Abstract
Very few attempts have been made to survey patient opinions, particularly regarding the use of residual biospecimens and health information in research, to clarify their values. We conducted a questionnaire survey that targeted outpatients of a university hospital to gauge their awareness levels and understand patient perspectives on research that uses these items. Few patients felt that obtaining individual consent for each research study was necessary. Most patients expressed the view that researchers should be obligated to inform them about the research use of their items and be subject to self-directed rules (including sanctions). The research community should try to obtain “societal consent regarding an opt-out system” from the public. A salient value-sharing-based governance structure is necessary for obtaining public trust.
Keywords
Research that uses residual biospecimens and health information after these are obtained for treatment purposes is essential for the development of medical treatment, diagnosis, and prevention strategies. With developments in genome informatics and other technologies including IT, the importance of research that makes use of both residual biospecimens and health information is anticipated to increase even further.
Currently, when collecting human samples (blood, tissue, etc.) or health information to construct a new biobank, or when conducting new research using biospecimens or health information, researchers must obtain explicit consent from patients, in accordance with the “Ethical Guidelines for Medical and Health Research Involving Human Subjects” (established by the Ministry of Health, Labour, and Welfare) in Japan (Ministry of Education, Culture, Sports, Science and Technology/Ministry of Health, Labour and Welfare, 2017).
On the other hand, when a research study uses existing residual biospecimens or health information for treatment purposes, the ethical guidelines require that (1) the researcher states in the research protocol that the study could not otherwise be conducted without the use of residual biospecimens and health information and that these will be used anonymously, (2) the researcher presents a research outline in a poster or website that clearly specifies that patients can withhold their permission, and (3) the research protocol is approved by an ethics review committee. In other words, researchers do not have to obtain explicit consent from each patient.
Some research targets diseases with poor prognoses, proposing to investigate residual biospecimens to identify any markers that could be used for early detection or genes responsible for the condition; in these cases, some patients are deceased and cannot be contacted to obtain clear individual consent, so this approach is fairly realistic. However, even if the research is presented via a poster, it is nearly impossible for patients to know that their residual biospecimens or health information will be used for this research, and even if they were alive and wanted to withhold their consent, they would be unable to do so. It is difficult, therefore, to assert that this method ensures respect for the human subjects (Helgesson, 2012; Hofmann, 2009).
Use of biospecimens and health information in research has been debated widely, and various approaches have been taken (Menikoff et al., 2017; Page et al., 2016). In recent years, many arguments and proposals have been put forth with regard to the handling of blanket consent or broad consent for unspecified research (Beskow, 2016; Parker, 2011; Petrini, 2010; Willison et al., 2007). In the context of biobank research, some have reported that biobank donors do not see the importance of obtaining explicit consent from an individual when using DNA information for other research studies (Kelly et al., 2015).
In addition, it is critical for research success, including that of biobank studies, to gain the trust of the general public (Hansson, 2005). Some studies indicate that the success of biobank research is contingent upon good governance in research rather than individual consent (Chalmers et al., 2014), one component of which is policymaking (O’Doherty et al., 2011). Of course, if this policy is not something that donors can accept, then it is meaningless. Policies must reflect and encompass patient perspectives, specifically with regard to how the patients and the public feel about residual biospecimens and health information, what kind of awareness they have about research using these items, and what hopes they have regarding the methods used to obtain informed consent. In addition, it is important to listen carefully to patients as they voice any concerns and their thoughts on necessary strategies, and ultimately create policies to reflect their concerns (Dresser, 2014; Dry et al., 2017). However, very few attempts have been made to survey patient opinions, particularly with regard to the use of existing residual biospecimens in research and health information for research purposes, to clarify their values.
To this end, we conducted a questionnaire survey that targeted outpatients of a university hospital to gauge awareness levels and understand patient perspectives on research studies that use these items, as well as propose the first step required to create a research policy and ensure that research is conducted on a foundation of trust from society.
