Abstract
The University of Kentucky has required that all researchers and research-eligible individuals complete RCR training every 2 years to ensure there is at least a baseline of RCR training throughout the wider research community. The overall goal is to create a research climate that fosters RCR across the institution for approximately 14,400 researchers and research eligible faculty, staff, and trainees engaged in research or creative work. A systematic data strategy was developed and implemented to identify individuals required to complete the RCR training and included real-time data tracking using data analytic tools and sophisticated data models. The RCR training consists of the completion of online asynchronous modules and an in-person RCR training event where case studies are discussed to apply the learning gained from the online modules and prepare participants to practice ethical decision-making in future real-world problem-solving. A train-the-trainer approach was implemented to conduct the in-person RCR training of all researchers and research eligible individuals. A library of interactive case scenarios was developed for the mandatory in-person RCR training applicable to various disciplines across the institution. A survey of participants on the RCR training provided empirical data from approximately 12% of participants. Over 72% of respondents indicated the training positively impacted their behavior. An initiative of this magnitude and reach is the broadest of its kind in the United States (US) and the article outlines the procedures and experiences in implementing the project.
Introduction
Many models of Responsible Conduct of Research (RCR) training have been developed and evaluated across the United States (US) (Katsarov et al., 2022; Todd et al., 2017b; Torrence et al., 2017). To our knowledge, none of them have encompassed training or central tracking of RCR training of this scale to include all researchers and research eligible individuals at an institution. RCR training has generally been developed to meet the mandate by US federal agencies as a condition of research funding (National Institute of Food and Agriculture, 2024; National Institutes of Health, 2023a; National Science Foundation, 2023). As many researchers do not have federal grants and research eligible individuals would not be under a requirement to complete RCR training, there is a disconnect in ethical research education. Also, despite the federal grant mandate, a lack of responsible research conduct continues to be an issue within the scientific community (Armond et al., 2021; Mousavi and Abdollahi, 2020; Oransky and Marcus, 2023; US Department of Health and Human Services, 2021, 2023a). The cost of such misconduct is multifold as it affects the institution, collaborators, trainees, the greater research community, and societal trust in science (De Melo-Martín and Intemann, 2018).
The University of Kentucky (UK) is a public land-grant research university in Lexington, Kentucky. It supports more than 200 academic programs spread across 16°-granting colleges, an honors college, a graduate school, and a library system. The campus consists of approximately 34,000 students and 27,000 employees. In the last several years, UK has received nearly 100 national rankings for excellence in academics, research, health care, and economic development. UK has a broad range of resources centered on a single campus in the heart of Kentucky. The wide array of programs allows multidisciplinary studies and fosters an environment of cooperative engagement across colleges, programs, and research endeavors. Over the past few years, UK has managed research misconduct cases revealing a lack of uniform RCR training throughout the institution. UK research misconduct proceedings have shown that excuses by researchers, against whom allegations are raised, include lack of training, different training, and lack of awareness of responsible conduct of research. Due to these claims a decision was reached that a comprehensive RCR program was needed to proactively educate a wider institutional research community. The anticipation is that education of researchers and research eligible individuals will result in an improved ethical research culture.
A rather comprehensive institutional RCR training and tracking system has previously been implemented by Duke University School of Medicine (DUSoM) (Simon et al., 2019) and comprised ~5000 researchers at the Duke University Medical Center, but did not comprise an all-inclusive Duke University researcher or research eligible audience, nor a central system that could be configured to track such a sizeable group. The UK mandatory RCR training ensures that all researchers and research-eligible individuals, regardless of whether they are federally funded or not, are trained in RCR and raises the overall institutional awareness of responsible conduct of research thus, making UK, to our knowledge, the first institution in the US to implement this type of wide-reaching RCR training.
This change in management practice is intended to improve research behavior and circumvent improper conduct which can lead to research misconduct proceedings. It can also prevent consequences of research misconduct for the institution, research collaborators, trainees associated with the person involved in research misconduct and most importantly, the public trust in science when RCR is not followed. The goals for the RCR required training are for all researchers and research eligible individuals to have baseline training in RCR principles. Accomplishing this goal involved the development of a centralized, scalable, self-service system that monitors, in real-time, both web-based and in-person case-based RCR training and compliance, a train-the-trainer component to enable training of all researchers and research-eligible individuals, a case-based in-person RCR training program and curriculum, and an email-based system for researchers to be reminded of their RCR training status as well as a system for exemption of the RCR requirement for eligible individuals. UK required RCR training for a wider research community consisting of researchers and research eligible individuals, is a first of its kind and important academically at any institution willing to adopt this encompassing system. An increase in RCR awareness is beneficial not only for the institution and the career development of researchers and research eligible individuals, but also for the general public in reducing the incidences of research misconduct that can erode the public’s confidence in research.
Materials and methods
A centralized tracking system for RCR activities
The technologies that support the culture of RCR awareness across campus used in this project include Microsoft SQL server, Tableau Software, ASP.NET C# programing, and Microsoft SharePoint.
The RCR data model is a collection of SQL database tables combined to produce a list of individuals at UK who are required to take RCR training. In addition, this database contains completion records directly from integrations with CITI. Data from the Institutional Review Board (IRB), Institutional Animal Care and Use Committee (IACUC), Office of Sponsored Projects Administration (OSPA), and finance databases are combined with UK personnel and student data from SAP HANA (institutional student data warehouse) to create the list of individuals required to take the training. Specific and detailed business rules were used to limit the datasets to individuals who are required to complete RCR based on the institutional requirement. These data tables are reloaded nightly Sunday through Friday to keep data current while also being considerate of database and report data-extract performance. Once data reloads are complete, Tableau Software is used to extract both RCR-required individuals and training records into a meaningful report.
In-person RCR train-the-trainer program
UK ORI staff recruited trainers across campus to aid with the substantial number of training sessions necessary to fulfill the in-person RCR training requirement. ORI staff met with either an Associate Dean for Research (ADR) or Department Chair to identify a suitable person(s) in the department/unit willing to be trained to deliver RCR education. 1 Trainers were also recruited via postings on various UK websites, email blasts, newsletters, and word of mouth from across campus. Trainers were onboarded by ORI staff by reviewing the RCR cases, discussion, and ethical decision-making points with them to ensure their understanding and ability to navigate the RCR curriculum and associated resources.
