Abstract
Objective:
Review the safety of fertility preservation through ovarian stimulation with oocyte or embryo cryopreservation, including cycle and medication options.
Evidence review:
A systematic review of peer-reviewed sources revealed 2 applicable randomized control trials and 60 cohort studies as well as 20 additional expert opinions or reviews.
Results:
The capacity for future family building is important for the majority of reproductive age people, despite life-altering medical or oncologic diagnosis. Modern fertility preservation generates a high rate of oocyte yield while utilizing protocols that can be started at multiple points in the menstrual cycle and suppressing supra-physiologic levels of estrogen. Finally, more than one quarter of fertility preservation patients will return to later utilize fertility services.
Conclusion:
For most patients, fertility preservation can safely be pursued and completed within 2 weeks without affecting disease severity or long-term survival.
Introduction
Fertility preservation has become a common part of the infertility community vernacular, and yet only a minority of patients diagnosed with cancer, medical conditions, or genetic predispositions that result in subfertility utilize this option. To ensure access to care for all at risk people, clear evidence of safety and best practices for success should be easily available to referring oncologists, immunologists, nephrologists, gynecologists, as well as treating reproductive endocrinologists.
This review assembles the evidence regarding safety of fertility preservation across a range of medical indications. It discusses ovarian stimulation protocols and adjuvants that can increase the chances of a patient’s future fertility while facilitating the rapid transition to appropriate medical treatments that increase the probability of years of future good health in which to pursue family building goals
Fertility preservation is a medical treatment field that encompasses preservation of gametes, embryos, and ovarian tissue cryopreservation. Ovarian tissue cryopreservation and in vitro maturation have, thus far, resulted in lower pregnancy rates and remain experimental with need for further evidence. 1 Thus, we will review the literature guiding embryo and oocyte cryopreservation.
Embryo cryopreservation through vitrification is an everyday part of modern-assisted reproductive care. However, of course, embryo cryopreservation requires fertilization of the oocyte either with a partner or sperm donor. This requirement limits the application of embryo cryopreservation to those who have an acceptable sperm source. For many women facing gonadotoxic treatment, such is not the case, and so embryo cryopreservation may limit reproductive autonomy making oocyte cryopreservation their best or only option.
Embryologists have strived since the early 1980s to arrive at safe clinical and laboratory techniques for cryopreservation of oocytes. Oocytes were first frozen and thawed to achieve a live birth in 1989. 2 Quickly the potential to preserve fertility in the face of gonadotoxic treatments was recognized, but oocyte quality and fertilization rates were initially concerning. 3 The experimental label was not removed by ASRM until 2012 after significantly increased cryopreservation survival rates were obtained following improvements in vitrification as opposed to prior slow freezing protocols. Now, it is time for our clinical communities to become aware of and utilize available and effective pathways for fertility preservation. In an era defined by the recognition of gender and racial inequities, we challenge the reproductive healthcare community to create safe and equitable fertility preservation care systems that ensure the opportunity to pursue a family after treatment with gonadotoxic medications, radiation therapy, or surgical excision.
After patients have fought to survive cancer or other major disease, the inability to get pregnant and deliver a baby can seem like the ultimate betrayal by their own bodies. Cancer survivors have a relative risk of infertility of 1.30–1.48 compared to their age-matched cohort.4,5
In many Scandinavian countries, fertility preservation is free of charge to all individuals at risk of sterility due to medical or surgical treatment. From 1998 to 2018, 73% of adult women who received counseling proceeded with ovarian stimulation and 27% of those survivors returned to utilize fertility services. 6 This return rate was consistent across patients with benign and malignant conditions. 6 In a US cohort, Dolmans et al. 7 showed a true come-back rate of 23%. This return rate to utilize cryopreserved oocytes is sufficient to recommend consideration of oocyte cryopreservation in almost all individuals who are about to undergo potentially gonadotoxic treatment.
