Abstract
This paper addresses psychological factors that might interfere with informed consent on the part of stable patients as potential early-phase clinical trial participants. Thirty-six semistructured interviews with patients who had either diabetes or gout were conducted. We investigated stable patients’ attitudes towards participating in a fictitious first-in-human trial of a novel intervention. We focused on an in-depth analysis of those statements and explanations that indicated the existence of psychological factors impairing decision-making capacity. Three main themes emerged: insufficient comprehension of the inherent logic of clinical trials (actual comprehension), the recourse to trust over comprehension (prioritization of trust), and visceral factors that override deliberative process (visceral factors). Overall, our results indicate a limited psychological capacity on the part of stable patients to meet the requirements of informed consent as set by Declaration of Helsinki. A redesigned informed consent procedure should take account of these psychological realities.
Keywords
Introduction
According to the Declaration of Helsinki (World Medical Association, 2013), the research participant must be “adequately informed” about the goals, risks, and harms involved in a given study and has to “understand the information.” Further, the participant should not be “in a dependent relationship with the physician” and should “agree freely.” However, several weaknesses in human decision-making have been found to undermine informed consent.
It has been questioned whether the way patients are typically informed ensures understanding (Eyal, 2011; Levy, 2014). Many weaknesses in human reasoning have been discussed, including motivated reasoning (Levy, 2014), social desirability bias (Redelmeier and Dickinson, 2011), and heuristics in general (Lee, 2011). Regarding informed consent to participation in an early-phase clinical trial, all of these are likely to be at play. This is because people mostly have little knowledge concerning new technologies and the logic of clinical studies and therefore are more likely to have a misconception regarding the therapeutic value of participation. A recent literature review found that participants in clinical trials have very limited understanding of the components of informed consent and of the nature of the study (Tam et al., 2015). Another review of participation in phase I clinical trials concluded that consenting participants displayed insufficient comprehension of the trial purpose as well as unrealistic expectations of the benefits and risks (Cox et al., 2006).
In addition, patients often suffer from psychological factors related to their illness, for example, fear or desperation, which can damage decision-making capacity (Boyd, 2015; Hewlett, 1996). Furthermore, the operation of visceral factors, which include drive states such as hunger, thirst, physical pain, and emotions, has been attributed to behavior that is contrary to a person’s self-interest (Loewenstein, 1996). Such drive states affect behavior by influencing brain functions and mental activities (Critchley and Harrison, 2013) and, ultimately, can override rational deliberation (Loewenstein, 1996). The strong presence of one visceral factor dominates all goals except striving to fulfil the visceral need; that often short-term need predominates decision-making and can cause patterns of behavior, which can lead to long-term harm (Loewenstein, 1996). Considering all these factors together, it has even been questioned whether informed consent is possible at all (Boyd, 2015). In fact, current research appears to validate the view that without adopting a more realistic conception of rationality, some bioethical principles will be difficult to realize in practice and “remain clumsy and often irrelevant” (Lee, 2011: 368).
Nevertheless, there has been little discussion concerning informed consent of stable patients to participation in early-phase clinical trials. Phase 1 clinical trials are traditionally carried out with terminally ill patients such as those suffering from cancer. Since chronically ill patients such as those diagnosed with diabetes or gout often have an almost normal life expectancy, they are at a higher risk of suffering from possible long-term side-effects of experimental treatment during their lifetime than patients with a terminal disease (Dresser, 2009) (although, for the same reason, stable patients have a higher chance of benefiting from a study’s findings in the long term). To justify early-phase clinical trials among stable patients, it is therefore even more important to minimize the risk that consent is unduly influenced.
