Abstract
Background:
Menstrual-related symptoms and menstrual cycle abnormalities linked to low energy availability can have detrimental long-term health consequences. However, longitudinal data regarding the prevalence trends of these conditions in elite athlete populations remain limited.
Objectives:
To investigate changes in the prevalence of menstrual-related symptoms and menstrual cycle abnormalities among Japanese female Summer and Winter Olympic athletes over 12 years.
Design:
A repeated cross-sectional study using retrospective data was applied.
Methods:
Data from Japanese female athletes competing in four Summer Olympic Games (Beijing 2008, London 2012, Rio 2016, Tokyo 2020 (held in 2021)) and three Winter Olympic Games (Vancouver 2010, Sochi 2014, PyeongChang 2018) were analyzed. Gynecological questionnaires assessed menstrual cycle status, symptoms, and medication usage. Trends were analyzed using logistic regression (main analysis) and generalized estimating equations (GEE) to account for repeated participation.
Results:
Among 954 athletes, 786 non-hormonal contraceptive users were analyzed. The prevalence of irregular menstrual cycles significantly increased over 12 years (odds ratio (OR) per cycle: 1.24, 95% confidence interval (CI): 1.13–1.37;
Conclusion:
Irregular menstrual cycles significantly increased among elite Japanese female athletes over 12 years. Furthermore, accounting for repeated participation revealed a significantly increasing trend in dysmenorrhea. These findings highlight the need for continued comprehensive gynecological support and education to optimize the health and performance of female athletes.
Plain language summary
Keywords
Introduction
Modern women experience 4–10 times more menstrual cycles in their lifetime than women living in pre-industrial societies, 1 and the risk of gynecological diseases increases concurrently with the number of menstrual periods, as typified by endometriosis. However, in recent years, awareness of the importance of menstrual management and treatment of gynecological diseases, which can improve the quality of life and reduce the economic burden associated with women’s health issues, has been increasing. Among athletes, the prevalence of menstrual-related symptoms during both the premenstrual and menstrual phases is high, reportedly 74.0%–82.4%,2,3 with many athletes experiencing symptoms particularly on the first-to-third days of menstruation.2,4,5 While the incidence of dysmenorrhea and premenstrual syndrome (PMS) may be lower in athletes than in non-athletes,2,4,6,7 these symptoms adversely affect not only perceived performance5,8 but also participation in competitions or training sessions. For example, Oxfeldt et al. 9 reported that 13% of Danish elite athletes missed or failed to complete planned training owing to menstrual-related symptoms.
Recent research suggests the impact extends beyond participation, potentially affecting objective physiological functions linked to injury risk. Studies indicate that hormonal fluctuations, particularly the decline in the premenstrual phase, can worsen the effect of neuromuscular fatigue on postural control, a factor relevant to non-contact lower limb injuries common in female athletes. 10 Although meta-analyses find only marginal objective performance decrements during the early follicular phase compared to other phases, they also highlight that symptoms such as dysmenorrhea, PMS, and poor sleep quality are associated with performance perceptions. 11 In addition to evidence suggesting that menstrual-related symptoms may affect performance, there is also evidence that these symptoms can influence sleep behavior and recovery outcomes, which in turn may impact performance capacity in elite athletes. 12 Given that performance effects may be mediated more by symptoms than direct hormonal changes,11,13 a comprehensive understanding of these symptoms is crucial for effective conditioning. Despite this, reports focusing specifically on the prevalence of these symptoms in athletes with natural menstrual cycles remain limited.