Objective
We reviewed the literature on safe management of fertility preservation through oocyte and embryo cryopreservation to facilitate decision-making, appropriate patient-selection, and treatment planning. The important management issues are addressed such as: expectations for cycle yield, impact on disease progression, protocol strategies, and adjuvants that enhance safety and success.
Methods
Search and selection strategy
A systematic literature search was conducted in PubMed, Embase, and Ovid MEDLINE from their establishment through June 2021. No language restriction was imposed. The following search terms were used: (oocyte OR embryo) AND (cryopreservation OR fertility preservation) AND (cancer or chemotherapy) AND (stimulation). The International Prospective Register of Systematic Reviews guidelines were followed (identification number CRD42021251821). The Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines were employed. Ethical approval by an institutional review board (IRB) and informed consent was not required for this systematic review. No financial support was provided. Figure 1 provides an overview of the literature search and selection process.
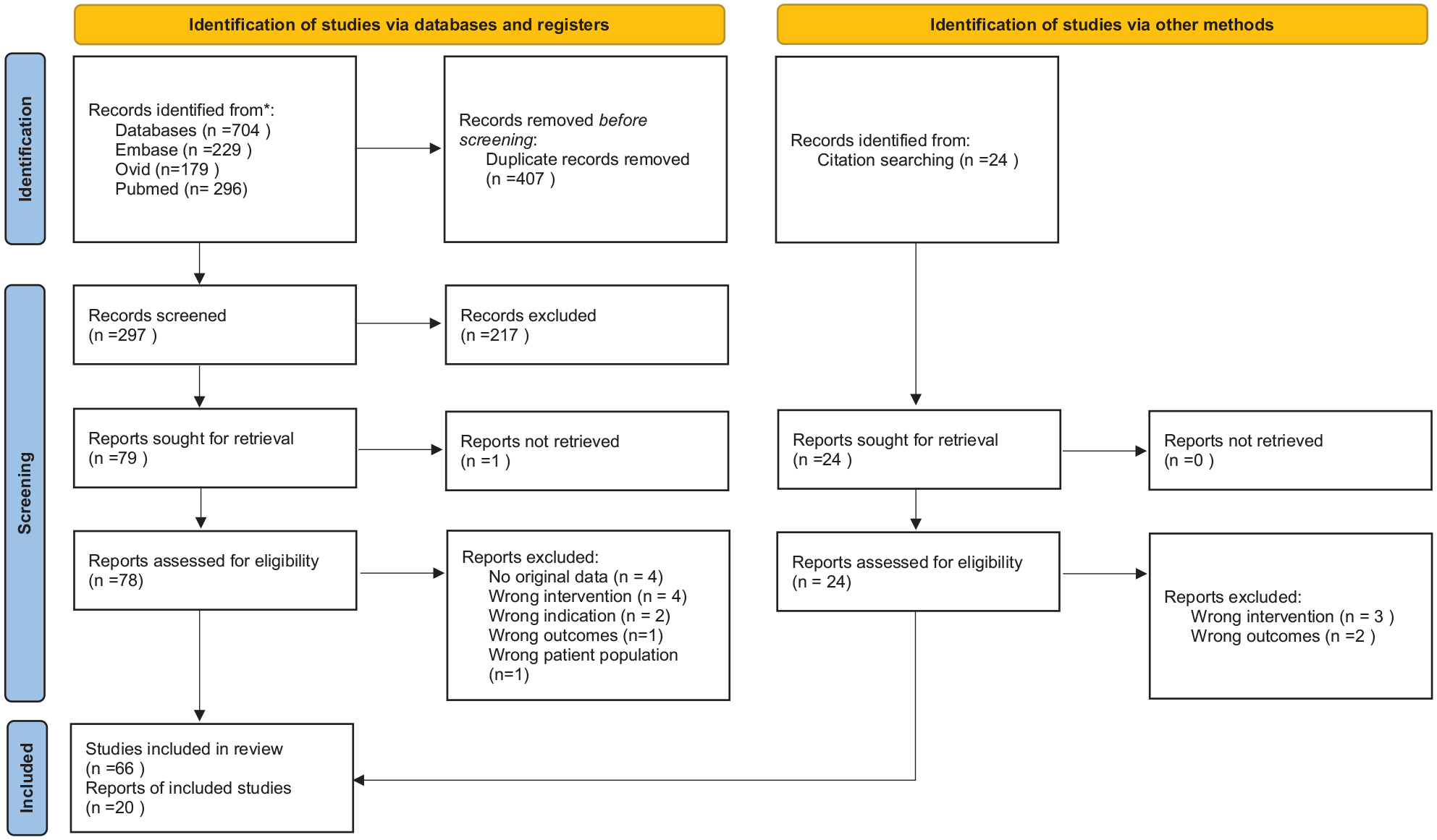
PRISMA 2020 flow diagram for new systematic reviews which included searches of databases, registers, and other sources.
