Abstract
Institutional review boards have a dual goal: first, to protect the rights and welfare of human research subjects, and second, to support and facilitate the conduct of valuable research. In striving to achieve these goals, IRBs must often consider conflicting interests. In the discussion below, we characterize research oversight as having three elements: (i) research regulations, which establish a minimum acceptable standard for research conduct; (ii) ethical principles, which help us identify and define relevant ethical issues; and (iii) virtue ethics, which guides the prioritization of relevant issues. We describe specific ways in which the lessons of virtue ethics suggest revisions to the IRB structure and review process, the education and training of IRB members, and the appropriate limits of regulations in research ethics oversight.
Keywords
Introduction
The primary charge to institutional review boards (IRBs) in the United States is to protect the rights and welfare of human research subjects (Code of Federal Regulations 21CFR56.102(g): FDA, 2013a). The mission of an IRB, however, is not to eliminate all possible risk, but rather to ensure that risks to subjects are outweighed by the sum of the expected benefit to the subject (if any) and the expected benefit to society in the form of knowledge to be gained (Code of Federal Regulations 45CFR46.111(2): FDA, 2013b). Given these dual goals of protecting human subjects and of supporting valuable research, IRBs must often consider conflicting interests in determining whether to approve a particular research study. The Institute of Medicine (IOM) has pointed out that other responsibilities that fall to IRBs (e.g. protecting institutions from legal risk) threaten these primary missions. The IOM concludes that the IRBs ‘should be reshaped and reformed to serve the role for which they were originally intended—ensuring participant protection through the careful ethical review of research protocols’ (Institute of Medicine, 2002: 70).
In our discussion below, we describe how the lessons of virtue ethics can help us reshape and reform IRBs to ensure that all research undergoes careful ethical review. We will argue that ethics principles and regulations, although vital, are not sufficient for excellent research oversight. Rather, principles and regulations present often-conflicting claims for the IRB to adjudicate. We therefore argue on behalf of a virtue ethics approach to structuring IRBs, training IRB members, and informing the optimal degree of regulation within which IRBs must operate.
What is virtue ethics?
Ethical problems are often complex and novel; they present themselves in unique contexts in which conflicting principles are at stake. Principle-based ethical theories provide guides to action. Virtue ethics, by contrast, focuses on the development of the character of the individual who must confront ethical dilemmas. Virtue ethics recognizes that resolution of difficult problems depends, above all, on the character (that is, on the virtues) of the people making decisions. It is important to note that, strictly speaking, virtue ethics is focused on character rather than on specific actions. Nevertheless, when someone chooses which actions to carry out, those choices reflect his or her character. Virtue ethics also strongly emphasizes that individuals can be trained in the virtues so that they develop excellent habits of mind. Such habits will best prepare an individual to choose optimally in any given context. Virtue ethics is based in Aristotelian philosophy and was a dominant influence in the classical period, though it was later eclipsed by principle-based ethical philosophies. Virtue ethics regained prominence in the latter half of the twentieth century, largely in response to Anscombe’s 1958 ‘Modern moral philosophy’ (Anscombe, 1958). Virtue ethics is based on a positive view of human nature, one that takes into account that humans are strongly predisposed to (a) recognize excellence in others whom they can take as role models and (b) gain fulfillment from a life lived virtuously (eudaimonia). Practicing virtues and habits of excellence are therefore self-reinforcing behaviors. Virtues, as conceptualized by Aristotle, represent ‘the mean by reference to two vices: the one of excess and the other of deficiency’ (Aristotle, translated 1886). As such, the idea of moderation and compromise are intrinsic to the theory.
Some have criticized virtue ethics on the grounds that virtue ethics is not sufficient to explain why people should act rightly, that virtue ethics is not necessary to account for the intrinsic value of the virtues, and that virtue ethics focuses on the agent’s flourishing or ‘admirability’ when, intuitively, our ethics should be motivated by the opportunity to improve the lives of others (Hurka, 2001). We do not address these criticisms in this article. We do not propose that moral action need be motivated exclusively or even primarily by a desire to increase in virtue, though we recognize that living virtuously is often rewarding and generally habit-forming. Rather than arguing for the primacy of virtue ethics over principle-based ethics, we believe that the two approaches complement each other. Below, we reframe research oversight, drawing specifically on this complementary relationship between principles and virtues.