Methods
Participants
The survey was conducted in December 2009 and targeted outpatients of the urology, gastroenterology, and breast surgery departments of a national university hospital in Japan. The author (M.Y.) asked each patient individually if they would be willing to participate in the questionnaire survey. Those who provided their consent to participate were given an explanation of the terms “residual biospecimens” and “health information” as well as the methods of anonymization and rules concerning ethical guidelines, after which the self-administered questionnaire survey was given to the participant to complete. Participants were informed that “residual biospecimens” were “blood and urine that were left over following a test,” and that “health information” comprised “test results, imaging data such as x-rays, and the written content of a medical chart.” Participant questions on the definitions of terms in the questionnaire items were addressed by the researcher, but any questions that could be interpreted as leading or manipulating their responses were not addressed. After the questionnaire was completed, the researcher (M.Y.) checked to ensure that no answers were missing and then collected the questionnaire.
Patient assessment
The questionnaire was created by the authors (M.Y. and K.S.) based on previous studies (Asai et al., 2002; Hamajima et al., 1998; Willison et al., 2007). The initial version of the questionnaire was given to multiple laypersons to obtain their responses, and the authors asked for their opinions on the suitability of the questions’ intent and ease of response. Following several revisions, the questionnaire was completed.
Participants were asked the following questions: “Is it acceptable to use residual biospecimens and health information in medical research?” (Acceptable/Unacceptable) and “Is consent necessary?” (Unnecessary/Consent is unnecessary, but disclosing information and providing an opportunity to refuse consent is necessary/Consent to use these in some form of medical research is necessary/Consent is required for each research study that is conducted).
The questionnaire also contained 13 questions on issues such as awareness and concern about research that uses residual biospecimens and health information (Agree/Neither agree nor disagree/Disagree). Similarly, participants answered 11 questions concerning the conditions for research use of these items (Necessary/Neither necessary nor unnecessary/Unnecessary).
Statistical analysis
Quantitative data were analyzed using JMP (4.0.5J). For descriptive data, we investigated the frequency distribution and rate for each variable. Missing data were considered “unanswered.” To characterize the association between two items, we conducted a chi-square test using cross tabulation, for which p < 0.05 was considered statistically significant. Free writing data were analyzed inductively by the authors (M.Y. and K.S.), and important items were extracted.
Ethical considerations
The present survey used an anonymous self-administered questionnaire, and none of the information collected contained any detail that would identify the individual. This survey was approved by the Research Ethics Committee of the X University Graduate School of Medicine and Faculty of Medicine.
Results
Survey population
Of the 361 patients asked to participate in the survey, 302 completed the questionnaire (response rate, 83.7%), and data from all responses were used in the analysis. Respondent demographic characteristics are summarized in Table 1. Respondents were 55% male and 44% female, with 35% in their 60s, 22% in their 70s, and 14% in their 50s (age range, 10-90 years). Previous experience with clinical research trials was recorded by 38% of the respondents.
Demographic and characteristics of the 302 survey respondents.

Note: The percentages do not all add up to 100 because the numbers were rounded off.
Willingness to provide residual biospecimens and health information for medical research
Those who responded that residual biospecimens and health information could be used by these studies comprised 92% and 84%, respectively. We observed no significant differences according to basic characteristics such as sex, age, medical department visited, frequency of hospital visits, or past experience with research participation (p > 0.05 for all).
Those who responded that they would donate their residual biospecimens but not their health information comprised 5%, 0.3% responded that they would donate their health information but not the residual biospecimens, and 3% responded that they did not want to donate either.
Necessity to obtain consent
Table 2 displays responses to the question, “If used for research, is patient consent necessary?” Of the respondents, 36% and 32% stated that “Consent is unnecessary” for residual biospecimens and for health information, respectively. Twenty-five percent and 24% of respondents stated that [consent is] “Unnecessary, but it is needed to publicize about conducting research using these items and to create opportunities for refusal (opt-out consent)” for residual biospecimens and for health information, respectively. Those who responded that researchers would “Need consent to use these items for unspecified medical research” comprised 26% and 27% for residual biospecimens and health information, respectively. Meanwhile, 10% and 13% of respondents said that researchers “Need consent to use these items for each research purposes” with regard to residual biospecimens and health information, respectively.