Data published only as abstracts in conference proceedings were reviewed, and attempts were made to contact authors for full details; the data were included if these details were made available. All peer-reviewed, published studies meeting the inclusion criteria for the primary outcomes of evaluating outcomes of stimulation and cryopreservation of embryos or oocytes prior to gonadotoxic treatment in female patients ages 14–45 years were evaluated in full-text form for inclusion. Studies were excluded if they focused on ovarian tissue cryopreservation, did not include novel data, or if the full text was not available. Two authors independently reviewed all citations and their abstracts to assess appropriate studies to be included. Conflicts were then reviewed with a third reviewer to achieve consensus.
Bias was assessed in all included randomized control trials and cohort studies. The Cochrane Risk of Bias 2 assessment tool and the Newcastle–Ottawa Scale were used, respectively. 8 The scoring is noted in Table 3 of the supplemental materials.
Results
The objective was to complete a systematic review of the fertility preservation process and opportunities to enhance safety and yield. In total, 62 studies met inclusion criteria, plus an additional 20 expert opinions or reviews contributed to the subsequent synthesis reported in this article.
Barriers to seeking safe fertility preservation
Expected ovarian stimulation outcomes
Many providers still share a perception of poor cycle outcomes in the setting of medically indicated fertility preservation. The first technical barrier has been oocyte survival following cryopreservation and subsequent pregnancy rate. However, growing evidence demonstrates a comparable live birth rate with vitrified oocytes compared to fresh oocytes. 9
Biologic concerns remain regarding malignancy and disease and the subsequent “detrimental impact on the quality and behavior of oocytes.”3,10 Multiple studies have now demonstrated that fertility preservation cycles in the setting of planned gonadotoxic therapy have resulted in oocyte yield consistent with an age-matched infertility cohort as well as people undergoing planned oocyte cryopreservation.11–14 This does not guarantee that all patients will have adequate ovarian response to fulfill their family planning desires. Cohort data do show that when adjusted for age and body mass index (BMI), people with systemic cancer may have a lower antral follicle count and require higher doses of stimulatory medications. 15 Part of a successful fertility preservation consultation is a tailored discussion regarding realistic expectations of both ovarian stimulation and post-chemotherapy ovarian reserve using markers like anti-mullerian hormone (AMH), antral follicle count and age to predict outcomes.16–18
Retrospective cohort data suggest a mean number of oocytes collected as 8.2 per cycle, with 6.1 of those being mature oocytes for fertilization or cryopreservation. 19 Furthermore, utilization of a cutoff of 1.2 ng/mL for AMH predicted a high likelihood of retrieval of 4 or greater oocytes following stimulation. 20 Finally, AMH values can help predict post-chemotherapy ovarian reserve and function. 21 Women diagnosed with early breast cancer who maintained regular menses following completion of chemotherapy had an average AMH of 2.5 ± 0.4 ng/mL compared to 0.7 ± 0.1 ng/mL for those who did not (p < 0.0001). All women in this cohort with a pretreatment AMH less than 1.9 ng/mL became amenorrheic during treatment. 22
Sometimes patients or the medical team may need to prioritize the first round of chemotherapy or pelvic surgery over fertility preservation. In these situations, subsequent oocyte yield will be lower, but many patients will still have a successful round of cryopreservation following a first round of chemotherapy. 23 Consideration should be given to stimulation for preservation as soon as possible in patients despite ongoing treatment, as AMH decreases and follicle-stimulating hormone (FSH) increases at 1, 6, 12, 24, and 36 months following chemotherapy.24,25 Providers may find the American Society for Clinical Oncology (ASCO) recommendations on fertility preservation and risk of permanent amenorrhea helpful when counseling patients. 26
Risk of delay in treatment, disease progression, or complication
When battling against the threat of mortality from cancer or the life-altering morbidity of lupus, vasculitis, or endometriosis, patients and care teams may have their focus absorbed by short-term disease treatment to the detriment of future survivorship goals. Physicians can provide objective counseling regarding future family opportunities following fertility preservation in conjunction with cancer or other treatment. Minimal, if any, delay in treatment is required for oocyte cryopreservation.