Complementary roles of virtues and principles
Neither principles nor virtues provide sufficient guidance in and of themselves. Resnik points out that ‘virtue theories do not give us clear instructions on how to handle conflicts between the virtues’ (Resnik, 2012). Similarly, principle-based ethical theories do not give clear instructions about how to handle conflicts between principles. For example, IRBs struggle to determine the extent to which incidental or secondary findings of uncertain significance should be communicated back to subjects. The principle of autonomy suggests that subjects have a right to information about themselves, whereas the principle of non-maleficence may direct one to avoid worrying subjects by providing extensive information that cannot be reliably interpreted. Practical wisdom occupies a central space in virtue ethics because of these inherent tensions. Although virtue ethics does not rank most of the virtues in constant hierarchy, most virtue ethicists from Aristotle on have recognized the primary role of practical wisdom. Practical wisdom allows virtuous individuals to recognize the morally salient features presented by a case in its particular context and to respond appropriately.
Aristotle’s description of practical wisdom has been translated as ‘good deliberation and sensitivity to salient factors (Aristotle, translated 1886). Adherents of principle-based ethics recognize the importance of practical wisdom as well, because correct application of principles ‘requires situational appreciation—the capacity to recognise, in any particular situation, those features of it that are morally salient’ (Hursthouse, 2013). Annas describes that the virtuous moral actor ‘responds directly to the situation in an intelligent way, one which takes account of all the relevant factors’ (Annas, 2011). Practical wisdom develops through experience and manifests as excellence in deliberation, helping people discern when a situation calls out for which particular virtues (Annas, 2011; Hursthouse, 2006; Russell, 2009). Further, as we shall discuss, practical wisdom is amenable to education and training.
Research regulations, principle-based bioethics, and virtue ethics each contribute uniquely and critically to excellence in research oversight (Figure 1). Research regulations are explicit rules that guide IRBs in their determinations. They derive from a canon focused on rules, duties, and responsibilities (National Commission for the Protection of Human Subjects, 1979; Nuremberg Military Tribunals, 1949; World Medical Association, 1964, last amended 2008). These regulations are necessary to ensure that research meets a minimum acceptable standard for research conduct, and to provide a means to enforce that standard (Martin and Lantos, 2005). Research regulations can prevent much wrongdoing, but they cannot address the specifics of every case, so they do not always suffice in novel situations. An IRB whose sole goal is to ensure compliance with regulations will fall short of its mission to protect human subjects and promote ethical and valuable medical research.
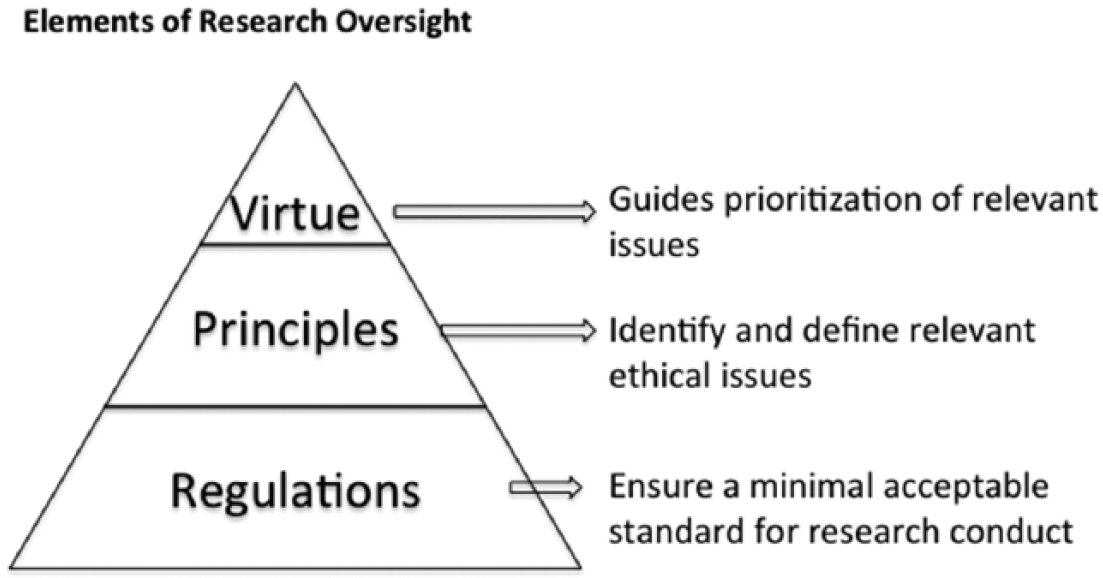
Elements of research oversight.