Since the 1990s, awareness has grown regarding menstrual cycle abnormalities like amenorrhea and irregular menstrual cycles in athletes. The “female athlete triad”—low energy availability (LEA) with or without disordered eating, hypothalamic amenorrhea, and osteoporosis—is well-known, 14 and can lead to long-term issues such as osteoporosis and infertility. 14 Recent reports indicate that amenorrhea is associated not only with these conditions but also with cardiovascular disease. 15 The International Olympic Committee has broadened this concept to relative energy deficiency in sport (REDs), acknowledging the multisystem effects of LEA,15–17 with hypothalamic amenorrhea being a key physical sign in 2014. 17 This concept of REDs applies to all athletes, not just female athletes. Medical and scientific data have since accumulated, leading to updates in 2018 and 2023.15–17 LEA is a state of insufficient energy available to maintain bodily functions and performance in athletes, which may lead to decreased performance. The syndrome of physiological and/or psychological dysfunction experienced by athletes exposed to prolonged and/or severe LEA is defined as REDs. Effects on reproductive function, such as amenorrhea and irregular menstrual cycles, are included as one of these physiological dysfunctions. 15 Incidence rates of amenorrhea and oligomenorrhea vary widely (6%–79%) depending on sport and level,2,18,19 highlighting the significant impact of LEA on athlete health and performance. Understanding temporal changes in the prevalence of these abnormalities is vital for developing effective countermeasures.
However, most existing research on female athletes suffers from methodological limitations, often relying on cross-sectional designs and focusing primarily on Western populations. 20 This limits our understanding of long-term trends and potential cultural or ethnic variations. Furthermore, factors influencing menstrual health may differ across populations; for instance, a study on Japanese university athletes identified longer training hours as a specific risk factor for severe dysmenorrhea, distinct from non-athletes, 21 underscoring the need for population-specific research. To the best of our knowledge, no study has investigated longitudinal changes in the prevalence of menstrual symptoms and abnormalities among Olympic athletes, particularly within a non-Western cohort. Given differing diagnostic criteria and cultural backgrounds globally, 2 examining temporal trends within a specific, high-level population using a consistent questionnaire offers unique insights. Such information can inform targeted management strategies, potentially reducing the burdens associated with menstruation and improving performance.
Therefore, we aimed to investigate changes in the prevalence of menstrual-related symptoms and menstrual cycle abnormalities, as well as gynecological consultation rates, among Japanese female Olympic athletes competing in summer and winter across seven consecutive Olympic Games (over 12 years).
Materials and methods
Study design
This study was a repeated cross-sectional study using retrospective data collected from mandatory pre-participation medical examinations conducted before seven consecutive Olympic Games between 2008 and 2021. This report adheres to the Strengthening the Reporting of Observational studies in Epidemiology (STROBE) guidelines for cross-sectional studies. 22
Participants and data collection
This study was conducted following the Declaration of Helsinki and was approved by the Institutional Ethical Review Board of the JISS (approval No. 038; category = Medical Research Using Medical Information). Written informed consent was obtained from the athletes for the use of the data provided in the questionnaires at the time of the medical check-up. For athletes under 18 years of age, we mailed the instructions and consent form in advance and requested that they bring a signed consent form from their parent or legal guardian on the day of the medical check.
Participants were Japanese female athletes who competed in four Summer Olympic Games (Beijing 2008, London 2012, Rio 2016, and Tokyo 2020 (held in 2021)) and three Winter Olympic Games (Vancouver 2010, Sochi 2014, and PyeongChang 2018). The study population comprised the entire cohort of athletes who underwent pre-competition medical evaluations at the Sport Clinic of the Japan Institute of Sports Sciences (JISS) during the specified period. No exclusion criteria were applied for the initial data collection. Data were collected using a gynecological self-report questionnaire (Supplemental Material 1) completed by athletes during their medical check-ups.
The questionnaire items included sports, discipline, age, menstrual cycle characteristics, presence or absence of dysmenorrhea, presence or absence of premenstrual symptoms (e.g., weight gain, edema, irritability, and breast fullness), subjective menstrual flow volume, use and type of hormonal preparations for menstrual management, and history of obstetric and gynecological consultations. Athletes self-reported medications prescribed by gynecologists, and nurses subsequently interviewed them to verify the information. Height and weight were measured using a DC-250 (TANITA, TOKYO, Japan).