If rapid referral occurs during ongoing oncologic evaluation, cancer can be fully evaluated while fertility preservation is discussed, coverage sought, and medications obtained. The mean duration of ovarian stimulation is only 11 days.12,27 Breast cancer is the most common malignancy to affect reproductive age females, and data have demonstrated that a delay of up to 12 weeks between surgery and chemotherapy has no effect on survival or recurrence.28,29
The need for early consideration for fertility preservation goes beyond malignancy and the many indications are reviewed in Table 1. Autoimmune conditions can have worse fertility outcomes and gonadotoxic treatment-dependent side effects, so affected individuals can also benefit from early referral for consultation. 30 Patients with endometriosis are another group at risk for infertility and have the additional risk of diminished ovarian reserve following surgery, with more surgeries increasing the risk of their being poor responders to ovarian stimulation.31–34
Conditions associated with increased risk of infertility due to diminished ovarian reserve and/or early menopause.

BEP= bleomycin, etoposide, platinum; BRCA= breast cancer gene.
Oncologists, infertility specialists, and patients alike worry about the risk of disease progression later. Cohort studies continue to add to the body of literature showing that patients who undergo ovarian stimulation show no increased risk of death or recurrence compared to those who chose not to pursue stimulation.39,48,53,66,72 This remains true even in hormone sensitive cancer when appropriate protocols are utilized. 39 Furthermore, if patients do plan pregnancy after completion of their therapy, these survivors should be reassured that these pregnancies do not affect long-term survival and recurrence risk.40,56,58 Of note, therapies like radiation may still affect uterine competence during pregnancy, particularly if exposure occurs around or before the time of puberty. 57
The concern regarding the likelihood of complications of fertility preservation can seem like a black box given the relative lack of familiarity with ovarian stimulation outside of the fertility community. The most common side effects of ovarian stimulation include local pain and bruising; gastrointestinal changes including nausea, diarrhea and constipation; hormonal symptoms to include mood swings, fatigue, breast tenderness, hot flashes and bloating; and finally, pelvic fullness and pain.
