Abstract
Background:
Health-related quality of life (HRQoL) measures may help incorporate patient preferences and deliver individualized care for older breast cancer survivors. However, few studies have used clinically meaningful thresholds to evaluate the various characteristics associated with HRQoL in older women.
Objectives:
To examine sociodemographic and clinical characteristics associated with HRQoL and minimal clinically important differences (MCIDs) among female breast cancer survivors aged ⩾65 years.
Design:
A cross-sectional study.
Methods:
We used 2006–2015 Surveillance, Epidemiology, and End Results data linked to the Medicare Health Outcomes Survey (SEER-MHOS) for U.S. women aged ⩾65 years diagnosed with stages I–III invasive breast cancer. Descriptive statistics were used to summarize data. Generalized linear regression models were fitted to identify characteristics associated with the HRQoL physical component summary (PCS) and mental component summary (MCS) scores from the Veterans RAND 12-Item Health Survey scale. Missing values were imputed using chained equations. A threshold of two points was used to identify clinically meaningful between-group differences.
Results:
The median age at diagnosis was 72 years (interquartile range: 68–76) among 3218 breast cancer survivors. Obesity (mean difference (MD): −2.42; 95% confidence interval (CI): −3.34, −1.49), older age (⩾80 years; MD: −2.05; 95% CI: −3.15, −0.96), difficulty with ⩾1 activities of daily living (ADL; MD: −11.70; 95% CI: −12.42, −10.99), and cardiovascular (MD: −2.27; 95% CI: −3.02, −1.53) and musculoskeletal disease (MD: −3.88; 95% CI: −4.67, −3.09) were associated with clinically meaningful lower PCS scores. Less than high school education (MD: −3.43; 95% CI: −4.53, −2.33), annual household income ≤$19,999 (MD: −4.08; 95% CI: −6.00, −2.16) and $20,000–$39,999 (MD: −2.71; 95% CI: −4.63, −0.79), no surgery (MD: −3.03; 95% CI: −5.33, −0.72), difficulty with ≥1 ADL (MD: −6.71; 95% CI: −7.52, −5.89), and obesity (MD:2.06; 95% CI: 1.00, 3.12) were associated with MCIDs in MCS scores.
Conclusion:
Clinically meaningful differences in HRQoL were observed across sociodemographic and clinical characteristics in older female breast cancer survivors. These results may help identify women in need of interventions to improve HRQoL post-diagnosis.
Plain language summary
We studied more than 3,000 women in the U.S. who were breast cancer survivors aged 65 and above. This study was conducted to learn various clinical (e.g., stage at diagnosis, weight status) and personal (e.g., race and/or ethnicity, age) factors that may determine a breast cancer survivor’s quality of life after a breast cancer diagnosis. We found that increased weight, older age, difficulty with normal day-to-day activities, and having conditions related to heart disease and arthritis were associated with lower physical quality of life. Lower socioeconomic status (e.g., income), no surgery, and difficulty with normal day-to-day activities were associated with reduced mental quality of life. These findings show that personal and clinical factors could meaningfully change the quality of life of older breast cancer survivors. Our findings may help identify women who are in need of interventions to improve physical and mental quality of life after breast cancer diagnosis.