For each in-person RCR case, an instructional guide was provided to the trainers with an overview of the case, learning objectives, lesson description, discussion and ethical decision-making points, questions, and suggested answers to the questions. In addition, references were provided should there be an interest from the trainers in learning more and if participants in the sessions had questions not addressed during the training session. A TEAMs page for the trainers was created to aid with questions, suggestions and case discussions.
A Qualtrics web-based software is used to create registration forms for the in-person training sessions. The forms include information on when and where the sessions will take place. Information is collected to enable the RCR training completion records in a Tableau report and creates a list of registrants for the trainers. The form has a workflow to send a confirmation to the registrant with the session information and a link to cancel their registration if needed. Trainers submit attendance to the RCR In-Person Training Attendance form in Qualtrics after each session. Once verified for accuracy, the attendance is automatically uploaded to the Research Training Database.
Development of in-person RCR training materials
Others found that a combination of online learning activities combined with in person learning was the most effective RCR training (Todd et al., 2017a, 2017b; Watts et al., 2017). Educational materials used for UK’s online RCR training program are the nationally recognized Collaborative Institutional Training Initiative (CITI) web based RCR modules (CITI Program, 2024). For the in-person RCR training, a library of interactive case scenarios presented in 2D and animated video formats, applicable to various disciplines across campus, was developed by the UK Office of Research Integrity (ORI) from various sources (American Physical Society, 2024; National Institutes of Health, 2023b; National Science Foundation, 2023; Washington University Bioethics Research Center, 2024; US Department of Health and Human Services, 2023b) and also included research ethics videos from federal ORI (US Department of Health and Human Services, 2023c). In addition, RCR cases were developed based on suggestions from the audience of the RCR in-person training events with some based on personal experience. The interactive cases were designed to generate discussion of institutional expectations pertaining to the subject matter and in addition ethical decision-making points with the goal of applying information learned from materials on the CITI platform. Todd et al. (2017b) found that successful courses involved active engagement of learners. RCR training that focuses on challenging the learners to develop ethical decision-making skills are more effective than abstract moral principles (Torrence et al., 2017).
Each format was planned with a broad scope of RCR areas including the NIH FY 2022 Updated Guidance: Requirement for Instruction in the Responsible Conduct of Research that recently added subject matter “Safe Research Environments” (National Institutes of Health, 2022). Each case is 2–3 minutes long and each one-hour in-person RCR session presents three (3) cases with approximately 15 minutes of discussion and ethical decision-making framework for each case. All cases included a set of reflection questions to generate discussion and interaction among participants, to practice applying ethical problem-solving skills and further develop these in the session for future real-world application. The cases illustrate ethically problematic situations in research. The goal is for trainees to discuss how to manage situations such as, for example, plagiarism, conflict of interest, mentoring, and discrimination issues.
To facilitate interaction and discussion, each in-person RCR session was limited to 25–30 participants. This type of deliberation has been found to be effective in promoting ethical sensitivity and moral judgment as well as behavioral learning (Katsarov et al., 2022).
Several software programs (Blocksmith XR, Vyond, Murf, and Adobe Captivate) were used to create the case-based resource library.
RCR centralized email notification system
An email notification system was created to automate communications about RCR compliance tailored to each individual. The UK ORI first designed email notification templates for each type of email notification (e.g. a communication for an individual whose training is expiring in 1 week). The UK Office of Research Information Services (RIS), which provides research IT technical support, then created an SQL Server Agent job that generates batches of emails on a weekly or bi-weekly schedule using the following process:
The job queries the RCR data model for individuals who fit the criteria of each email type.
The job inserts the recipient’s name and email address into the correct email template for that type of communication and writes the individual’s email to a database table as a new record. The record has a field that distinguishes whether the email has been sent or not. This database-driven approach to email automation allows for a log of all emails sent.
Then the job sends emails by iterating through the table of email messages and sending all emails that are marked as unsent. Once the emails are sent, their records are dated and marked as sent.
Participants who have completed the training receive certificates of completion and a resource document link.
Exemption from RCR training process
Individual(s) who are of the opinion they should be exempt from the required RCR training can submit an exemption request form. The determination process occurs via a review by an ad hoc committee. The ad hoc committee will interphase with college leadership in deliberating exemptions monthly (if needed) followed by written dissemination of a decision to the individual, the program director or Department Chair, and their ADR. An example of exempt RCR training requirements include, for instance, graduate school curricula that do not have a research or scholarly components.
Survey of in person RCR training participants
Participants’ feedback was sought by deploying a survey electronically via email 1 year post implementation of the required RCR training initiative. Questions included topics of applicability of the training, influence on ethical decision making, ability to relay learned information to others, and impact on behavior.
Development of the research resource document for In-Person RCR training participants
The in-person RCR training sessions resulted in a comprehensive document on resources for trainees. The document was compiled based on interactions with the trainees and questions posed in the sessions. The resource document serves as a central location for relevant policies, resources, and points of contact for various issues/topics that researchers at the institution may need to refer to throughout their careers. It also allows researchers to find further reading to aid with the in-person session information retention. The document, along with an attendance certificate, is linked in the completion certificate email received by all in-person RCR trainees post session attendance. The resource document is also posted on the ORI website for researchers to access as needed.
Results
The required RCR training and tracking system incorporated a variety of technologies including the currently required CITI web-based RCR modules, an integrated database for tracking and monitoring RCR activities, the train-the-trainer sessions with instructional guides, the development of a resource library of RCR cases for in-person training, a process for exemption of the RCR required training, a survey of participants and a research resource document.
RCR data model
A lack of completion of the required RCR training by researchers triggered the idea of a centralized tracking system for completion of the required CITI modules and planned in-person RCR training. Previously no single data source could identify all researchers and research eligible individuals required to complete the RCR training. The creation of the tracking system incorporated a variety of technologies including the development of an integrated database, reporting, and sophisticated processes to support automated communication. All current technologies and those developed assist in creating an effective and efficient RCR program across all of the institution’s campuses. These technologies have increased efficiency in monitoring the progress of mandatory training, training deadlines, and annual training requirements to ensure all researchers and research eligible individuals complete the RCR training. The central tracking system is based on an SQL-server database infrastructure and employs a variety of data elements from several different databases. This centralized system brings together disparate data sources, including human resource data for payroll on sponsored grants and contracts from the institution’s central enterprise system (SAP), protocol data from the IRB, IACUC and OSPA from our internal grants management system. All data sources were developed on the backend to create reportable tables based on specific business rules and can be used in reporting platforms and tools, such as Tableau Server, to deploy interactive tracking and self-service reporting across the campus.