In order to contribute to filling this research gap regarding factors that might interfere with informed consent on the part of stable patients as potential early-phase clinical trial participants, we conducted an exploratory qualitative study. The aim was to identify and analyze psychological factors that could interfere with stable patients’ ability to provide informed consent to participation in a clinical trial. Accordingly, we chose a group of patients who are suffering from significant disease, but who are not close to death as in first-in-human trials involving new cancer drugs. We addressed issues where consent is related to the trial rather than to clinical treatment, because the lack of direct benefit from participation in research poses particular challenges to the validity of consent, and even more so in early phase clinical trials. Performing an in-depth analysis of explanations that indicated the existence of psychological factors determining decision-making, we identified three main themes: (a) actual comprehension; (b) prioritization of trust (over comprehension); and (c) visceral factors. We argue that these themes are of cardinal importance when thinking about informed consent, and even more so when consenting to participation in an early-phase clinical trial.
Methods
We conducted 36 semistructured interviews with patients who had either diabetes or gout and asked for their reactions to a fictitious first-in-human trial participation. The interviews were conducted during a time period of six months from December 2014 to May 2015. We focused on an in-depth analysis of those statements and explanations that indicated the existence of psychological factors that may interfere with an unfettered decision-making. Informed consent documents and the study protocol were reviewed and approved by the local ethics committee (Nord-West and Central Switzerland Ethics Committee, EKNZ) before the recruitment began.
Study Tool
The interview guide consisted of 10 open questions, each with several subquestions prompting participants on details, for example, current health status, knowledge regarding technology, and opinions on research participation. During the semistructured interview, the researcher provided additional information about a novel intervention by reading a case study (Rakic et al., 2017) as well as showing information graphics on the intervention and on clinical trials in general. The hypothetical example presented during the interview depicted the implantation of autologous and modified cells (removed from a person and later implanted into that same person). The field of new studies using synthetic biology (SB) devices was chosen as these questions are under-researched despite presenting important challenges that are typical for future medicine. The interviews were part of a larger project investigating cutting-edge biotechnology, but our focus here was on potential research participants’ (i.e., stable patients’) attitudes and knowledge towards research and their decision-making process when confronted with a possible research participation rather than on novel interventions. Participants were informed that this treatment could eventually be able to cure their illness one day (though not necessarily for them directly). In that case no further medication would be needed. The example was openly described as a hypothetical one. Concomitantly, all participants were systematically informed about how early-phase clinical trials (e.g., phase I, II, and III) work and asked for their potential willingness to and reasoning for participating in such a trial.
Sample
The qualitative study was carried out with a convenience sample of 36 patients suffering from either gout or diabetes and were in a stable stage of health when they were asked for participations. This group was chosen as being representative of stable patients because they are not near death but report decreased quality of life (Franco et al., 2007; Scire et al., 2013).
Recruitment and data collection
Collaborating physicians agreed for the interviewer (R.N.) to approach potential participants in their outpatient clinic at Basel University Hospital. As university hospitals recruit regularly patients for research, these patients are thus likely to have been approached for informed consent previously not only in a clinical, but also in a research context.
During the recruitment phase the interviewer (R.N.) was notified about scheduled consults for stable outpatients with diabetes or gout. When such consults were scheduled the interviewer (R.N.) visited the clinic and personally approached patients at their appointment. Potential participants were informed about the study and interested patients were invited to participate. To be included, patients had to be over the age of 18, diagnosed with and treated as outpatients for diabetes or gout, and mentally competent to understand and consent to our interview. This was evaluated by the interviewer (R.N.) prior to obtaining a written informed consent. All interviews were conducted in Swiss-German. Based on participants’ preferences interviews were conducted at the participants’ home or in the hospital, where a separate room was available. Interviews were transcribed verbatim, translated into standard German, and fully anonymized.