Keywords
Introduction
In the past three decades, the 5-year relative survival rate for breast cancer has increased from 57.3% to 92.0% in women aged 65 years and older.1,2 Despite these improvements in survival, older breast cancer survivors continue to report lower health-related quality of life (HRQoL)3,4 due to higher comorbidities, symptom burden, and decreased cognitive function.5–7 Moreover, financial strain associated with increasing cancer care costs could reduce both mental and physical quality of life among older breast cancer survivors.8,9
Currently, there are several HRQoL assessment tools available to describe the physical and mental health of women diagnosed with breast cancer.10–14 For example, the Functional Assessment of Cancer Therapy–Breast (FACT-B) instrument measures five domains of HRQoL (physical, functional, social/family, emotional, and breast cancer-related well-being). 10 The FACT-B instrument has been used to recommend therapeutic and home-based multi-dimensional interventions for breast cancer survivors.15,16 Moreover, studies of HRQoL often report a minimal clinically important difference (MCID) identifying the smallest difference in HRQoL scores perceived as beneficial (or harmful) by patients and/or clinicians which could warrant a change in patient management.17,18 Accordingly, even a small difference in HRQoL scores may represent clinically meaningful variations in the functional ability and/or mental well-being of individual patients.19,20 While several studies have identified individual characteristics associated with HRQoL among older breast cancer survivors, to our knowledge none have comprehensively evaluated these characteristics in relation to MCIDs.21,22
Studies on individual sociodemographic (e.g., age, race, ethnicity) and clinical (e.g., weight) characteristics associated with breast cancer outcomes could inform the development of individualized interventions for breast cancer prevention, screening, treatment, and survivorship.23–25 For example, current clinical guidelines recommend cessation of mammography screening in women aged ≥ 75 years due to increased risk of competing mortality and breast cancer over-detection with increasing age.26–29 However, currently over 50% of community-dwelling older women (aged ≥ 75) report screening within 2 years. 30 Therefore, clinicians have developed clinical decision tools to increase knowledge on the benefits and harms of mammography screening in older women.25,31 Furthermore, studies show that Black and Latina women are less likely to receive genetic counseling and testing services for hereditary breast and ovarian cancer in the United States. 32 These services are important as Black and Latina women show higher breast cancer mortality rates compared to non-Hispanic White women. 26 Therefore, culturally tailored interventions are currently being developed and tested to increase uptake of genetic counseling and testing among Black and Latina women in the United States.33,34 Similarly, clinical characteristics may also help inform the development of individualized clinical decision tools to support breast cancer survivorship care. 23 For example, decades of studies have linked physical inactivity and obesity to poorer survival among breast cancer survivors.35–37 This data has informed the development of individualized interventions to improve physical activity and health outcomes among breast cancer survivors.38–41
Similarly, information on the individual characteristics associated with HRQoL and MCIDs may help identify older breast cancer survivors in need of individualized interventions to improve HRQoL. 21 For example, lower HRQoL in women with multiple comorbidities, low income, education, or obesity could highlight the need to develop targeted interventions to improve HRQoL among those women.42–44 However, there is limited data on MCIDs in older breast cancer survivors. Therefore, the overarching goal of this study was to evaluate sociodemographic and clinical characteristics associated with HRQoL and MCIDs in older women (⩾65 years) diagnosed with breast cancer in the United States.
Methods
Data source and study population
Data was obtained from the National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) cancer registry data linked with the Centers for Medicare and Medicaid Services’ Medicare Health Outcomes Survey (MHOS) from 1998 to 2017. This is collectively known as the SEER-MHOS linked data resource. 45 SEER collects cancer incidence and survival data, as well as population-level demographics and tumor characteristics, from cancer registries representing approximately 48% of the U.S. population.45,46 MHOS is a randomly sampled longitudinal survey of Medicare Advantage enrollees to assess health plan effectiveness and quality of life.45,47 The analytic period was restricted to 2006 to 2015 as this was the most recent period of complete linked SEER-MHOS data including all the variables considered in our analysis. Consistent with prior research, we selected women with a diagnosis of invasive breast cancer from SEER who had completed at least one MHOS survey.22,48–50 Exclusion was made for those who were aged <65 years at the time of diagnosis, diagnosed with distant or in situ breast cancer,42,51–53 institutionalized, had end-stage renal disease, died within 6 months of the survey, or had missing data for physical component summary (PCS) score, mental component summary (MCS) score, or difficulties with activities of daily living (ADLs; Figure 1). The final sample included 3218 female breast cancer survivors aged ⩾65 years diagnosed with localized and regional breast cancer.

Inclusion and exclusion criteria.