RCR data model—validation and reliability of collected data
Validating the pilot RCR data model centered around four major areas: the HR researcher personnel report, HANA student report, RCR training exemptions, recipients of the automated RCR email communications, and the RCR Tracking report in Tableau Server available to end users, which include Associate Deans for Research and Center Directors.
Unit testing occurred at the referenced database level to validate that the personnel listing from each research area was accurate and met the established criteria. For example, the IRB protocol on which a person was listed in the IRB database must be in either submitted or active status and the person must not have been removed from the protocol in the current version. Once unit tests were completed, personnel lists from each area were combined into a one record per person table that became the cumulative personnel list required to take RCR training. This table uses several fields to identify which referenced database(s) each individual is listed in, which in turn helps provide administrators with context for why the individual is required to take the training. The table had to be validated to meet the following criteria:
• One distinct record per person required to take the training.
• Each personnel record had to reflect membership in at least one of the referenced databases to be in the cumulative RCR personnel list.
When the above criteria were met, the RCR Tracking Tableau report was built using the cumulative RCR personnel as the base table of its data source.
To ensure the institution has an efficient and manageable system to require RCR completions, institutional leaders including upper administration, and senior administrators, at the college and departmental levels are provided with the necessary data tools to manage the RCR lifecycle. The SQL data tables created and maintained in the tracking system by RIS are critical to putting real-time data into the hands of decision-makers (Figure 1). SQL Server data tables are used in a variety of reporting tools created using Tableau Software, an online deployable real-time data analytics platform. The analytics allow leaders across the institution to see real-time data on several aspects of the RCR-required program:
Aggregated counts by college and department of individuals required to complete RCR training and completion percentages.
Employee details, including contact information and course completion data for easy access to records and completion dates.
Individual details related to why an individual may be required to complete RCR. This includes their status as faculty, staff or trainee listed on a protocol, or paid on a sponsored project—or any combination of the attributes that make an individual required to complete the RCR training.
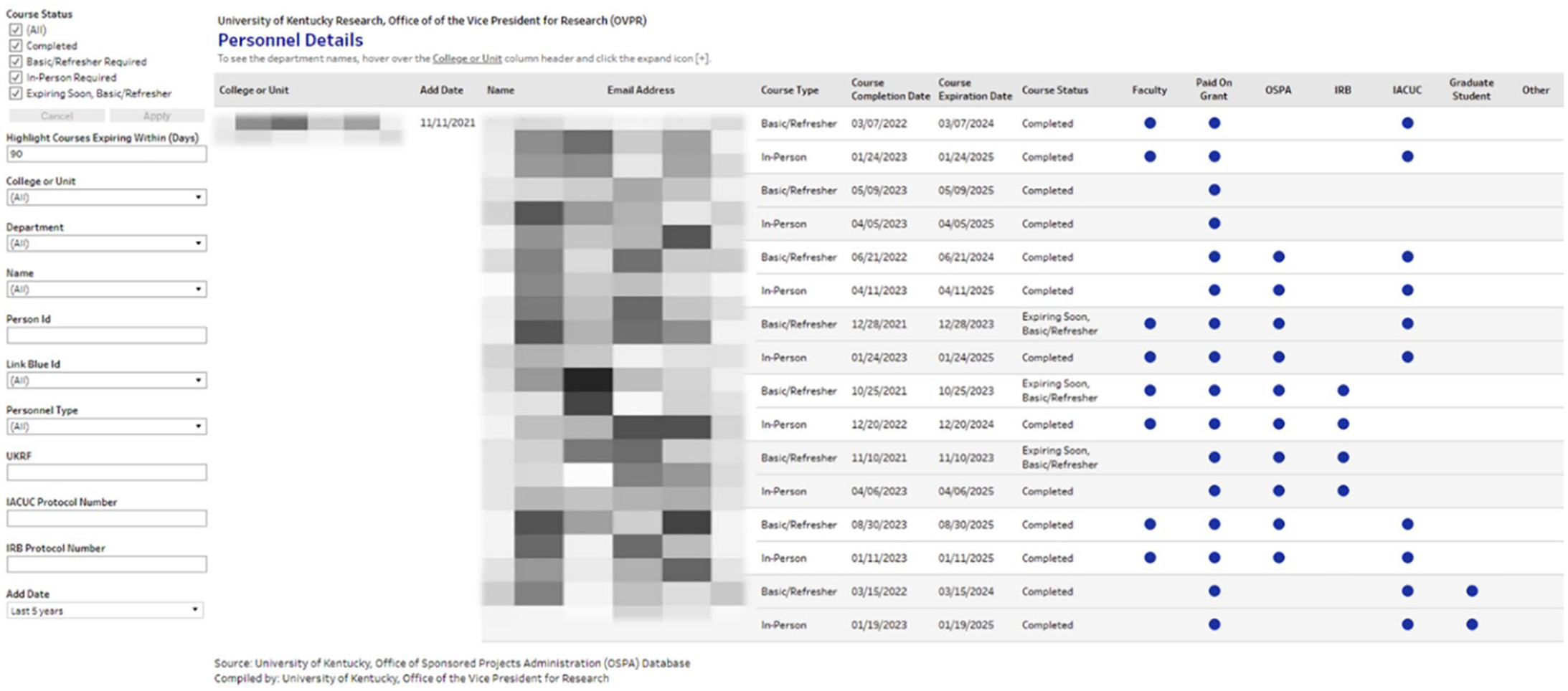
Individual researcher RCR completion details.
The RCR Tracking report in Tableau was created to unite the cumulative RCR personnel list with existing training records in a meaningful interactive interface for end users. In addition, the reporting module in Tableau Server allows for actionable regular reporting via email to campus leadership (Figure 1).
Train the trainer program
The development of the instructional materials for the in-person RCR training included a train-the-trainer program to manage the substantial number of researchers and research eligible individuals to train. UK ORI staff conducted initial training with individuals from various departments across campus to develop the train-the-trainer program. UK ORI staff presented the in-person RCR case studies, reviewed issues presented in each case and provided an instructional manual containing a summary of the case, potential questions, and suggested answers to the questions as well as additional questions time permitted in the training session. The instructional manual also contained resources for the trainers and trainees should they desire further learning. A Teams Channel was developed to aid trainers with resources and to provide a venue for trainers to raise questions and enable discussions related to the training. Training also included information on registration and attendance tracking of participants. The trainers then provided in-person RCR training to their respective departments/units using the 2D and animated videos along with the instructional resources. Some trainers volunteered to train researchers and research eligible individuals in additional departments/units as well.
