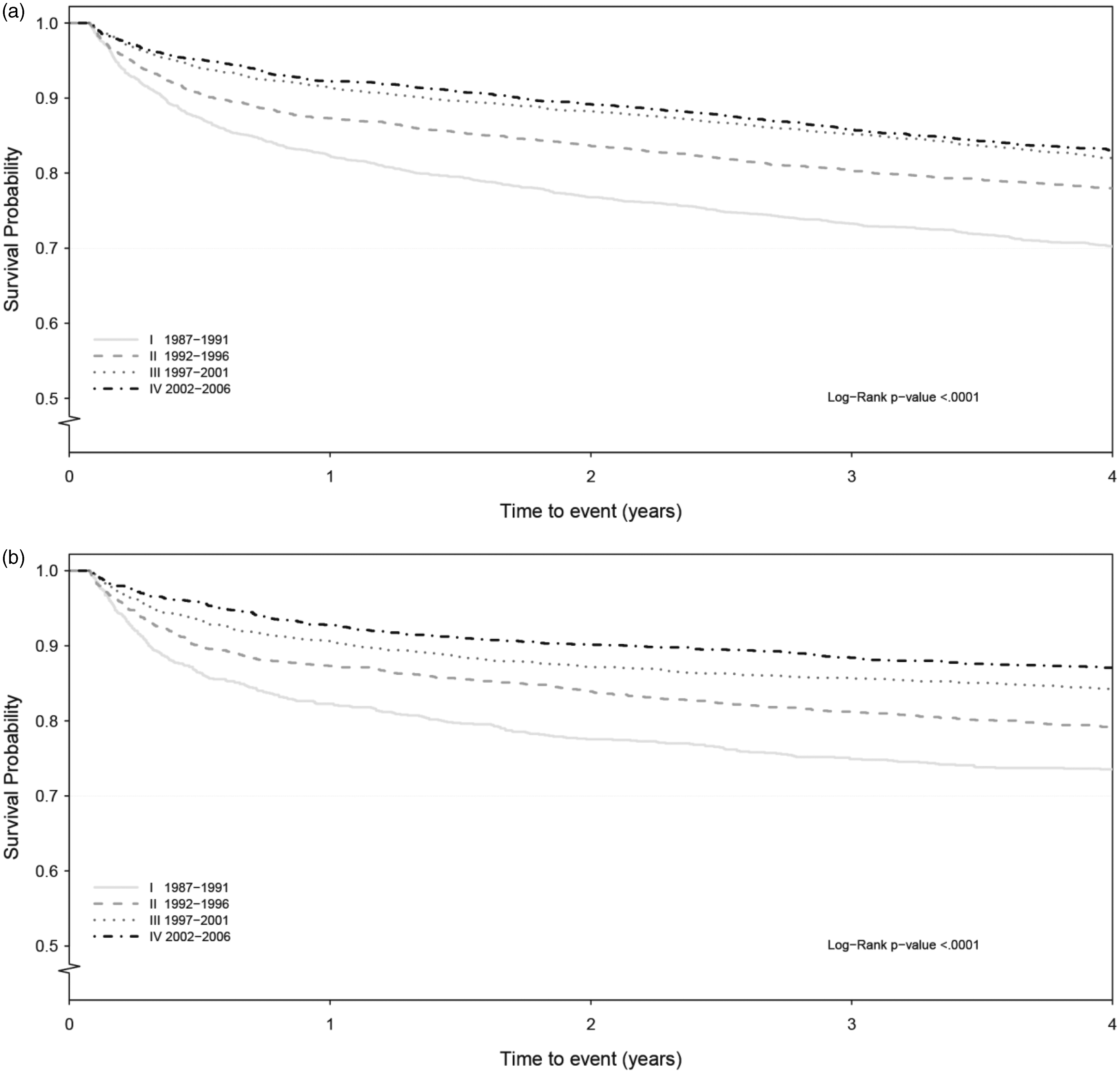
Figure 2 shows the cumulative risk of recurrent IS. The cumulative risk after four years was 11.8% (95% CI 10.55–13.25) in men and 9.8% (95% CI 8.40–11.46) in women during the last five-year period (2002–2006). Stroke-free survival after four years improved from 70.2% (95% CI 68.25–72.11) to 83.0% (95% CI 81.56–84.38) from the first to the last period in men and from 73.5% (95% CI 70.83–76.05) to 87.1% (95% CI 85.40–88.57) in women (Figure 3).
