Abstract
Conduct of clinical research involving single patient subjects has a relatively long history. The notion of ‘N-of-1 trials’ first emerged in the 1980s, introducing a method for evaluating the impact of alternately exposing individuals ‘on’ and ‘off’ a particular treatment. In the years since, interest in conventional, randomised, ‘N-of-1 trials’ has fluctuated, though recent advancements in data science, remote monitoring technologies and, more broadly, the ‘personalised’ medicine era has seen renewed engagement with the methodology. Amongst patient cohorts with cancer conduct of such trials has, for the most part, been uncommon, given the methodology is best suited to study of chronic, stable conditions. In recent years however, there has been growing attention within oncology research communities to the idea of ‘N-of-1 trials’. In part, this has been driven by the need to accommodate increasingly granular levels of molecular heterogeneity in cancer research. However, use of the label ‘N-of-1 trial’ has been applied to a variety of research endeavours involving single participants, with varying resemblance to conventional definitions of an ‘N-of-1 trial’. In this paper we review recent cases in which the term ‘N-of-1 trial’ has been operationalised in relation to clinical research amongst adult cancer populations. We then consider both the epistemic and ethical implications of individual-patient research endeavours in oncology. We conclude by noting the phenomenon represents a new frontier for those engaged in ethical oversight, as well as other stake-holders in cancer clinical research, including clinicians, researchers and health-care consumers.
Keywords
In 1986, the New England Journal of Medicine published an article that reported findings observed following exposure of a single individual to alternating periods on and off a medication for the management of asthma (Guyatt et al., 1986). This paper, entitled ‘Determining Optimal Therapy – Randomized Trials in Individual Patients’, is often referenced as a pivotal point in the history of ‘N-of-1 trials’, however other examples of single participant experiments are also evident earlier than this, for example in psychology (Mirza et al., 2017).
From the 1980s onwards, interest emerged in leveraging N-of-1 trial methods, traditionally conceived of as ‘single patient, multi-cycle crossover trial[s]’ (Collette and Tombal, 2015), for refining treatment approaches across a range of chronic, stable conditions; with respiratory, musculoskeletal, and neuropsychiatric conditions (e.g. Attention Deficit Hyperactivity Disorder (ADHD)) featuring most commonly (Gabler et al., 2011; Hawksworth et al., 2024). Such efforts were later facilitated by the development of several coordinated N-of-1 trials ‘services’, in jurisdictions such as Australia, Canada and the United States (US) to reduce burdens on individual clinicians in conducting such studies (Chalmers et al., 2019; Kravitz et al., 2008). In the years since, the popularity of N-of-1 trials has waxed and waned. In part, this has been due to a variety of well-described obstacles to wide-scale implementation, such as resource intensity (including both costs and expertise requirements; Chalmers et al., 2019; Kravitz et al., 2008; Wilmont et al., 2024), as well as regulatory (Weinreich et al., 2017) and/or ethical uncertainties about when to categorise such entities as ‘care’ versus ‘research’ (Cen et al., 2016; Irwig et al., 1995; Mirza et al., 2017; Stunnenberg et al., 2020). In recent years, however, methodological advances in data science, statistics, and technologies for remote patient monitoring (Lillie et al., 2011); as well as refinements in understanding and treatment of disease (i.e. the ‘precision medicine’ movement), have led to a resurgence of interest in this trial design.
In this commentary, we will review traditional conceptualisations of N-of-1 trials, as well as recent interest in so-called ‘N-of-1 trials’ in oncology. We will then outline their ethical and epistemological implications, using adult oncology as an example.
Background – Traditional, randomised N-of-1 trials
The phrase ‘N-of-1 trial’ has been used to describe a variety of different approaches to single-patient research (Porcino et al., 2020). However, as will be subsequently outlined, it is important to note that the term, used correctly, refers to a specific methodological approach. Conventionally defined, an ‘N-of-1 trial’ is a randomised trial consisting of ‘repeated cycles of treatment challenge[s] (e.g. A-B, A-B) in a single participant. . . i.e., a single patient, multi-cycle crossover trial’ (Collette and Tombal, 2015). N-of-1 trials may or may not use placebos alongside active comparators, be open-label or use blinding and can incorporate varying numbers of treatment cycles (Gabler et al., 2011; Hawksworth et al., 2024). This understanding of N-of-1 trials has been described as ‘type 1 evidence’ in evidence-based-medicine hierarchies (Chalmers et al., 2019), given the capacity to generate high levels of internal validity (Wilmont et al., 2024). This is also the design to which consensus statements for reporting and conduct of N-of-1 trials apply (e.g. 2015 ‘CONSORT (CONsolidated Standards Of Reporting Trials) extension for reporting of N-of-1 trials’ (Vohra et al., 2015), and the 2020 ‘SPENT (SPIRIT (Standard Protocol Items: Recommendations for Interventional Trials) extension for N-of-1 trials’ (Porcino et al., 2020)).
