Abstract
In this paper, we discuss challenges associated with implementing a policy for Ancillary Care (AC) for related and unrelated (serious) adverse events during an Ebola vaccine trial conducted in a remote area of the Democratic Republic of the Congo. Conducting clinical trials in resourceconstrained settings can raise context-related challenges that have implications for study participants’ health and wellbeing. During the Ebola vaccine study, three participants were injured in road traffic accidents, but there were unexpected difficulties when trying to apply the AC policy. First, because of the nature of the adverse events, the insurer refused to cover the costs. Second, the AC policy did not address treatments by traditional medicine, even though traditional medicines are frequently used and highly trusted in the study community. This highlighted a contrast between the researchers’ well-intentioned AC approach and the participants’ legitimate preferences. The way in which researchers should address their responsibility to provide AC is not straightforward; it requires contextualization. Our experience highlights the importance of involving community representatives and the local ethics committee to ensure development of an AC policy that is culturally and ethically appropriate. Additionally, the insurance contract should clearly stipulate which adverse events are linked to the trial participation, and thus eligible for coverage, to avoid controversies when claims are made.
Keywords
Ensuring adequate provisions for research-related harm is one of the primary responsibilities of researchers and research sponsors, overseen by Ethics Committees (ECs). According to the Council for International Organizations of Medical Sciences (CIOMS), aspects like the informed consent procedures, caring for participants’ health needs during and after research, compensation for participation, and indemnity in the event of research-related harm, require due attention and tailored measures to each particular study site (CIOMS, 2021). The CIOMS guidelines also state that researchers have an obligation to care for participants’ health needs, and that in resource constrained settings (RCS), providing medical care beyond the scope of the specific clinical trial should not be seen as undue influence on trial participation. Moreover, CIOMS explicitly refers to the provision of ancillary care as a way to balance the burdens and benefits of research.
Ancillary care (AC) is the care provided to trial participants when a medical event is not related to the research question or intervention (Belsky and Richardson, 2004; Pratt et al., 2013). When clinical trials are conducted in RCS, the provision of AC might be seen as a legitimate benefit of research participation (Haire and Ogundokun, 2014). Unfortunately, CIOMS does not further elaborate on how the provision of AC should be translated into practice across different research contexts and settings, leaving the issue open to interpretation, discretion, and the benevolence of researchers.
To the best of our knowledge, there are no internationally binding regulations on AC, and clear guidance is lacking for ECs to evaluate the adequacy of AC provisions (Kapumba et al., 2022). Furthermore, the Democratic Republic of Congo (DRC), where our study was based, does not have any specific legislation or guidelines on this topic either. Hence, the sponsor and principal investigator (PI) of the Ebola vaccine trial designed and put in place their own measures to ensure adequate AC, trying to take into account the contextual conditions in the study area. In this article, first, we provide an overview of the study and context; second, we present the AC policy developed for this study; and third, we focus on some challenges experienced when implementing the AC policy for two mobility-related events.
The vaccine study and context
Under the sponsorship of the University of Antwerp, with the University of Kinshasa as PI, an Ebola vaccine trial (NCT04186000) was set up at the end of 2019 in Boende, a remote Ebola endemic region in the DRC. Overall, 699 health care providers (HCPs) were recruited to this study which aimed to better prepare the region for future outbreaks. The study vaccination schedule was as follows: a two-dose heterologous vaccine regimen was administered, with a 56-day interval between both doses, followed by a booster dose either 1 or 2 years after the initial dose (Larivière et al., 2021). The HCPs were enrolled for a period of 30 months; the study was completed in October 2022 and analyses are ongoing at the time of writing.
Before the start of the trial, the team developed a community engagement strategy that was carried out by anthropologists of the University of Kinshasa. Several community engagement and sensitization efforts (e.g. meetings and workshops) were made to inform potential participants, address possible fears, mistrust, or preconceived notions amongst the population about the Ebola vaccine trial and, a year after the start, about the COVID-19 pandemic. Local political and health authorities were involved, as well as representatives of the civil society, non-governmental organizations and potential participants. Additionally, the study team consulted and collaborated with the local health authorities to help anticipate the potential impacts of the trial activities on the HCPs’ duties in the community (Zola Matuvanga et al., 2022).