Cumulative risk of recurrent IS in men and women divided by period. (a) Men 18–54, (b) women 18–54. Stroke-free survival in men and women divided by period. (a) Men 18–54, (b) women 18–54.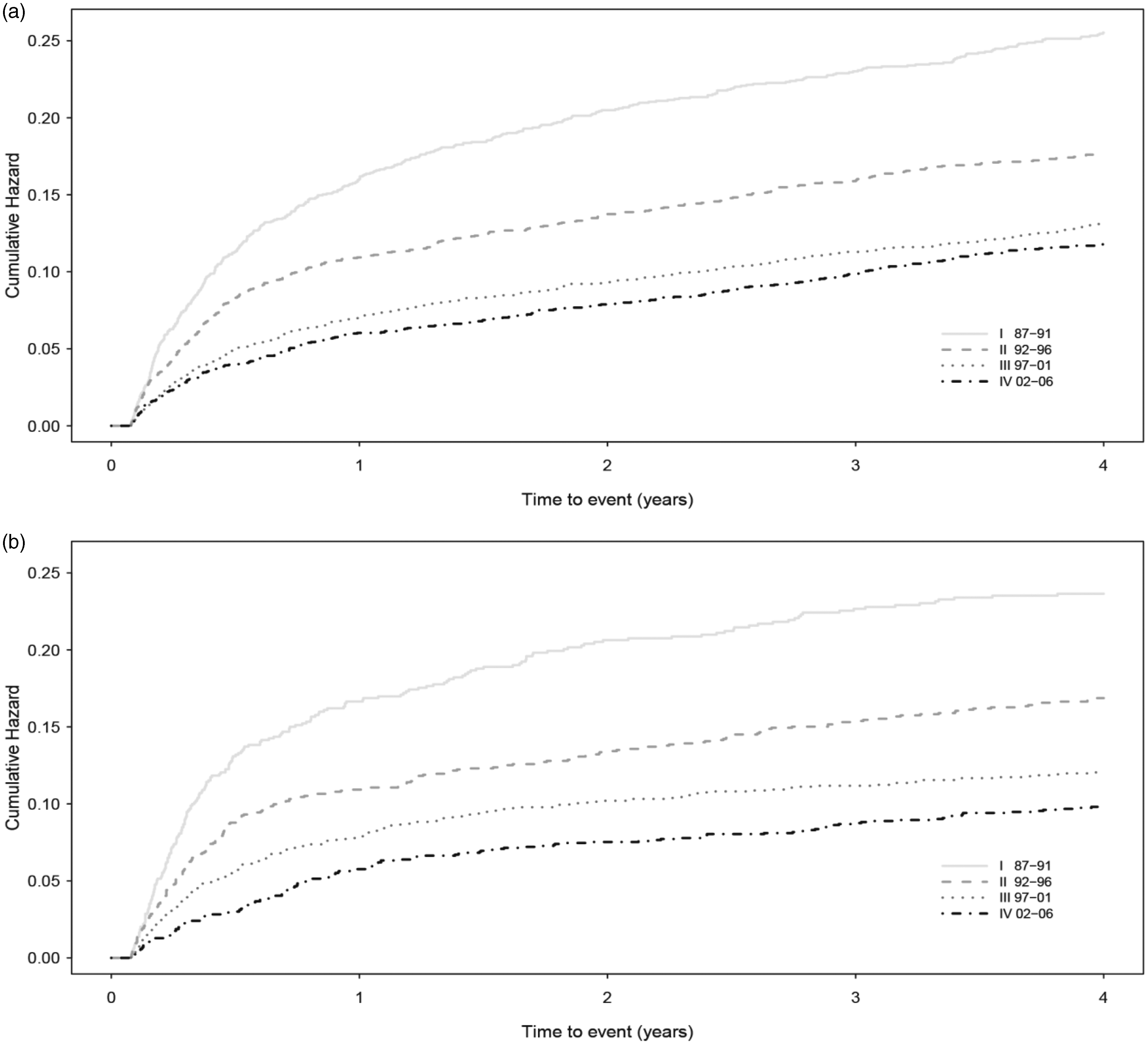
Discussion
Our study showed that the risk of recurrent IS has substantially decreased over time in younger stroke patients. The majority of the decline in risk occurred within the first months, but this effect was largely sustained over the first 2–3 years. The four-year cumulative risk of recurrent IS was 11.8% in men and 9.8% in women during the last five-year period (2002–2006), which is slightly higher than that in other previous studies.10,11,14 In addition, we observed that stroke-free survival improved throughout the study period, but despite these improvements, almost one in six patients had either died or had a recurrent stroke within four years in the last period.
In a report from the population-based Monitoring Trends and Determinants of Cardiovascular Disease Stroke Incidence Registry, a 36% decline in recurrence of stroke was observed in 6700 stroke patients when comparing the periods of 1995–1998 with 2004–2008. 15 In a Scottish study by Lewey et al., stroke patients who were hospitalized in 2001 had a 27% lower risk of recurrence compared with individuals who were hospitalized with a stroke in 1986. 16 However, none of these studies have separately investigated temporal trends in younger IS patients.
To the best of our knowledge, this study is one of the largest studies conducted on recurrent IS in younger patients with IS. In the present study, we found that the four-year risk of recurrent IS declined in both men and women during the study period. Similar trends were observed in most age groups over time except for the youngest group (18–25 years). However, the results should be interpreted with caution since the numbers of recurrent events were limited. Consistent with previous reports, we found that the risk of recurrence of IS was highest during the first several months and up to a year after the index event, regardless of time periods.10,14 After the first year, the rate of recurrence declined and was less than 2% annually during the last period (2002–2006). Additionally, we showed that most of the decrease in risk occurred within the first six months to one year. No improvements were observed at the time intervals of 2–3 years and 3–4 years in men. However, women showed a more complicated pattern, with a significant decline in risk after six months at a time interval of 1–2 years during the last period and until the third period (1997–2001) at 6–12 months and 2–3 years. Again, these results should be interpreted with caution because the number of recurrent IS cases was limited.
