Abstract
Life sciences research is increasingly international and data-intensive. Researchers work in multi-jurisdictional teams or formally established research consortia to exchange data and conduct research using computation of multiple sources and volumes of data at multiple sites and through multiple pathways. Despite the internationalization and data intensification of research, the same ethics review process as applies to single-site studies in one country tends to apply to multi-site studies in multiple countries. Because of the standard requirement for multi-jurisdictional or multi-site ethics review, international research projects are subjected to multiple ethics reviews of the same research protocol. Consequently, the reviews may be redundant and resource-consuming, whilst the opinions delivered by ethics committees may be inconsistent both within and across jurisdictions. In this article, we present findings based on interviews conducted with international experts in research ethics on the topic of ethics review mutual recognition. We explore the issues associated with ethics committee review of multi-jurisdictional data-intensive research projects, identifying current problems, real-life experiences, and potential solutions that are both bottom-up (via researchers, participants and publics) and top-down (via statutory regulation), as well as challenges in achieving both. On the whole, participants recommended multiple changes to the current ethics review regime for data-intensive international research with the aim of reducing inefficiency and inconsistency. But, the changes recommended differ in terms of degree and scope. In general, participants stressed that key drivers of success in a reformed system should be strong leadership (on the ground and in government) and demonstration of value.
Keywords
Introduction
Research ethics guidelines recommend that multi-jurisdictional research be evaluated by research ethics committees (RECs) in all relevant jurisdictions, or indeed at all sites where the proposed study will take place (Coleman et al., 2015). The rationale for this approach is that it reflects regulatory requirements for REC review in each jurisdiction, and, from a policy and normative perspective, that RECs in each site or jurisdiction are best attuned to ‘local sensitivities’, reflecting the socio-legal environment of the local site. It is assumed that, among other things, risk and benefit assessment, consent procedures, local population recruitment strategy, knowledge of laws and regulations, and understandings of research participants’ rights and interests are best placed in a localized expert body rather than through some other arrangement that could neglect these local issues. Moreover, from a historical perspective, most life sciences research was interventional and conducted on only a few participants by individual researchers at a local institution. In such a context, local knowledge of both researcher and study population could be important (Stark, 2012). The historical impetus and underlying normative rationales have endured. Consequently, many countries’ regulatory and policy instruments require
Yet, there is long-standing recognition that the research ethics review system is operating sub-optimally internationally and in many areas of research (Dove et al., 2014, 2016; Klitzman, 2015; Schneider, 2015; Stark, 2012; van den Hoonaard and Hamilton, 2016). Because of the requirement for local REC review, research projects engaging multiple sites are subjected to multiple ethics reviews of the same research protocol. Consequently, the reviews may be redundant and resource-consuming. The opinions delivered by RECs, however, are often not redundant; rather, they are inconsistent (Abbott and Grady, 2011; Angell et al., 2006; Resnik, 2014; Vadeboncoeur et al., 2016). Because each REC may have distinct rules, forms, procedures and practices for review, divergent opinions outside a range of reasonableness can result. Replicate REC reviews have not been shown to improve the quality of participants’ well-being and could indeed inhibit important research (Dove et al., 2016). The issue is particularly challenging in international data-intensive research projects,
1
which, compared to interventionist clinical research
The coordination of international data-intensive research projects thus raises important practical challenges for the regulatory framework of research ethics review. This is a timely topic to explore because research involving humans is increasingly international, indirect (as it is more data-intensive) and complex (Kaye et al., 2012). Researchers work in multi-jurisdictional project teams or formally established research consortia to recruit data-providing participants (or work with already-collected datasets) and conduct research at multiple sites. A question arises as to whether there are multi-jurisdictional (i.e. international) work-arounds or solutions for the challenges associated with REC reviews at each site or jurisdiction for a data-intensive research project. On this point, academics recently have called for more empirical research to guide policies that seek to institute alternative ethics review models for multicentre studies (Ervin et al., 2015).
Substantively addressing this issue is the objective of the Ethics Review Equivalency (ERE) Task Team of the Global Alliance for Genomics and Health (GA4GH), of which the authors are members. 2 The GA4GH is an international non-profit organization of over 1000 individual and organizational members whose mission is to accelerate the international sharing of genomic and clinical data to improve human health. The ERE Task Team is exploring several innovative ethics review tools, especially ‘mutual recognition’ of ethics review between RECs for international data-intensive research projects. Mutual recognition can be defined as the acceptance by RECs of the outcome of each other’s review such that further (full) local ethics review is unnecessary. We exclude work in the clinical trials domain, which is led foremost by the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH). As members of the GA4GH, we believe that if society is to maximize the potential of modern computing power that gathers individual, patient and population data into large datasets, linking them to enable precision medicine, we must move beyond the current system that emphasizes independent and siloed local ethics review to one that works with all stakeholders to develop models for mutual recognition between RECs. Such models, supported by ongoing dialogue with a wide range of stakeholders and shared policies, guidance and frameworks, could facilitate ethical research, clarify the processes of evaluating data-intensive research projects, and better balance the interests of science, society and data-providing research participants.
In this article, we present findings from a qualitative research project in which we explore the issues associated with REC review of multi-jurisdictional data-intensive research projects, identifying current problems, real-life experiences, potential work-arounds and challenges therein. To our knowledge, no similar projects have been undertaken and reported in the literature. The driving research questions for this project were to explore what problems (if any) arise in REC review of multi-site and multi-jurisdictional data-intensive research projects, how these problems can be effectively addressed through both bottom-up solutions (e.g. via researchers, participants and publics) and top-down solutions (e.g. via statutory regulation), and what challenges the potential solutions themselves may create.
Methods
Semi-structured interviews
Qualitative research was seen as the best approach to learn about specific case studies – that is, real-life examples of international research projects that encountered RECs in multiple jurisdictions – and uncover what those experiences were like, and whether any key lessons were learnt. One-on-one, semi-structured interviews allowed us to gather experts’ in-depth, detailed points of view and to explore real-life processes of multi-jurisdictional ethics review. Semi-structured interviews were also seen as the best method to converse with people to get a sense of their lived world as a stakeholder in research ethics and, together with the interviewee, produce practical knowledge about multi-jurisdictional ethics review (Arksey and Knight, 1999; Brinkmann and Kvale, 2015; Ritchie and Lewis, 2003). Further, conversing with experts in the field of research ethics review was seen as a beneficial method to draw out insights and perceptions into research ethics oversight, as well as to gather insight on and interpret the problems of international ethics review and uncover potential solutions to those problems. The goal of this project, as in many qualitative research projects, was not to obtain statistical representation, but rather to obtain theoretical representation to reflect particular features of the sample population of research ethics review experts.