For the analysis of dysmenorrhea, PMS, heavy menstrual bleeding (HMB), and menstrual cycle abnormalities, athletes continuously using hormonal preparations (oral contraceptives (OCs) or progestin preparations,
Diagnostic criteria for menstrual-related symptoms
Dysmenorrhea was defined as pathological symptoms accompanying menstruation according to the Japan Society of Obstetrics and Gynecology (JSOG). 23 This definition is broadly consistent with those used by the International Federation of Gynecology and Obstetrics (FIGO) 24 and the American College of Obstetricians and Gynecologists (ACOG), 25 which emphasize menstrual pain with or without accompanying systemic symptoms. Although pain severity and tolerance vary among individuals, there are currently no internationally established diagnostic criteria based on objective clinical findings. Therefore, in this study, athletes who experienced menstrual pain severe enough to require the use of analgesic medication (answering “Severe; medication is required”) were classified as having dysmenorrhea. This pragmatic definition aligns with methodologies used in large-scale epidemiological and observational studies in both Japanese and international populations,21,26,27 ensuring comparability of methods.
PMS was defined according to the JSOG criteria (“mental or physical symptoms that persist during the luteal phase (for 3–10 days before menstruation) and subside or disappear at the onset of menstruation”), 28 specifically when athletes reported experiencing symptoms like weight gain, edema, irritability, or breast fullness only before menstruation. HMB was defined as subjective reporting of their menstrual blood volume as “heavy,” based on the options “light,” “normal,” or “heavy.” Because only 10 athletes had not yet experienced menarche, a number considered insufficient to meaningfully influence the analytical results, these athletes were included in the statistical analyses of menstrual-related symptoms, including dysmenorrhea, PMS, and HMB.
Diagnostic criteria for menstrual cycle abnormalities
Based on previously reported diagnostic criteria, 29 a menstrual cycle lasting 24–38 days was defined as usual. Cycles <24 or 39–90 days were defined as irregular menstrual cycles, and cessation of menstruation for ⩾3 months as secondary amenorrhea. Athletes aged 13–14, 15–17, and ⩾18 years who had not reached menarche were defined as having absent menarche, delayed menarche, and primary amenorrhea, respectively.
Statistical analysis
Descriptive statistics are expressed as numbers (%) and mean ± standard deviation (SD). Comparisons of participant characteristics (age, height, and weight) across the seven Olympic Games were performed using the Kruskal–Wallis test, as the Shapiro–Wilk test indicated that the data were not normally distributed in some Games. Post hoc pairwise comparisons were conducted using Dunn’s test with Bonferroni correction. To assess temporal trends in the prevalence of menstrual cycle abnormalities, menstrual-related symptoms (dysmenorrhea, PMS, HMB), specific PMS symptoms, and gynecological consultation rates across the seven Olympic Games, logistic regression was used as the primary analysis. The Olympic Games sequence (Beijing 2008 = 1, Vancouver 2010 = 2, London 2012=3, Sochi 2014=4, Rio 2016=5, PyeongChang 2018=6, Tokyo 2020 = 7) was treated as the sole continuous independent variable; no other covariates were included in the models. Results are presented as odds ratios (ORs) per Olympic cycle (one cycle increment) with 95% confidence intervals (CIs). Additionally, a sensitivity analysis using generalized estimating equations (GEE) was conducted to account for the potential correlation of data from athletes who participated in multiple Olympic Games (repeated participation). The GEE models assumed a binomial distribution with an exchangeable correlation structure. Due to the extremely low prevalence of primary amenorrhea and delayed menarche, these outcomes were excluded from GEE analyses. Statistical significance was set at
Results
Participant characteristics
This study included Japanese female athletes who participated in seven Olympic Games from the Beijing 2008 Summer Olympics to the Tokyo 2020 Summer Olympics (held in 2021). Table 1 shows the characteristics (mean ± SD) of the participants, including age, height, and weight, for each Games. While no significant difference in age was observed across the seven Games, significant differences were found in height (
Characteristics of the study participants.