A deontological, or rule-based, approach dominates modern, western, research ethics discourse. This approach is grounded on the four ethical principles delineated by Beauchamp and Childress: respect for autonomy, non-maleficence, beneficence, and justice (Beauchamp and Childress, 2001). Attention to these fundamental principles provides a common language, focusing and organizing research ethics debates.
Unfortunately, different principles frequently make contradictory demands of us. For example, the principles of autonomy and non-maleficence may collide when determining whether a new, experimental drug should be made available for human trials. Advocates for early availability cite patients’ ‘right to try’, whereas proponents of a more cautionary approach invoke non-maleficence because they fear the harms that may befall patients if they are given a drug whose safety and efficacy have not been fully established. Principle-based ethics systems do not make it clear how to adjudicate between these competing principles (MacFarlane, 2009).
Consequentialism, by contrast, is based on the idea that right choices are those with the best overall consequences. Utilitarianism, popularized by John Stuart Mill, characterized the relevant consequence as happiness, but emphasized that happiness is manifest in higher and lower forms, and that the higher forms should be weighted more heavily (Mill, 1998). Consequentialism makes an appearance in research regulations and research ethics when we stipulate that the potential benefit of research (including future benefits to individuals not involved in the research) must be proportionate to the risks borne by study subjects.
However, neither a deontological nor a consequentialist approach provides sufficient direction when faced with competing or incommensurable claims or consequences. Virtue ethics shows us that weighing competing, incommensurable claims need not present an intractable problem. The primary question virtue ethics asks is not ‘what potentially contradictory commandments must I obey?’ but ‘how should a virtuous person act in this situation?’ Virtue ethics offers the potential for more flexible and creative solutions than principlism or consequentialism alone (Gardiner, 2003).
As Martin and Lantos point out, ethics should aim to discriminate ‘between what is good and what is excellent’ (Martin and Lantos, 2005: 63). Virtue ethics reminds us that the character, or the virtues, of the IRB members and chairs will determine how well IRBs will adjudicate between competing claims when regulations are open to interpretation. ‘It is the agent who interprets principles, selects the ones to apply or ignore, puts them in an order of priority and shapes them in accord with his life history and current life situations’ (Pellegrino and Thomasma, 1993: 19). When IRBs encounter complex cases in which principles come into conflict with each other, we depend upon the informed, intelligent deliberation of IRB members for optimal resolution. In other words, we rely on their virtue.
Undeniably, the call of one virtue or another may lead to different courses of action. For example, courage may inspire approval of a human study of a new drug, whereas the virtue of temperance may suggest that more work on animal models would be necessary first. The virtue of honesty may direct one toward disapproving a proposal involving covert observation of social phenomena, whereas the virtue of fairness may suggest that covert observation is necessary to identify and address societal problems.
The simple guide to ‘act virtuously’ does not always suggest a solution to an ethical dilemma (Resnik, 2012). The summary, ‘act virtuously’, though, is an oversimplified interpretation of virtue ethics. Virtue ethics focuses on the importance of developing the habits of mind and character to engage and resolve ethical dilemmas while embracing, not forsaking, ethical principles. Training in the virtues, particularly in the virtue of practical wisdom, helps us to assign the proper weight to competing claims in a particular context so that we can most appropriately adjudicate between them. The individual principles may be seen as claimants, and the virtuous research ethics professional as a judge determining how best to balance competing claims. A rule-based or even a consequence-based approach to research ethics may provide clearer solutions to questions of precisely how much animal data should precede human testing, or under precisely what circumstances deception is permissible, but the resultant loss of flexibility would impede scientific progress without improving human subject protections.
Specific changes suggested by a virtue ethics approach to research oversight
Resnik describes that there are ‘four ways of promoting research integrity in an institution: education, policy development, policy enforcement (also known as compliance), and leadership.’ (Resnik, 2012). When we argue that incorporating a virtue ethics paradigm will improve the performance of IRBs, we are not merely advancing the indisputable claim that more discerning IRB members will be better at their jobs. Rather, we suggest specific, concrete refinements that address education, policy development, and leadership (though we do not address compliance in this article). In the following sections, we argue for virtue ethics inspired revisions to (i) how IRBs are structured, (ii) how IRB members are trained and educated, and (iii) how we determine the appropriate limits of regulations in research ethics oversight.