Outcome variables
The outcome variables of interest were the t-standardized PCS and MCS scores from each individual’s first survey post breast cancer diagnosis. Consistent with previous studies, use of first survey after diagnosis ensures comparability across patients.22,49,50 These scores were provided from the Veterans RAND 12-Item Health Survey (VR-12) scale.54,55 The VR-12 uses eight scales measuring mental health, social functioning, bodily pain, vitality, general health perceptions, physical functioning, and role limitations due to emotional or physical health, to generate the component scores. 55 Higher scores reflect greater levels of physical and mental well-being. 56 This scale has been validated for use in older adults.57,58
Covariates
Sociodemographic characteristics
Sociodemographic characteristics included self-reported marital status (married, never married, divorced/separated/widowed), race and/or ethnicity (non-Hispanic American Indian/Alaskan Native, non-Hispanic Asian/Pacific Islander, non-Hispanic Black, Hispanic, other or multiracial, non-Hispanic White), education level (some college or more, high school, less than high school), annual household income ($0–$19,999, $20,000–$39,999, $40,000–$79,999, $80,000 or more), and smoking status (not at all, some days, every day). Non-Hispanic American Indian/Alaskan Native, Asian/Pacific Islander, and individuals reporting other or multiple races were used to create a single “other” category due to small sample sizes within each group. While these populations are demographically and culturally distinct, aggregation allowed for retention of participants and model convergence.
Clinical characteristics
Clinical characteristics included age at diagnosis (65–69, 70–74, 75–79, ⩾80 years), SEER historic stage (localized, regional), estrogen receptor (ER) status (negative, positive), receipt of surgery (none, lumpectomy, mastectomy, excision of biopsy site), year of diagnosis, and years from diagnosis to survey. We also included weight status measured using body mass index (underweight (<18.5 kg/m2), normal (18.5–24.9 kg/m2), overweight (25–29.9 kg/m2), obese (⩾30 kg/m2)), 59 difficulty with at least one ADL (yes/no), and comorbidities, including pulmonary disease, cardiovascular disease, hypertension, diabetes, and musculoskeletal disease (yes/no). Musculoskeletal disease was defined as having arthritis, sciatica, or osteoporosis.
Statistical analyses
Distributions of sociodemographic and clinical characteristics were summarized using descriptive statistics. Univariate analyses were conducted on PCS and MCS for sociodemographic and clinical characteristics. There were missing data for 12 of 17 variables (Table 1). Missing data were imputed using multiple imputation by chained equations (MICE), assuming missing at random. MICE generated missing values in 20 imputed datasets and provided pooled regression estimates. A least absolute shrinkage and selection operator (LASSO) was also used to assess model fit, overall accuracy, and variable selection for the final models.60,61 Covariates for final multivariable models were selected using previous literature and expert opinion.62,63 Generalized linear models, with an identity link function and normal distribution, for PCS and MCS scores included education level, race/ethnicity, annual household income, marital status, age at diagnosis, weight status, ER status, receipt of surgery, and difficulty with ⩾1 ADL, year of diagnosis, historical stage, time from diagnosis to survey, smoking status, and comorbidities. Pooled fit statistics were created for the PCS score (adjusted R2 = 0.40, root mean square error (RMSE) = 9.28, Akaike information criterion (AIC) = 7.31) and MCS score (adjusted R2 = 0.15, RMSE = 10.61, AIC = 7.58) imputation models.
Sample characteristics (n = 3218).

PI: Pacific Islander; AI: American Indian; AN: Alaska Native; SEER: Surveillance, Epidemiology, and End Results; IQR: interquartile range.
Categories were suppressed when the number of cases or events in any cell was less than 10 to reduce the likelihood of a breach of confidentiality.
Weight status represented by body mass index categories: normal (18.5–24.9 kg/m2), underweight (<18.5 kg/m2), overweight (25–29.9 kg/m2), obese (⩾30 kg/m2).