A total of 954 athletes were eligible for inclusion in the analysis, of whom 168 were continuously using hormonal preparations (OCs or progestin-only preparations). Therefore, analyses of menstrual-related symptoms and menstrual cycle abnormalities were conducted in the remaining 786 athletes (635 Summer Olympians and 151 Winter Olympians; Beijing 2008:
Percentage of menstrual cycle abnormalities
Figure 1 illustrates the prevalence of menstrual cycle abnormalities at each Olympic Games. Overall, 22.5% of the 786 participants reported some form of menstrual cycle abnormality (primary amenorrhea: 1.8%; delayed menarche: 1.5%; secondary amenorrhea: 5.3%; irregular menstrual cycles: 13.9% (Supplemental Table 2). Fourteen athletes had not yet experienced menarche by age 15. The sports disciplines of the 68 athletes reporting amenorrhea (sum of primary, secondary, and delayed menarche) are shown in Table 2. A high proportion was observed in aesthetic and endurance sports, particularly in rhythmic gymnastics (66.7%,

Trends in the prevalence of menstrual cycle abnormalities among Japanese female Olympic athletes (2008–2020). The stacked bar chart illustrates the prevalence (%) of menstrual cycle abnormalities (primary amenorrhea, delayed menarche, secondary amenorrhea, and irregular menstrual cycles) among Japanese female Olympic athletes not using hormonal preparations at each Olympic Games (Beijing 2008, Vancouver 2010, London 2012, Sochi 2014, Rio 2016, PyeongChang 2018, and Tokyo 2020). Values within each segment indicate the specific prevalence (%) for that abnormality. Numbers in parentheses below the
Sports disciplines of amenorrheic athletes.

Temporal trend analysis using logistic regression models revealed that the prevalence of irregular menstrual cycles significantly increased across the seven Olympic Games (OR per cycle: 1.24, 95% CI: 1.13–1.37;
Prevalence of menstrual-related symptoms
Figure 2 shows the prevalence of menstrual-related symptoms among the 786 athletes not using hormonal preparations. Overall, 24.3% experienced dysmenorrhea, 66.9% reported PMS, and 9.2% reported HMB. PMS was the most prevalent symptom (Supplemental Table 4).

Trends in the prevalence of menstrual-related symptoms among Japanese female Olympic athletes (2008–2020). Line graphs show the trends in the prevalence (%) of dysmenorrhea (solid line, circle marker), PMS (dashed line, square marker), and HMB (dotted line, triangle marker) among Japanese female Olympic athletes not using hormonal preparations at each Olympic Games (Beijing 2008–Tokyo 2020). Numbers in parentheses below the
Temporal trend analysis using logistic regression models indicated no significant trends in the prevalence rates across the seven Games for PMS (
Prevalence of specific PMS symptoms
Trends in the prevalence of four specific PMS symptoms (irritability, edema, weight gain, and breast fullness) are presented in Figure 3. The overall prevalence rates among the 786 participants were highest for weight gain (38.8%), followed by irritability (34.4%), breast fullness (31.9%), and edema (23.7%; Supplemental Table 5).