The DRC health system is heavily dependent on external aid and out-of-pocket contributions from patients (Institute for Health Metrics, 2022; World Health Organization, 2015). In many sub-Saharan African countries, community health insurance schemes have been created to provide access to essential health care for the most vulnerable, but they suffer from operational obstacles that hinder their successful roll-out (De Allegri et al., 2009). Some scholars point to uncertainty about whether these schemes always facilitate access to care; the poorest cannot afford the user fees and remain excluded from care (Wiysonge et al., 2017). Thus the capacity of health insurance mechanisms, either formally implemented (via government or private companies) or organized at community level, to enhance the quality and affordability of healthcare, remains weak (CIOMS, 2021; Dawkins et al., 2021). As in other RCS, access to good quality health care and to effective health insurance schemes remains a challenge throughout the DRC, particularly in rural areas (Criel et al., 2020; World Health Organization, 2015). Should diseases or injuries require specialized medical care, those who have the personal financial means can access the facilities in the capital or closer (provincial) cities where the appropriate medical provisions are available. However, the majority of the population cannot afford self-funded referrals and remains dependent upon the local health services, which may be unable to provide the needed care. Local services are generally very basic; there can be shortages of essential medical equipment or skilled and qualified staff, or they may be difficult to reach due to logistical challenges such as long distances and lack of public transport. This situation creates a context-related health vulnerability, caused by the lack of access to quality health care services, that can have a direct impact on participation in clinical research, and on the measures that researchers should take to ensure appropriate health care and protection of research participants.
AC policy in the Ebola vaccine trial
To facilitate a non-arbitrarily approach and support the care needed for both adverse events and serious adverse events ((S)AEs) of participants, a context-specific algorithm was developed with clear criteria and decision-making principles (Lemey et al., 2021). We refer to the International Conference for Harmonization of Technical Requirements for Pharmaceuticals for Human Use to define AE and SAE as follows:
An AE is a medical occurrence which does not necessarily have a causal relationship with the investigational product.
An SAE ‘is any untoward medical occurrence that at any dose results in death; is life-threatening; requires inpatient hospitalisation or results in prolongation of existing hospitalisation; results in persistent or significant disability/incapacity; is a congenital anomaly/birth defect; is a medically important event or reaction’ (European Medicines Agency, 2004).
Officially introduced in November 2021, when the study was already ongoing, the newly developed AC policy was presented during a one-on-one informed consent procedure conducted before participants’ next scheduled study visit. All participants were informed and consented to the terms (e.g. for reimbursement) and possible support options provided in case of a medical event. They were advised to report any (S)AEs to the study team, whether or not they could be considered solicited events, and for which a possible support outcome would be determined. The algorithm provided guidance for covering (medically and financially) all medical needs beyond research intervention-related harm – an element appraised by the EC – and including events that were treated offsite. The algorithm policy was also applied retrospectively, where possible, to non-related SAEs that had occurred between the start of the trial and issuing the AC policy. The development and specific modalities of this AC approach have been described by Lemey et al. (2021). The algorithm was developed through a collaborative process between the Sponsor, based in Belgium, and the PI, based in Kinshasa, DRC. The lack of direct involvement of local health staff, community members or lay people from the study area may have contributed to the implementation challenges described in this paper; an element which the research partners will take to heart in future studies.
When developing this policy, injuries occurring from ‘actions and displacements directly linked to trial participation or during trial activities on site’ were classified as related to study participation. For compensation, these cases were perceived to fall under sponsor responsibility. In accordance with Good Clinical Practices (GCP) a ‘no fault’ policy insurance was taken out (Belgisch Staatsblad, 2004; ICH, 2016; Ministère de la Santé, 2011).