On the necessity to obtain consent (n = 302).

Patient perspectives on conducting medical research with residual biospecimens and health information
Table 3 displays the responses to 13 questions pertaining to respondent awareness and concern about the medical research use of residual biospecimens and health information. Approximately 90% of respondents selected “Agree” to the statements “Medical research is necessary for the development of medicine,” “Medical research is useful to society,” and “Medical research benefits future patients.”
Patient perspectives on conducting medical research with residual biospecimens and health information (n = 302).
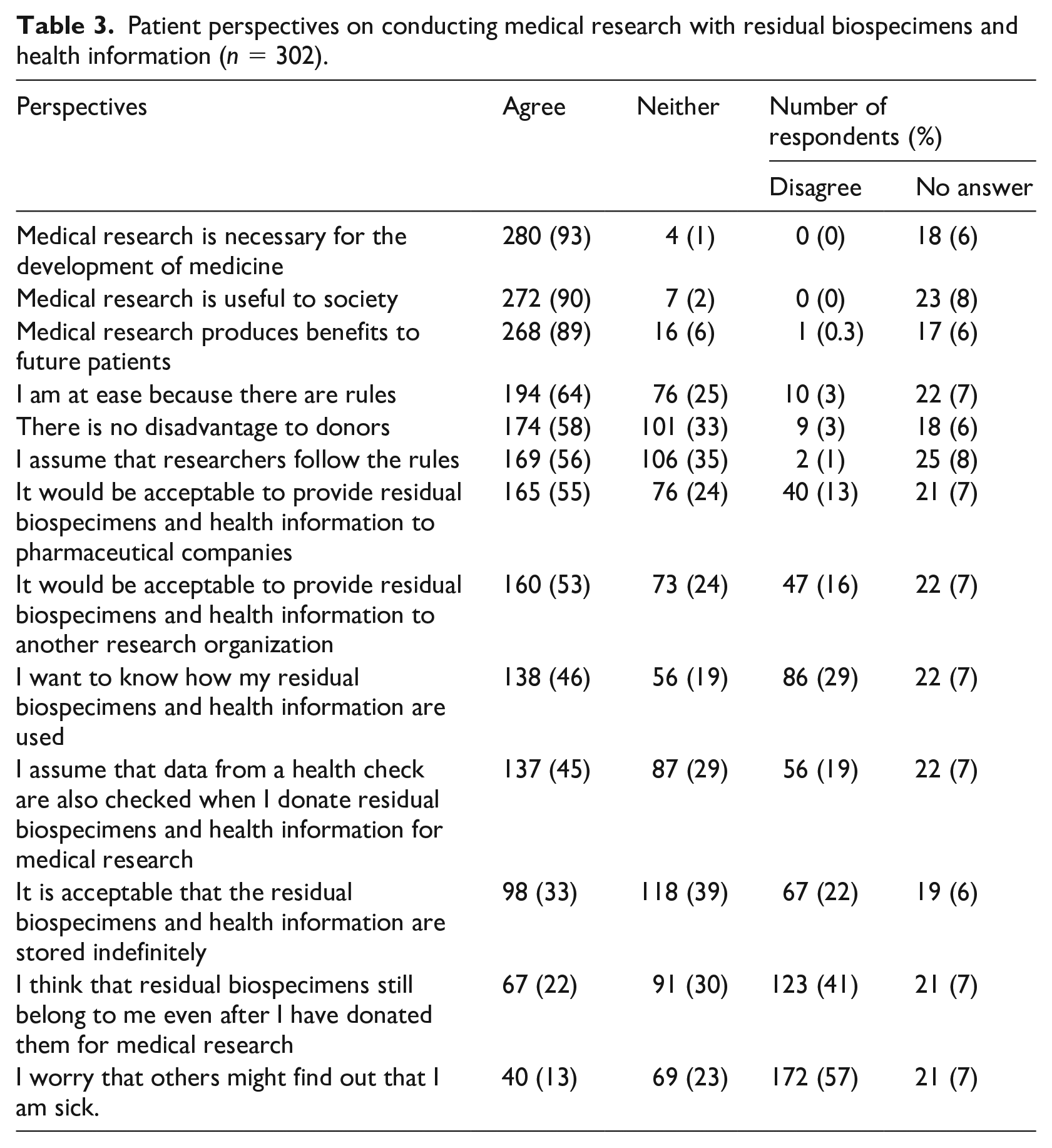
Regarding the rules for conducting research, 64% responded “Agree” to the statement, “I am at ease because there are rules,” and 56% responded “Agree” to the statement, “I assume that researchers follow the rules,” while 35% responded with “I don’t know either way.”