Interview guide design
As these interviews were semi-structured, an interview guide was designed. Interviewing people identified as ‘experts’ meant that the questions had to assume a baseline of significant knowledge about research ethics review. After outlining a brief introductory statement on the research project, emphasizing confidentiality and confirming consent to audio-record and transcribe the interview, the interviewer (the first author: ESD) asked for brief background information of the interviewee, including their involvement in ethics review and a summary of their current activities. The interview then proceeded to explore three broad topics as follows.
The objective of the first section was to learn about the interviewee’s experience with multi-jurisdictional research projects. We hoped to draw out rich information about a particular case study or two, where possible – letting the interviewee select a project considered relevant, and then deep-diving into it. The objective of the second section was to learn about some of the possible general problems encountered in multi-jurisdictional ethics review. We wished to know if participants viewed the current system as a ‘problem’ and, if so, why – and what kinds of ‘problems’ they identified in particular. The objective of the third section was to determine whether participants were aware of any ‘work-around’ solutions to the perceived problems (if they identified any problems), either based on their own experiences with research projects or through general awareness. We also wanted to broach different models the ERE Task Team had developed, including three emerging models (reciprocity, delegation, federation) of ethics review mutual recognition (Dove et al., 2016) 3 and receive feedback from participants regarding the feasibility and support of these models.
Sampling, selection and analysis
Prior to recruitment, ethics approval was obtained from the Edinburgh Law School Research Ethics and Integrity Committee. We then engaged in strategic sampling (Mason, 2002) to identify individuals (a) who could speak to multi-jurisdictional ethics review from different jurisdictional perspectives, and (b) who had experience with a particular research methodology (i.e. data-intensive research), whilst (c) also keeping in mind the project’s resource limitations. Ease of access also drove the sampling strategy. Therefore, we also used convenience sampling and, after the initial interviews, snowball sampling, to select further participants with the aim to be representative of various global regions (Patton, 2002).
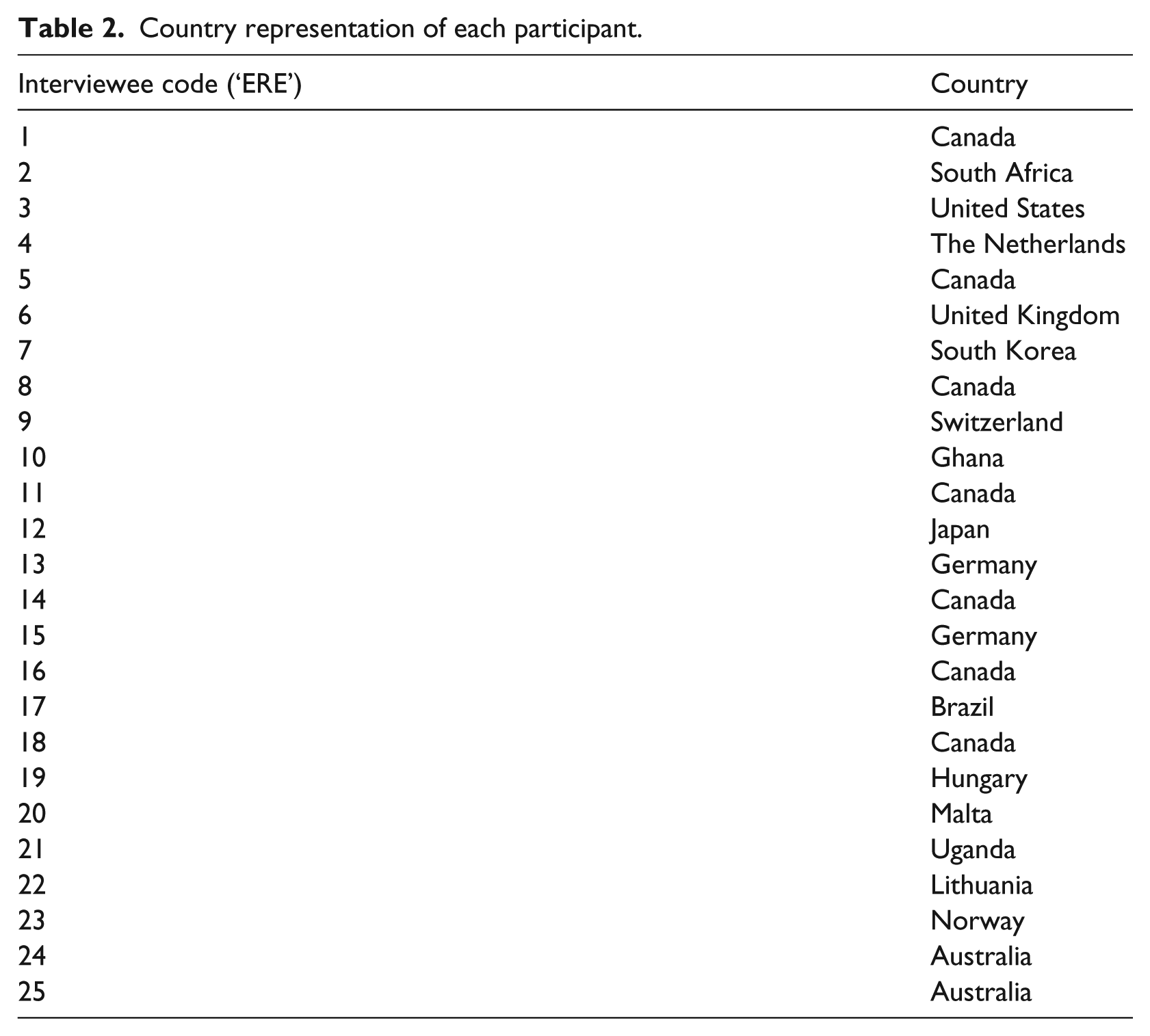
Twenty-five individuals, of the twenty-nine contacted, responded and agreed to be interviewed in English. All but two of the interviews were conducted via Skype; the other two interviews were conducted in person. These 25 individuals came from different jurisdictions that reflect different socio-legal and research environments. Several participants were also current or past REC members themselves. Whilst there is some overlap, the professional and geographic distributions by continent are reflected in Table 1. Table 2 provides the specific country and interview ID of each participant. The average interview time was 57 minutes (ranging from 35 minutes to 82 minutes). Interviews were audio-recorded with a digital recorder; audio files of the interviews were professionally transcribed in intelligent verbatim and then checked against the recordings. Transcripts were manually coded using qualitative thematic analysis, which offers theoretical freedom and flexibility to yield rich and detailed, yet complex, accounts of data (Braun and Clarke, 2006).