IRB structure and review process
A clinical research ethics consortium, convened by the Berman Institute of Bioethics, identifies the lack of an ‘effective mechanism for IRBs to address major ethical issues’ as among the foremost problems with the current research review system in the United States (Emanuel et al., 2004). We propose that a clear definition of the roles and responsibilities of the people contributing to IRBs should be defined so that IRBs can excel in their oversight of research involving human subjects. Specifically, IRBs should be organized so that IRB members educated in research ethics principles and trained in the relevant virtues can best deliberate and resolve major ethical issues (Figure 2).
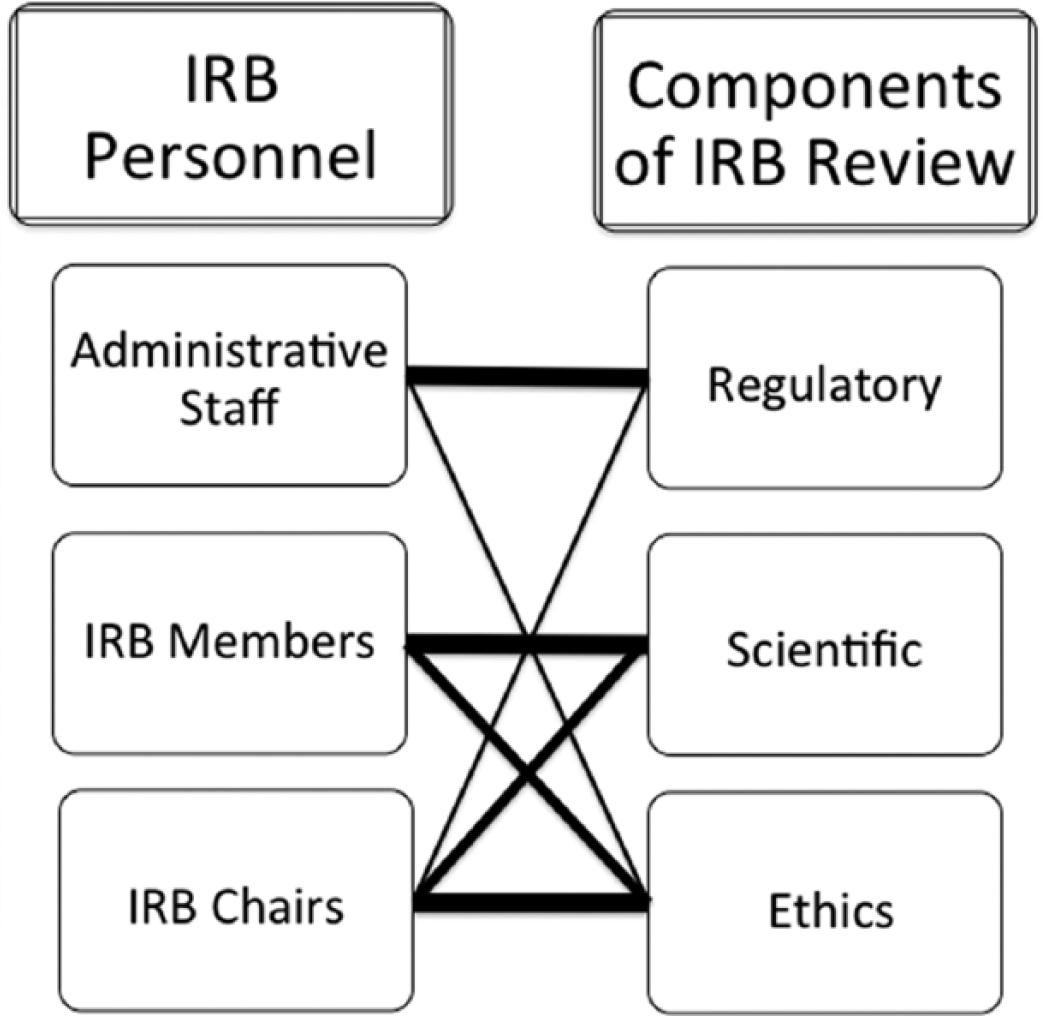
Schematic of IRB organization.