Trends in the prevalence of specific PMS symptoms among Japanese female Olympic athletes (2008–2020). Line graphs show the trends in the prevalence (%) of specific PMS symptoms: (a) irritability, (b) edema, (c) weight gain, and (d) breast fullness among Japanese female Olympic athletes not using hormonal preparations at each Olympic Games (Beijing 2008–Tokyo 2020). Numbers in parentheses below the
Temporal trend analysis using logistic regression models showed a significant decreasing trend for breast fullness across the seven games (OR per cycle: 0.89, 95% CI: 0.83–0.95;
Rate of gynecological consultations
Figure 4 displays the trend in the gynecological consultation rate among all participating athletes (
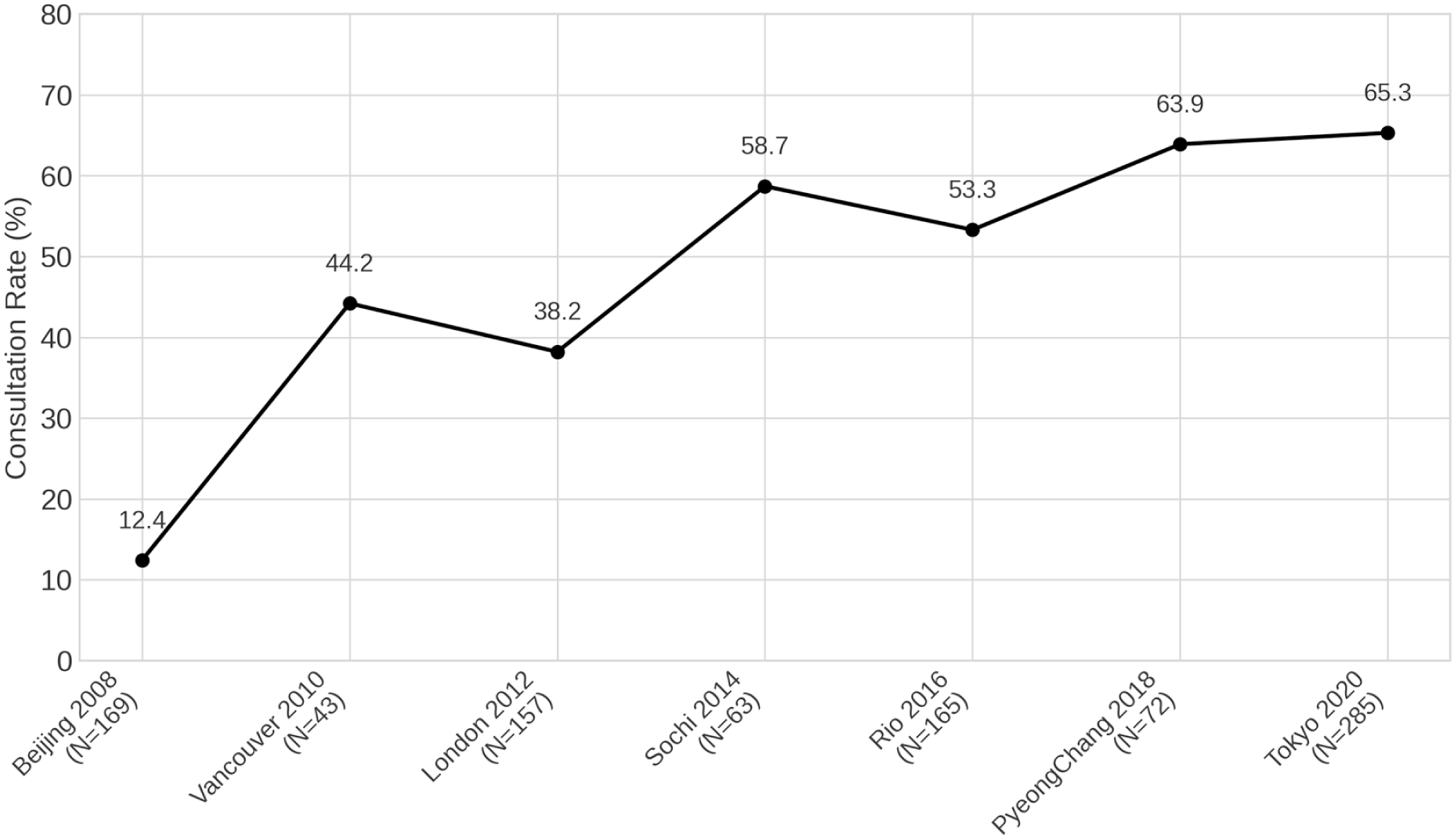
Gynecological consultation rate among Japanese female Olympic athletes competing in different Olympic Games (2008–2020). The line graph shows the trend in the gynecological consultation rate (%) among all participating Japanese female Olympic athletes (including users of hormonal preparations) at each Olympic Games (Beijing 2008–Tokyo 2020). Numbers in parentheses below the
Logistic regression analysis confirmed that this increasing trend was highly statistically significant (OR: 1.42, 95% CI: 1.33–1.52;
Discussion
We investigated changes in the prevalence of menstrual-related symptoms (i.e., dysmenorrhea, PMS, and HMB) and menstrual cycle abnormalities among female athletes with natural menstrual cycles who participated in the past seven Olympic Games. Our key findings revealed that the prevalence of menstrual cycle abnormalities, particularly irregular menstrual cycles, significantly increased over the 12-year period. Regarding menstrual-related symptoms, while the main analysis for dysmenorrhea showed a borderline trend, the sensitivity analysis accounting for repeated participation (GEE) revealed a significantly increasing trend. In contrast, no significant temporal trends were observed for PMS or HMB, although the prevalence of PMS remained consistently high. Among specific PMS symptoms, only breast fullness showed a significant decrease over time. Amenorrhea was most prevalent among athletes in aesthetic sports. Concurrently, the rate of gynecological consultations among all participating athletes increased dramatically during the study period.
Dysmenorrhea
The average prevalence of dysmenorrhea among athletes over the seven Olympic Games was 24.3%. While the main analysis showed a borderline trend (