Analysis
The overall aim of the larger study was to inductively explore stable patients’ attitudes towards participation in first-in-human trials for a new treatment. We applied thematic analysis (Braun and Clarke, 2006), not using a preexisting theoretical framework but applying a bottom-up method. Identification of themes was performed via a stepwise procedure. First, we read the interviews in order to familiarize ourselves with the data and to obtain preliminary ideas in an inductive way. Second, these ideas were discussed among the team members to produce initial codes from the data. Third, we went through the previously produced codes searching for possible themes that we agreed on, thereby narrowing the overall research question. In a fourth step, all themes were critically reviewed before, and in a fifth step, the final codes were defined, and all the interviews were coded with the particular focus on the defined themes that are presented in the results section. Codes were implemented by hand using a specifically formatted word document. We used both semantic themes at an explicit level (prioritization of trust) and latent themes at an interpretative level (actual comprehension, visceral factors).
Results
The participants interviewed in this study were representative of stable patients suffering from chronic disease who are regularly exposed to situations requiring informed consent. The sample includes 27 male and 9 female patients suffering from chronic disease (21 from diabetes and 15 from gout). Their mean age is 66 years (SD 15.7), most (
When analyzing participants’ explanations of why they would or would not be willing to participate in the presented first-in-human trial, we identified three emerging themes that indicate how stable patients’ decision-making capacity regarding participation in early-phase clinical trials could be determined by psychological factors: (a) actual and often insufficient comprehension of the inherent logic of clinical trials (actual comprehension); (b) prioritization of and recourse to trust as a key feature of patient–physician relationship (prioritization of trust); and (c) awareness of the potency of visceral factors, for example, pain, which might interfere with their capability to provide autonomous consent to clinical trials (visceral factors). However, these themes were identified through the analysis and thus were not preconceived a priori.
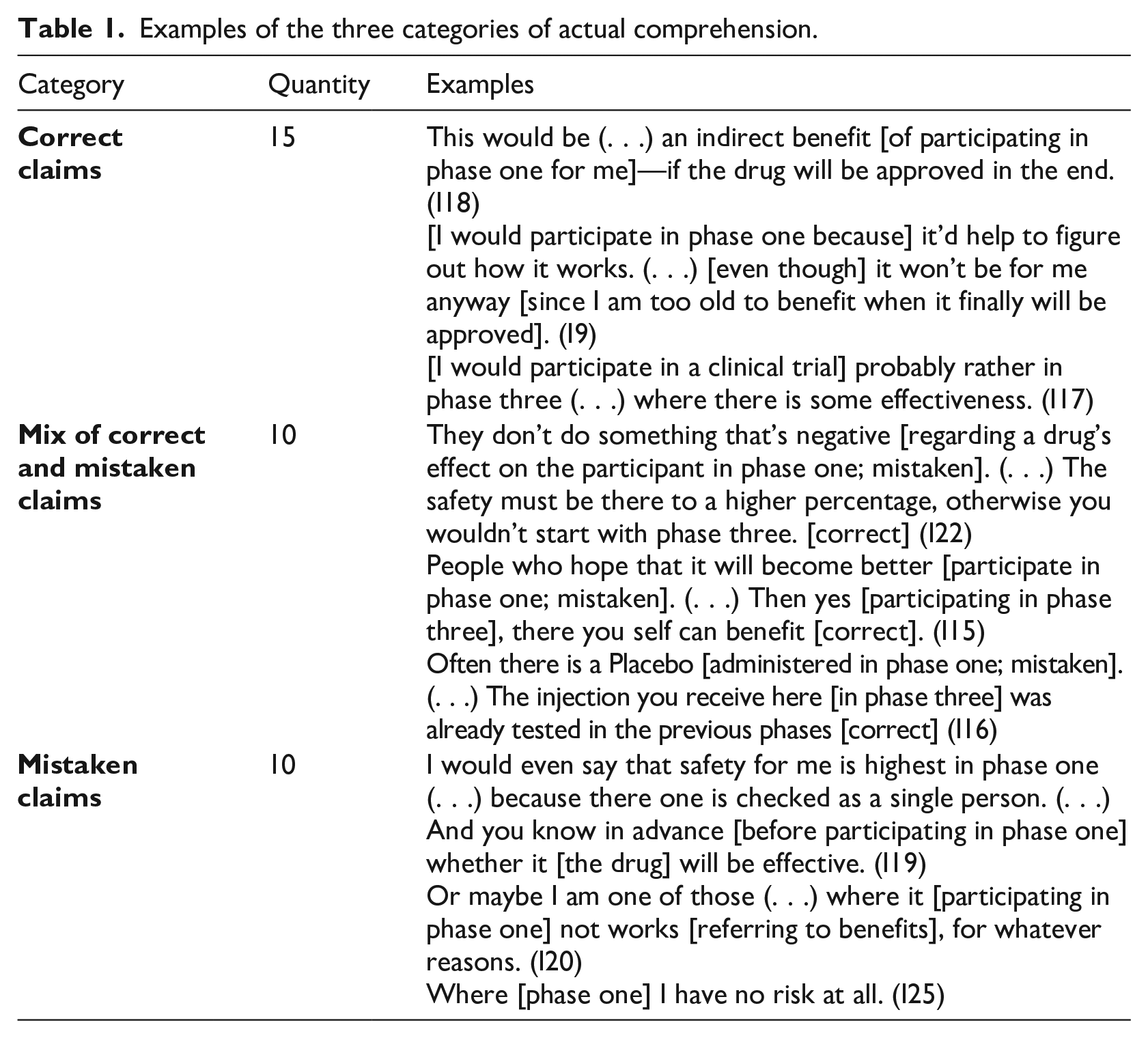
Actual comprehension
A person’s comprehension of the logic of clinical trials requires, among other things, at least minimal cognitive capacity. Understanding is itself a mental phenomenon related to the object in question and is
Examples of the three categories of actual comprehension.
Prioritization of trust
In many interviews, trust in the physician was mentioned as a way of compensating for a lack of understanding and knowledge. Frequently, participants expressed their difficulties in understanding medical information. A majority of participants (
Examples of the two categories of the prioritization of trust.

Visceral factors