Development of case study library
The in-person RCR training program contains a resource library of case studies, in different formats and subject matters to suit the RCR educational needs of departments and centers across the institution. Our in-person case study curriculum covers seven (7) areas (Table 1), and the UK-selected asynchronous CITI modules cover six (6).
RCR areas in the library of case studies for in-person RCR training.

The animated videos and 2D case study library initially included 12 case studies and have continued to grow as more scenarios are created and now comprise 43 case studies (Figure 2) in addition to eight (8) cases from the DHHS ORI website. A wider range of RCR topics have previously been found to be more effective (Watts et al., 2017). Some scenarios originate from participant suggestions and are sometimes based on their personal experiences. The cases are short, 2–3 minutes long and leave open some interpretation by the audience of the situations portrayed to encourage discussion and ethical decision-making points. The subject matter is broad and reflective of the many research areas at the institution and allows for approximately 15 minute discussion periods per case. In-person RCR sessions are capped at 25–30 participants per session to allow enough time and opportunity for participant discussions.

Examples of in-person RCR cases developed for the RCR case library.
Individual departments or centers can request personalized training sessions for their academic area to ensure applicable topics are provided to each research group.
Researchers and research eligible individuals complete the CITI RCR training modules and the in-person RCR training every two (2) years to stay compliant with the requirement and retain their understanding of RCR.
University-wide RCR training notification system
A university-wide notification system was developed to alert researchers of their RCR training status (Figure 3). This notification system uses data to accurately prompt individuals when their expiration date for RCR training is approaching and when training is incomplete.
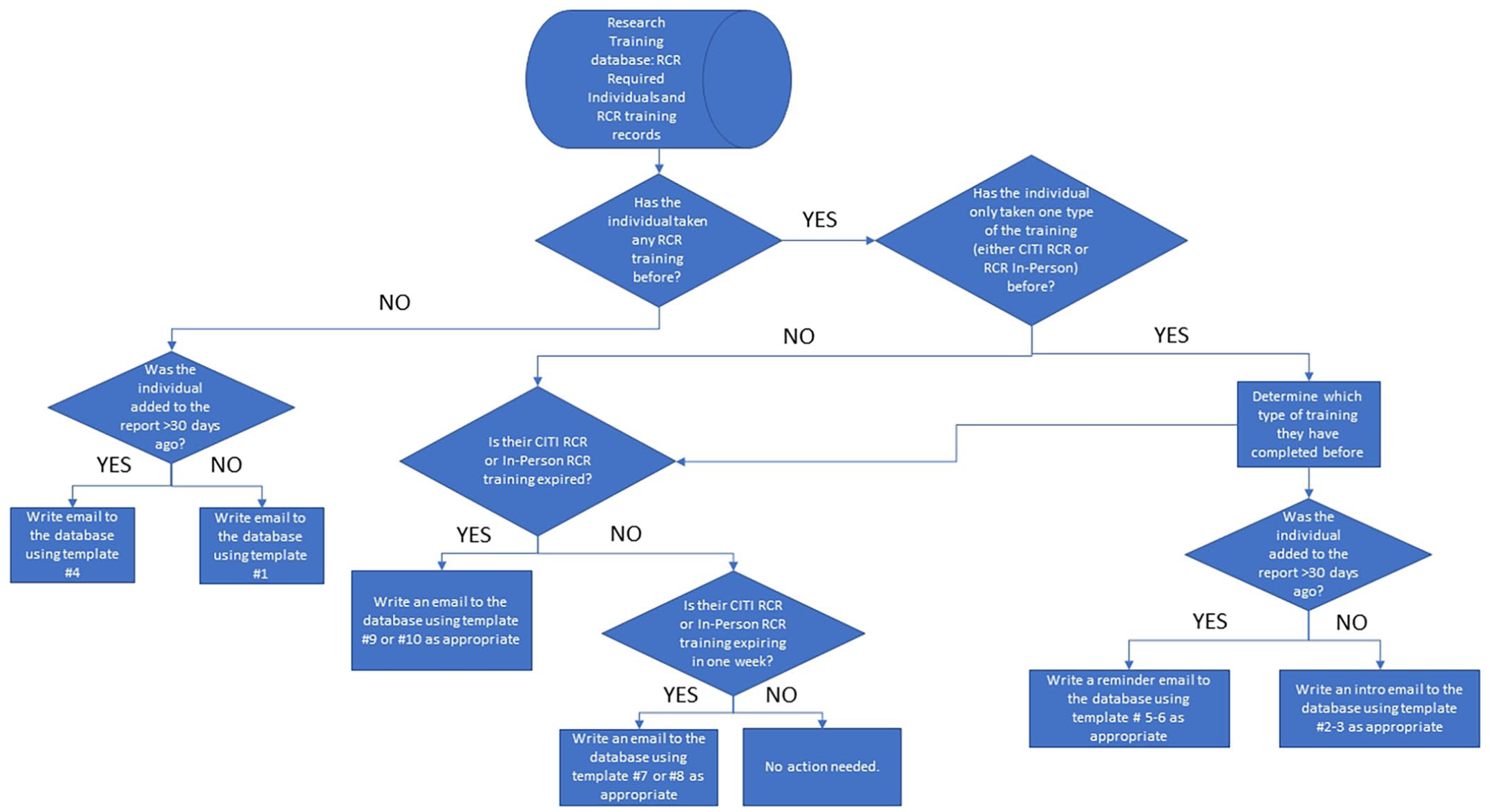
Types of email notifications designed for varying levels of RCR compliance.
Database queries were developed to dynamically determine the recipients for 10 types of communications designed for varying levels of RCR compliance (Table 2).
Types of email notifications designed for varying levels of RCR compliance.

Exemption from required RCR training
An exemption request process was developed for faculty, staff, and graduate students with credible reasons why they should not be required to complete the mandatory RCR training.
Any UK employee or trainee who is designated to complete the RCR training but believes they should be exempt from the process can complete the online Exemption Request form. Once completed the individual will submit the form for a decision through an institutional determination process.