IRB review is made up of three overlapping and interactive components: regulatory, scientific, and ethical. Similarly, the work of the IRB is performed by three interactive groups of people, administrative staff, IRB members, and IRB chairs.
The primary focus of the IRB administrative staff should be compliance with regulations. An excellent IRB staff will ensure that all required components of the protocol and the consent form are present before the protocol proceeds to board review. For example, the IRB staff should identify special populations affected by the research, prompting the board to make the required determinations regarding inclusion of vulnerable populations. The administrative staff should also take primary responsibility for ensuring that all approved protocols are fully compliant with research regulations. This responsibility will involve close collaboration with IRB members and chairs, as certain regulatory determinations (e.g. pediatric and pregnancy risk categories) require medical expertise in conjunction with regulatory knowledge.
The foremost question in reviewing proposed human subjects research is determining whether the risks to human subjects are justified by the potential benefits to the subjects and the value of the knowledge to be gained. Board reviewers are primarily responsible for understanding and evaluating these risks and potential benefits. In addition, IRB members must be proficient in the design of protocols and the practical conduct of medical research, as they will frequently have the opportunity to identify modifications to study design that enhance protection of research subjects without hindering the acquisition of knowledge. IRB members should be selected in large part on the basis of scientific expertise and research experience in addition to a commitment to research ethics.
We posit that the most important job of the IRB chair is to identify ethical challenges posed by research protocols, and to guide deliberation. The IRB chair must clearly and explicitly articulate the ethical principles that relate to a given proposal, particularly noting when and how principles come into conflict. The IRB chair and other experienced IRB members should serve as role models for less experienced reviewers. By this, we mean not only that these personnel should possess practical wisdom and the other virtues critical for IRB members, but also that they should carefully explain their reasoning and articulate their arguments to enhance the training of newer members as they learn to deliberate. As Annas explains, ‘The ability both to teach and to learn a skill thus depends on the ability to convey an explanation by giving and receiving reasons’ (Annas, 2011: 19).
Meetings of the convened board should focus on research ethics, directly informed by knowledge of the science. It is neither possible nor desirable to separate ethics review from scientific review, because where there is more important knowledge to be gained more risk is justified. We therefore disagree with the proposal by the Institute of Medicine that scientific review of research proposals should be separated from ethics review (Institute of Medicine, 2002: 75–76).
Training and education
Research ethics training is mandated for researchers who undertake studies using human subjects, but there is currently no mandated program for the training of IRB members. We conceptualize research ethics education for IRB chairs and members as having three components: (i) knowledge of research regulations; (ii) instruction in research ethics principles; and (iii) training of the virtues most essential to IRB members, through the practice of IRB deliberation.
Principles of research ethics can be introduced via readings and live or on-line lectures, but IRB members require mastery of these principles that is better fostered through analysis of case studies, ideally in small groups. Understanding the definitions of autonomy, justice, and beneficence does not suffice; rather, research ethics professionals must understand how and why these principles apply in diverse situations so that they can use them to resolve ethical dilemmas.
A review of scientific ethics training programs demonstrated that the effectiveness of many programs was low. The most successful programs were ‘case-based, interactive and allowed participants to learn and practice the application of real-world ethical decision-making skills’ (Antes et al., 2009). The training of new IRB members should consist of practice in the following: (a) identifying the principles at stake in a particular case; (b) identifying how these principles may come into conflict; and (c) discussing how different decisions might adjudicate these conflicts. Real-life cases that challenge IRBs offer valuable opportunities for education. These exceptional cases will change over time, with the evolution of medical and research practices. The value of discussing challenging cases is enhanced when members are taught to pinpoint areas of conflict and to articulate how they would resolve the conflict. This approach aligns with Edmund Pellegrino’s description of high quality bioethics education: ‘When properly taught, [ethics education] demands that reasons be given for moral choices, that opposing viewpoints be given an adequate response, and it encourages the laying bare of underlying prelogical assumptions in any ethical argument’ (Pellegrino, 2007: 179).
Education of IRB members should be designed to foster the virtues that promote excellence in research review. Education informed by virtue ethics requires a focus on reasons and explanations. ‘It is the drive to “give an account” of what we are doing, to ask for and give reasons, that enables us to become virtuous, rather than merely habituated to act in certain ways’ (Annas, 2011: 54).