Visceral factors determine behavior, cognition, and emotions (Critchley and Harrison, 2013). Assessing the power of visceral factors, Loewenstein notes that “immediately experienced visceral factors have a disproportionate effect on behavior and tend to ‘crowd out’ virtually all goals other than that of mitigating the visceral factor” (Loewenstein, 1996: 272). When questioned about the circumstances in which they would be more likely to participate in a first-in-human trial, most interviewees recognized the powerful influence that visceral factors might have on their decision. Some participants stressed the point by admitting that they would even participate in literally every trial at a certain level of pain, where all considerations or worries are “trumped” or overruled by the wish to escape the pain. Almost two out of three patients interviewed (
Examples of visceral factors.

Discussion
The doctrine of informed consent requires the disclosure of all relevant information. A valid consent needs to fulfil three criteria: adequate information, voluntariness, and mental capacity (Eyal, 2011; World Medical Association, 2013). Overall, our results indicate that several types of psychological factors seem to interfere with stable patients’ decision-making to an extent that the requirements of informed consent would not be met as set by Declaration of Helsinki (World Medical Association, 2013).
Actual comprehension
The interviews reveal that, despite the fact that a case study, graphics, and detailed explanations were used, patients’ actual comprehension was frequently insufficient. More than half did not understand the logic of clinical trials and therefore would not be “informed enough” to meet the requirement of informed consent. In light of this shortcoming, simply increasing the amount of information provided will not necessarily be successful (Eyal, 2011; Manson and O’Neill, 2007). Also, providing more information would prolong the already highly time-consuming procedure of informed consent. Others have proposed that to enable true and independent informed consent in the clinical context, comprehension needs to be improved by various measures, for example, by giving enough time to the patients and using a patient advocate (Hewlett, 1996) or neutral discloser who has enough time to “translate” the medical information (Appelbaum et al., 1987), in general by effective communication between physician and patient (Manson and O’Neill, 2007), by providing opportunities to ask questions (Sreenivasan, 2003), or by a neutral educator talking one-on-one to the potential research participant (Flory and Emanuel, 2004). In sum, the finding from our qualitative study emphasizes the difficulty in ensuring that stable patients’ consent is “adequately informed” and that the “potential subject has understood the information” (World Medical Association, 2013). One explanation for the lack of comprehension is that a two-step process from information to consent would have been needed. We offered as much time as desired during the interview, since there was no set time limit for questions and answers during the interview. Nevertheless, participants did not process the information adequately and were exposed to more information that they were able to assimilate during the time of the interview. This clearly has practical implications. Our study suggests that even offering as much time as needed for explanations might be insufficient for many if the science of the trial is highly complex. We therefore recommend a two-step process to facilitate true informed consent. If researchers can ensure greater comprehension, we think it is their obligation to do so.