There are several possible explanations for the decline in recurrent IS. Improved treatment of cardiovascular risk factors, such as blood pressure levels and smoking rates over time, likely contributed to better survival and less severe stroke cases.17,18 The use of antihypertensive, antiplatelet, anticoagulation, and lipid-lowering drugs in secondary prevention is important for reducing the risk of recurrent IS. 19 Prescription of these medications has increased over time. 20 However, Glader et al. showed that persistent use of secondary prevention medications (e.g. statins and warfarin) decreased within the first two years after IS. 21 A discontinuation of secondary preventive medication after discharge from hospital has previously been shown to be a risk predictor for recurrent events among younger IS patients. 11 Therefore, a poor adherence to medication over time might explain why no significant improvements were observed for men and women at the time interval of 3–4 years. In addition, previous studies have shown that patients who are treated at stroke units have a better outcome than those who are treated at a general ward, 22 especially for those <65 years of age. 23 The positive effect on outcome may be the result of a multidisciplinary team effort with focus on early treatment, better adherence to medications, and rehabilitation in stroke patients.21,24 In Sweden, the number of stroke units has increased for the past two decades and a majority of all patients are now being treated at a stroke unit. 25 It is our belief that improved secondary preventions and better accessibility to stroke units over time have most likely contributed to a better prognosis after an IS.
In comparison to the MONICA and the Scottish study with older patients, we found a slightly larger decline in the risk of recurrent IS over time.15,16 Our result does not necessarily indicate that secondary prevention treatments have been more effective on younger IS patients. Instead, possible explanations for the differences in the risk decline may be that older patients have more complications, comorbidity, higher case fatality rate, and a poorer recovery rate than younger individuals. In addition, younger, as opposed to older, patients may receive a more thorough investigation and careful follow-up.
Overall, an increase in comorbidities was observed throughout the study period. This finding could be explained by better clinical reporting, partly because of changes in financial incitements over the years. 12 Dyslipidemia has previously been reported to be a prevalent risk factor among younger IS patients and a potential predictor for recurrent events.14,26 The validity of lipid disorders in the Swedish IPR is low and most likely underreported. 12 Therefore, it was not included in the present study. The marked increase in hypertension during the study period may partly be owing to changes in definition (currently > 140/90 mmHg) by the World Health Organization. 27
There are some limitations and strengths to our study. A strength of our study is use of the IPR. This registry has near-complete coverage of all hospitalized stroke cases in Sweden which is required to identify the large population needed for IS cases because it is an uncommon diagnosis in younger adults. A limitation of our study is the potential variation in diagnosis of stroke that has been set by physicians over time. However, imaging diagnostics were routinely used throughout the study period, and these largely exclude hemorrhagic stroke and cerebral malignancies. Clinical data on severity of stroke, neurological impairment, functional recovery, medications, and other hospital treatments were not available, which limits the interpretation of the data. Another potential limitation is that there is no specific coding for recurrent IS in the IPR. However, to account for this, we used a strict protocol and selected only those cases that occurred after 28 days, specifying a time window of at least two days after discharge to avoid false-positive cases.
Conclusions
The risk of recurrence in younger patients with IS has decreased over time, particularly during the first year after an IS. However, younger patients have a substantial risk of recurrent IS, and a high proportion still either die or have a new stroke over a four-year period. Because younger stroke patients have more to lose than older patients in terms of quality of life and disability-free remaining lifetime, development of new strategies and improvements of secondary preventions including medications and lifestyle changes should be prioritized. To further improve outcome, improving understanding of the needs of younger stroke patients and recognizing that they might be different from older patients are important.
Footnotes
Authors’ contribution
KWG: Wrote the article and has been involved in the analysis and interpretation of the results.
LB: Involved in the interpretation of the results and made critical revision of the manuscript. Final approval has been given.
CHS: Made critical revision of the manuscript and has given final approval.
SN: Made critical revision of the manuscript and has given final approval.
TZS: Performed the statistical analysis, made critical revision to the manuscript, and has given final approval.
CJ: Made critical revision of the manuscript and has given final approval.
KT: Made critical revision of the manuscript and has given final approval.
AR: Involved in the interpretation of the results and made critical revision of the manuscript. Final approval has been given.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Swedish Research Council (grant numbers 2013-5187 and 2013-4236); the Swedish Heart and Lung Foundation (grant number 2012-0325); and the Swedish Council for Working Life and Social Research (grant number Epilife 2006-1506), the Health & Medical Care Committee of the Regional Executive Board, Region Västra Götaland, and the Swedish state under the agreement between the Swedish government and the county councils concerning economic support of research and education of doctors (ALF-agreement).