Specific methodological requirements for conventional, randomised N-of-1 trials include: (a) that the intervention is directed towards a chronic, relatively stable condition (such that any change in the outcome of interest is most likely due to the intervention rather than evolution of the underlying disease state); (b) the intervention has a short half-life with quick onset/offset of action (to facilitate efficient washout between alternating cycles of treatment, and avoid any carry-over effects of the preceding intervention); (c) there are recognised measures to monitor the outcome/s of interest and (d) the intervention is unlikely to modulate the subsequent biology or behaviour of the disease (such that later treatment cycles are then applied to an altered disease state – which would complicate interpretation of the treatment effect; Chatters et al., 2024; Guyatt et al., 1990; Kravitz and Duan, 2022; Mirza et al., 2017; Nikles et al., 2011). Notwithstanding these ideal characteristics, a variety of more recent analytic techniques have been proposed to try and accommodate both deterioration in disease state (e.g. during evaluation of ‘symptom science’ interventions in palliative care settings (Nikles et al., 2011)), and to handle ‘carry-over effects’ of interventions with slower washout (e.g. by putting less weight on outcomes derived shortly after a switch of treatment cycle; Duan et al., 2013; Kravitz and Duan, 2022).
N-of-1 trials differ from observational ‘case reports’ (and series) by virtue of being hypothesis-driven, and conducted according to prospectively-designed protocols (Ankeny, 2017; Figure 1a). They differ from group ‘crossover trials’, which also involve exposing participants to alternating series of intervention/s, because the goal of cross-over trials is to determine the group-level effect of a study intervention/s, whereas the focus of an N-of-1 trial is to determine the treatment effect in a single individual (Guyatt et al., 1990; Figure 1b). This also distinguishes N-of-1 trials from conventional, non-crossover, parallel-group randomised controlled trials (RCTs), which also aim to estimate population-level treatment effects (Figure 1c).
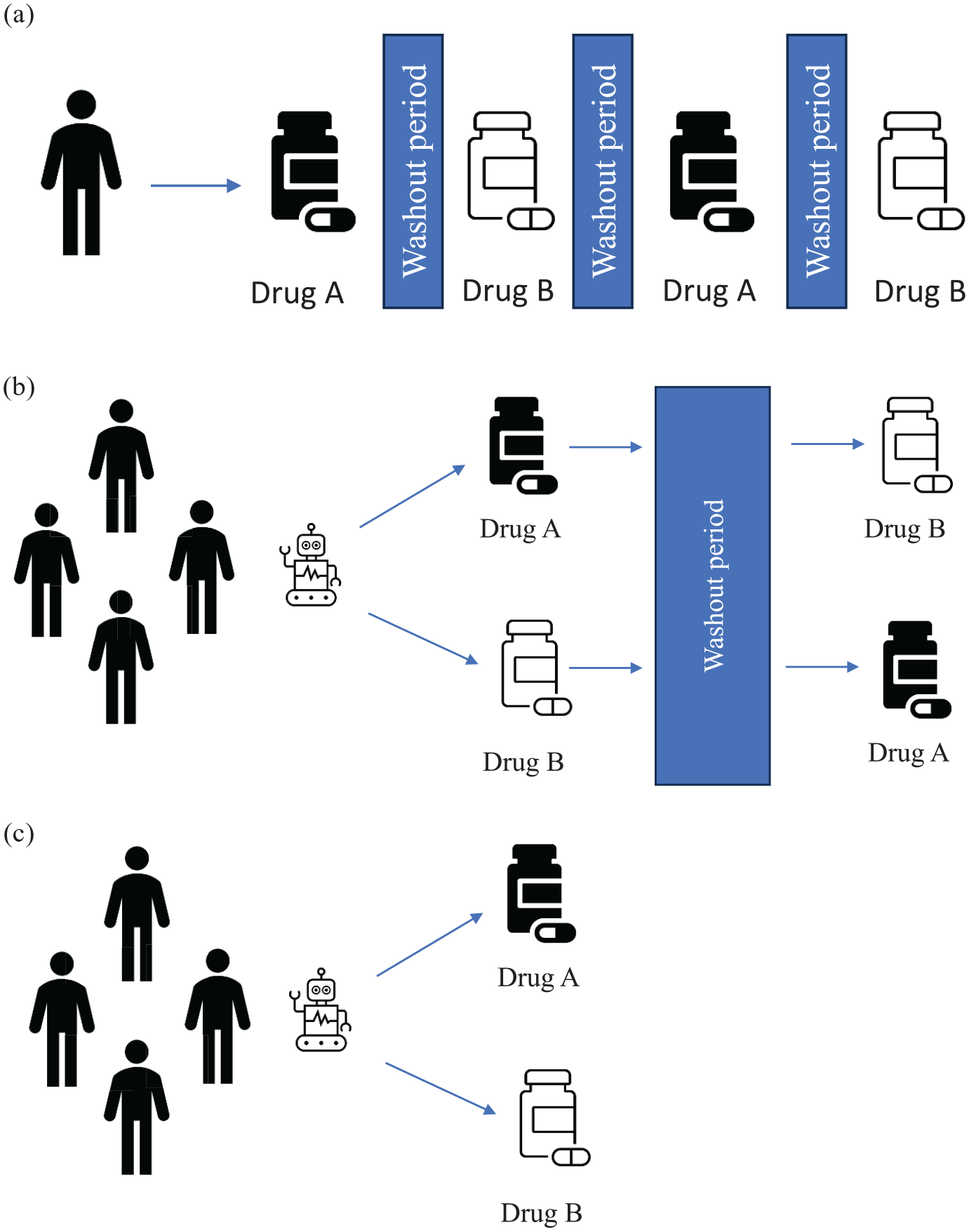
(a) N-of-1 Trial. A single participant receives alternating sequences of treatments which, conventionally, would involve randomisation ± blinding. N-of-1 trials are designed to identify the individual-level effect of an intervention (noting multiple individual N-of-1 trials can be aggregated to identify the population-level effect). (b) Group Crossover Trial. Following recruitment participants are randomly allocated (e.g. by a computer) to one study arm or the other, involving different sequences of treatments. Group crossover trials are designed to identify the population level effect of an intervention sequence. (c) Parallel Group Randomised Controlled Trial (RCT). Following recruitment participants are randomly allocated (e.g. by a computer) to one study arm (which, in a phase 3 RCT, would typically involve an ‘experimental’ and standard-of-care/‘control’ arm). In some cases, it may be possible for participants who experience disease progression on the ‘control’ arm to ‘cross-over’ to the ‘experimental’ treatment. RCTs are designed to identify the population-level effect of an intervention.