Prioritization of trust
Given their perceived inability to fully understand, as well as their preexisting lack of knowledge, stable patients could decide to (or feel to some extent forced to) “trust” medical authorities. What has been denominated as “trust” functions as a kind of psychological reassurance. By labeling the “decision” to do what a physician suggests as “trust” patients would reframe their situation as if they had a choice. This strategy of restoring freedom of choice, even if imaginary and not real, helps to overcome their discomfort in a situation of helplessness. Whether this could be seen as genuine trust(ing) is highly questionable. Many patients interviewed value trust generally as a necessity in a physician–patient relationship. However, this relationship should not result in any form of coercion, for example “forced trust” without any alternatives. The latter is no longer the result of experiences with the physician or of the physician’s reliability or trustworthiness. Such limited voluntariness clearly questions the value of any consent that has its foundation in forced trust. On the other hand, it would be paternalistic to force patients to be fully informed if they prefer to trust their physician. But preferring to place trust in the physician has to be one option in a realistic alternative between trusting and actually comprehending on the one hand and not trusting and actually comprehending on the other hand.
Emphasizing the importance of correct understanding among patients, O’Neill (O’Neill, 2004: 18) states that “the only trust that is well placed is given by those who understand what is proposed, and who are in a position to refuse or choose in the light of that understanding.” It is thus not informed consent per se, but psychologically realistic requirements of informed consent that improve understanding and thereby promote trust. O’Neill claims that informed consent requirements “play (. . .) a role that can have distinctive importance in maintaining trust” (O’Neill, 2004: 151). Here she refers to the normal clinical context, where the goal is to give patients treatment that will help them. Do her arguments also apply in the research context, where the goal is to generate useful knowledge rather than to directly benefit the research participant? If patients decide to place trust in their physicians because they do not adequately understand different treatment options, that may be a reasonable choice because doctors in the clinical context aim to benefit patients. But if patients place trust in physicians because they do not adequately understand the proposed trial, as seems to be the case with some participants of our interview, this trust is misplaced because the aim is not to benefit them, but to study the new intervention. In effect, this is a different variant of the therapeutic misconception. Normally, the misconception is that research participation will benefit the research participant; here, the misconception that physicians can be trusted with the decision arises
In terms of informed consent, a distinction was drawn between a forward-looking and a backward-looking rationale (Eyal, 2011). The forward-looking rationale refers to restoring societal trust via using informed consent procedures. Thus, informed consent can restore or even enhance trust. Yet, this requires a redesign of informed consent such that it better meets humans’ actual capacities. The case of a patient who does not understand information sufficiently will not enhance trust, even despite full disclosure of information. The backward looking rationale “defends informed consent as an intrinsically valuable way to honor the trust that the patient has placed in the physician” (Eyal, 2011: 2). Given the fact that many patients in our sample reported that they were situationally forced to place trust, a redesigned informed consent could better serve this post-hoc justification. Although the trust is initially forced, it then could simultaneously be seen as substantiated. A redesigned procedure, namely more tailored to psychological factors, provides physicians with more trustworthiness. Finally, our finding regarding the prioritization of trust challenges the requirement that informed consent should be “particularly cautious if the potential subject is in a dependent relationship with the physician” (World Medical Association, 2013).