Participant demographics (

Country representation of each participant.
Limits
Our qualitative research project has some limitations. First, we interviewed only individuals we identified as ‘experts’ (which we took to mean academics, regulators or policy-makers, and life sciences researchers) in research ethics and data-intensive research. This therefore excluded perspectives from other critical actors such as participants and researchers in other domains who could offer additional insight into problems, challenges and solutions relating to ethics review of international data-intensive research. As our research focused on the perspectives of experts in the field of data-intensive research, the views expressed may not be representative of those in another research context. However, some participants felt more comfortable discussing other areas of research, such as clinical trials, and therefore our findings may have resonance outside the confines of data-intensive research as we define it. Second, the framing of our research project and several of the interview questions may have meant that those who had a particular story to tell or a particular interest in ethics review were more likely to share their insights of perceived problems and work-arounds. Third, Canada is somewhat overrepresented in our sample, largely due to the snowball sampling method. Although we believe that our Canadian participants offered insightful perspectives on navigating ethics review in a federal country and in a country known for its strong data-intensive and internationally collaborative research environment, it is unknown how this distribution would compare to other countries in terms of density of research activity or experience with ethics review problems and the solutions associated with mutual recognition. Finally, although qualitative data provide valuable insight into conceptually nuanced topics such as ethics review, our findings are not externally generalizable per se. This said, we believe our thematic findings can generate ‘theoretical generalisability’ (Mason, 2002); in other words, they can be used to generate hypotheses and queries for subsequent investigation.
Results
Participants communicated to us that although there are multiple
In the remainder of this section, we unpack the problems, challenges and solutions identified by the participants.
Problems
All participants stressed that the current system in place for reviewing the ethical acceptability of international data-intensive research suffers from problems that impact the system’s efficiency and robustness. Indeed, participants identified over 25 distinct issues they considered to be ‘problems’ with the current system (either stated explicitly as a ‘problem’ or through use of a similar term). The underlying thread in all the distinct problem areas identified is the notion of
Variability, inconsistency and duplication
Not surprisingly, given its common presence in the empirical literature (e.g. Angell et al., 2006; Caulfield et al., 2011; Ferguson and Master, 2016), participants frequently expressed concern about the amount of variability and inconsistency in decisions or opinions rendered by RECs across sites within the same jurisdiction or across several jurisdictions. Participants also criticised the duplication of ethics reviews, leading to bureaucracy and significant time delays in research projects. The general sentiment expressed was that RECs conduct the same kind of review based on the same project, leading to waste. Participants emphasized this point well by referencing their own difficult experience with multi-jurisdictional or multi-site research projects even within their own country, either as a project manager or member of a REC: We submitted [our application] to ten sites and we had ten hospitals in [an Australian state], we needed to submit … this is our [research] protocol back in 2007/2008. We wanted to get this up and running within a year, knowing it would probably take us that long to get all the protocols sorted out, so we still had to fill in the local 12-page documented questions that were relevant to whatever local ethics committee. (ERE25)
Participants found it unclear what the additional reviews contributed by way of ethics rather than local site-specific
Lack of communication
Lack of communication between RECs, especially across multiple jurisdictions, was identified by participants as a cause of inefficiency and variability in the system. If RECs fail to adequately communicate with one another, it is difficult for them trust each other, much less have insight into the decision rendered by another REC on the same research project (and the reasons thereof), and for RECs to learn about best practices from one another. Some participants expressed frustration that much knowledge about the ethics of data-intensive research is not shared across jurisdictions, and that REC administration is detached from the deep scholarship on research ethics generally. Others commented that it is difficult for RECs to view the opinions rendered by other RECs. Because the underlying reasons are not commonly shared (particularly outside the country), an internationally shared body of knowledge cannot easily be built.
Lack of expertise
As mentioned above, another cause of systemic inefficiency and variability identified by several participants was that REC members lack sufficient training or expertise, especially with respect to understanding the issues associated with data-intensive science, including the challenges it presents to traditional notions of ‘specific’ consent and the need to link data to multiple datasets – possibly in other jurisdictions – for research discovery. Participants found that, speaking of RECs generally, there is a crucial role for training of REC members (particularly in research ethics and data-intensive science) and for REC members to have a good orientation as to what their mandate is. Some RECs, particularly in lower-and-middle-income countries (LMICs), were seen as apprehensive to emerging or ‘grey’ areas of science such as genomics, in part due to either the lack of any regulation or guidance in the subject area, or regulatory restrictiveness that thwarted the REC’s ability to be innovative in their review processes.
Challenges
Despite the many problems identified in the current system, participants suggested innovative work-arounds (as discussed below in the section on Solutions), but often whilst simultaneously acknowledging challenges in trying to reform the status quo. Indeed, participants listed more than 30 distinct challenges, which are grouped into several categories discussed below. The common underlying thread across the identified challenges was the difficulty in building trust between RECs such that each could rely on the review undertaken and decision made by another.
Local context and sensitivity
By far the most significant challenge identified was the pervasive notion of locality, specifically local context and sensitivities. Likewise, the literature is replete with discussion about the importance (or not) of local knowledge and decision-making (e.g. Barchi et al., 2014; Klitzman, 2015; Wainwright and Saunders, 2004). Several participants noted that it is difficult to involve local communities in ethics review processes in a meaningful way, and to assume that RECs can adequately represent such sensitivities. Further, some participants pointed out that there is disparity between the ethics review system in the Western world, where governments and research institutions have invested heavily in operational resources and professional staff to assist RECs, and the ethics review system in LMICs, which remains understaffed, under-resourced and largely under-informed about emerging data-intensive science areas such as genomics.