The determination process occurs via a review by an ad hoc committee. The ad hoc committee interphases with college leadership in deliberating exemptions monthly (if needed) followed by written dissemination of a decision to the individual, the program director or Department Chair, and their ADR. If those requesting an exemption from the training requirement are, or become, part of the eligibility criteria, the exemption designation is overridden from the tracking software, and the RCR training requirement is enforced (Figure 4).
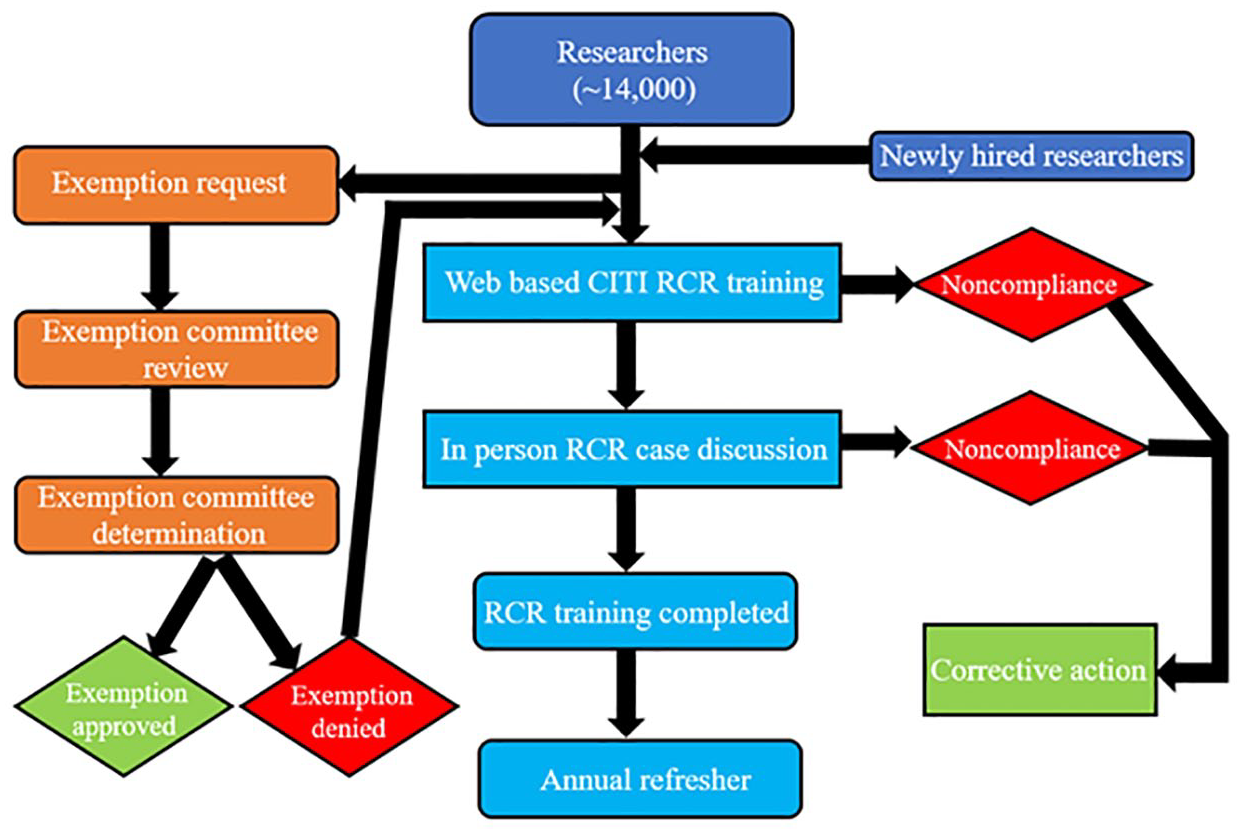
University of Kentucky’s mandatory RCR training process.
Individuals that could be exempt from the RCR training requirements include, for instance, graduate school curricula that do not have a research or scholarly component.
Corrective actions are implemented for individuals who fail to complete the required training by the deadline. Penalties depend on the affected personnel and their role in research.
Assessment of completions of required RCR training