Approximately 60% of respondents selected “Agree” to the statement, “There is no disadvantage to donors.” In addition, 13% of respondents selected “Agree” to the statement, “I worry that others might find out that I am sick.”
Although 55% of survey participants responded positively, stating that “It would be acceptable to provide residual biospecimens and health information to pharmaceutical companies and other research institutions,” 15% were opposed to this.
Roughly 45% of respondents selected “Agree” to the statements, “I want to know why my samples or information were used” and “I assume that my health is also checked when my samples or information is donated for medical research.”
With regard to the indefinite storage of samples and information, 33%, 39%, and 22% responded with “Agree,” “Neither,” and “Disagree,” respectively.
Regarding the issue of whether these items would constitute patient property, 22% of respondents stated, “I think that my samples are still my property even after I have donated them for medical research,” while 41% of respondents stated, “Disagree.”
Necessary conditions for research conducted using residual biospecimens and health information
Table 4 displays respondent perspectives with regard to whether or not any of the 11 conditions are required when conducting research using residual biospecimens and health information.
Necessary conditions for research conducted using residual biospecimens and health information.
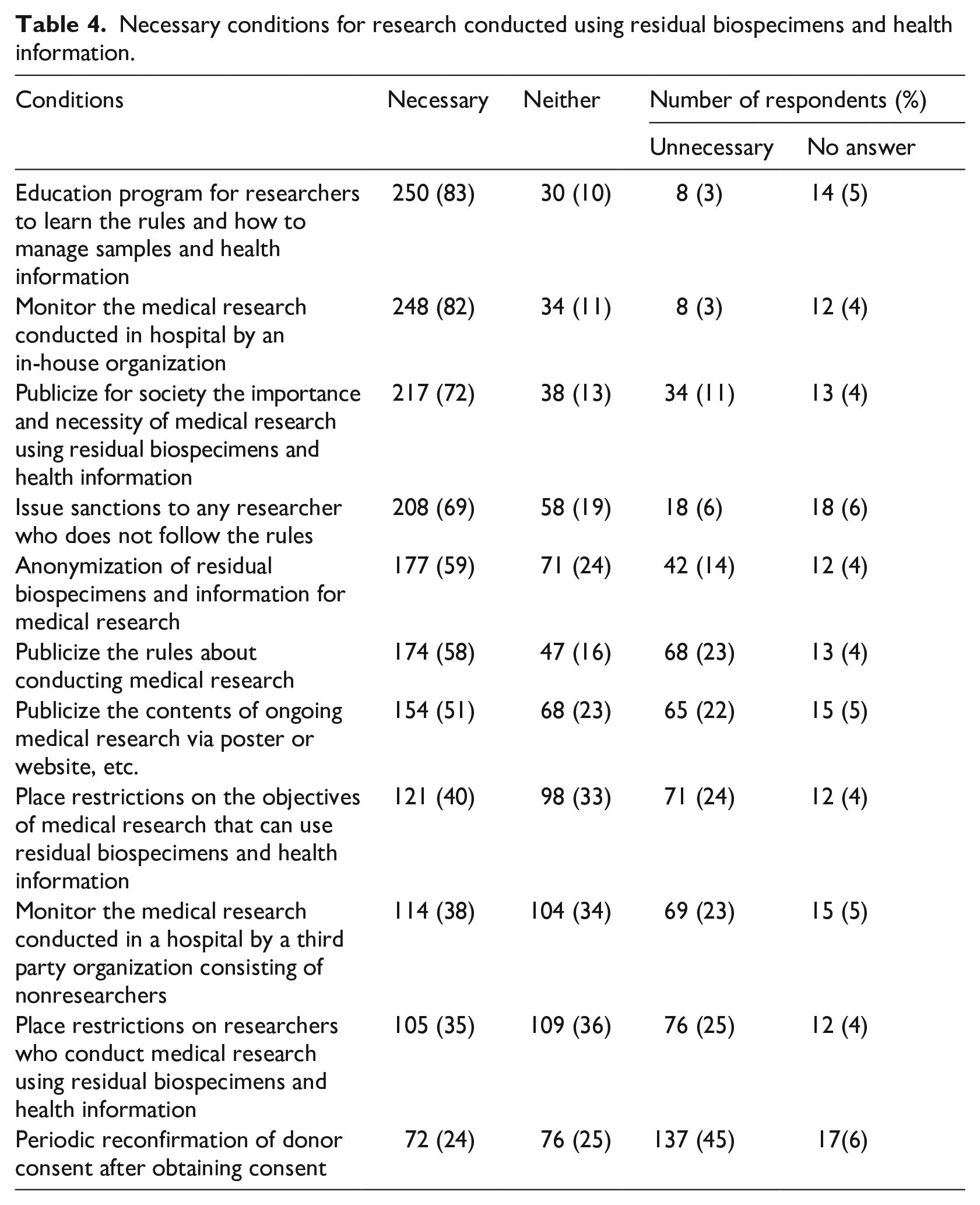
Approximately 80% of respondents requested an “Education program for researchers to learn the rules and how to manage samples and information” and “Monitoring by an in-house organization of the medical research conducted in the hospital.”
Approximately 70% of respondents requested “Publicizing the importance and necessity of medical research using residual biospecimens and health information for society” and “Sanctions on any researcher who does not follow the rules.”
The condition of “Anonymization of samples and information for medical research” was deemed necessary by 59% of the respondents, while 14% stated that they were unnecessary.
The conditions of “Publicizing the rules for conducting medical research” and “Publicizing the contents of ongoing medical research via a poster or website, etc.” were deemed necessary by 58% and 51%, respectively, and unnecessary by 23% and 22%, respectively.