Local context was a hybrid concept in our research findings: some thought it to be an insurmountable challenge to systemic reform; others thought it also to be a current ‘problem’ in the system that caused inefficiency. Few thought it could be addressed with a clear solution. For example, ERE6 found that, in her years working in international research consortia, local sensitivities were important, if difficult, to acknowledge: … the questions that come out of an individual country often times take you by surprise. ‘Oh, that’s what you worry about’, or, ‘Gosh, you know, I hadn’t thought about that’ … You just don’t know what’s important to a country, if I can use that as a kind of a global setting, or even peoples within a country. It’s completely different and harmonization is blooming difficult when it comes to that kind of thing. You just have to spend a lot of time finding out what is important to different groups.
Participants felt that there are vast cultural differences between RECs, reflecting in part how different laws or policies govern the ethics review process. Participants from Europe further stressed that linguistic differences between countries can affect the style of communication, and that the nature of ethics review is very much linked to the way one deliberates on ethics issues.
Some participants noted the tremendous cultural variation even within the same country. ERE10, for example, commented that the REC in the Ghanaian capital city, Accra, might not ‘necessarily understand the local culture and some of the traditions that might influence research participation [in other parts of Ghana], some of the issues that people might have about participating in genetic and genomic studies’. For this reason, she felt that local RECs were likely to identify pertinent ethical issues better than a distant REC ‘because of the closeness of the committee to the research site and also to the research participants’, but she conceded that they ‘haven’t tested this in any way to see whether it is really the case’. She felt that a ‘joint ethics review’ in Ghana is possible, ‘as long as the local ethics committee is not left out’ in order ‘to still get those inputs from someone who would be able to bring these local perspectives to the review process’. ERE10 mentioned this could be done through a community representative participating in the joint review, but as ERE21 from Uganda noted, defining a ‘community representative’ is a difficult endeavour (especially in an intensely varied culture environment such as many places in Africa), and it remains unclear how and whether this approach can be scaled up beyond just one country or region.
Others, however, questioned whether local context and sensitivity was really an insurmountable challenge, and whether it was more rhetoric than reality: The thing that I’m always puzzled [about local context and sensitivity], though, is that if that’s the claim, where’s the evidence? … It becomes a procedural difference rather than a substantive difference. I don’t think it’s defensible to maintain procedural differences because it’s just that you do things slightly differently. Well, that’s just a nonsense to say that that should stand in the way of a more efficient review. (ERE4)
Similarly, ERE3 stated: ‘I think it’s a bit of a mistake and fantasy to believe that those committees are actually functioning only as local representatives of their institution. They’re not.’ Some participants described local context and sensitivity as a ‘nightmare’ for reforming the system, but felt it was a ‘cloak’ that REC members and others often hide under as an excuse to avoid thinking about systemic reform. These participants felt that the issue was really about trying to maintain local (i.e. specific REC) precedent and control rather than local sensitivity, an observation that has been evidenced in the empirical literature on RECs (e.g. Klitzman, 2015; Stark, 2012).
Attitude and power dynamics
Participants suggested that institutions, RECs and members thereof are reluctant to cede power and authority to another REC and therefore could pose an obstacle to any model seeking to streamline ethics review. Some spoke of both anxiety and attitude issues amongst REC members because of uncertainty whether they will make the ‘right’ decision, or because they think their decision will be ‘more right’ than other RECs. In the opinion of these participants, RECs have always been bodies of power that control research and thus are reluctant to move towards a system of ethics review mutual recognition where they have to cede some control and thereby trust the review of another REC: Every REB thinks that they do the best job or that they are the ones that nobody can review it the way that they do. It’s a little bit of an ego and it plays into it. Also there’s the general feeling that, well, if we’re not seen as an REB that could be the REB of Record, then we’re going to lose our share of the research or we’re going to lose the caché that’s associated with having a ‘qualified’ research ethics board. (ERE16)
ERE7 stated that accreditation systems, whilst a possible solution to improving the overall quality of RECs, could also exacerbate power dynamics, either because non-accredited RECs are viewed as untrustworthy or because even accredited RECs feel that they must always take charge of reviewing a research protocol in order to maintain their accreditation standing.
Another important power issue concerns the relationship between the Western world and LMICs. Participants, particularly from the African continent, raised concerns about double standards (e.g. an expectation that Western RECs might have to be ‘recognized’ by African ones, but not the other way around), and wondered whether RECs at different institutions in previously disadvantaged areas of a country or region would follow due diligence in making decisions. In this context, emphasis was placed on the importance of trusting that the research enterprise will promote the interests of
Regulatory complexity, jurisdictional politics and sovereignty
Systemic reform of ethics review is challenged by the extant laws and politics within each jurisdiction. Not only do jurisdictional politics and sovereignty present challenges to designing models of mutual recognition, complex or restrictive regulations also can make delegation or centralization of review cumbersome or illegal (i.e. creating potential regulatory liability). Participants recalled situations where they had engaged in discussions with RECs in other jurisdictions to query whether they could do a ‘facilitated review’ (i.e. forego further full committee review and rely on a full committee review done by one REC), but ultimately could not, ostensibly because of jurisdictional issues such as regulations requiring that full committee review be done by one or several RECs in their own jurisdiction.
Participants noted that some regulatory frameworks concerning, for instance, privacy and data protection, require ethics reviews be conducted in the local jurisdiction, preventing reliance on or delegation to reviews conducted elsewhere. Some lamented the additional, non-ethics-related roles imposed on RECs (e.g. acting as a privacy overseer for an institution) that make shifting to a non-local ethics review model challenging. Because of the challenge of regulatory complexity (or restrictiveness) and sovereignty, many stressed the importance of having a regulatory authority at the highest level of government – if not international authorities such as the World Health Organization (WHO) – to push for change and endorse the legality of reliance on reviews conducted by RECs in trusted jurisdictions. Others, however, held little faith in the ability of regulatory authorities, national councils or governments, or supranational unions to push for ethics review mutual recognition. For these participants, actors at this level are not necessarily the right actors to facilitate systemic change.