N-of-1 trials have a number of potential strengths. In both RCTs and crossover trials, extrapolating from the group to a particular individual can be problematic if, for example, the group-level treatment effect is actually discordant with the effect observed in that specific participant (i.e. the ‘ecological fallacy’; Kyr et al., 2021). Use of an N-of-1 trial design may therefore be particularly advantageous in circumstances when it might be expected that significant ‘heterogeneity of treatment effects’ may exist across a population in response to a given intervention (Chatters et al., 2024). Heterogeneity that arises from genuine ‘biological variability’, is best recognised as a type of ‘variability that carries information’, and so of interest in research (rather than, for example, erroneous variability as a consequence of random ‘noise’, and in need of statistical management) (Kyr et al., 2021). As such, N-of-1 trials also offer a mechanism for identification of ‘exceptional responders’ to a particular intervention (both poor and superior), which may in turn serve as a prompt for further investigation (LeBlanc and Marra, 2015). Finally, conduct of N-of-1 trials may also have a role in generating evidence in settings in which conduct of RCTs are infeasible, for example due to difficulties with recruitment (e.g. rare diseases, palliative care settings (Lillie et al., 2011), or for patient groups less often eligible for RCTs due to comorbidities (Vohra et al., 2015)).
N-of-1 trials have also been described by some to function as ‘promising clinical decision tool[s]’ (Duan et al., 2013) which contribute to the idea of a ‘learning health system’ (Duan et al., 2013). This is because for individual patients (and their clinicians), the N-of-1 design provides the opportunity for objective feedback regarding response to an intervention (Lillie et al., 2011). Further, as clinicians have arguably always conducted informal ‘trial and error’ investigations in routine practice (Schork, 2015; Wilmont et al., 2024), formalising such efforts via N-of-1 trials has been described as an ‘ethical’ step forward (Schork, 2015). For some individuals, participation in an N-of-1 trial may also be perceived more favourably than involvement in other types of research, given they offer exposure to the range of study interventions under investigation (Kyr et al., 2021). This is in comparison to standard RCTs, in which participants are allocated to one or other of the study arms, with opportunity for crossover dependent on the specific RCT.
Whilst a single N-of-1 trial is concerned with understanding the treatment effect in a particular individual, a group of N-of-1 trials may also be able to explore questions such as ‘does the individual treatment effect vary between patients?’ (Chatters et al., 2024) Various methods for aggregating data generated from N-of-1 trials have been proposed including Bayesian and ‘random effect’ modelling approaches (Kyr et al., 2021; Nikles et al., 2011; Punja et al., 2016). Data generated from a series of N-of-1 trials may be advantageous over conventional RCT data for several reasons. First, aggregation of multiple N-of-1 trials has the potential to offer evidence of an intervention’s effect at both the individual patient as well as population level (Punja et al., 2016). Second, the fact that individuals act as their own controls in N-of-1 trials means that smaller sample sizes are required than for standard, parallel-group RCTs (Nikles et al., 2011). Finally, it may be noted that N-of-1 trials represent just one aspect of recent and broader interest in attending to individual-level treatment effects in clinical research. For example, recent efforts amongst statistical methodologists to disentangle treatment effects pertaining to individuals participating in RCTs are notable (Lamont et al., 2018), including via leverage of artificial intelligence (AI)-assisted techniques (Jaki et al., 2025).
Recent interest in ‘N-of-1 trials’ in cancer medicine
Historically, N-of-1 trials have been considered of limited value in oncology because cancer is usually a progressive condition with limited opportunities for ‘trial and error’. As such, inclusion of placebos or ‘washout periods’ in which agents are withheld may be undesirable (Duan et al., 2013), and perhaps even unethical (Selker et al., 2022). Furthermore, random assignment of alternating treatments may be inappropriate where treatment sequencing is known to be important because exposure to earlier systemic therapies is recognised to influence the subsequent biology and/or behaviour of an individual’s cancer (e.g. by exerting selective pressures on different clones of cells). Finally, in the absence of disease progression or treatment toxicity it would not be usual, and potentially problematic to switch or cease an individual’s existing systemic therapy. Consequently, N-of-1 trials have not traditionally been considered possible for the evaluation of systemic therapies in oncology. Even for the evaluation of supportive care and/or ‘symptom science’ interventions this type of trial design has been utilised infrequently in oncology, with a review of published randomised N-of-1 trials from 2011 to 2023 (Hawksworth et al., 2024) identifying only three publications (Kronish et al., 2019; Mitchell et al., 2015; Nikles et al., 2015) pertaining to cancer populations.