MCIDs were used to determine the smallest meaningful change in HRQoL useful for determining prognosis and symptom management. 64 Consistent with previous literature, a two-point difference in MCS or PCS scores was considered a minimally important difference in breast cancer survivors.65–67
Sensitivity analyses
Estimates from multiple imputations were compared to a complete case analysis. For the complete case analysis, individuals with missing data for covariates in the multivariable models (i.e., annual household income, marital status, education, smoking status, ER status, surgery type, weight status, cardiovascular disease, diabetes, pulmonary disease, hypertension, musculoskeletal disease) were excluded.
All analyses were conducted in Stata, version 18.0. 68 The National Institutes of Health Institutional Review Board approved this work, which was deemed exempt research due to use of de-identified pre-existing data. This research followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guidelines (Supplemental Table 1). 69
Results
The final sample included 3218 older breast cancer survivors (Figure 1). The sample had a median age of 72 years (interquartile range (IQR): 68–76) and a median time from breast cancer diagnosis to first MHOS survey of 5.9 years (IQR: 2.5–9.4; Table 1). Most women were non-Hispanic White (56.6%) and were divorced, separated, or widowed (56.6%). A higher proportion of survivors had some college education or more (41.1%) and an annual household income between $0 and $19,999 (34.3%). Most women reported not smoking at all (92.3%) and under half of women reported no difficulty with ⩾1 ADL (45.9%; Table 1).
The mean PCS and MCS scores across the sample were 36.4 (standard deviation (SD): 12.0), and 51.2 (SD: 11.5), respectively. Univariate analyses showed MCS and PCS scores were associated with race, education, income, age at diagnosis, weight status, difficulty with ⩾1 ADL, and cardiovascular and musculoskeletal disease (Supplemental Tables 2 and 3). In the univariate analyses, surgery type was only associated with MCS, while marital status was only associated with PCS.