Primary dysmenorrhea is believed to result from uterine contractions and ischemia induced by prostaglandin (PG) E2 and PGF2α. 30 High-intensity exercise may diminish PG secretion,2,18,30,33 potentially explaining the lower prevalence in athletes compared to non-athletes, although the exact mechanism remains unclear. In this study, dysmenorrhea was defined by the self-reported need to take painkillers “every time” for menstrual pain. While objective assessment of dysmenorrhea is challenging, 2 our findings indicate that approximately one in four athletes relied on painkillers, a rate that did not decrease despite the significant rise in gynecological consultations over the same period, as well as in the prevalence of hormonal preparations use. 34 This suggests that while self-management with analgesics may be common, underlying issues contributing to dysmenorrhea might persist, highlighting a continued need for awareness and access to effective medical interventions beyond simple pain relief.
Premenstrual syndrome
The prevalence of PMS among athletes varies widely in the literature (reported ranges 8.6–59.6%).
2
Our study found a high average prevalence among Japanese athletes (66.9%), which might be influenced by the diagnostic criteria used based on the JSOG definition
28
compared to other international standards. PMS can significantly impact athletes, potentially leading to alterations in training schedules
35
or changes in functions like postural control.35–37 Our analysis focused on four specific symptoms (irritability, weight gain, edema, and breast fullness). While the overall prevalence of PMS did not show a significant trend, we observed a significant decrease specifically in the reporting of breast fullness (
Heavy menstrual bleeding
The average prevalence of HMB in this study was 9.2%, falling within the wide range (3.4–42.1%) reported in a systematic review by Taim et al. 2 This finding was potentially affected by the subjective assessment method used. HMB is particularly relevant for endurance athletes due to the risk of iron deficiency anemia impacting oxygen transport capacity.2,40 However, research on HMB prevalence and its impact remains limited compared to dysmenorrhea and PMS, partly due to difficulties in objectively quantifying menstrual blood volume.2,41 Similar to the overall PMS prevalence, we found no significant temporal trend in HMB prevalence. Future studies utilizing objective tools like the Pictorial Blood Loss Assessment Chart2,41 could provide more accurate insights into HMB trends and impacts in elite athletes.