Virtue ethics teaches the importance of role models in the development of virtue. Following role models, or doing what a virtuous person would do, is not mere mimicry. Rather, as Annas writes, ‘Doing what your role model does is not repeating that action, but coming to understand what they were doing, in performing that action, and being able to do that yourself, perhaps in a quite different way’ (Annas, 2011: 23). Russell suggests that virtue ethics can ‘guide action not so much by telling agents what to do as by directing their development into ethically mature agents who are better able to tell for themselves what to do’ (Russell, 2009: 3). Virtues are learned from role models, then refined and advanced through practice and experience. Small-group discussions, led by IRB chairs or other experienced research-ethics professionals, should focus on the process of deliberation, on identifying salient factors and discussing the implications of the different ways one might adjudicate among them. The importance of chairs and experienced IRB members in training newer IRB members cannot, therefore, be overstated.
As newly appointed IRB members develop a degree of mastery of the ethical principles involved in research regulation, they should begin participating actively in IRB sessions, so that they can gain further proficiency, learning from role models, and from their own experiences in deliberation. IRB experience facilitates the pattern recognition skills that comprise much of the virtue we refer to as practical wisdom: after engaging in a discipline or an activity over an extended period of time, an intuition develops based on shared aspects of different experiences that can be enormously helpful in identifying and analyzing problems and in finding their solutions.
In addition to training IRB members, a robust research ethics education program must include the broader community of researchers. The importance of the ‘responsible investigator’ was emphasized by Henry Beecher, and has been reiterated and reemphasized by MacFarlane (Beecher, 1966; MacFarlane, 2009). Research ethics education can reach this broader community through several means. The usual on-line training courses for human subject protections and good clinical practice provide an important basic exposure to research regulations and research ethics. All investigators must successfully complete these before their research on human subjects is approved. However, virtue ethics’ emphasis on the importance of role models and articulacy in deliberation suggests additional opportunities.
Ideally, the scientific members of the IRB will be practicing researchers, drawn from different departments within academic institutions. IRB members should be encouraged to share their expertise and advocate for the responsible conduct of research when they interact with their colleagues in the research community, outside of the IRB. When presenting or discussing their own research, IRB members have the opportunity to discuss ethics questions and their resolution so that a culture of thoughtful ethical deliberation will diffuse throughout each IRB’s institution.
IRB chairs also have the opportunity to educate investigators when they communicate the decisions of the IRB. The value of a clear report of the IRB deliberation process and careful articulation of the IRB’s reasoning in this context should not be overlooked. In addition, IRB chairs should encourage investigators proposing ethically challenging research to attend the IRB meetings when their research protocols will be discussed so that they can participate in deliberation about whether and how the challenges can be addressed and so that they will better understand the ethical concerns of the IRB in the future.
Regulations
Research regulations should ensure an acceptable standard of research conduct, without unduly constraining the research endeavor. The dangers of erring on either side of this balance are real: under-regulation fails to protect the interests and welfare of research subjects, whereas overregulation impedes the conduct of valuable, ethically sound research studies. A virtue ethics approach begins with the understanding that no regulatory code or set of guiding principles is sufficient for optimal resolution of all ethical dilemmas. A set of regulations that attempts to anticipate and manage every contingency will be too inflexible to respond to unforeseen circumstances and can only hamper an IRB’s pursuit of excellence. Rather than trust in edicts, virtue ethics would encourage us to put our trust in IRBs staffed by people who are well-trained in the virtues and dedicated to navigating the best possible path through a complex and changing landscape.
Medical research is a rapidly evolving field, with new ethical challenges emerging routinely. Properly trained IRB members will be best positioned to reach optimal decisions when they are given freedom to exercise their judgment as regulators of research ethics. IRBs can best assure integrity in medical research when research regulations allow IRBs the flexibility to enact creative solutions to new problems as they arise.