Traffic accidents
HCPs were recruited from the Boende health zone (Figure 1). According to the DRC Ministry of Health, a health zone is a geographically well-identified space covering a theoretical population of 100,000–150,000, and is responsible for delivery of comprehensive and continuous health care (Ministère de la Santé, 2012). The Boende health zone is 10,775 km² in size, roughly the size of the Gambia, and currently has a population of 316,145 (Ministère de la Santé, 2021). The area, intersected by the majestic Tshuapa river, is characterized by poor road infrastructure and a lack of public transport for moving through the dense tropical forest. Many trial participants had to travel long distances to reach the study site, with dugout canoes and motorcycles as the most common and affordable means of transport at their disposal (Zola Matuvanga et al., 2022). To compensate the participants for participation in the research fairly, financial reimbursement was foreseen to cover time spent and travel expenses. The amounts had been discussed with local health authorities and community representatives, and were approved by the Comité National d’Ethique de la Santé – the EC appointed by the Ministry of Public Health in DRC (Saleh et al., 2020; Zola Matuvanga et al., 2022). Given the geographic and logistical circumstances, it is perhaps not surprising that three participants had been involved in motorcycle accidents, with medical consequences and the need for care. As Table 1 indicates, case B was considered to be unrelated to the investigational product or to trial participation, as the accident did not occur while travelling to or from the trial site before/after a scheduled study visit. However, in line with the AC approach that was implemented, the medical care for this unrelated SAE was covered by the study funds. As the traffic accidents of participants A and C happened during essential travel to partake in the trial activities, the sponsor and PI declared them ‘related to trial participation’. This paper focusses specifically on the latter cases, because they led to deviations from the study’s AC policy, and we can draw from them several lessons for improving the policy.
Boende health zone.
Serious adverse events caused by traffic accidents.
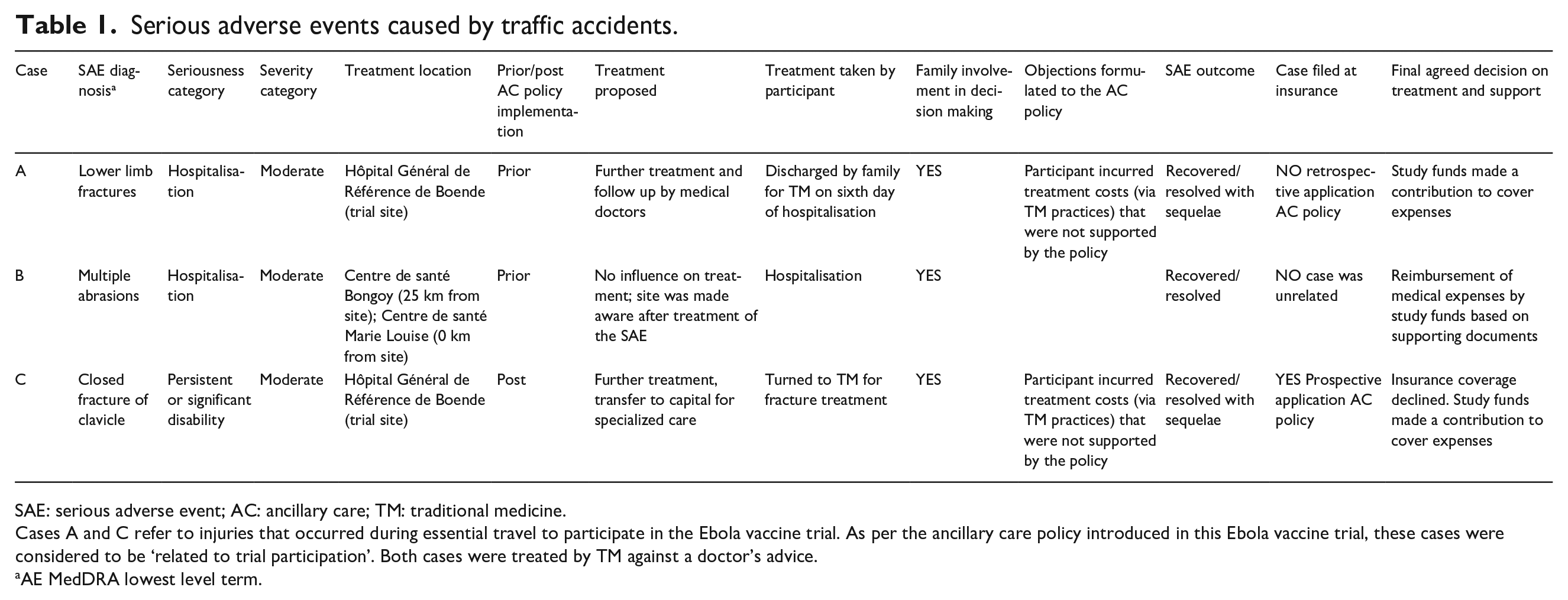
SAE: serious adverse event; AC: ancillary care; TM: traditional medicine.