Breast cancer survivors with older age (⩾80 years; mean difference (MD): −2.05; 95% confidence interval (CI): −3.15, −0.96), obesity (MD: −2.42.; 95% CI: −3.34, −1.49), difficulty with ⩾1 ADL (MD: −11.70; 95% CI: −12.42, −10.99), cardiovascular (MD: −2.27; 95% CI: −3.02, −1.53), and musculoskeletal disease (MD: −3.88; 95% CI: −4.67, −3.09) had a clinically meaningful lower PCS score compared to those with younger age (65–69 years), normal weight status, no difficulties with ADL, no cardiovascular, and no musculoskeletal disease (Figure 2). Statistically significant lower PCS scores were associated with older age (70–74, 75–79 years); however, these differences were not clinically meaningful.
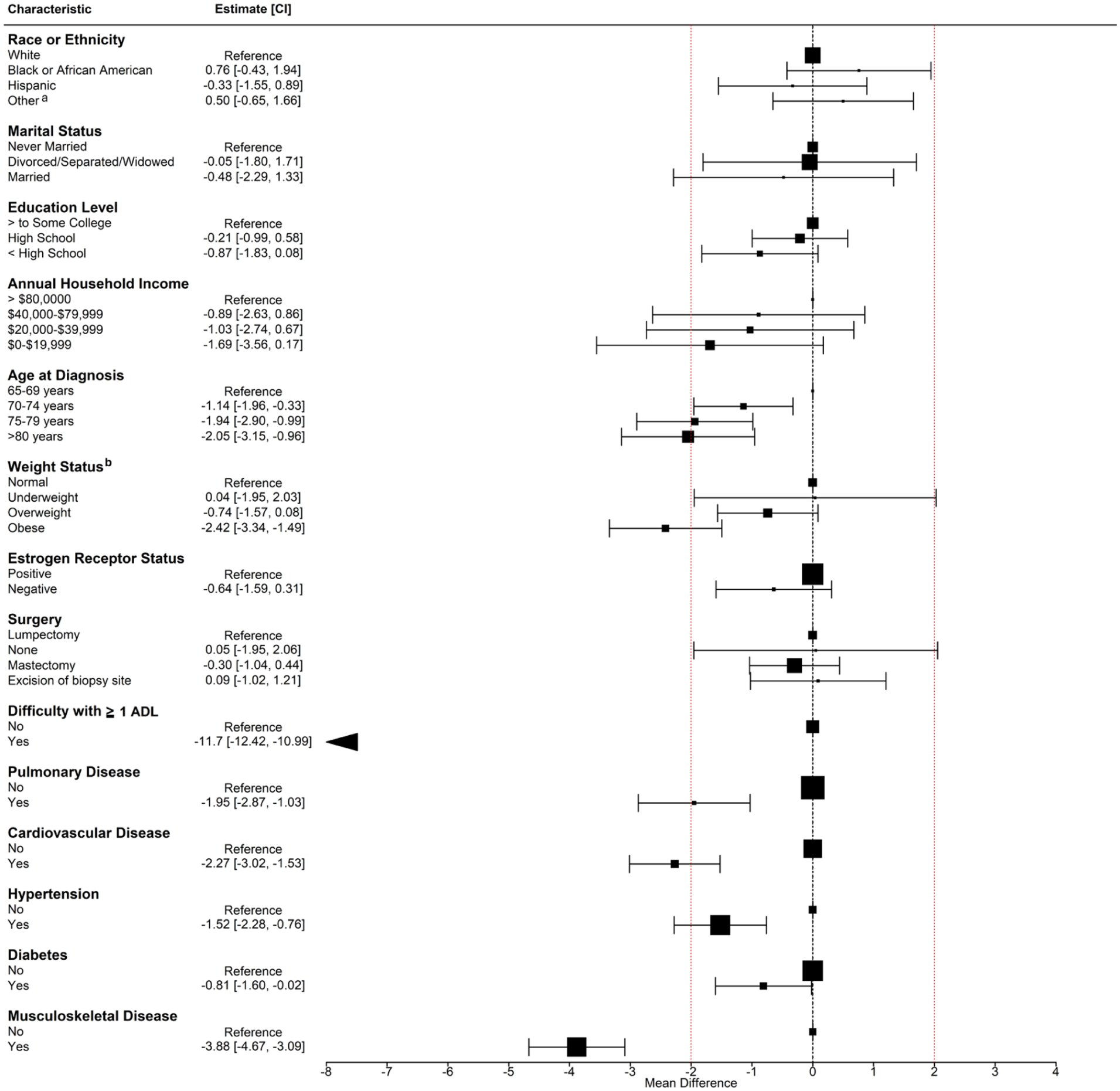
Sociodemographic and clinical characteristics associated with the physical component summary score for older women (⩾65 years) diagnosed with localized and regional breast cancer.
Breast cancer survivors who were married (MD: −2.25; 95% CI: −4.33, −0.17), or who had less than high school education (MD: −3.43; 95% CI: −4.53, −2.33), annual household income between $0 and $19,999 (MD: −4.08; 95% CI: −6.00, −2.16) or $20,000–$39,999 (MD: −2.71; 95% CI: −4.63, −0.79), no surgery (MD: −3.03; 95% CI: −5.33, −0.72), or difficulty with ⩾1 ADL (MD: −6.71; 95% CI: −7.52, −5.89) had a clinically meaningful lower MCS score compared to survivors who were never married, had some college education, income ⩾ $80,000, a lumpectomy, and no difficulties with ADL (Figure 3). Additional analysis showed that women who did not receive surgery were more likely to be aged ⩾80 years and/or report poorer general health compared to those who underwent surgery (Supplemental Table 4). Obesity (MD: 2.06; 95% CI: 1.00, 3.12) was associated with a clinically meaningful higher MCS score compared to normal weight status. Statistically significant lower MCS scores were associated with high school education and cardiovascular disease; however, the differences did not meet the clinically meaningful threshold.