There is a further challenge in constitutional politics reflected in the composition of certain countries. Participants in Canada and Australia noted that because health is governed at the provincial/state level, there was a gap in leadership in pushing for federated or centralized ethics review across the entire country, much less outside it. At the same time, the federal government has to respect the politics of the provinces/states to prevent encountering resistance: I think it’s always difficult to do things nationally, to tell people what to do … Anything you do federally has to come in respecting what’s happening in those provinces and taking the attitude, ‘how can we facilitate and help this, what can we do to make things work and improve things across the country?’ If you come in with, ‘we’re going to coordinate all of you’, that’s not working. It’s not going to work. (ERE18)
Some participants observed that jurisdictional sovereignty and local law impact on all areas, inside and outside life sciences research, and present significant challenges to harmonization. This is not to say that participants considered ethics review harmonization to be impossible as opposed to challenging. Rather, many stressed that harmonization – that is, working towards mutual recognition of elements of a robust and trustworthy ethics review and similar processes – should be distinguished from global ‘standardization’ that could, for example, impose a single consent template or research protocol template worldwide, in addition to the multiple (and wildly differing) templates that already exist (Jackson and Larson, 2016) which could create or exacerbate liability concerns in each country.
Solutions
Despite the many problems, participants suggested multiple solutions to reform the system, even if some carried challenges themselves. Quite often, participants qualified their discussion of potential solutions by advocating two overarching tenets: (i) the need to demonstrate value (i.e. show that a different way of conducting a multi-jurisdictional review is beneficial for different stakeholders); and (ii) the need to have leadership from both the bottom up (e.g. within specific projects or institutions) and top down (e.g. within regulatory agencies or government ministries). Most of the solutions identified by participants can be grouped into the categories of improved communication, harmonization and operational planning, as follows.
Improved communication (frequency and platform)
Many participants spoke of the need to bring various stakeholders together, ideally in-person (but also possibly through internet web fora), to talk through the issues and explore alternative arrangements for multi-jurisdictional ethics review. The suggestions ranged from holding face-to-face meetings among RECs a few times a year to discuss scientific questions, legal questions and ethical questions to determine if there are discrepancies and different opinions among RECs, to more established arrangements for sharing of information among RECs, which is seen by some as a prerequisite to creating a more centralized or coordinated ethics review system. Several recommended having a much stronger forum for dialogue about ethics, and engaging all institutions involved in research, as both are seen as an essential step towards achieving mutual recognition.
Harmonization
Participants suggested various ways of harmonizing processes of ethics review. This included developing a so-called ‘placemat’ where a common ethics review form (with sublayers of guidance) is provided for REC members when conducting reviews. It also included fostering associations for RECs that could run training programmes, drafting guidance documents and establishing fora for RECs to build trust in each other. This exists in some jurisdictions already. For example, the UK’s Health Research Authority has created a common ‘ethical review form’ for National Health Service REC members to consult when undertaking reviews (Health Research Authority, 2015). Others suggested that regulatory authorities (or some other competent body) create and disseminate model (i.e. template) reciprocity agreements for RECs to use, ideally for establishing reciprocity across jurisdictions and not just within. Still others suggested that countries try to harmonize (but not standardize in a formulaic way) their definitions of key terms such as a ‘research database’, as well as the governance arrangements for RECs that address issues such as composition of committees and how the reviews should be undertaken. Such harmonization would cater to some cultural variation to avoid making the ethics dialogue between members and committees artificial, but nonetheless firm up trust in the reviews undertaken by other RECs in other jurisdictions.
Operational planning
Operational planning, or due diligence of various operational aspects before launching a new model of ethics review (be it federated or delegated arrangements), was viewed by many as crucial both for establishing the legitimacy of an alternative model (e.g. a newly established central REC or a REC that has been tasked with reviewing on behalf of others), and for maintaining the model’s success. ‘Operational planning’ as discussed by participants often translated into money, a regulatory mandate, space, staff (i.e. administrative support), a secretariat and software (including common online IT platforms for sharing information seamlessly, including perhaps with a standardized research application form). To establish meaningful ethics review mutual recognition, the first step according to many is to focus on implementation, developing interoperable infrastructures (such as shared software) and building the processes into the already-developed administrative processes for each institution.
Another interviewee also felt ‘ethics review equivalence alone is not enough’ and that the importance of the operational aspects of any streamlining initiative could not be stressed enough (ERE14). She referenced the initiative undertaken by the Canadian province of Quebec, which has instituted a more streamlined ethics review model since 1994. However, it was not until the province implemented a recently revised multi-centre model with a complete infrastructure surrounding it that improvements in efficiencies started to be noted on the ground and in collected data metrics. Thus, ERE14, along with others, stressed that in promoting greater efficiencies in multi-jurisdictional REC review, it is crucial to deliberate and even pilot test a model or arrangements beforehand to determine how it might work (or not work) in practice.
Bottom-up approaches
Participants, particularly those sceptical of systemic change being led from the top through government, thought that significant change in the near term more likely would come from the bottom up, where international research projects can build a critical mass of data to develop several good case studies of successful implementation of mutual recognition. Through these case studies, further pilot studies could be tested that would further accumulate data to lead to evidence-based policy for governments to then implement on a systemic level. Participants thus suggested ‘starting small’ in developing mutual recognition. Each jurisdiction (e.g. a province, state or country) could work towards having a single ethics review for the whole jurisdiction – some said ideally on a voluntary basis to foster greater support – and then building up from there eventually between two countries and then beyond. By starting small, evidence can be gathered and, through an iterative process, improve the mutual recognition model(s).
Regulatory endorsement and accreditation
Even though many participants felt that systemic change gradually would be led by projects on the ground and expressed some scepticism about political leadership, many equally felt that there was a critical role to be played by governments, regulatory authorities or similar bodies, policy-makers and research leaders. The common sentiment was that both elements were needed: bottom-up at the REC and administrative level, and top-down from senior research leaders and government. Some even went further and advocated new legislation to change the current system. For example, ERE6 referenced the 2002 EU Clinical Trials Directive as an example of successful legislative change that overhauled the UK’s previously much-maligned REC system, most notably by instituting a single UK-wide ethics opinion for most health-related research. According to her: ‘It’s the only way. It was the [Directive] that really drove something a little bit more formal here in the UK, and people being fed up with not being able to get any work done.’ Similarly, ERE23 said that the best way to reform the system, at least in the EU, was for new EU legislation to be passed, as ‘it is important to find those solutions and perhaps committees also need to be in a sense forced to accept a more international system’.