Cases A and C refer to injuries that occurred during essential travel to participate in the Ebola vaccine trial. As per the ancillary care policy introduced in this Ebola vaccine trial, these cases were considered to be ‘related to trial participation’. Both cases were treated by TM against a doctor’s advice.
AE MedDRA lowest level term.
Participant A
A few months into the trial activities and before the implementation of the AC policy, the frontline research team was informed by the hospital emergency services that participant A was involved in a traffic accident and was diagnosed with a lower limb fracture. The participant had just left the site after having received the second vaccine. As participant A was driving the motorbike, a link to the investigational product (which could have influenced the accident) could not immediately be ruled out. After thorough medical assessments and queries about the circumstances of the traffic accident, this event was reported as unrelated to the investigational product. At the hospital a plaster splint was administered and the participant was kept in hospital. After the sixth day of hospitalization, family members discharged the participant, against the doctor’s advice, and sought treatment for the fractures from traditional medicine (TM). After a worsening of the condition, the participant returned to the hospital for treatment, but the fractures never healed fully. Thus, the outcome of the SAE was deemed ‘recovered with sequelae’. When the AC policy was implemented in 2021, the participant requested compensation for the incurred medical expenses retrospectively. As the motorcycle trip was essential to participate in the trial, the sponsor and PI considered this case to be related to trial participation. However, the fact that the fracture was treated by TM against the study team advice, rendered the participant ineligible for insurance coverage.
The resolution dialogue with this participant was straightforward and an agreement was reached jointly between the study staff and the participant. First, the hospital care would be reimbursed, but the costs for TM would not. Second, the costs for the subsequent hospital care following deterioration would be partially met because the self-discharge was explicitly against the doctor’s advice. The medical expenses were reimbursed from the trial budget, which helped the participant to repay loans taken to cover treatment costs.
Participant C
The SAE was caused by a motorcycle accident that took place 85 km from the General Reference Hospital of Boende. Participant C, being the passenger of the vehicle, was traveling to the trial site for a planned visit. The diagnosis was a closed fracture at the level of the clavicle. Although suffering from this medical condition, the participant proceeded with the trial’s visit that day. As it took place after the study AC policy had been implemented, the sponsor and PI immediately considered the event related to trial participation. Subsequently, a report and claim were submitted to the insurance company.
However, subsequent negotiations with the participant and the family concerning the medical care and support were lengthy and complex, and spread over several days. Given that the local care available in Boende significantly diverges from adequate standards for optimal treatment, and that this injury was assessed to be related to trial participation, the sponsor and PI agreed that extra medical care should be sought. Therefore, it was suggested that the participant transfer to Kinshasa for specialised outpatient treatment. However, this suggestion raised several concerns related to the indirect costs of the referral. For example: Where will I stay? Where will I eat? How will I get to the hospital? Who will pay for my garde malade (caregiver)? These concerns were difficult to address, as modalities for referral to the capital had not been foreseen for the AC policy and the related budget. This clearly revealed a gap in the policy that needs further consideration and improvement for future studies. Additionally, the frontline researchers did not anticipate, nor were they prepared, for negotiations that extended beyond the usual relationship between the study and participants. Some members of the participant’s family also contested the description of the rights of participants and responsibilities of the trial team as described in the study information for informed consent. The involvement of the extended family and the community added complexity to discussions about the individual’s health options and best interests.