In recent years there has been growing interest in the possible applications of an ‘N-of-1 trials’ approach in cancer medicine, ranging from ‘in passing’ mentions (Adashek et al., 2023; Catani et al., 2017; Fountzilas et al., 2022, 2024; Fountzilas and Tsimberidou, 2018; Gupta et al., 2017; Subbiah, 2023; Subbiah et al., 2025; Zardavas and Piccart-Gebhart, 2015) to dedicated commentaries on the topic (Collette and Tombal, 2015; Gouda et al., 2023; Kyr et al., 2021; Silvestris et al., 2017). This has arisen in large part because of changes in understanding and classification of cancers. Advances in the molecular understanding of cancer, including via an ever-expanding plethora of ‘omics data (i.e. genomics, proteomics, transcriptomics, metabolomics etc.; LeBlanc and Marra, 2015), has shed light on significant heterogeneity in cancer. Such heterogeneity exists between patients (even if they share the same tumour type), as well as over the course of an individual patient’s experience of having cancer (i.e. temporally, across an individual’s disease trajectory, or spatially between primary and metastatic tumour sites). In some cases, this has prompted a shift away from tumours being classified and treated according to histologic site-of-origin in favour of molecular classifications (Subbiah et al., 2024b). In 2017, for example, the immunotherapy pembrolizumab became the first agent to receive ‘tumour agnostic’ approval by the US Federal Drug Administration (FDA), for patients with tumours characterised by microsatellite instability-high (MSI-H) or mismatch repair deficiency (dMMR), and five further ‘pan-cancer’/‘histology-independent’ approvals have since followed (including both immuno- and targeted therapeutics, directed at specific genetic and other molecular alterations; Subbiah et al., 2024b). Advances in molecular understanding of disease have prompted new nosological classifications of some cancers, in which, as one commentator has claimed, ‘soon every patient will have an ultra-orphan disease’ (Berry, 2015). These changes have been especially prominent in some tumour areas, a notable example being non-small cell lung cancer, which is now sub-divided according to expression of a variety of therapeutically actionable oncogenic driver mutations and/or other biomarkers (e.g. EGFR, ALK, ROS1, RET, HER2, KRAS G12C gene alterations, PDL-1 expression, amongst others). The complexity of such heterogeneity only increases upon disease progression, given the wide range of possible resistance mechanisms (and permutations) that may emerge following exposure to systemic anti-cancer therapies (Catani et al., 2017).
Division of cancer, at diagnosis and progression, into smaller and smaller categories makes it increasingly difficult to conduct traditional large-scale RCTs. In this setting, some trialists have called for a reorientation in the way in which cancer research and drug development is undertaken, arguing that ‘next-generation trials’ (Fountzilas et al., 2022) should move away from ‘trials that emphasize optimizing benefit for the group as a whole to those trials that optimize benefit for individual patients’ (Fountzilas et al., 2024). Conceived otherwise, there should be a shift in focus from ‘drug-centred’ to ‘patient-centred’ trials (Fountzilas et al., 2022), whereby instead of matching the right individual to the trial the focus should instead be on finding the right agent for the individual (based on their cancer’s molecular and/or biomarker profile, and other characteristics; Fountzilas et al., 2024). In this vein, advocates of N-of-1 trials in oncology have described such entities as ‘the epitome of precision medicine’ (Fountzilas et al., 2024).
Clarifying the meaning of so-called ‘N-of-1 trials’ in oncology
Importantly, recent calls for N-of-1 trials in oncology have generally not referred to conventional, randomised N-of-1 trials but rather to a broader range of research methods that centre on the individual patient. In this section, we will consider the various types of activities to which the ‘N-of-1 trial’ label has been applied in the academic oncology literature. Table 1 offers further detail for those interested in the specific scientific and clinical features of these endeavours, and the associated ethical complexity. Importantly, only the first study (
Examples of studies in oncology to which the label ‘N-of-1 trial’ has been applied.

PDL-1 IHC: Programmed Death Ligand-1 immunohistochemistry; TMB: tumour mutation burden; MSI: microsatellite instability; ctDNA: circulating tumour DNA.
Functional Assessment of Chronic Illness Therapy-Fatigue Scale (FACIT-F) and the Wu Cancer Fatigue Scale (WCFS).
Defined in study as ≥ 50% 2-year cancer-associated mortality.
The ‘matching score’ consisted of the number of ‘matched’ alterations (excluding variants of unknown significance) divided by total identified alterations (Sicklick et al., 2021).
Per the treatment-naïve study – two-drug combinations generally started at ~50% of usual dose, and three-drug regimens ~33% of usual dose (Sicklick et al., 2021).