As mandatory training is required every 2 years and was implemented in the Fall of 2022, the first cohort completing RCR was tracked in December of 2023 (Table 3) to ensure compliance with both CITI RCR modules and in-person RCR training requirements.
Assessment of RCR training completion in December 2023 (not including the number of graduate students ~2000).
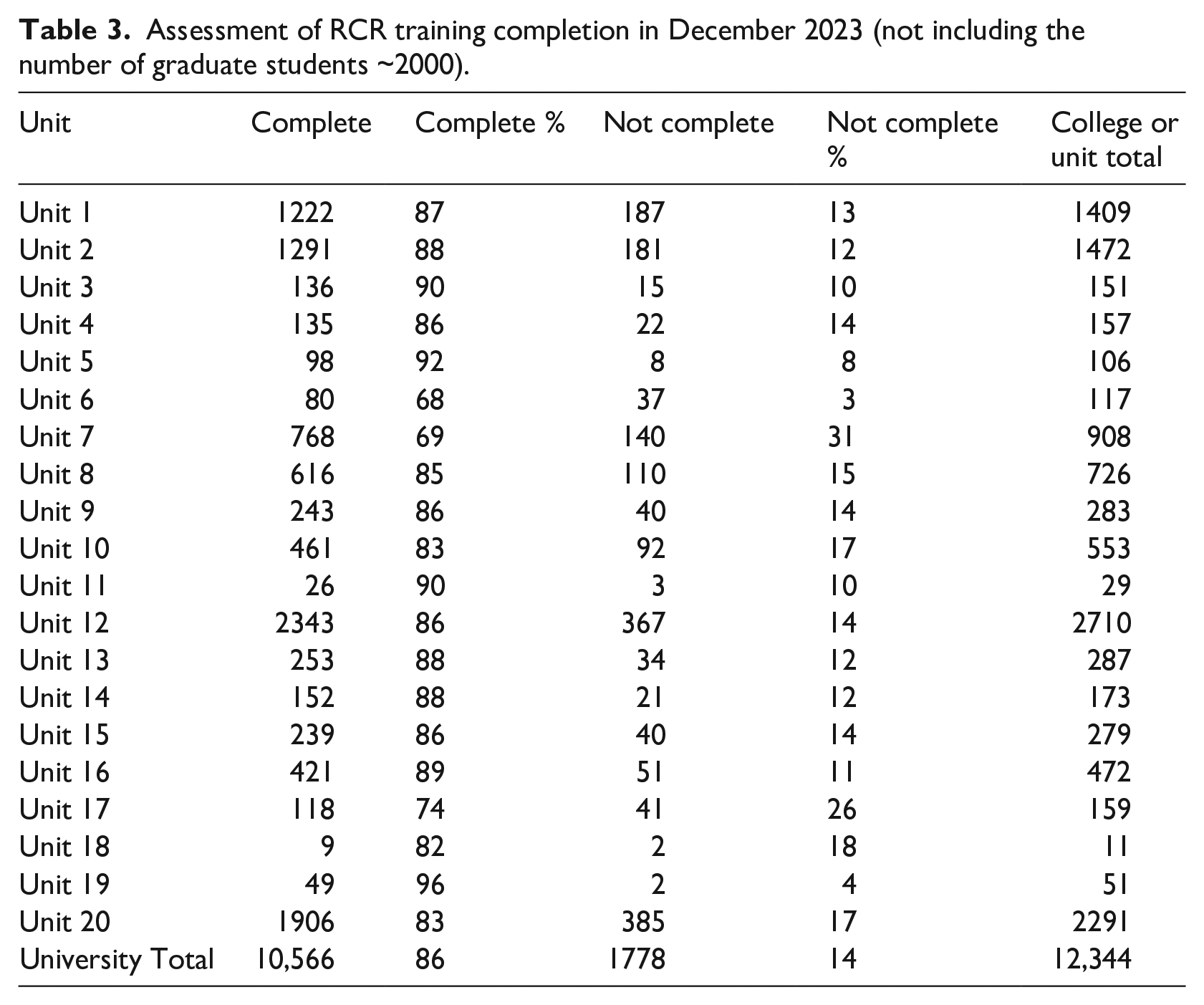
One year post implementation of the required RCR training we observed an overall completion rate of 96%. 14,400 Researchers were required to take the RCR training. Graduate students (approximately 2000 individuals) were added again in the Fall of 2023 (usually not listed over summer months due to a pause in graduate stipends).
Assessment of the required RCR initiative
To evaluate the overall effectiveness of the in person RCR training, a survey instrument was deployed 1 year into the RCR training requirement. The survey response rate was ~12% and provided valuable information on the RCR training initiative (Figure 5). Over 65% of the respondents felt the training was at least moderately applicable and almost 59% thought the resource documents were at least moderately useful.
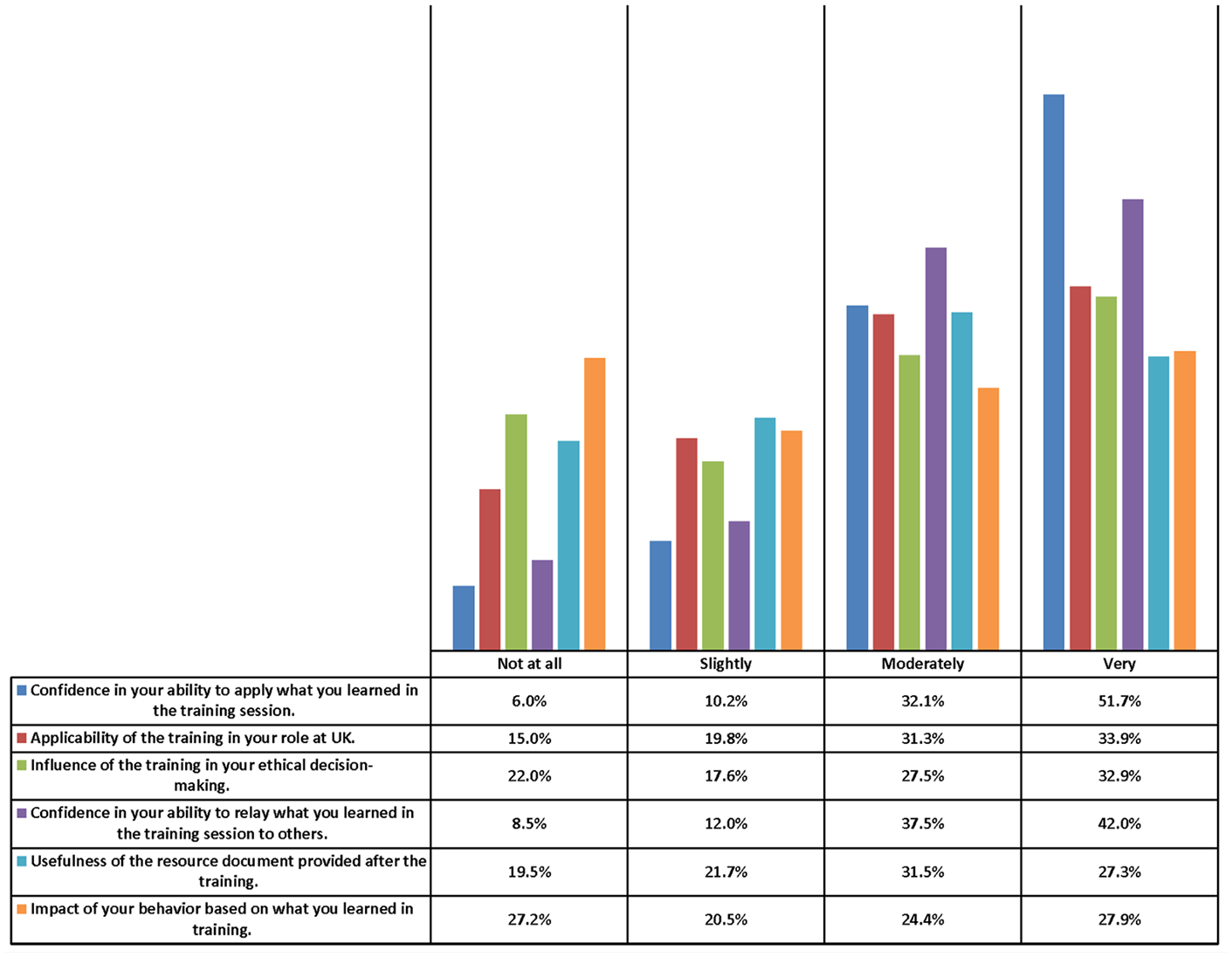
Survey results on the required RCR training initiative.