Participant C ultimately declined the proposed referral. The family members insisted on having the fracture treated with TM, which are widely utilised in the study area. Ultimately, a financial contribution, equivalent to that of participant A, was agreed for partial reimbursement of costs. Following up on this SAE, the study team learned that participant C had sought treatment by TM and that the fracture had a malunion, making the SAE outcome ‘recovered with sequelae’. Meanwhile, the insurance company ruled this case inadmissible as only cases with a clear causality to the investigational product qualified for reimbursement.
These two cases highlight some ethical challenges related to clinical researchers’ responsibilities in RCS, which are divided and discussed below in three categories; (1) local health practices, (2) insurance implications and (3) AC considerations in RCS. In what follows, the term ‘researcher’ refers to both the principal investigator conducting the study, as well as the sponsor responsible for the vaccine trial. Table 2 provides an overview of the different challenges encountered within these three categories, the study’s response, and recommendations for future studies.
Challenges, responses and recommendations.
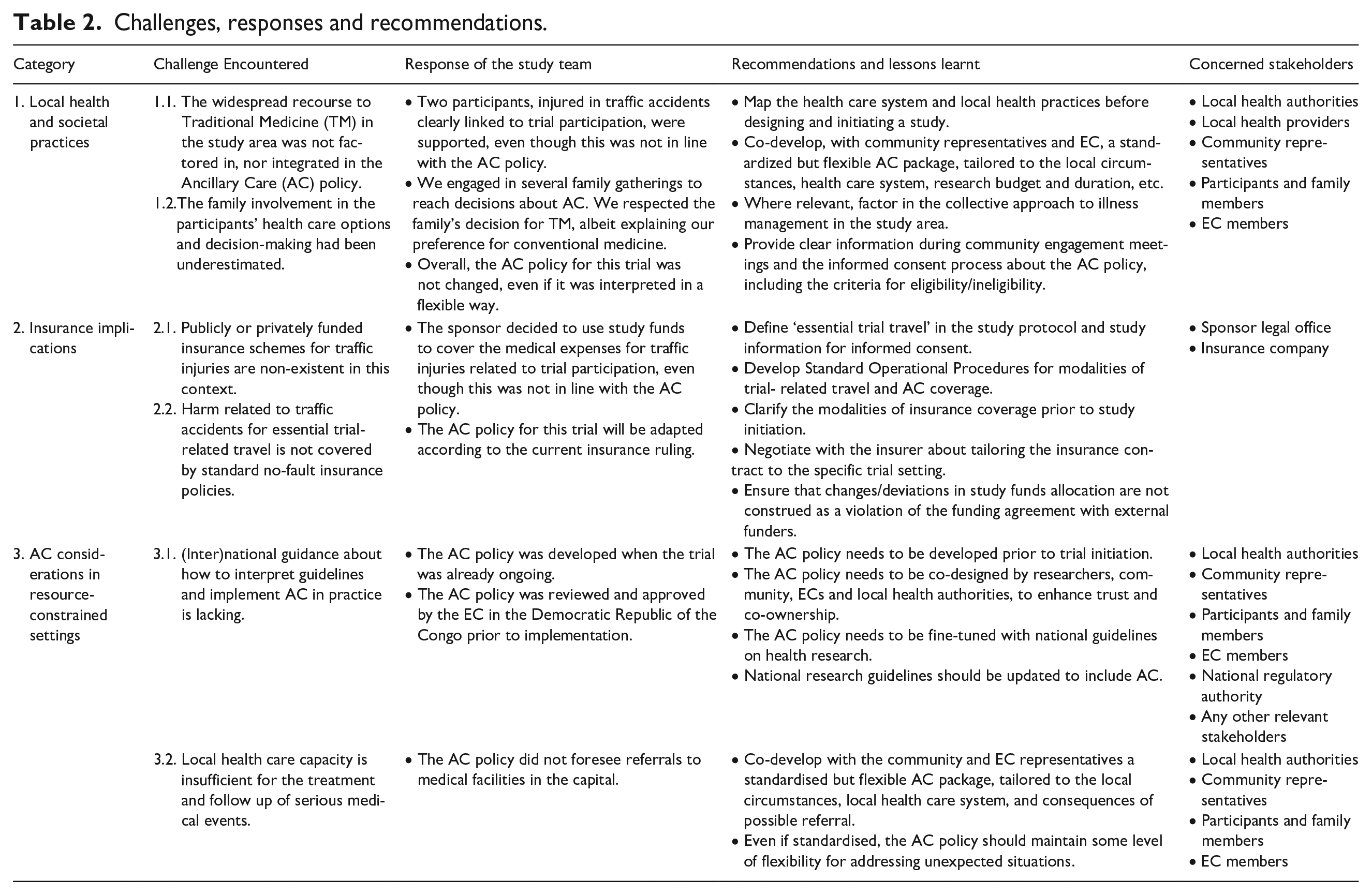
Local health and societal practices
Some of the local staff, as sub-investigators in the trial, were (indirectly) involved in the development of the AC policy, providing guidance and advice to the PI on what would be feasible and acceptable in the study area. However, when implementing the policy, the sponsor and PI rapidly realised that some contextual factors had not been duly integrated. In this case, that the population in the Boende region frequently turned to TM for health care needs (Table 2, 1.1). In the DRC, the Ministry of Health refers to TM as an essential part of the healthcare provision that at times can be the only choice (in absence of formal health structures), as well as incurring lower costs (Ministère de la Santé, 2018; Muela et al., 2000; Sato, 2012). Here, like in many African countries, TM is part of an established health infrastructure (Amzat and Razum, 2018; World Health Organization, 2013). This requires reflection on whether TM should be included in an AC, which does not seem to be addressed explicitly in current ethics and GCP guidelines.