Menstrual cycle abnormalities
Compared to some international reports,
2
the overall prevalence of amenorrhea and oligomenorrhea in our cohort appeared relatively low initially. However, the most striking finding of our study was the significant increase in irregular menstrual cycles (OR per cycle: 1.24,
Female figure skaters have been shown to experience significantly greater physical pressure compared to male figure skaters, 47 and reports indicate they exhibit inappropriate attitudes and behaviors regarding eating habits and weight management. 48 This suggests that underlying sociocultural and historical factors may also contribute to worsening LEA. 48 This observed increase in menstrual cycle abnormalities likely reflects complex, interacting factors prevalent during the study period (2008–2020). Intensified training loads and trends toward early sport specialization may have increased the physiological stress and risk of LEA among elite athletes. 49 LEA is recognized as a primary driver of functional hypothalamic amenorrhea (FHA) and other menstrual dysfunctions by suppressing the hypothalamic–pituitary–ovarian (HPO) axis. 50 Recent research confirms that even short-term severe LEA can impair performance and alter physiological stress responses, such as increasing cortisol and oxidative stress, highlighting its profound impact. 51 The rise in menstrual abnormalities may thus signal an increasing prevalence or severity of LEA and its broader health implications encompassed by the REDs framework 13–15. 42
Furthermore, the study period coincided with the rapid rise of social media. The constant exposure to idealized athletic bodies on social media platforms can intensify social comparison and pressure for a certain physique, potentially contributing to disordered eating behaviors or intentional caloric restriction to achieve perceived performance benefits, thereby increasing LEA risk. 52 Psychological stress itself can also disrupt the HPO axis. 50 Therefore, the increasing trend in menstrual abnormalities might represent a physiological manifestation of the compounded physical and psychosocial stressors faced by modern elite female athletes. Addressing these abnormalities requires tackling not only energy balance but also the wider training and sociocultural environment. Continued screening for LEA/REDs using validated tools like the REDs Clinical Assessment Tool (REDs-CAT2) 50 and management by multidisciplinary teams remain essential. 15
It is crucial to recognize that the consequences of amenorrhea extend beyond bone health. Recent evidence highlights significant cardiometabolic risks associated with FHA. It has been reported that women with FHA exhibit impaired endothelial function, a marker of preclinical cardiovascular disease. 53 This is supported by a meta-analysis confirming that physically active women with amenorrhea have reduced flow-mediated dilation and unfavorable lipid profiles compared to eumenorrheic controls. 54 Mechanistically, LEA may increase oxidative stress, further compromising vascular health. 51 These findings underscore the importance of early intervention for menstrual cycle abnormalities to protect athletes’ long-term cardiovascular health.
Gynecological consultation rate
Another key finding was the dramatic and highly significant increase in the gynecological consultation rate among all participating athletes, rising from 12.4% in 2008 to 65.3% in 2020 (OR per cycle: 1.42,
Second, there may have been a reduction in the stigma surrounding menstruation and gynecological health in elite sports. Historically, athletes often did not seek help for issues like amenorrhea due to normalization of the condition (“a sign of hard training”), embarrassment, or prioritizing performance over health. 57 Increased public discourse, prominent athletes sharing their experiences, and a greater societal focus on women’s health may have helped dismantle these barriers. 57
Third, qualitative research indicates ongoing challenges and unmet needs regarding menstrual health support in elite settings, including athletes feeling that support is minimal or data collected is not effectively utilized, alongside communication barriers with staff. 58 The increased consultation rate might reflect athletes proactively seeking specialized care to address these needs, possibly facilitated by improved access to multidisciplinary support teams within the national training centers. Therefore, the rising consultation rate likely represents a convergence of factors: potentially increasing health issues (like irregular menstrual cycles), improved health literacy and awareness driving help-seeking, and possibly a more supportive environment enabling athletes to address previously neglected concerns.