In recognition of the fact that the term ‘N-of-1 trial’ is being operationalised with reference to a range of research entities, Selker et al. have suggested the need to consider the possibility of different categories of ‘N-of-1 trial’ (Selker et al., 2023). According to their proposal, ‘Type 1’ refers to a conventional, randomised N-of-trial, whereas a ‘Type 2 N-of-1 trial’ consists of a ‘single-cycle pre-post design’, in which observation is undertaken before and after administration of a novel agent in one individual (Selker et al., 2023). Importantly, a ‘Type 2’ design does not necessarily require the condition to be chronic and stable, and is therefore best suited to evaluation of novel, ‘personalised’ therapeutics in individuals with conditions that are imminently life-threatening, and for which no other approved therapies are available (Selker et al., 2023). In articulating their proposal, Selker et al. consider the example of individualised genetic interventions (e.g. oligonucleotide therapy), arguing that having a paradigm for conceptualising ‘Type 2 N-of-1 trials’ will become increasingly important to patients, researchers, clinicians, regulators and funders (Mullard, 2020; Woodcock and Marks, 2019), particularly in relation to rare and ultra-rare diseases (Selker et al., 2022).
An example of Selker et al.’s ‘Type 2’ variant is described in Table 1,
Other instances in which the label ‘N-of-1 trial’ has been operationalised in oncology include both single-case and group research programmes involving development of novel combination therapy approaches customised for individual recipients. Regarding the former, Subbiah et al. report on the results of their experience tracking the development of resistance to selpercatinib, in an individual with RET-altered advanced medullary thyroid carcinoma (Subbiah et al., 2024a; Table 1,
Epistemic implications
The increasing emphasis on ‘N-of-1’ research studies in oncology raises two key epistemic questions. These are: how do we categorise such entities as forms of evidence in relation to and comparison to other forms of evidence, and how do we make use of insights from such studies in subsequent research?
Whilst conventional, randomised ‘N-of-1 trials’ (i.e. Table 1,
Most evidently, as noted above, N-of-1 studies support ‘precision’ and ‘personalised’ medicine, where the goal is ‘to achieve improved individual-level description, prediction and, ultimately, control for that specific [N-of-1] unit’ (Hekler et al., 2019). Heckler et al advocate for conceptualisation of a ‘small data paradigm’, focussed on specific ‘N-of-1 unit[s]’, which they argue is particularly advantageous for conditions characterised by high degrees of ‘complexity’, ‘multi-causality’, and which are ‘dynamic’ over time (Hekler et al., 2019). Their proposal coheres with the notion, outlined earlier, that diversity among individuals should not be discounted as mere ‘anecdote’ (LeBlanc and Marra, 2015) or statistical ‘noise’, but rather recognised as ‘variability that carries information’ (Kyr et al., 2021). Whilst there would be limitations to solely relying on a ‘small data paradigm’ (e.g. limitations in ‘transportability’ of knowledge beyond the N-of-1 unit, with a need to continuously ‘relearn’ for each unit (Hekler et al., 2019)), this is not Hekler et al.’s proposal. Rather, they are advocating for greater appreciation of the necessity for both ‘small’ and ‘big’ data methods, in assembling ‘consilient knowledge of complex health phenomena’ (Hekler et al., 2019). Possible examples of a combined ‘small’ and ‘big’ data approach might include leveraging of the engineering concept of ‘digital twins’ (Mollica et al., 2024; Subbiah, 2023). Here, pertinent biological, molecular and other characteristics of an individual patient are incorporated in a ‘digital twin’ model which then forms a digital platform for conducting various computer-based simulations to test the utility of different drug candidates (i.e. ‘in silico’ testing). The more promising approaches to treatment can then be evaluated ‘in vitro’ in patients themselves (Björnsson et al., 2019).
Proposals such as Hekler et al.’s raise important questions about the need for ‘generalisability’ in order for a study to be considered ‘research’. This matters because a commonly proposed method for distinguishing between ‘care’ and ‘research’ is that clinical research aims to produce findings that have sufficient ‘external validity’ that conclusions can be drawn that may benefit future patient populations (Johnston et al., 2025). The emergence of ‘N-of-1’ research approaches force us to ask whether generalisability must be a necessary feature for every clinical research methodology. In an attempt to resolve this in relation to research into purposely-engineered and genuinely individualised interventions for ultra-rare genetic disorders (‘Type 2 N-of-1 trials’ per Selker et al.’s taxonomy (Selker et al., 2023)), Johnston et al. assign the label ‘research-treatment hybrids’ (Johnston et al., 2025). They note that the goals, methods, funding and oversight of such entities may reflect both ‘care’ and ‘research’ (Johnston et al., 2025), and simultaneously produce generalisable knowledge and aim to benefit the individual patient (Johnston et al., 2025).
In grappling with the applicability (or lack thereof) of the notion of ‘generalisability’ in the development of the individualised antisense oligonucleotide, ‘Milasen’ (Kim et al., 2019), Johnston et al. (2025) invoke the concept of ‘scope validity’. Originally proposed by philosopher Lara Keuck, the idea here is instead of focussing on ‘external validity’, and a ‘totalising assessment of generalisability’, the objective instead is ’to specify targets where the results of a study will be locally generalisable’ (Keuck, 2024). (Keuck, 2024). Accordingly, regarding the ‘Milasen’ example, Johnson et al. (as well as others (Kane et al., 2021)) conceptualise the experience described as a proof-of-concept for a novel ‘algorithm’ for therapeutic development (Johnston et al., 2025). In this manner, returning to oncology, it could be argued that prior reporting of the methodological approach to the development of seletrectinib (Drilon et al., 2017), afforded ‘scope validity’ for the approach subsequently taken with selpercatinib (Subbiah et al., 2018; see Table 1, footnote to
Ethical implications
Beyond epistemic considerations, N-of-1 studies in oncology also introduce a range of ethical issues. Some of these overlap with those that have been described previously in relation to traditional, randomised N-of-1 trials, for example ongoing questions surrounding the nature of ethical oversight that is most appropriate (Cen et al., 2016; Crowden et al., 2015; Irwig et al., 1995; Stunnenberg et al., 2020). Where N-of-1 studies diverge from conventional, ‘Type 1’ designs, new challenges arise and it is not clear that traditional mechanisms for ethical review are yet prepared for engagement with such issues. We will now briefly review some well-known concepts in research ethics and the nuances of their application in relation to N-of-1 studies in oncology.