Visceral factors
Patients anticipated that visceral factors would influence their behavior. It is important to note that the potential influence of a visceral factor on future behavior (e.g., of a present or anticipated gout attack) is usually underestimated (Loewenstein, 1996). Accordingly, patients, although aware of the huge influence of visceral factors, would still underestimate their actual future power. Therefore, their decision-making could be even more biased, when immediately experiencing a visceral factor, than they could presume. The actual influence of the visceral factor will thus be higher than the predicted effect. Moreover, stable patients may have a long history of recurring suffering. The accompanying feeling of powerlessness could cause a kind of therapeutic desperation, a state of hopelessness regarding a possible cure for the disease. Since emotions are classified as visceral factors (Loewenstein, 1996), desperation or hopelessness is another visceral factor in addition to the anticipated suffering. Therefore, desperation may increase the likelihood of a stable patient to participate in a clinical trial when pain is not immediately experienced. Generally, at a moderate level visceral factors are always present. At a higher level, or in patients whose resistance is somewhat limited, visceral factors can overrule any decision-making (Loewenstein, 1996). That means that, even if a patient shows the highest possible cognitive capacities, visceral factors are more powerful and easily overrule any otherwise rational deliberation. With respect to stable patients who only anticipate their own suffering and even more to immediately suffering patients, this finding challenges the requirement of voluntariness for informed consent (World Medical Association, 2013), because both the anticipated suffering and the immediately experienced might interfere with free volition. Although a patient has the right to decide based on visceral factors, in cases of strong visceral factors, any free deliberation can be overruled by those factors and the given consent would not fulfil the requirement of being “free.”
Strengths and Limitations
This study has several strengths. It is the first qualitative study to explore the potential of psychological factors to constrain the ability to give valid informed consent to participation in clinical trials, and the data that emerged are rich and informative. The data and analysis thus shed light on a topic that has been neglected in the literature. However, our study also has a few limitations. First, the interview was conducted with a convenience sample in only one hospital in the German-speaking part of Switzerland. Therefore, the question of transferability of our findings arises. However, there is no reason to believe that the sample has unique traits when compared with the population of stable patients. As described in the methods section, we defined criteria to request participants to counteract researcher bias and recruited them directly in the hospital to avoid further selection biases due to indirect request through physicians. The fact that only a few patients addressed refused to participate in the interview suggests an absence of self-selection effects due to voluntary enrolment. Furthermore, gender differences in the prevalence of either diabetes and gout give us no reason to suggest a gender bias within our sample (Guariguata et al., 2014; Zhu et al., 2011).
Nevertheless, different settings possibly lead to different psychological states, and stable patients at different disease stages or severities might have given different responses. The fact that our case study concerned a SB intervention may have influenced responses, but our hypothetical trial example enables extrapolation of our results to other informed consent situations because it presents sufficiently new and complex information, that is, clearly tests patient understanding and mechanisms such as trust that are used to cope with difficulties in understanding. While some trials might present more simple information, our own experience as members of research ethics committees (BE; DS) has convinced us that the majority of clinical trials are not easy to understand, even for expert committee members.
Second, as pointed out in our discussion, many participants decided to (or felt to some extent forced to) “trust” the medical authorities given their perceived inability to fully understand. What participants denominated as “trust” therefore functioned as a kind of psychological reassurance. Although another sample might find other ways of compensating, it is highly questionable if these would serve as sufficient substitutes for individual reasoning and therefore not interfere with the ideal form of reasoned medical decision-making, as argued in the discussion section.
Third, social desirability (Crowne and Marlowe, 1960) and the therapeutic misconception (Appelbaum et al., 1982) are likely to be at play. To reduce their influence, we informed about anonymization and confidentiality by telling participants that there was no advantage due to participation in our interview before, at the beginning of, and when needed during the interview. Hence, there is no reason to believe that participants’ answers are affected by anticipated benefits. Finally, a bias may have been caused, since we did not determine inter-rater reliability. However, to further ensure the reliability of the results, generated themes were critically discussed in group meetings (Wirtz and Caspar, 2002).