More participants, however, spoke in favour of regulatory
Some thought that national regulatory authorities could work together to establish international systems of accreditation, qualification or certification of RECs to encourage mutual trust in each other’s processes of review. ERE21, for instance, found that the accreditation system created in Uganda in 2009 vastly improved problems of ethics review capacity, consistency and logistics in the country. But whilst some participants saw accreditation (or a similar approach) as a useful step towards establishing mutual trust in RECs, others were cautious in touting its benefits. As one stated: ‘What’s the problem we’re trying to solve? Because if you ask the folks in Quebec, they’ve had a kind of accreditation system of [RECs] since 1994, but it hasn’t helped with efficiency and getting reviews faster until they’ve actually put a mechanism in place to support the infrastructure and the actual operations’ (ERE14). The concern from several participants was that accreditation could actually exacerbate a tick-box mentality where RECs merely follow the regulations, and following regulations does not necessarily mean that the REC is protecting participants any better. Thus, they emphasized caution in advocating too strong a top-down approach in imposing ethics accreditation. As there is as yet no sound evidence that shows accreditation alone better protects participants or improves the efficiency of ethics reviews, several participants advocated a ‘staged approach’ to establishing a baseline quality or standard in reviews and then moving toward full accreditation only if there is sufficient evidence to do so. Nonetheless, participants felt that it should be possible to work towards an agreement for equivalent protection.
Mutual recognition models
Participants were supportive of ethics review mutual recognition models discussed in a recent article (Dove et al., 2016), namely the delegated model (also known as a ‘REC of Record’ model), the reciprocity model (often conducted through bilateral reciprocity agreements or MOUs between institutions), and the federated model of ethics review. In this latter model, which was the most commonly cited by participants, institutions, funders or regulators/governments create through agreement a central, specifically formulated multi-jurisdictional REC (which could be comprised of REC representatives of each involved site). The agreement may state that the multi-jurisdictional REC assumes compliance functions and adheres to internationally recognized ethics policies as well as applicable regulatory requirements. The multi-jurisdictional REC could manage the liability of local institutions (e.g. through several or proportionate liability or ‘no fault’ insurance schemes) (Townend et al., 2016). The agreement may also stipulate agreed-upon principles, forms, operating procedures and standards that allow for robust ethical scrutiny and continuing review. One option is that local sites may provide input on site-specific matters to the multi-jurisdictional REC, and the ethics review opinion provided by the multi-jurisdictional REC may be accepted or rejected by all participating local sites.
Participants often advocated this model more than others, for various reasons. Some suggested that reciprocity agreements between institutions can be impractical (as they govern relationships between only two or several institutions rather than entire jurisdictions), and that a ‘REC of Record’ (i.e. delegation of full review to a single REC) model could present challenges where not all sites are involved in projects (hence it would be more ad hoc) or in similar ways, and therefore there can be resistance to having everything go to one REC and not others. A federated REC comprised of the leading experts from various regions in data-intensive science, or even a variation of this, such as a ‘joint REC’ with joint chairs and members of two or more institutions, was seen as a promising approach to addressing concerns of local context, both reducing duplicative reviews and building trust amongst RECs, institutions and jurisdictions. Some thought that this model could be pilot-tested in one jurisdiction and then scaled up, depending on the evidence of success. As ERE10 told us: ‘An ideal situation would be a kind of joint review, where if the project is going to be conducted at three sites in Ghana, then some members of the three committees could come together and conduct a joint review of that project, instead of letting it go through all the IRBs.’ This interviewee added that it would be important for such a REC to still receive input from an individual who could bring local perspectives to the review process. However, other participants acknowledged challenges in a federated REC model, including membership composition and the potential loss of, rather than recognition of, ‘local sensitivities’.
Discussion
Almost all participants spoke of the regime of multi-jurisdictional ethics review as being comprised of interconnected problems, challenges and solutions set within overarching themes –
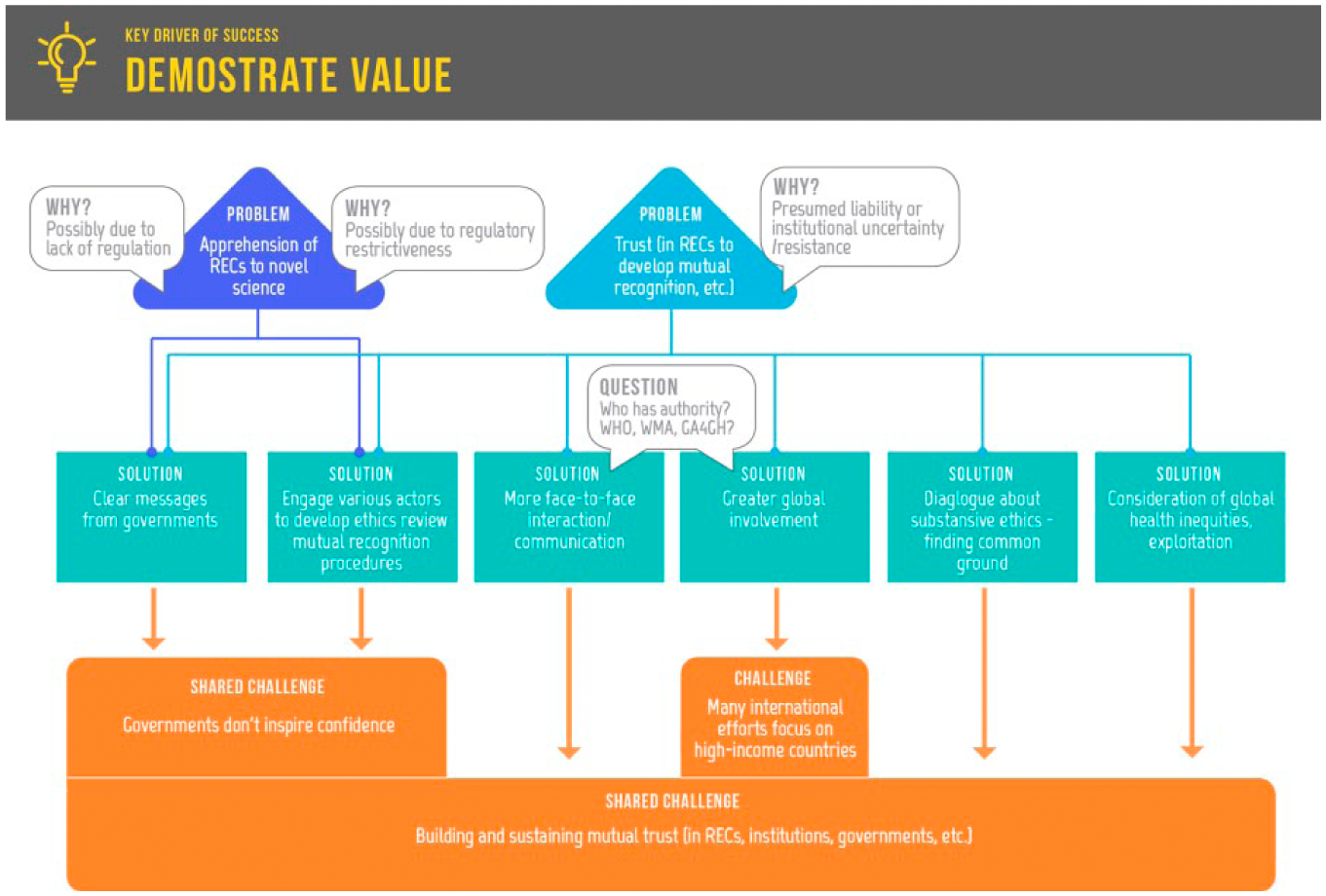
Addressing the problems of lack of trust and apprehension to novel science.