A majority of researchers and research eligible individuals were very confident in their ability to apply what they learned in the training sessions and over 83% were at least moderately confident. Additionally, almost 80% were confident in their ability to convey what they learned to others, which indicates that not only are trainees getting the message, but they should have a positive indirect impact on their colleagues and mentees. Researchers and research eligible individuals’ ethical decision-making and impact on their behavior after the training was less significant, but still over 60% were at least moderately influenced by the training. The fact that 22% were not influenced at all could mean that they are aware of and engaged in ethical research practices and ethical decision making before the RCR training was required. The other possibility is a lack of interest in the subject matter or a lack of retention of the materials presented. Over 72% of respondents felt the training impacted their behavior. These results were extremely positive, especially for a required training, and hopefully will yield positive results in reducing research misconduct at the institution.
Open ended feedback from the surveys, aside from some dissatisfaction with the mandatory nature of the training, included suggestions for departmental centralized training to ensure cases applied to a particular research area. Another suggestion was to have groups according to career stage such as postdoctoral students in separate groups from junior faculty who should be in separate groups from research staff. Several respondents requested more advanced cases for senior research faculty who may encounter complex cases of research misconduct. Finally, attendees voiced that the experience of the trainers has an impact on the responsiveness of the audience in the discussion sessions. This would align with the finding by Watts et al. (2016) in one prior review, that RCR courses were more effective if the trainers had good expertise in the area taught.
Continued assessment of the required training includes the preference of each case scenario for in-person instruction, whether the cases are broad enough in subject matter content, whether case studies are explored sufficiently in-depth, whether discussions generated are in line with the planned awareness, and whether there are other topics for which cases need to be developed such as cases including artificial intelligence (AI) and the regulations applicable to this rapidly evolving area. Developing more advanced cases is under consideration but may alter the number of cases per training session as these cases would involve more complex issues and discussions.
Moving forward, the initiative of the required RCR in-person training every 2 years will be managed by each College at the institution. Reviews have found that offering courses to an audience of learners from different areas in academia is not as effective as if they are separated by academic area (Watts et al., 2017). Courses previously considered effective are those that target specific groups of learners (Mulhearn et al., 2017; Todd et al., 2017a). A recent literature review on effectiveness of RCR courses however found that courses that were offered to learners from multiple academic areas were as effective as those offered to a focused academic group (Katsarov et al., 2022).
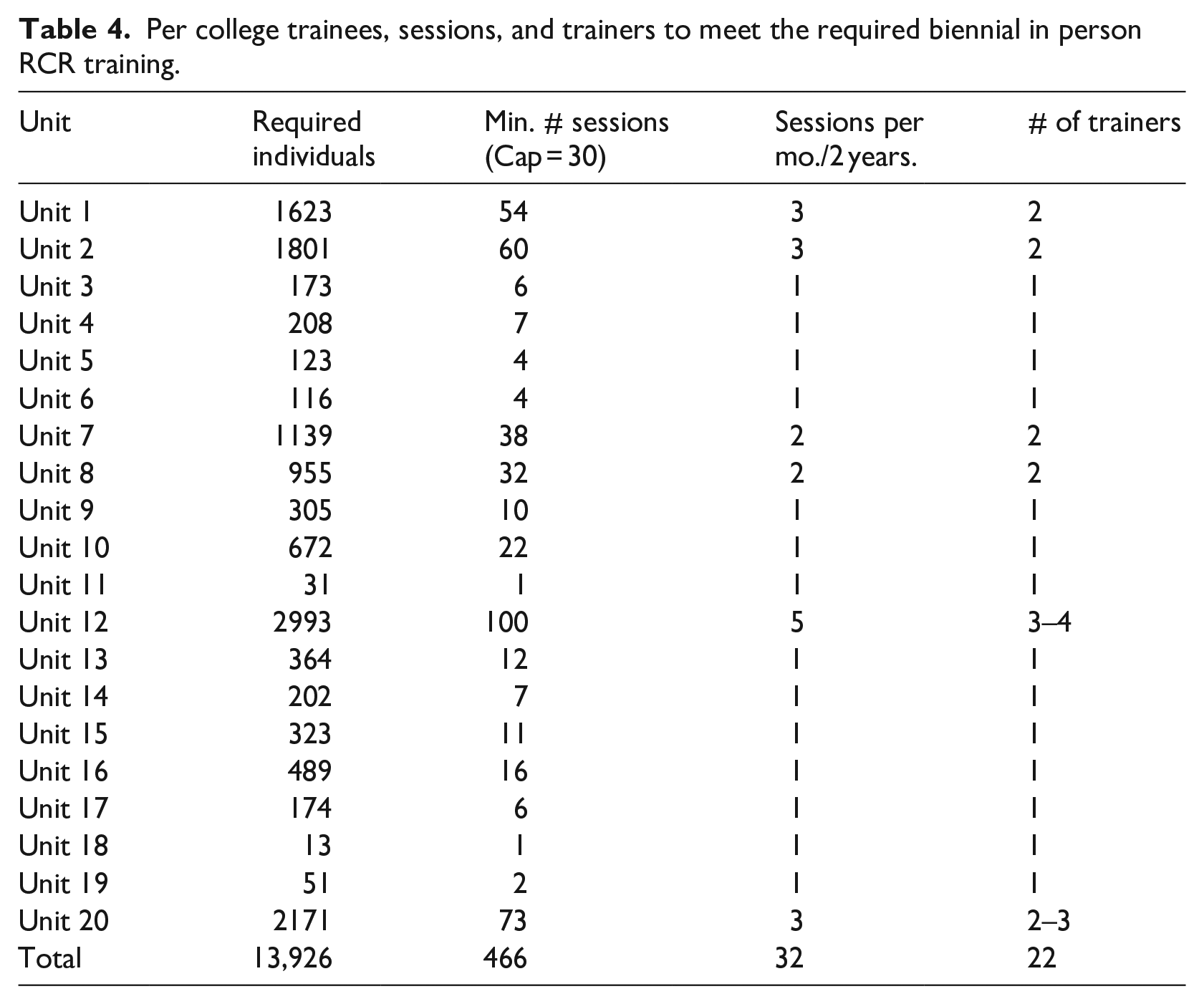
Each College will be providing trainers and training researchers, and research eligible individuals in their college. Colleges have been provided with the number of individuals to be trained, suggested number of trainers as well as the number of sessions to offer over the next two (2) years to provide the training (Table 4).