“Restrictions on the research objective” or “Restrictions on researchers” were deemed necessary by 40% and 35% of the respondents, respectively, and neither by 33% and 36%, respectively. Finally, “Monitoring by a third party organization” was deemed necessary by 38% and neither by 34%.
In contrast, the condition “Periodic reconfirmation of donor consent after obtaining consent” was deemed necessary by 24% and unnecessary by 45%.
Discussion
In this survey, we focused on the research use of residual biospecimens and health information, which were residual in the hospital after treatment. Our respondents felt that the use of residual biospecimens and health information in medical research is a necessity, and the majority demonstrated the willingness to donate their own items to research. Most respondents felt that individual consent is not required. However, they also clearly stressed the need for stringent conditional use to ensure privacy protection, education of researchers about management of these items, disclosure of research conducts and outcomes, and a clear demonstration of the necessity of the research. In addition, they expressed the desire for a structure that would monitor the research, as well as rules that would issue sanctions on those who deviate from the rules.
Positive perspectives on medical research and donating residual biospecimens and health information
These findings of patients demonstrating positive opinions concerning the donation are consistent with previous study results (Botkin et al., 2014; Mello et al., 2018; Page et al., 2016). The positive nature of these perspectives may have been enhanced by the fact that the survey was conducted at a university hospital, or because 30% of respondents had experienced clinical trials. In addition, most respondents felt that research benefits future patients, so their perspectives may be based on altruistic feelings. On the other hand, it should be noted that some respondents were hoping for personal benefits, based on the fact that 45% of them expected that a health check could be done.
Based on these findings, we conclude that researchers are obligated to design scientifically meaningful research and achieve results, and to explain clearly to the general public that the university hospital is conducting medical research using their residual biospecimens and health information, not only for maintenance and training, but also for routine medical research, as responsible researchers. Researchers must also clearly explain that the value in conducting the medical research is to be extended to society and not necessarily to benefit the donors themselves.