As the SAE of participant A happened before the implementation of the AC policy, we can hypothesise that the choice of TM might have been avoided had the AC policy been available upfront. However, the study team observed that the family evaluated the outcome of the initial treatment of the fracture within the hospital as insufficient. This concern may have contributed to their decision to turn to TM. Additionally, the lack of specialized medical equipment at the hospital made it also difficult for the fracture of participant C to be confirmed or ruled out. Although the AC policy was already in place when this SAE occurred, it became clear that, for certain medical conditions such as fractures, TM remains a deep-rooted and preferred tradition and custom (Ministère de la Santé, 2018).
Moreover, the level of family involvement was very important when it came to an individual’s health choices (Table 2, 1.2). In both cases, the participant was either discharged by family members while hospitalized or was urged to refrain entirely from treatment by conventional medicine and seek TM instead. This was a significant reminder of the complexity of consent in this context, where individual consent is shaped and enriched by the contribution of the family members and other trusted persons. As pointed out by Muela et al., ‘people’s economic behaviour in treatment-seeking seems to follow the basic logic that the more an illness affects a social group, the more will the group be involved in illness management’ (Muela et al., 2000).
Notwithstanding a national DRC programme in place since 2001 that guides and regulates TM, these practices are generally poorly-known by those working in the conventional medicine sector, who may be concerned by the lack of (documented) efficacy and safety data for herbal medicine (Amzat and Razum, 2018; Ernst, 2004; Ministère de la Santé, 2018), and by the lack of formal training and regulation of traditional health practitioners (Amzat and Razum, 2018; Chatfield et al., 2018; Smith, 2008). The combination of lack of knowledge of TM and concerns about potential harms were influential factors for the sponsor and PI when considering the inclusion of (some) TM practices in the study AC policy. Based on this experience, we recommend that researchers map, recognize and respect the use of and trust in TM in communities in RCS. Also, as they cannot speak with authority regarding forms of healthcare that they are not familiar with, researchers must balance paternalistic inclinations to protect from harm, with respect for autonomy, both during the informed consent process, and when proposing a therapeutic approach for specific events. Otherwise there is a risk that the participants may feel coerced to opt for conventional medicine, rather than protected from possibly sub-optimal or inadequate cures, and the differences in perceptions and opinions could be exacerbated rather than reconciled.