More serious but uncommon complications include ovarian hyperstimulation syndrome (OHSS) and thromboembolism which in current practice occur in 1%–5% and 0.19% of stimulation cycles, respectively.70,73 OHSS is characterized by increasing fluid shifts due to vascular permeability and leakage into the extravascular space; this can lead to accumulation of fluid in the abdomen (ascites) and in severe cases lead to pleural effusions, respiratory complications, and even acute renal injury. Planned cryopreservation cycles reduce the risk of OHSS by avoiding subsequent exposure to endogenous human chorionic gonadotropin (HCG) which occurs with a developing pregnancy. 67 This can be reduced even further through utilization of a gonadotropin-releasing hormone (GnRH) agonist to trigger final oocyte maturation instead of HCG. 38
Thromboembolisms are known to be increased in oncologic conditions at baseline and should be considered during the elevated estrogenic, hypercoagulable state associated with ovarian stimulation. The annual rate of thromboembolism during oncologic treatment ranges from 0.9% in breast cancer, to 3.7% in Hodgkin’s and non-Hodgkin’s lymphoma, and 4.2% in ovarian cancer. 67 By avoiding OHSS, this risk is likely decreased, but no trials currently exist to guide fertility preservation-specific prophylaxis. 67 ASCO guidelines do not routinely recommend antithrombotic prophylaxis for outpatient procedures.26,74
From a different perspective, De Groot et al. 75 found that controlled ovarian stimulation prior to chemotherapy in breast cancer patients not only demonstrated no increase in grade II/IV side effects but may have a protective effect for mucositis and constipation. Table 2 offers a summative review of the most common questions and current interpretation of the available literature.
Key take home questions and answers.

Risk of bias assessment.

Current stimulation strategies
Traditional ovarian stimulation protocols start stimulation shortly after initiation of menses which allows for synchronization of natural follicular recruitment with medication-driven oocyte stimulation. While the broad range of options for stimulation is beyond the scope of this review, we will focus on stimulation strategies that do not await natural menses and utilize the precious resource of time in these medically motivated patients. These options are also reviewed in Figure 2.

Stimulation cycle options based on time at presentation with plan for rapid initiation of stimulation.
Random start
Random start stimulation strategies advocate for immediate initiation of stimulation medications utilizing letrozole 2.5 mg/day and recombinant FSH 150–300 IU/day or human menopausal gonadotropins (HMG) 150–225 IU/day with early start of GnRH antagonist dependent on baseline ultrasound and cycle date.44,46,61,64 This minimizes patient delay and can proceed without a significant manipulation of standard stimulation medications.
Luteal phase
Luteal phase stimulation specifically denotes ovarian stimulation in the luteal phase of the menses, in other words, 1–3 days following ovulation. This protocol can be the random start protocol as listed above, or an alternate as described by Chen et al. This strategy includes a combination of letrozole 2.5 mg/day and HMG 225 IU/day. A non-androgenic progestin in the form of medroxyprogesterone acetate 10 mg can be added on stimulation day 12 if follicles are greater than 14 mm but not yet ready for trigger to avoid onset of spontaneous menses. This alternate luteal strategy has been shown in at least one large trial not to have an increase in birth defects or complications following subsequent frozen embryo transfer. 69
Dual stimulation
Dual stimulation was developed to enhance oocyte yield in patients with poor ovarian stimulation, this condition may also describe older patients with a medical indication for cryopreservation. The two published versions of the protocol have moderate differences, but both allow for stimulation and retrieval in the follicular and luteal phases of the cycle to enhance total oocyte yield and may be started at a wide range of points within the cycle.54,62
Vaiarelli and colleagues 45 recommend luteal priming with estradiol 4 mg/day with initiation of injectable stimulation on cycle day 2 with FSH 300 IU/day and recombinant luteinizing hormone (LH) 150 IU/day. GnRH antagonist was added on identification of a follicle measuring ⩾13 mm through the development of a mature cohort. When that occurs, GnRH agonist was utilized for trigger and rapid luteolysis. Repeat stimulation utilizing the identical protocol was started 5 days after oocyte retrieval.
Kuang and colleagues 62 instead advocated for a mild first stage of stimulation on cycle day 3 with clomiphene citrate 25 mg/day and letrozole 2.5 mg/day. Letrozole is continued for 4 days, and clomiphene citrate is continued through to oocyte trigger. HMG at a low dose of 150 IU/day is started on cycle day 6 and continued every other day until a mature cohort is achieved, and final oocyte maturation is triggered with a GnRH agonist and ibuprofen 600 mg. Stimulation is quickly started again the day following oocyte retrieval with HMG 225 IU/day and letrozole 2.5 mg/day. Letrozole is continued through identification of a 12-mm follicle. Medroxyprogesterone acetate 10 mg was added on stimulation day 12 through oocyte retrieval to prevent premature onset of menses. Finally, GnRH agonist and ibuprofen 600 mg was employed for cycle-completing oocyte-maturation trigger. Table 4 maps out key safety concerns and mitigation strategies.