Most patients do not require individual consent for using their items for medical research purposes
Current ethical justification in the use of existing residual biospecimens or healthcare information in medical research is rooted in the attainment of individual consent. In Japan, if the research is deemed valuable to society, a research ethics committee could approve the use of residual biospecimens and health information in the research conditionally, such as if the researchers disclose the fact that this study will be conducted in house via a poster or website (Ministry of Education, Culture, Sports, Science, and Technology/Ministry of Health, Labour and Welfare, 2017). However, it is impossible for patients to be notified of the research, because the notification period may create a large gap in the timing of visiting patients to whom this would apply. This survey revealed that many patients would allow the use of their samples and information without explicit consent for each research purpose, and felt that periodic reconfirmation of their consent was not necessary. However, at the same time, they desired to have information concerning the fact that the hospital was conducting medical research.
Based on these situations, we recommend that an opt-out method is provided for those who wish to refuse donation. However, researchers are also obligated to the public (including outpatients) to visualize how the residual biospecimens and health information obtained through treatment will be used, in what type of research, and the necessity of these medical research studies (Saha and Hurlbut, 2011). In other words, the research community must obtain societal consent (Sears, 1980). This could be achieved more readily if the hospital or research community were to require attainment of societal consent as a policy (Resnik and Elliott, 2016).
Patients require appropriate conditions for allowing their biospecimens to be used by pharmaceutical companies
In contrast to previous studies, which found that research subjects and the public had concerns about the use of residual biospecimens for profitable objectives (Beskow and Dean, 2008; Dresser, 2014), roughly half of our survey respondents stated that they would allow their biospecimens to be used by a pharmaceutical company, but seemed to indicate that this was contingent upon the condition that the research objective contributed to the public interest.
Therefore, we surmise that it will be important for policies to specify that research may be conducted at third party institutions to contribute to the public interest, including pharmaceutical companies. In addition, organizations should publicize information about the types of ongoing research studies and their results on the website in a way that would be easily understood by the public (Petrini, 2010).
Patients request stringent conditions for conducting medical research
Respondents did not see immense significance in obtaining consent for each research study, but this did not mean that they were consenting to unconditional use. Rather, they desired that the samples and information were used in appropriate ways, and that a means to ensure the appropriate use was in place.
A large majority of respondents reported that they highly value a system to oversee and ensure that the research is being conducted in appropriate ways, researchers are educated about the rules, and sanctions are imposed on any researcher who deviates from these rules. Supervision and imposing of sanctions seem to give rise to peace of mind due to the presence of a restraining power (“Since the researchers will be disadvantaged if they break the rules, they will not do such a thing.”). In this manner, respondents desire a structure that will enable them to donate their items with peace of mind. However, it has been found that conditions mediated by fear of sanctions or mutual supervision may be less likely to cultivate “trust” as readily. What should be emphasized here is that trust must be obtained and maintained by clearly stating the provision of autonomous supervision in a research policy (Nakayachi and Watabe, 2005). Our survey respondents seem to desire independent supervision by the researchers themselves and to have the rules clearly specified to patients. This highlights the importance of establishing a governance structure so that research institutions will independently ensure that the research is conducted appropriately and clearly reveal the content of that governance structure to the patients. It is also necessary for researchers to create a research governance structure based on an understanding and consideration of patient values. This allows patients to gain an understanding that researchers share their deeply held values, and thereby approve a system in which researchers are allowed to use these items. This salient value-sharing-based governance structure is necessary for obtaining patient and public trust (Nakayachi and Cvetkovich, 2008).
Recommendations for medical research using residual biospecimens and health information
Our survey results indicate that the research community should provide transparent information in order to enable outpatients and the public to recognize that they are routinely conducting medical research using residual biospecimens and health information. This transparency is likely more important for future patients relative to seeking an ideal consent process agreed upon by all stakeholders. In other words, we recommend that the research community should try to obtain “societal consent regarding an opt-out system” from the public.