Sociodemographic and clinical characteristics associated with the mental component summary score for older women (⩾65 years) diagnosed with localized and regional breast cancer.
Sensitivity analyses
The results for the PCS and MCS scores were similar across the multiple imputation and complete case analyses (Supplemental Tables 5 and 6).
Discussion
This is one of the first studies to evaluate sociodemographic and clinical characteristics associated with clinically meaningful differences in HRQoL in a nationally representative sample of older non-metastatic breast cancer survivors. Our results show obesity, older age at diagnosis, being married, having difficulty performing ADL, no surgery, cardiovascular and musculoskeletal disease, lower household income, and education were associated with clinically meaningful differences in HRQoL among older breast cancer survivors. Our findings are consistent with previous studies indicating that weight and age are associated with statistically significantly lower physical HRQoL.70,71 However, our findings extend prior studies in evaluating the individual characteristics associated with MCIDs in HRQoL in older women.
Our results show that older women (≥ 65 years) with obesity, difficulty engaging in at least one activity of daily living, cardiovascular, and musculoskeletal disease showed lower clinically meaningful physical components of HRQoL. Studies suggest that tailored exercise interventions for older women could help increase HRQoL among these women. 72 Furthermore, our results showed that women with lower income and education had clinically meaningful lower mental components of HRQoL. Previous studies have shown that worry about cost of healthcare, insurance coverage, and affordability of basic needs could increase stress, depression, and anxiety among cancer patients which could be reflected in the low MCS scores in our study.73,74 While further research is needed to better understand various factors contributing to lower MCS scores in older breast cancer survivors with low income and education, previous studies also support the development of interventions to mitigate financial toxicity in this population.75,76
Interestingly, we found that not receiving surgery was associated with a clinically meaningful lower mental HRQoL in comparison to those who received lumpectomy. These findings differ from previous studies showing that surgery is associated with lower HRQoL, potentially mediated through body image disturbance. 77 Upon additional analysis, we found that women who did not receive surgery were more likely to be aged ⩾80 years and/or report poorer general health. These differences could partially explain lower mental HRQoL in this group reflecting disease severity or terminal disease associated with older age and/or poor general health. Further research is warranted to understand the underlying characteristics contributing to higher HRQoL observed among older breast cancer survivors who received surgery.
Consistent with prior literature, obesity, cardiovascular disease, and musculoskeletal disease were statistically significantly associated with physical HRQoL among breast cancer survivors.78,79 Furthermore, in this study, we found these differences met the minimal clinically important threshold. Chronic inflammation, disability, and low perception of physical health associated with obesity may explain the lower HRQoL scores associated with obesity.80,81 Moreover, comorbidities such as cardiovascular and musculoskeletal disease may affect HRQoL through poor functional status, bodily pain, and/or fatigue.82–84 There are several tools that consider the relationship between comorbidities, weight status, and survival to support clinical care.85,86 However, currently there are no tools that incorporate the effects of clinical characteristics on HRQoL to guide clinical decisions for breast cancer survivors. Development of these tools may help integrate HRQoL measures into clinical care to meaningfully improve HRQoL among older breast cancer survivors.