Scientific merit and social value
A central ethical question raised by N-of-1 studies, and arguably the one that underpins all subsequent questions, surrounds the scientific merit of such undertakings and their capacity to advance science and practice in oncology. In the pivotal paper ‘what makes research ethical?’, ethicists Ezekiel Emanuel et al., consider ‘scientific’ and/or ‘social’ value as a moral requisite, underpinned by the importance of ensuring fairness with respect to allocation of available resources (Emanuel et al., 2000). In clinical research resources need to be carefully allocated including research funding, researcher expertise (alongside other roles, such as care provision, teaching and/or competing research involvement and time) and availability of participants (particularly if the target population is rare). Challenges related to resource intensity for the design and conduct of traditional N-of-1 trials have been well described (Kravitz et al., 2008), and it may be anticipated that similar considerations would apply to other types of N-of-1 studies, including those outlined in oncology.
Assessment of social value and scientific contribution will presumably differ between and within stakeholder groups, who will have different understandings of the concept and different ideas about epistemic value. In this regard, Selker et al. note the critical importance of broad and integrated multidisciplinary engagement, including with researchers, clinicians, ethics committee members, regulators and funders, and consumer representatives, to ensure that N-of-1 studies are ‘useful’ and ‘sustainable’ in the ‘healthcare ecosystem’ (Selker et al., 2022, 2023). To this end, it is noteworthy that, to date, discussion in the literature in favour of ‘N-of-1 trials’ in oncology have emerged from several, at times overlapping, author groups, and in reference to some tumour streams more so than others.
Relationship of N-of-1 studies to research, care and ‘innovative care’, and implications for ethical oversight
The question raised above as to whether N-of-1 studies are best characterised as ‘research’ or ‘care’ has ethical as well as epistemic implications because it determines when formal ethical oversight is required, and what kinds of oversight may be necessary. It could be argued, for example, that informal ‘trials of therapy’ conducted by clinicians in the context of routine care can avoid formal institutional ethical review, whereas activities more on the ‘research’ end of the spectrum should proceed to institutional review board (IRB) evaluation per other clinical research methodologies (Figure 2). Amongst traditional, ‘Type 1’ N-of-1 trials this question remains the subject of longstanding debate (Cen et al., 2016; Crowden et al., 2015; Stunnenberg et al., 2020), including frustration amongst some who have argued that, at times, ‘unnecessary’ reviews and lack of consensus on this matter contribute to unhelpful roadblocks to conduct of such trials (Chalmers et al., 2019; Kravitz et al., 2008). However, notwithstanding the challenges of disentangling research and care in oncology more generally (Heynemann et al., 2023), it is our contention that each of the cancer-related ‘N-of-1’ studies discussed here are best categorised forms of research. (Of note, ethicists Kane et al. share this view in their description of similar N-of-1 research studies applied to genetic disorders, which they term ‘pre-post trials’ (Kane et al., 2021)). For each of the entities we have described (Table 1,