Second, to create a workable opt-out system, we recommend that the research community autonomously develops research policies that are transparent (Nakayachi, 2008). These approaches may lead to heightened trust in medical research from the public (Mello et al., 2018). Research using residual biospecimens and health information could be possibly presumed as a kind of biobank. If so, the case of acceptance by the opt-out system applies only for low-risk research. That said, where there is significant potential for harm to individual interests or values, such as through genetic analyses of single-gene disorders, HIV-related research, or the derivation of an induced pluripotent stem cell line, etc., explicit consent must be obtained before research can begin (Parker, 2011). In principle, it is not acceptable to use residual biospecimens and health information for high-risk research and research that requires additional specimens, because patients are not notified about the details of the risk and purpose of such research. This acts an essential deterrence from participating in clinical research or biobank.
Third, we recommend that the research policy includes the following key components: (i) the medical research using residual biospecimens and health information obtained for medical practice is important and necessary to improve medical treatment or prevention for future patients; (ii) there is no direct benefit to donors if research outcomes are observed; (iii) a research ethics committee evaluates the importance and legitimacy of the research; (iv) an explanation and exemplification of the types of ongoing research and research outcomes are provided to the public; (v) researchers are well-educated about the rules of conducting research, (vi) donor privacy and rights are protected, (vii) the method of use is in third party settings, including commercial use, (viii) details of rules on conducting the research (including sanctions) are provided, (ix) methods used to manage and audit/oversee medical research studies using residual biospecimens and health information in house are established, and (x) management of intellectual property and rights for the research findings are ensured.
Lastly, at the national level, we recommend that Japan establishes a fundamental law to protect donors/patients, as well as that for the management and transferring of human tissues and medical information. At this point, Japan has no fundamental law regarding the treatment of human tissues for any purpose; these include research, as well as education, training, display and disposal, etc., even though the guidelines concerning research on human or human tissues do exist in Japan, as do laws about sharing de-identified medical information for large-scale records research. Finally, fostering professionalism among the researchers themselves is essential for these guidelines and laws to function.
Our recommendations based on this survey are summarized in Figure 1.

Recommendation for creating an opt-out research governance system in medical research using residual biospecimens and health information.
Conclusions
Our survey clearly reveals that patients responded positively to the research use of residual biospecimens and health information. Most respondents indicated that consent for use in each individual research study is unnecessary. However, these positive attitudes were also conditional, in that they required that a strict governance structure was present to ensure autonomous protection of patient rights in the research that was being conducted.
The results of this survey emphasize the need to notify patients and the public that their items are used for research under a governance structure with self-directed rules.
The research community should try to obtain “societal consent regarding an opt-out system” from the public. A salient value-sharing-based governance structure is necessary for gaining public trust.
Footnotes
Acknowledgements
The authors thank the respondents of this questionnaire survey, the various departments of the national university hospital we approached for this study, and also PROEDIT_JAPAN (http://www.proeditjapan.co.jp/) and Editage (![]() ) for English translation and proofreading. This survey was supported in part by a Grant-in-Aid for Scientific Research (17K08916) from the Ministry of Education Culture, Sports Science, and Technology (MEXT), Japan, and the Uehiro Foundation on Ethics and Education. The content described in this manuscript originate from the authors’ personal ideas and does not represent the organization.
) for English translation and proofreading. This survey was supported in part by a Grant-in-Aid for Scientific Research (17K08916) from the Ministry of Education Culture, Sports Science, and Technology (MEXT), Japan, and the Uehiro Foundation on Ethics and Education. The content described in this manuscript originate from the authors’ personal ideas and does not represent the organization.
Declaration of conflicting interests
M.Y. and M.S. declare no competing interests. K.S. is a member of the Bioethics Committee of the Japanese Society for Regenerative Medicine, a member of the Certified Special Committee for Regenerative Medicine of Gamagori City Hospital and Yamaguchi University, and a member of the Research Ethics Review Committee for Human Embryonic Stem Cell Research of Sumitomo Chemical Company, Limited.
Funding
All articles in Research Ethics are published as open access. There are no submission charges and no Article Processing Charges as these are fully funded by institutions through Knowledge Unlatched, resulting in no direct charge to authors. For more information about Knowledge Unlatched please see here: ![]() .
.