Limitations
This study has several limitations that should be acknowledged when interpreting the results. First, although uniform criteria were applied for symptom evaluation, the self-report questionnaire used in this study was designed for clinical screening and has not been psychometrically validated. This reliance on self-reporting potentially introduces recall bias. However, the questions regarding menstrual status and symptoms were based on standard gynecological clinical practices. It was recently demonstrated that retrospective questionnaires tend to overestimate menstrual symptoms compared to daily monitoring via mobile applications. 59 To address this, future research should adopt prospective tracking methods using digital tools and adhere to the standardization proposed in the recent UEFA consensus statement on menstrual cycle tracking. 60 Second, the definitions relied on subjective assessments, such as reporting menstrual volume as “heavy” for HMB, and the use of painkillers (“every time”) for dysmenorrhea, the latter potentially influenced by factors other than pain severity, such as individual pain tolerance. Third, this study diagnosed PMS based on the definition established by the JSOG. Diagnostic criteria for PMS vary by country. For example, since these criteria differ from the diagnostic criteria proposed by the ACOG, this point must be considered when comparing prevalence rates between countries. In addition, the assessment of PMS symptoms was limited to four specific items (irritability, edema, weight gain, and breast fullness), which may not fully capture the full spectrum of symptoms associated with PMS. Fourth, while menstrual cycle abnormalities like amenorrhea and irregular menstrual cycles were identified, their underlying cause (e.g., LEA or other conditions) could not be definitively determined, as hormonal tests or ultrasonography were not part of the standard medical check-up protocol. Fifth, the study population consisted of elite Japanese female athletes, a particular group; therefore, caution should be exercised when generalizing these findings to athletes of different competitive levels, nationalities, or to the general female population. Sixth, as this was a retrospective analysis of existing medical records, no a priori sample size calculation was performed. Finally, this study did not assess menstrual health literacy, menstrual product use, or mobile application use for menstrual tracking. As these factors are essential for self-management and early detection of disorders, future research should examine them to develop more comprehensive support systems for female athletes.
Conclusion
Over the past 12 years, while the prevalence of PMS remained consistently high among elite Japanese female athletes not using hormonal preparations, the prevalence of irregular menstrual cycles significantly increased. Furthermore, accounting for repeated participation revealed a significantly increasing trend in dysmenorrhea. Concurrently, gynecological consultation rates rose dramatically among all athletes. These findings underscore the ongoing need for comprehensive gynecological support and education tailored to optimize the health and performance of female athletes. Enhancing athletes’ awareness of their own menstrual cycles and equipping them to recognize and communicate abnormalities to medical professionals remains crucial. Furthermore, coaches and support staff require improved understanding and open communication regarding menstruation to support athletes facing related issues effectively. Continued efforts in education and medical access are vital for addressing these persistent and emerging health challenges in elite female sports.
Supplemental Material
sj-pdf-1-whe-10.1177_17455057261440253 – Supplemental material for Changes in the prevalence of menstrual-related symptoms and menstrual cycle abnormalities among Japanese Summer and Winter Olympic athletes over the past seven Olympic Games
Supplemental material, sj-pdf-1-whe-10.1177_17455057261440253 for Changes in the prevalence of menstrual-related symptoms and menstrual cycle abnormalities among Japanese Summer and Winter Olympic athletes over the past seven Olympic Games by Sayaka Nose-Ogura, Rika Kawabe, Yuki Nakamura, Mariko Nakamura, Anna Tomori, Katsuyoshi Shirai and Kohei Nakajima in Women's Health
Footnotes
Acknowledgements
The authors would like to thank the athletes who participated in this study and the staff at the JISS for their support in data collection.
Ethical considerations
This study was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional Ethical Review Board of the Japan Institute of Sports Sciences (JISS) (approval No. 038; category: Medical Research Using Medical Information).
Consent to participate
Written informed consent was obtained from all athletes for the use of data provided in the questionnaires at the time of the medical check-up. For athletes under 18 years of age, instructions and consent forms were mailed in advance, and participants were required to bring a signed consent form from a parent or legal guardian on the day of the medical check-up.
Author contributions
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