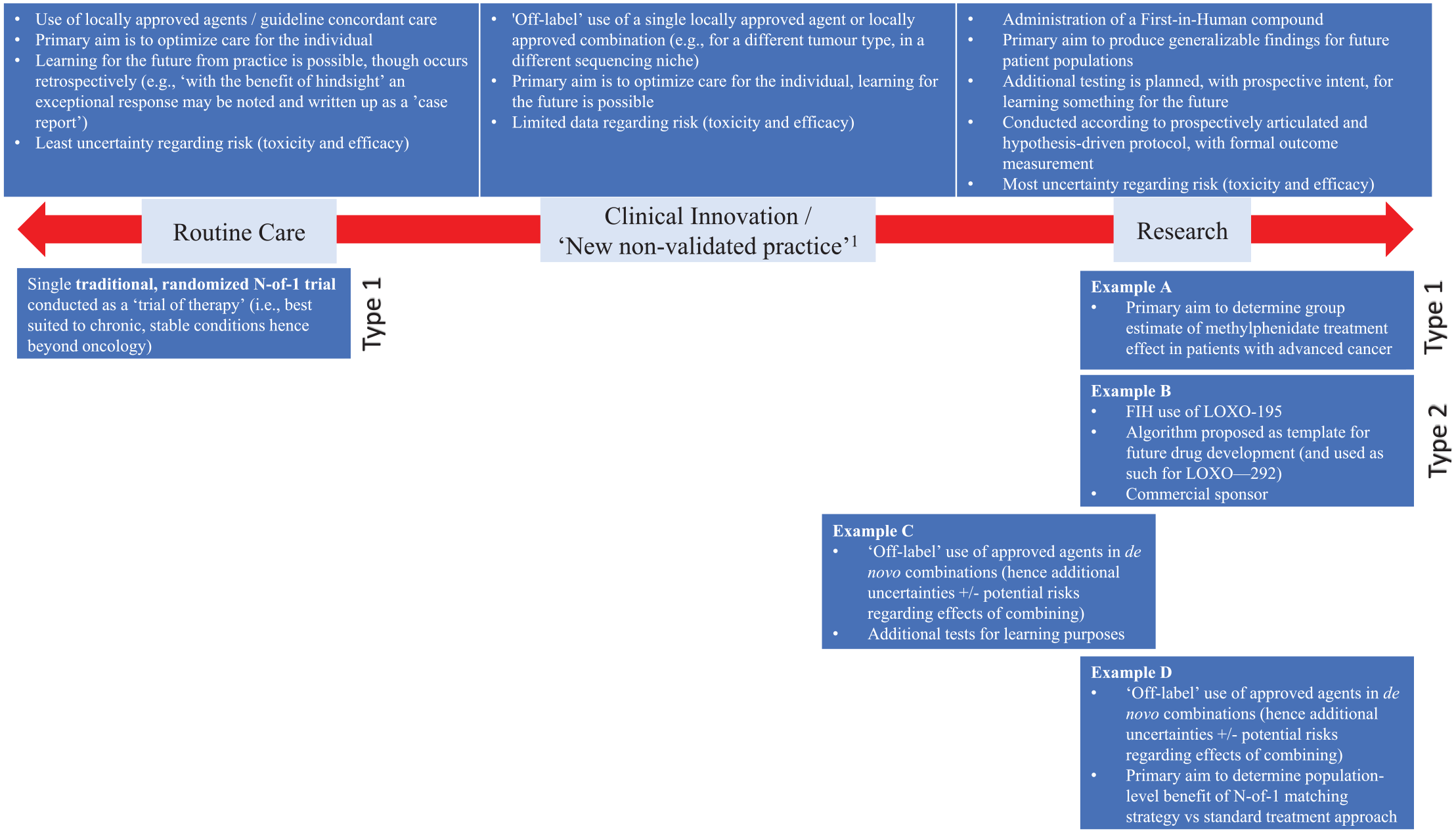
The overlapping domains of routine care, clinical innovation, and research in relation to ‘N-of-1’ studies in oncology.
The prospective goal for learning, albeit in varying ways, also distinguishes the aforementioned ‘N-of-1’ entities in oncology from usual conceptualisations of ‘clinical innovation’ (also referred to as ‘innovative care’ and/or ‘new non-validated practice’ (Mastroleo and Holzer, 2020; Wiersma et al., 2023)). In ‘innovative’ care, contrary to ‘routine’ care, interventions are administered in a novel fashion, however the primary aim remains to optimise care for the individual patient (Mastroleo and Holzer, 2020; Wiersma et al., 2023). While ‘off-label’ drug use, whereby agents are prescribed outside their locally-approved indications, is a type of clinical innovation (Wiersma et al., 2023) and the ‘N-of-1’ study described in Table 1
Returning to the nature of oversight required for ‘N-of-1’ research studies in oncology, the next question that arises is whether traditional IRBs are presently equipped for the task, or alternative oversight mechanisms are required. Whilst prior research has sought to map understanding and attitudes towards conventional N-of-1 trials amongst IRB members (Cen et al., 2016; Samuel et al., 2016), we are not aware of analogous research in oncology. Three particular functions of IRBs merit special attention in relation to N-of-1 studies in oncology: ensuring adequate informed consent, protecting participants from risk, and identifying conflicts of interest.
Informed consent
Voluntary and informed consent, as well as the opportunity to withdraw participation later on, are both considered moral requisites of clinical research (Emanuel et al., 2000). For N-of-1 studies, attention to suitable mechanisms for withdrawal will require particular attention by IRBs, given that participants know (rightly) that the research ‘depends on’ them and they may feel a sense of loyalty to the researcher, particularly if also their treating clinician. To the extent that N-of-1 research entities involve novel undertakings, but simultaneously tailoring, in various ways, to the characteristics of the individual participant, they also raise questions as to how informed consent procedures should be framed. Previously, we have written on the notion of the ‘therapeutic misconception’ in the increasingly intertwined research-care interface in oncology (Heynemann et al., 2024). We have noted, as have others (Subbiah and Kurzrock, 2025; Weber et al., 2015), that in some cases, it may be reasonable for researchers, patients and clinicians to conceive of participation in clinical research as, simultaneously, opportunities for ‘treatment’ (e.g. in the advanced cancer setting when standard options have been exhausted). Notwithstanding, in the N-of-1 context, it will be important that the novelty of the undertaking being offered is clearly articulated, and there is recognition that uncertainty (regarding risks and efficacy) is greater than would be the case in routine care scenarios (Figure 2).