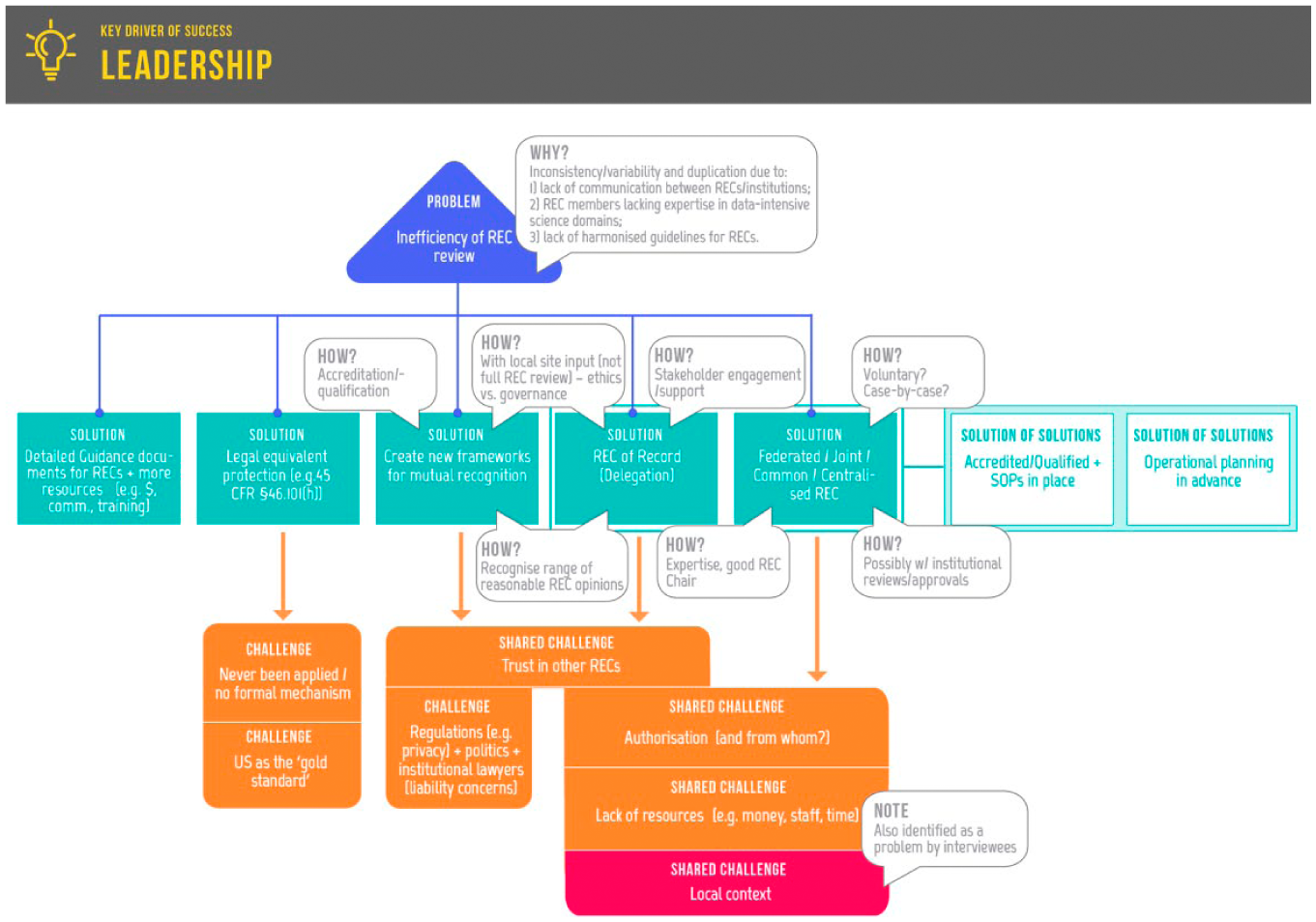
Addressing the problem of systemic inefficiency.
In our discussions, we encountered a general sense of concern about the present ethics review system, which has also been expressed in the literature (e.g. Ferguson and Master, 2016). This said, all participants were of the opinion that ethics review is a critical component of research involving humans (even if such research only involves humans indirectly) and should not be seen by researchers as merely a hurdle to get past.
The foremost concern was
A few participants were sceptical of systemic reform – particularly those in Europe with experience in EU policy-making. A few suggested that at least one full REC review should be undertaken in each relevant country, though some were open to the idea of regional REC review, e.g. an ‘EU REC’ in Brussels. Most advocated that greater information-sharing between RECs in different jurisdictions should occur so that reviews between them could be informed by the other; this would be an improvement that need not require a completely different model of ethics review. Several participants also stressed that RECs are only one (albeit important) component in data-intensive research (McDonald et al., 2011). In the data-intensive research context, other actors such as data access committees, funders, institutions, regulatory authorities, and privacy/data protection authorities and officers also must be brought into discussions about systemic reform.