Per college trainees, sessions, and trainers to meet the required biennial in person RCR training.
Discussion
While UK has encouraged RCR training and ethical conduct of research in the past and expects that graduate students and junior faculty are mentored by major professors, faculty advisors and senior faculty, it has proven inconsistent between disciplines and departments across the institution. The implementation of an RCR tracking and notification system to verify individual researcher RCR training compliance coupled with newly developed RCR instructional resources and a training plan, ensures the wider audience of all researchers and research eligible individuals at our institution receive in person RCR training and complete the CITI RCR modules to understand the expectations regarding maintaining an ethical research environments across all departments and colleges at the institution.
The mandatory RCR training encompassing the wider audience of all researchers and research eligible individuals at an institution, and a central tracking system of RCR completion, addresses any lack of RCR training by researchers and research eligible individuals, which could impede a climate of integrity, conduct, quality, and reliability of research. A universal requirement of RCR education for all researchers at an institution regardless of whether they are federally funded or not is essential in establishing a baseline of RCR training to improve research ethics knowledge and a dynamic RCR climate. Accountability on the part of researchers is heightened and not having RCR training is not a viable excuse for irresponsible conduct of research.
UK as an institution invested in this mandatory RCR education and central tracking program to ensure its success in baseline RCR training of researchers by providing support from the top administration down. The central tracking system is a combination of computer databases already existing at the institution, plus a new Research Training database. Administrative support for this project also includes working with managers of the existing institutional computer databases. Trainers are volunteers from most Colleges. Development of the library of case studies requires monthly subscription fees to computer programs enabling the 2D and animated videos.
Both DUSoM and UK inspire to create an all-inclusive environment of responsible conduct of research by including a broad audience in their RCR education activities. Similarities between the DUSoM and UK RCR training programs are that they are required and that a computer program was developed to track who is required to take the RCR training along with a system issuing reminders and completion data. The differences are that the DUSoM program encompassed only the DUSoM research faculty and staff and not all Duke University researchers and research eligible individuals. UK requires the expansive audience of all researchers and research eligible individuals at the entire institution to complete the RCR training. The centralized computer system developed by UK to track all individuals required to take the training, reminder emails and completion data is quite sophisticated as it again includes the entire institution. The DUSoM program offered online, in-person or test-based RCR training alternatives while UK, in addition to the online CITI RCR training, ensured in-person discussion of responsible conduct of research areas by requiring in-person RCR training. Face-to face learning offers a depth of deliberation and interaction not found in online courses where content can be brushed over or skipped.
There are three critical barriers to mandating RCR training and tracking compliance through a central tracking system are researcher culture and institutional infrastructure. The first barrier is a deficiency of the institution’s infrastructure to track completed required RCR training through a centralized campus-wide system. The target population is in constant flux due to extramural funding growth at the institution resulting in new hires and more individuals added to research protocols. This, along with enrollment changes of graduate students, creates challenges or barriers to effectively monitor RCR requirements for, in our case, 14,400 individuals on a campus of approximately 34,000 students and more than 27,000 full-time employees.
The system configured to track the status of the institutional requirement of RCR training for all researchers and research eligible individuals is central to the institution’s mission to create a responsible conduct of research culture. While CITI does provide RCR training notifications and certificates, the institution’s central system tracks every researcher in all departments across campus and includes prompts to researchers and research eligible individuals on when and what RCR training is required. The system enables departments to view and track internally who has completed or needs to complete mandatory RCR training.
The second barrier is behavioral and based on the reluctance and/or disinterest of researchers to complete RCR training when not mandated by a federal agency. To emphasize the importance that all UK researchers, regardless of discipline or funding, complete the RCR training, UK has been transparent regarding the institution’s recent research misconduct cases. These cases illustrate a lack of or inconsistent RCR training or ethical research awareness. In addition, UK broadened the definition of RCR to include scholarly activity as some departments did not consider themselves included in the RCR requirement, but their scholarly or creative work could result in research misconduct if researchers are not appropriately trained in RCR. The inclusion of all scholarly and creative activity in the definition of RCR for researchers and research eligible individuals also encompasses a broader research community. The transparency of the institution regarding recent research misconduct cases and the broadened definition of RCR has improved the understanding of who is required to complete RCR training and why the training is important.
A third barrier is measuring effectiveness of the mandatory RCR training initiative. More robust quantitative and qualitative methods to assess the effectiveness of the training are needed. We issued an anonymous post session survey to all in person RCR training attendees reflecting on their perceptions. The measure included self-reported usefulness of the training. While the feedback is helpful, the results were not objective determinants of enhanced ethical conduct.
The required training is to be completed every 2 years to ensure researchers’ RCR training stays current and that a baseline of understanding of RCR expectations remain constant across the research community. Assessment of the RCR training requirement is important to guide the institution in further development of the RCR training. The aim of instituting comprehensive RCR training to a wider institutional audience is to optimize RCR conduct and prevent research misconduct and the associated societal and institutional consequences.
The requirement is, to our knowledge, the first of its kind in terms of magnitude and is hopefully a model of how to encourage RCR awareness across a wider institutional research community and an increase in the public trust in research.
The impact of mandatory RCR training is an improvement of overall institutional awareness and accountability that all researchers and research eligible individuals have completed baseline training in RCR. This achieves a climate of responsible research conduct from top administration down to students. UK has the support of the upper administration, starting with the President, who have also completed RCR training to emphasize the importance of everyone participating in the training. Proper mentoring is crucial for the development of responsible researchers who are capable of self-regulating their ethical conduct.
As universities across the US grapple with maintaining research cultures informed by the most robust ethical guidelines, UK’s recent implementation of a first-of-its-kind comprehensive RCR education program to a wide audience comprised of researchers and research eligible individuals offers one approach that can be modeled at other institutions. Given that 14,400 researchers were trained in a relatively brief period of time, UK’s efforts signal an important commitment to fostering a research climate that will hopefully reduce incidents of research misconduct in the long term. The authors have provided the methodology that informed their implementation of required RCR training as an act of transparency so that institutions could adapt it to their own context. The authors hope that an encouraged use of comprehensive RCR training at institutions across the US will lead to improved ethical research conduct and fewer cases of misconduct across institutions who choose to adopt these measures.
Footnotes
Research ethics/Ethics approval information
The study underlying this article was determined not to require institutional research ethics approval by the University of Kentucky Institutional Review Board.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