Evaluation of risk
Another challenging role for IRBs is to determine what constitutes ‘reasonable’ risk. In the N-of-1 research context, several unique challenges may be anticipated. First, in cases where an N-of-1 design is leveraged as part of early ‘first-in-human’ clinical translation efforts (e.g. Table 1,
For conventional N-of-1 trials, beyond oncology, particularly those undertaken outside medicalised settings (Duan et al., 2022), involving the participant in decisions regarding aspects of the N-of-1 trial design, outcomes of interest and recognising the participant as a genuine partner and/or collaborator in research is considered best practice (Selker et al., 2022). Whilst the degree to which this is possible evidently depends on complexity of the trial design and understanding (as well as interest) of the specific individual, this is nonetheless also a pertinent consideration for IRBs considering other types of N-of-1 studies. This means that during their deliberations, IRBs will need to be cognisant of the fact that tolerance of varying degrees of risk differs between individuals, as well as across individuals’ lifespans. For example, following a cancer diagnosis, particularly when no standard-of-care treatments or conventional trial opportunities remain, empirical (and anecdotal) data would suggest that individuals are more likely to accept higher degrees of risk (Gaskin et al., 2004). Whether standard IRBs are currently equipped to manage such nuance, and whether more clinically-oriented oversight committees would be better placed to do this, will be important questions for researchers, regulators, patient advocates and clinicians to consider going forward. To this end, ensuring efficiency of review will also be important, noting that many individuals considering participation in such studies will, presumably, be doing so in the setting of limited and/or no other standard therapeutic or other clinical trial options.
Identification (and appropriate management) of possible conflicts of interest
Beyond review of risk to participants, it may also be noted that receipt of independent and peer review may also afford desirable protections to clinician-investigators and/or sponsors of such entities (especially, for example, in the event of subsequent unfavourable treatment responses, or unanticipated adverse events; Kane et al., 2021). The importance of independent review is also important insofar as identifying conflicts of interest held by clinician-investigators undertaking N-of-1 studies (whether real and/or perceived conflicts). This is especially important in N-of-1 studies in which proximity to and/or involvement of industry may be more notable. For example, conduct of an N-of-1 study might be made possible by a clinician-investigator procuring access, off-label to an agent, via a pharmaceutical company. Alternatively, industry partners may be involved in N-of-1 studies involving development of novel agents, in which commercial implications follow should an agent be ‘successful’. To this end, ensuring that participants are appraised of any potential conflicts of interest as part of the informed consent process, will be an important function of IRB oversight.
The importance of terminology
While we, and others (Gouda et al., 2023; Kyr et al., 2021), have attempted to taxonomize the kinds of cancer-related studies referenced as ‘N-of-1 trials’ in the literature to date, there is ongoing confusion and inconsistency in the way the phrase is used. In the absence of definitional clarity, it is possible people may use the phrase to claim the advantages that come by calling something a ‘trial’, as well as, potentially, invoke the association with ‘type 1 evidence’ that rightly belongs only to conventional, randomised N-of-1 trials. In the contemporary ‘precision’ and ‘personalised medicine’ era, it may also be anticipated that calling something ‘n-of-1’ will carry more weight amongst educated consumers (probably more so than other terms such as ‘pre post trial’ (Kane et al., 2021) or ‘single-patient experimental design’). Consequently, we urge those commenting on ‘N-of-1’ research studies to be precise with their language. Use of ‘single-participant experimental design’ may be helpful to articulate to prospective participants regarding designs involving ‘first-in-human’ elements. Whilst it could be debated whether or not the entities described should even be labelled as ‘experiments’ (rather than, for example, merely ‘an exhaustive data collection regarding a single patient’s journey’ (Collette and Tombal, 2015)) we suggest that their prospective, hypothesis-driven nature merits this label. Otherwise, at a minimum, denoting something as ‘type 2’, as Selker et al. propose, at least signals difference in some way to the conventional definition (Selker et al., 2022, 2023).
Conclusion
Whilst it might be anticipated that N-of-1 studies in oncology will remain infrequent compared to more conventional trial designs, and predominantly used as a ‘last resort’ in the setting of rare tumour types, or uncommon situations (e.g. extensive pre-treatment), navigating the ethical oversight of such entities nonetheless represents a new frontier. As noted, broad engagement and education of stakeholders, across disciplines and from diverse backgrounds, will be important in guiding further theoretical and empirical work needed to conceptualise the boundaries of such entities. To this end, not discussed in this paper, but clearly salient for discussion going forward is how the concept of equity of access to participation in N-of-1 research entities might be understood, noting such undertakings are most likely to occur at academic centres where resources and expertise are concentrated. In future it may also be that other single-participant design variants emerge, with other nuances to consider. For example, beyond scope of this paper, but bearing some parallels in the sense of being a form of single-participant research, are examples of clinician-investigators undertaking ‘self-experimentation’ (Forčić et al., 2024). Such instances are likely to be rare, though often attract wide interest, and raise unique ethical implications (Corbyn, 2024). Finally, the very idea that there might be something of future use to learn from ‘n = 1’ represents a major shift in the approach to evidence generation in medicine, to which we may conclude that the populist adage ‘less is more’ has arguably never been more salient.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Sarah Heynemann is supported by the National Health and Medical Research Council (NHMRC) postgraduate scholarship scheme (2022/GNT2021953).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Tom John: Honoraria/Advisory (BMS, AstraZeneca, Amgen, Roche, Pfizer, Takeda, Boehringer Ingelheim, MSD, Merck, Puma, Specialised Therapeutics, Gilead, Seagen), Travel/speaker fees (AstraZeneca, MSD). Sue-Anne McLachlan: Honoraria/Advisory (BMS). Sarah Heynemann: Travel fees (AstraZeneca). Jennifer Philip: Speaker fees (Roche). Wendy Lipworth/Ian Kerridge: Nil.