In general, virtue ethics is more adaptable to different contexts and changing circumstances than are written codes of conduct (MacFarlane, 2009), and the freedom of IRB members to exercise their judgment is particularly important in novel situations where practical wisdom has the greatest role to play. Genomic testing guidelines provide a useful example of the need for regulatory restraint. Genomic research is a rapidly changing field, taking place in a society with rapidly evolving, community-specific knowledge and attitudes. Properly educated IRB members are ideally situated to identify nuances in proposals, and to work with researchers to develop optimal, case-specific approaches to return of incidental or secondary findings. As facility with these studies evolves, a body of precedents will emerge, gradually defining best practices. Regulations can then be put in place to ensure that minimal standards are met, allowing for IRBs and researchers to customize approaches when appropriate. The approach we are advocating here is sharply at odds with that proposed by Knoppers et al. (2015), at the conclusion of their clear and informative review of the laws and policies guiding the return of results from whole genome sequencing. These ethicists call for ‘anticipatory governance’ of the return of secondary (incidental) findings (Knoppers et al., 2015). We believe that such anticipatory governance is not desirable.
Which virtues?
There are almost as many lists of important virtues as there are philosophers interested in the concept of virtue. Plato identified wisdom, temperance, justice and courage as cardinal virtues (Plato, 360 BCE). Aristotle expanded the list to: justice, courage, temperance, magnificence, magnanimity, liberality, gentleness, prudence, and, especially, wisdom (Aristotle, 350 BCE). Some more modern ethicists posit that ‘role-differentiated virtues’ exist for different professions (Oakley, 2013). Pellegrino and Thomasma (1993) offer a list of virtues for physicians. MacFarlane offers quite a different list of virtues most appropriate to researchers (MacFarlane, 2009). In the same work, MacFarlane argues that virtues are ‘contingent on the social and historical contexts’ and so they vary from culture to culture. Others disagree about the significance of profession-specific virtues: McIntyre contends that the virtues are independent of professional skills, and Annas’ theory of virtue is explicitly focused on the virtue of individual people (Annas, 2011; MacIntyre, 1981).
We have argued above that the primary virtue necessary for IRB members and chairs is practical wisdom. The ability to discern which ethical questions are most pertinent, and when the specifics of a case demand prioritization of one or another principle it is invaluable. Beyond positing the centrality of practical wisdom, we will not attempt to create a definitive list of the virtues required for excellence in research oversight. However, the challenges routinely faced by IRBs suggest certain virtues that should be recognized and fostered.
IRBs must review proposed research that impacts two distinct groups of stakeholders: the future subjects of the research and the investigators conducting the research. The virtue of empathy allows reviewers to envision a research study from the perspective of a research subject or surrogate, understanding what information is required for subjects to make appropriate and informed decisions about research participation. The virtue of self- and group-awareness allows members to recognize when implicit biases and assumptions as well as intra-group interactions and relationships may interfere with optimal deliberation. The virtue of courage allows members to voice concerns about a protocol that may originate from within the member’s own department, for example, or to voice an opinion that differs from the majority of the board.
Finally, IRB members, and particularly IRB leadership, must be resolute. The lure of scientific discovery is strong, and the temptation will always exist for expedient solutions in study design or recruitment strategies. A resolute chair will engage directly and extensively with the researchers as well as with his or her board to resolve problems, but will not yield to expediency if an ethically sound approach cannot be found.
Potential objections
Several questions our proposal might raise are discussed below.
Is eudaimonia sufficient to motivate and direct virtuous behavior?
Virtue ethics is based on the premise that people can recognize excellence and that they are motivated to achieve excellence. How well that generalization applies to humanity as a whole is a very legitimate question, but we believe that this optimistic view does apply to individuals who choose to serve on IRBs. IRB membership is largely self-selecting for people who sincerely want to advance responsible research. These individuals are already sufficiently interested in research ethics to contribute significant time and energy toward helping to ensure that valuable research is carried out in a context that protects human subjects. We are optimistic that eudaimonia will motivate and guide IRB members to engage in the thoughtful practice necessary to cultivate the virtues and skills that IRB work requires.
We believe that IRB members will engage even more strongly with the research ethics endeavor if given appropriate training and opportunities to use their judgment. Pellegrino emphasizes the critical need for a ‘moral foundation’ for professionalism. Without a ‘moral foundation’, he writes, ‘the professions do indeed become simply occupations, jobs, or means of livelihood like any other’ (Pellegrino, 2007: 61). Our proposals that IRBs focus on virtue and that IRB members be given more autonomy should reinforce each other. Greater autonomy increases the rewards and motivational utility of eudaimonia, which in turn will help IRB members to achieve excellence as research ethicists.