Conclusion
Overall, our results suggest that the stable patient’s decision to participate (or not) in an early-phase clinical trial is affected by various psychological factors. A considerable number of the patients interviewed did not understand the presented trial sufficiently to meet informed consent requirements. Our study presents three potential constraints to stable patients’ decision-making capacity. First, insufficient comprehension can compromise informed consent procedures. Second, facing insufficient comprehension and little knowledge, trust is used instead of understanding. Third, even high decision-making capacity can be easily overruled by visceral factors. Informed consent procedures should take these factors into account. However, implementation is complicated since in the usually limited period of time in the clinical context as well as in the way research assistants are trained there is neither enough focus on those problems, nor on practices that could help to overcome them. The available evidence underlines that human beings in general are often incapable of being the rational, purposive, and deliberate individual who consents in accordance with informed consent requirements. As we have no reason to believe that our informed consent procedure nor our sample of stable patients are particularly unique or vulnerable per se, we think it is justified to conclude that the opinions expressed by our interview participants are not fundamentally different from those of many other stable patients.
What is known about the psychological nature of human beings and the limitations of decision-making could lead to a shift to a new paradigm of bioethics. Examining the field of bioethics, Lee fosters the debate on taking humans’ bounded rationality more seriously into consideration, thereby arguing (Lee, 2011: 368) to “adopt the more nuanced notion of rationality from behavioral economics.” In addition, he questions the assumption of a patient who is “clear-headed, far-sighted, informed, and eager to made decisions.” Yet, this often leads to a dichotomized concept of rationality that, in turn, is often applied to patients, a claim that seems to be supported by empirical findings (Hermann et al., 2014). However, the main metaethical premise behind his argument is that modern bioethics has to become what he calls “behavioral bioethics.”
Along similar lines, we would like to argue for a reconceptualization of informed consent toward a more modest and humble account (Manson and O’Neill, 2007), in both the academic sphere of ethics and in daily practice. Our results could be interpreted as lending support to this view. We support the call for behavioral bioethics and conclude that this nexus of empirical findings and bioethical considerations is necessary in order to achieve a more mature bioethics with psychologically realistic theorizing and a realistic choice architecture for physicians (Lee, 2011) as well as to avoid use of ideal principles that cannot be met by human beings. The bioethical assumption of a (fully) rational person is untenable in light of the behavioral sciences. A gradual view of human rationality should be applied instead of the dichotomy of competent versus incompetent, rational versus irrational. Bioethical thinking has tended to be characterized by overestimating human rationality. Instead, bioethics needs to come to terms with what is labeled as psychological realism (Levy, 2014) and should address the actual human being, who is, compared to the traditional addressee, hyporational. Psychological primacy does not mean psychological autocracy, but these changes will ensure that we will avoid putting bioethics and the applicability of its principles in practice on a procrustean bed, because bioethical principles will have their foundation in what humans are actually capable of and not in what ethics wishes they were ideally capable of.
Informed consent in clinical trials may be at higher risk than previously thought of being neither correctly informed nor free. Several strategies can be used to avoid this risk. First, we could simply accept lower standards of informed consent. Second, we could put more resources into research to provide (more) accurate information to potential research participants. Third, and most importantly, a better understanding of psychological factors can be translated into effective communication interventions that are likely to improve potential research participants’ comprehension in informed consent and thereby also facilitate respecting a patient’s autonomy (Schenker et al., 2011).
Footnotes
Declaration of conflicting interests
The authors declare that there was no involvement by the supporting source or any other conflict of interest in respect to the research, authorship, and/or publication of this article.
Funding
All articles in Research Ethics are published as open access. There are no submission charges and no Article Processing Charges as these are fully funded by institutions through Knowledge Unlatched, resulting in no direct charge to authors. For more information about Knowledge Unlatched please see here: ![]() .
.