Mapping out the safety strategies.

Adjuvants that may enhance safety and outcomes
Letrozole
Letrozole, an aromatase inhibitor, is commonly used in medical therapy for breast cancer. Its utility in ovarian stimulation results from both the increase in follicular recruitment along with the decreased peak estradiol level seen during cycles in which it is used.43,59,64 Initial cohort studies attempted stimulation with letrozole from cycle start compared to tamoxifen.63,64 The letrozole cohort had both significantly enhanced oocyte yield compared to control or tamoxifen stimulation (7.8 ± 0.9 compared to 2 ± 0.3 and 6 ± 1, respectively) and lower estradiol levels (380 ± 57 pg/mL compared to 419 ± 39 pg/mL and 1182 ± 271 pg/mL, respectively). 64 Follow-on studies have demonstrated appropriate pregnancy and live birth rates. 43 This has led to utilization of letrozole not only in estrogen-receptor sensitive cancers but also in other conditions that might be aggravated by a hyper-estrogenic state such as autoimmune diseases and preservation prior to gender-affirming therapy.
GnRH agonists
Previously mentioned in stimulation strategies, an injection of GnRH agonist for final oocyte maturation was initially explored to enhance safety in patients at high risk of OHSS. The brief flare and rapid luteolysis greatly reduces the occurrence of moderate to severe OHSS in a high responder cohort, particularly when appropriate caution in stimulation dosing is exercised, no HCG is added, and no fresh transfer is planned. 68 Furthermore, in a breast cancer preservation cohort, a GnRH agonist led to a more rapid drop in estradiol post trigger while enhancing the number of mature oocytes retrieved. 65 Collaborating physicians should also be cautioned not to administer long-acting GnRH agonist in the 1–2 weeks following retrieval as this may create a prolonged cascade of stimulation.35,76 The GnRH agonist utilized in individual protocols may vary by country and professional preference. The literature supports the use of leuprolide acetate 1 mg, triptorelin pamoate 0.2 mg, or buserelin acetate 1 mg.55,62,65,68,69,77,78 Another option, if GnRH agonist trigger is not available or recommended, is consideration of a double dose (a.m. and p.m. administration) of GnRH antagonist the day prior to HCG trigger. 37
Dopamine agonists
The recent review by Tang et al. 36 reports that dopamine agonists have been shown in 16 randomized control trials to decrease the risk of moderate to severe OHSS. Cabergoline, quinagolide, and bromocriptine have all been shown effective, but may increase the risk of other adverse gastrointestinal side effects. As these medications are widely available and not employed until the decision to move forward with final oocyte-maturation trigger, they can be considered for a last layer of protection for at risk patients.
Antithrombotic prophylaxis
As reviewed previously, routine use of antithrombotics, such as low molecular weight heparin, is not recommended routinely with ovarian simulation or in outpatient care in oncologic conditions. The best prophylaxis is likely avoidance of OHSS. However, if significant concern arises, antithrombotics may be considered for windowing around the time of retrieval. 67
Progestins
Progestins may be used to prime a patient prior to cycle start, to suppress ovulation, or may simply be left in place due to the presence of a previously placed intrauterine device (IUD).