Might an emphasis on virtue ethics protect human subjects less well than principles and regulations-based approaches?
Clinical research comes with a long history of ethically poor decisions and inadequate oversight. For that reason alone, research regulations will always be necessary to provide a baseline ethical and legal standard. Calls for less regulation must come from within a profession firmly committed to developing and maintaining the capacity to bear this trust. Regarding medical ethics, Pellegrino writes: ‘The tragic outcome of lax virtue is to invite increased societal control over the profession by means of externally imposed rules and regulations. These not only shrink professional latitude, as noted above, but also diminish the professional as a person’ (Pellegrino, 2007: 178). Our proposal that excellence in research ethics requires limited regulation and greater latitude for IRBs to exercise their judgment can only be considered in the context of a firm commitment to IRB training and education and to deliberating within a virtue ethics framework.
Will different IRBs be more variable in their standards and decisions if they rely on virtue ethics?
Several ethicists have described and criticized the variability in IRB ethics reviews (Freedman et al., 1993; Shah et al., 2004; Silverman et al., 2001), particularly because variability in review can hinder or complicate the conduct of multi-site studies (Kahn et al., 2014; Petersen et al., 2012; Ravina et al., 2010). We acknowledge that encouraging flexibility in regulations and reliance on board members’ thoughtful deliberation may result in more heterogeneity among IRBs. We strongly support the use of centralized IRB review for multi-site trials, which can mitigate this concern (Emanuel et al., 2004). For single-site studies, we contend that thoughtful deliberation will yield more consistently high-quality reviews than will reliance on detailed rules, albeit at a potential cost of more heterogeneity. To the extent that heterogeneity is identified, the cases in question should prompt further discussion, helping to inform ongoing debates in research ethics.
Evidence and assessment
We have suggested three ways in which the lessons of virtue ethics suggest revisions or refinements to the current research regulation paradigm: (i) changes in the structure of the IRB; (ii) focus on virtue ethics in the education of IRB members; and (iii) avoidance of overregulation of research practices. We must ask, then, what are the measurable outcomes by which we can evaluate the impact of these changes?
In their 2002 report, Integrity in Scientific Research: Creating an Environment that Promotes Responsible Conduct, the Institute of Medicine concludes that ‘the principal mode for evaluation of the effectiveness of an integrated program should be based on self-assessment and peer review … in the context of institutional accreditation’ (Institute of Medicine, 2002: 112). This self-assessment and peer-review approach is consistent with that used by the IRB accrediting organization, Association for the Accreditation of Human Research Protection Programs (AAHRPP, http://www.aahrpp.org).
Specific components of the IRB system are amenable to quantifiable assessment. Surveys have been used to assess the utility of training in the responsible conduct of research in preparing participants to recognize, avoid, and respond to research misconduct (Plemmons et al., 2006). Powell and colleagues describe a method to evaluate a responsible conduct of research course, in which decision-making skills are evaluated through individuals’ responses to brief scenarios (Powell et al., 2007). Evaluation and discussion of this type of qualitative assessment would be time-consuming, and we do not propose that it replace current research ethics training for investigators. We do believe it would be valuable for IRB members, however, perhaps as a yearly group session. We expect that incorporating the lessons of virtue ethics into the training of IRB members will improve quantifiable decision-making skills.
Investigators are a further potential source of quantifiable feedback for the IRB. Periodic evaluation of the IRB by investigators can reveal whether investigators see the review process as fair and thoughtful, whether the results of IRB deliberations were communicated to them in a clear and helpful manner, and whether investigators think that the process improved the science or the ethics of the research.
Conclusions
As medical technology and research methodologies evolve, IRBs are continually faced with novel research ethics quandaries. As such, there is an increasing need to ensure articulate and robust research ethics deliberation. Research ethics regulations are necessary to set a minimal legal and ethical standard for the protection of human subjects, and principles make essential ethical claims on IRB decision-making. These two elements are foundational to research oversight, but excellence in research oversight requires that IRB leadership and members develop pertinent virtues and that they be given freedom to exercise them in adjudicating these claims and interpreting these regulations. A research oversight program that draws heavily upon virtue ethics will be optimally positioned to face emerging challenges and to ensure that human subjects are appropriately protected as researchers work to advance medical knowledge.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