This is seen as a particularly important issue within data-intensive research, as some express concern that data-sharing is less ‘REC-impeded’ than it is privacy-officer impeded, and that data privacy legislation is disharmonized across jurisdictions and interpreted by privacy officers at different authorities or institutions in rather different ways (Dove and Phillips, 2015). For these participants, concerns about an inefficient ethics review system stretched to concerns about research governance processes for data-intensive research (and data-sharing) as a whole (Shabani and Borry, 2016; Shabani et al., 2015).
Regarding the seemingly intractable challenge of local context and sensitivities, several participants suggested that this could be addressed through community engagement exercises and in separating research ethics issues from research governance issues. For example, one interviewee discussed how a federated REC operating in a jurisdiction still permits local sites (institutions such as hospitals) to have a role to play in considering local aspects, where they can submit ‘centre-specific’ applications once the main jurisdiction-wide application has been reviewed and approved. This mechanism is seen to give institutions the chance to do tweaking and provide local input on governance matters, which reduces duplication and inefficiencies in the ethics review(s).
Finally,
Conclusion
The aim of this qualitative research project was to gather perspectives on real-life experiences with navigating ethics review of multi-jurisdictional data-intensive research projects, problems that arise in the course of this navigation, and how these problems can be effectively addressed, if at all. In this article, we highlighted the themes of systemic inefficiency and trust, as well as various problems, solutions and challenges in building a system of ethics review mutual recognition. This system can be designed through various models built on the dual tenets of demonstrating value and leadership (both bottom-up and top-down). Future studies should explore the niche area findings such as: the ‘challenge’ of addressing local context; the possibility of tailoring ethics review mutual recognition models depending on the sensitivity of the data to be collected; specific tools to foster trust between RECs and institutions; metrics to measure the quality of REC review (Nicholls et al., 2015); the difficulties associated with jurisdictional sovereignty, politics and regulation; and the notion of ‘power’ amongst different actors (institutions, RECs, regulators, sponsors and researchers) and how it impacts on ethics review (Hemminki, 2015).
Whilst many of the problems identified by participants have been known and discussed for years, as has the theme of systemic inefficiency, we share our participants’ belief that we are at an ‘inflection point’ for change, and share optimism for various ways forward for short-term gain, as well as long-term solutions. In this sense, there appear to be recent promising avenues for systemic reform. The GA4GH has developed an ‘Ethics Review Recognition Policy’ document to foster recognition of extra-jurisdictional ethics reviews and improve the consistency thereof, as well as to promote efficient and responsible health-related data sharing for human health and wellbeing. The policy could perhaps eventually be expanded to other types of international biomedical research. 4 A similar but broader initiative has been undertaken by the WHO (World Health Organization, 2011). Whilst a policy obviously cannot address more structural issues such as laws and monetary resources for RECs, it can establish a baseline for building trust in the processes undertaken by RECs in different jurisdictions, including in parts of the world that would want assurances that subsequent data uses for research would not lead to further disempowerment of local scientists and populations.
Also, participants pointed to successful existing initiatives in several jurisdictions that incorporate elements of mutual recognition, including: Clinical Trials Ontario; the Ontario Cancer Research Ethics Board (OCREB); the NIH’s move to require use of a single institutional review board for multi-site research in the US (National Institutes of Health, 2016) and a similar initiative in the US federal government (Federal Register, 2017); and the Australian federal government’s National Approach to Single Ethical Review of Multi-centre Research. The logical and achievable next step would be to scale up these existing initiatives extra-jurisdictionally. With Clinical Trials Ontario, it could mean working to achieve mutual recognition with an ethics committee in neighbouring Quebec or Manitoba; in Australia, it could mean pilot testing mutual recognition with an ethics committee in New Zealand. Research consortia may also, in consultation with relevant competent regulatory authorities, seek to pilot test a different type of mutual recognition model and determine its effectiveness even between two neighbouring and trusted jurisdictions (e.g. US and Canada or Norway and Sweden). There is also a role for international and regional bodies such as the WHO, GA4GH, UNESCO, EU and the European Network of Research Ethics Committees (EUREC) to play. We believe that these organizations have the legitimacy, expertise and authority to bring various actors together to share best practices and knowledge, foster trust, and work to design or endorse frameworks of mutual recognition.
At the same time, science groups, sponsors, funders and research consortia should continue to demonstrate leadership on the ground by developing innovative solutions to ethics review and sharing lessons learnt to develop best practices in areas such as conditions for release of data and data access procedures. Regulatory authorities can and should do more to encourage institutions and research organizations to use mutual recognition models when appropriate. Finally, we advocate longer-term orientated solutions such as: continued building of national and regional ethics committee networks and structures that can be integrated into an international network to build communication and trust; greater capacity building of RECs (particularly in LMICs); the development of internationally supported certification or accreditation processes for RECs and/or REC members to improve overall quality and trust; the development of interoperable IT systems for sharing research application-related documents (including an HTML-based standard research application form and an online application portal), REC opinions and registries of approved projects; greater separation of ethics issues from governance issues/empirical questions (e.g. local participant recruitment, site suitability, investigator competence), the latter of which should be addressed by local, non-REC bodies; further harmonization of regulation in areas such as data protection and in understandings of consent or authorization for using data for research purposes; and more international research ethics meetings to stimulate in situ dialogue on substantive ethics issues and harmonization of processes.
A deeper question remains, however: if we work to achieve harmonization of
Footnotes
Acknowledgements
The authors thank the interview participants for their generous time and effort in contributing to this qualitative research project, and Amy Xiaoying He (UX Design, Health and Life Sciences, Intel Corporation) for her excellent assistance in preparing the figures. The authors thank their fellow members of the Ethics Review Equivalency (ERE) Task Team of the Global Alliance for Genomics and Health (GA4GH) for their insightful comments on previous drafts of this article. The authors also thank the participants in the European Network of Research Ethics Committees (EUREC) meeting in September 2016 in Helsinki, and participants in the Biobanking and Biomolecular Resources Research Infrastructure (BBMRI) meeting on ethics review mutual recognition in September 2016 in Vienna for their useful comments on the research project’s findings.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research was funded by Intel Corporation. Intel Corporation had no influence in the design of the research project nor in the data analysis or conclusions drawn. The authors acknowledge the ERE Task Team funding support of the Wellcome Trust and the Public Population Project in Genomics and Society (P3G). The first author is supported by a Wellcome Senior Investigator Award ‘Confronting the Liminal Spaces of Health Research Regulation’ (award no. WT103360MA). The personal views of the authors may not reflect those of their institutions.