Endogenous progesterone is utilized in luteal stimulation to prevent loss of the follicular cohort prior to retrieval.62,69 Progestin-primed ovary stimulation extends this into follicular stimulation with the use of daily medroxyprogesterone acetate 10 mg starting on cycle day 3.42,47,71 This cost-effective option has been utilized in overweight, obese, and polycystic ovarian syndrome patients to good effect. 42 It may be an excellent adjunct in patients with endometrial hyperplasia or cancer. An ongoing trial is underway to compare the effectiveness for prevention of premature LH surges even in a cohort of poor responders. 47
IUDs have grown in popularity due to their effectiveness both as a contraceptive as well as control of heavy menstrual bleeding. If noted prior to fertility preservation, IUDs should be left in place as they do not affect cycle performance in women undergoing ovarian stimulation. 50
Other supplements
Poor quality evidence exists for use of over-the-counter supplements to improve oocyte yield or quality. Some limited trial data showed improvement in oocyte number, fertilization, and high-quality embryos in low-prognosis young women with 60 days of treatment with coenzyme Q10. 51 However, the short timeframe for treatment in fertility preservation likely minimizes any possible benefit.
Low-dose aspirin has become a common part of the obstetric armamentarium. A recent meta-analysis concluded that its use may improve clinical pregnancy rate, but that current data did not show its use affected oocyte yield, fertilization rates or live birth. 52
Discussion
The medical community has had increasing recognition of the importance of the loss of fertility that often occurs in addition to the morbidity caused by malignancies, autoimmune disease, metabolic and genetic disorders, and other medical conditions that are gonadotoxic in their course or their treatment. 1 However, fertility preservation services have been deployed unevenly and generally insufficiently. Our results echo the opinions of multiple medical organizations including but not limited to ESHRE, ASRM, ACOG, and FIGO that fertility preservation can be safely used in most patients and should be made available for these women.1,41,60,79
In patients with gynecologic cancer, 77% express clinically significant levels of distress in relation to loss of fertility or impaired fertility. 49 This despair is echoed in young patients across the diagnostic spectrum. Success in treating cancer has created a new focus on the quality of life after cancer, and therefore, a demand for quality fertility preservation care. 24
At this time, evidence is largely limited to cohort studies. These are often limited in number of participants and heterogeneity of diagnosis and baseline characteristics. Trials in the general infertility population do offer insight to safer strategies. However, the wider the utilization of fertility preservation, the greater we can voluntarily enroll patients in studies to clarify these areas of ambiguity.
This need for fertility preservation services extends beyond the oncologic community to individuals affected with other diseases notorious for their systemic inflammatory effects and associated gonadotoxic treatments. Vasculitis, lupus, and nephritis all disproportionately affect reproductive age females and place them at higher risk of menstrual irregularity and fertility failure.80–85 Medical predisposition and risk should be identified, and patients supported and treated. Societal barriers need to be overcome. Insurance coverage of fertility preservation services varies widely by country and state of residence. The financial burden associated with gamete preservation for oocytes compared to sperm reflect the complexity but also serve as another example of a “pink tax” that discriminates against women, especially with respect to reproductive healthcare.
In the face of potential mortality for their patients, we hope this review helps treating physicians reach safely across medical subspecialties to think not only of their patient’s survival, but also to the life and family choices that most of them desire, and that are every individual’s right.
Supplemental Material
sj-docx-1-whe-10.1177_17455065221074886 – Supplemental material for Oocyte and embryo cryopreservation before gonadotoxic treatments: Principles of safe ovarian stimulation, a systematic review
Supplemental material, sj-docx-1-whe-10.1177_17455065221074886 for Oocyte and embryo cryopreservation before gonadotoxic treatments: Principles of safe ovarian stimulation, a systematic review by Meghan CH Ozcan, Victoria Snegovskikh and G David Adamson in Women’s Health
Supplemental Material
sj-docx-2-whe-10.1177_17455065221074886 – Supplemental material for Oocyte and embryo cryopreservation before gonadotoxic treatments: Principles of safe ovarian stimulation, a systematic review
Supplemental material, sj-docx-2-whe-10.1177_17455065221074886 for Oocyte and embryo cryopreservation before gonadotoxic treatments: Principles of safe ovarian stimulation, a systematic review by Meghan CH Ozcan, Victoria Snegovskikh and G David Adamson in Women’s Health
Footnotes
Author contribution(s)
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