Insurance implications
The study site is located in a very fragile setting. The social protection mechanisms usually available in high-income countries, as well as insurance coverage for private and public transport, are not present. From the viewpoint of the insurer (located in a high-income country), injuries from traffic accidents should be covered by other publicly or privately funded insurance schemes. Indeed, this corresponds to the usual arrangements in a clinical trial’s insurance policies, based on a strict interpretation of GCP guidelines; ‘[. . .] the Sponsor should provide insurance or should indemnify (legal and financial coverage) the investigator/the institution against claims arising from the trial, except for claims that arise from malpractice and/or negligence’ (ICH, 2016). However, other insurance schemes are virtually non-existent in this remote, poor context, and it is not realistic to expect participants in our trial to settle claims and disputes according to these practices (Table 2, 2.1).
In the case of participant C, the sponsor’s and PI’s assessment of trial-related harm did not align with the sponsor insurance. For the sponsor and PI, a causality link was justifiable given the specificities of the study setting, and the fact that participants would not have undertaken this trip if they had not been a study participant. According to the insurer, and in line with the usual interpretation of a clinical trial insurance, only injuries caused by the investigational product could be covered, assuming that what happens along the route to and from the study site is not considered to fall within sponsor and study responsibilities (Table 2, 2.2). However, the sponsor and PI felt that they had a moral responsibility vis-à-vis the trial participants, that goes beyond the legal responsibilities, to which that may be held accountable. In fact, the ICF stipulates that participants are required to travel to the site to participate in research, and that they will be reimbursed for the travel expenses. This implies a clear mutual commitment, where participants will travel, and the study will provide the travel budget and any travel-related support that may arise.
The sponsor had not explicitly requested the inclusion of trial-related travel in the insurance coverage, nor was this clearly stipulated in the study protocol and study standard operating procedures. In future studies, this should be negotiated upfront. However, we cannot take it for granted that insurers will accept this interpretation of study-relatedness, nor that it will be without a significant increase of the cost of the insurance policy. Therefore, more guidance is needed in GCP and ethics guidelines, to understand how the usual interpretation of trial-relatedness and insurance coverage may or should be adapted in RCS, to address full moral responsibility vis-à-vis socioeconomically vulnerable research participants.
Our experience also indicates that sponsors and researchers need more knowledge and support in contractual matters, in particular about the stipulations of insurance contracts for clinical trials conducted in RCS. Otherwise, there is a risk that a study insurance remains a costly requirement that does not fully protect trial participants in real-life circumstances.
AC considerations in resource-constrained settings
Binding national regulations on AC in clinical health research, as well as guidance on its translation into practice, are lacking in DRC (Table 2, 3.1). The sponsor and PI of this trial designed the AC policy to ensure that they would meet their responsibilities to study participants, and in the knowledge that the local care available in Boende was not the highest attainable standard of health in the country (Table 2, 3.2). They recognised that relying on the local care would, in some cases, be insufficient, and thus not morally defendable. Their concerns highlight the need for broader ethical reflection on researchers’ responsibilities in RCS. The CIOMS guidelines, for instance, point out that ‘research or investigators cannot, in general, be held accountable for unjust conditions prevailing where the research is conducted, but they must refrain from practices that are likely to worsen unjust conditions or contribute to new inequities’ (CIOMS, 2021). Referring a participant to the capital for medical care (cf. case C) was not originally foreseen in the AC policy, but given that optimal care in Boende was unfeasible, this deviation seemed the correct approach.
Furthermore, providing free quality (ancillary) care, that would not be continued after the trial, can exaggerate expectations; interventions that are not usually one of the treatment options for the majority of the population can create inequalities in the community. Also, adopting a different approach for medical events related and unrelated to the trial may create confusion and tension across study participants and within the community. This needs careful rethinking in the next revision of the AC policy for transparency, consistency and equity.