Household income was associated with clinically meaningful differences in both mental and physical HRQoL. These results indicate that it is important for healthcare systems to address increased costs associated with cancer treatment. Survivors who experience a high financial strain may forego medical care, be unable to afford basic needs, and/or work longer hours, which can contribute to worsened HRQoL.87,88 We also found that less than high school education was associated with worse mental HRQoL compared to those who completed some college education. Educational attainment may lead to desirable occupational opportunities, career growth, increased social support and capital, and fewer reported symptoms of emotional distress.89,90 Therefore, consideration of patients’ education level, together with their economic status, may help clinicians account for the social and economic realities influencing HRQoL. In this regard, clinicians may consider tools like the COST (comprehensive score for financial toxicity) measure to assess and support early interventions related to financial burden.91,92
There are several strengths in this study. First, we conducted one of the first studies evaluating individual characteristics associated with clinically meaningful differences in HRQoL scores among older breast cancer survivors using a population-based dataset. Second, SEER-MHOS provided data from across the U.S. representing older (⩾65 years) breast cancer survivors seen in real-world settings, which may increase the generalizability of our findings to older breast cancer survivors enrolled in Medicare Advantage plans. Third, since SEER-MHOS includes data on other cancers (e.g., lung, prostate, colorectal), our findings may be replicated for different cancer sites. Finally, our study provides information for an understudied population of older breast cancer survivors (≥ 75) who are often excluded from clinical trials.93,94
Limitations
Our study presents several limitations. The number of women for non-Hispanic American Indian/Alaska Native women, other or multiracial women, and those living in rural areas were too small to provide quality of life estimates for these groups. 22 Future studies should consider using data sources that have larger sampling of these individuals. Additionally, this study focused on women aged ⩾65 years who were enrolled in Medicare Advantage programs, limiting generalizability of results to women with traditional Medicare fee-for-service coverage, younger breast cancer survivors, or women living in regions not covered by SEER. Although our analysis included localized treatment variables such as surgery and radiation, the absence of data on systemic therapies such as hormonal therapy, targeted therapy, or immunotherapy limited us from evaluating the full spectrum of treatments.
Conclusions
HRQoL and MCIDs could vary based on individual sociodemographic and clinical characteristics among older breast cancer survivors. These findings may help develop novel individualized interventions for older breast cancer survivors with lower socioeconomic characteristics, obesity, lower functional status, and comorbidities such as cardiovascular and musculoskeletal disease. However, further research is needed to inform the development and implementation of these interventions in clinical settings.
Supplemental Material
sj-docx-1-whe-10.1177_17455057261432623 – Supplemental material for Clinical and sociodemographic characteristics associated with health-related quality of life among older breast cancer survivors
Supplemental material, sj-docx-1-whe-10.1177_17455057261432623 for Clinical and sociodemographic characteristics associated with health-related quality of life among older breast cancer survivors by Jacob Schneider, Kaitlyn M. Wojcik, Emma Tian, Oliver W. A. Wilson and Jinani Jayasekera in Women's Health
Supplemental Material
sj-docx-2-whe-10.1177_17455057261432623 – Supplemental material for Clinical and sociodemographic characteristics associated with health-related quality of life among older breast cancer survivors
Supplemental material, sj-docx-2-whe-10.1177_17455057261432623 for Clinical and sociodemographic characteristics associated with health-related quality of life among older breast cancer survivors by Jacob Schneider, Kaitlyn M. Wojcik, Emma Tian, Oliver W. A. Wilson and Jinani Jayasekera in Women's Health
Footnotes
Acknowledgements
We thank Dr. Roxxane Jensen for her support in providing methodological and design consultation on the use of the SEER-MHOS linked dataset.
Ethical considerations
This study was approved by the National Institutes of Health Institutional Review Board and was considered exempt research based on use of de-identified pre-existing data.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Division of Intramural Research at the National Institute on Minority Health and Health Disparities of the National Institutes of Health (ZIA MD000022, PI Jayasekera). The contributions of the NIH authors were made as part of their official duties as NIH federal employees, are in compliance with agency policy requirements, and are considered Works of the United States Government. However, the findings and conclusions presented in this article are those of the authors and do not necessarily reflect the views of the NIH or the U.S. Department of Health and Human Services. This study used data from the SEER-MHOS linked data resource. The interpretation and reporting of these data are the sole responsibility of the authors. The authors acknowledge the efforts of the National Cancer Institute; the Centers for Medicare & Medicaid Services; Information Management Services (IMS), Inc.; and the Surveillance, Epidemiology, and End Results (SEER) Program tumor registries in the creation of the SEER-MHOS database.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The SEER-MHOS datasets analyzed during the current study are not publicly available due to the risk of re-identification. The data are available to investigators for research purposes, but approval is required to obtain the data.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