Additionally, the scope of researchers’ AC responsibilities during the research study is questioned when injuries are treated by TM. Does the duty of care for participants continue if medical guidance is not accepted? To what extent are they then accountable if there is deterioration of a condition? How can researchers best balance the need to respect a person’s autonomy, with the duty to protect them from unnecessary harm? The latter is a common question in medical ethics, whereby a respect for autonomy of a competent individual, whether or not embedded in collective interests or values, can conflict with the principles of beneficence and nonmaleficence (The National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research, 2014; Varkey, 2021). This ethical trade off illustrates the complex moral issues that can arise in research conducted in a RCS (Stuttaford et al., 2014). Reconciliating the two principles is a difficult exercise in practice, and the field researchers should not be left to decide which obligation prevails. Local ECs (where available) or community advisory groups might be able to help balance these obligations in a manner that is culturally acceptable and within the scope of a study’s accountability for participants.
Our experience confirms that robust community engagement, co-ownership and communication is essential in all aspects related to a clinical trial. When designing the next review of the study’s AC policy, we foresee engagement for meaningful dialogue not only with the community, but also with the EC representatives, in order to share our experiences with the AC policy in this study and discuss possible improvements. Their recommendations could help us to anticipate the expectations of participants, determine the responsibilities of researchers, and find ways to deal with future conflicts between different ethical obligations.
Conclusion and recommendations
The context in which research takes place can challenge research teams in many ways, but these challenges can, at least partially, be anticipated by good practices. First, researchers should map the available local health care options and involve the community in the development of adequate ancillary health care provisions for participants during research. Secondly, when TM is embedded within local health care infrastructures, related perceptions and expectations should be factored into the study’s AC policy. Thirdly, insurance contracts should be fair, transparent and contextualised to the local circumstances. Lastly, ECs could, in addition to critically reviewing the study provisions for AC and assessing the cultural and ethical appropriateness, also require researchers to explicitly report back on its implementation in the trial via yearly and final reports. Regulatory and ethics guidance in this regard would support sponsors and researchers to implement relevant and respectful AC measures. All these elements would contribute to enhancing trust in the research, to fairly address participant’s and community’s expectations, and to clearly frame the responsibilities of global health researchers.
Footnotes
Acknowledgements
The authors gratefully acknowledge the hard work and continuous dedication of the trial staff at the Hôpital Général de Référence de Boende throughout the Ebola vaccine trial.
Authors’ contributions
All authors were involved in the conception and development of this paper. HM and TZ represented the Principal Investigator in the negotiation process with the concerned participants; LB and EE were highly involved as representatives of the local medical hospital staff; on certain occasions GL represented the Sponsor during researcher-participant discussions. GL wrote the manuscript. RR and HM shared senior authorship. All authors read and approved the final manuscript.
Funding
All articles in Research Ethics are published as open access. There are no submission charges and no Article Processing Charges as these are fully funded by institutions through Knowledge Unlatched, resulting in no direct charge to authors. For more information about Knowledge Unlatched please see here: ![]() . This project has received funding from the Innovative Medicines Initiative 2 Joint Undertaking (IMI2 JU) under grant agreement No 800176. This Joint Undertaking receives support from the European Union’s Horizon 2020 research and innovation programme, European Federation of Pharmaceutical Industries and Associations (EFPIA) and the Coalition for Epidemic Preparedness Innovations (CEPI).
. This project has received funding from the Innovative Medicines Initiative 2 Joint Undertaking (IMI2 JU) under grant agreement No 800176. This Joint Undertaking receives support from the European Union’s Horizon 2020 research and innovation programme, European Federation of Pharmaceutical Industries and Associations (EFPIA) and the Coalition for Epidemic Preparedness Innovations (CEPI).
Ethical approval
The Ebola vaccine trial (ClinicalTrials.gov: NCT04186000) was approved by the National Ethics Committee of the Ministry of Health of the DRC (n°121/CNES/BN/PMMF/2019) and the Ethics Committee of the University Hospital of Antwerp/University of Antwerp (19/14/177). The ancillary care policy within the Ebola vaccine study obtained approval of the DRC National Ethics Committee (n°231/CNES/BN/PMMF/2021) in March 2021.
