Abstract
Background:
Dysmenorrhoea, a common gynaecological issue in women, is characterised by lower abdominal cramps during menstruation impacting quality of life (QoL).
Objectives:
This study evaluates the effectiveness, utilisation and safety of Buscopan® Plus (hyoscine butylbromide (HBB) Plus: 10 mg HBB + 500 mg paracetamol) for self-treating menstrual pain.
Design:
This non-interventional, prospective cohort real-world study included women with menstrual pain registered with Carenity, an online patient platform by Else Care SAS.
Methods:
Eligible German patients with the mean (standard deviation) age of 34.0 (8.0) years were enrolled between 15 September 2020 and 15 June 2022 and grouped into primary analysis or safety cohorts. A maximum of one to two tablets (thrice daily) were allowed, limited to 3 g paracetamol per day. Primary outcomes were pain intensity difference by mean change and percentage variation in numeric rating scale (NRS) score, percentage of NRS responders and time to pain relief; safety was a secondary outcome. All parameters were measured at 15 min, 30 min, 1 h, or 4 h following the first HBB Plus intake.
Results:
Of 1733 eligible women, 94% provided written consent and completed the baseline questionnaire, 56% met all inclusion criteria, 10% completed all intake assessments (primary analysis cohort) and 33% had ⩾1 dose of HBB Plus (safety cohort). Percent decrease in NRS score from baseline was 11.1%, 29.5%, 42.4%, 53.4% and 65.8% at 15 min, 30 min, 45 min, 1 h and 4 h (p < 0.001 at all-time points), respectively. Overall, 90.8% patients were categorised as responders up to 4 h post HBB Plus intake. Median time to first perceptible pain relief and meaningful pain relief was 30.0 and 37.5 min, respectively. In the safety analysis cohort, five non-serious treatment-emergent adverse events were reported.
Conclusion:
HBB Plus effectively reduced menstrual pain within 4 h of treatment and was well-tolerated, providing a viable option for self-management of pain and improving QoL.
Plain Language Summary
Period pain, also called menstrual cramps, affects many women and often goes unnoticed or untreated. Our study examined how effective and safe Buscopan® Plus (which contains hyoscine butylbromide [HBB] and paracetamol) is in everyday use. The study included women in Germany, with an average age of 34, who experienced menstrual pain. They first filled out a questionnaire and then took HBB Plus tablets (1–2 tablets up to three times a day) when they had cramps. We found that 91% of the women felt pain relief within four hours, with the first signs of relief starting after about 30 minutes. On average, noticeable pain relief was felt after 38 minutes. Pain levels steadily decreased at each time point measured—15 minutes, 30 minutes, 45 minutes, 1 hour and 4 hours after taking the tablets. Among women monitored for safety, the medicine was well tolerated, with only a few mild side effects. These results suggest that HBB Plus may be a helpful self-care option for women with menstrual pain, allowing them to return to their daily activities with much less discomfort.
Introduction
Dysmenorrhoea is the medical term used to describe menstrual cramps or period pain and is caused by the contraction of the uterus as it sheds the endometrium. This process is driven by rising prostaglandin levels, resulting in increasing uterine contraction and pain. Menstrual pain is experienced in the pelvis and may radiate into the lower back and thighs; this typically starts a few days before menstruation and persists for 48–72 h. Inhibition of prostaglandin production reduces the intensity of uterine contraction and, consequently, the pain experienced.1,2 Factors such as women’s age, number of childbirths (parity) and the use of oral contraceptives (OCs) decrease dysmenorrhoea, whereas high stress increases the risk of dysmenorrhoea. 3 Dysmenorrhoea is classified into two categories: primary dysmenorrhoea (PD), which is not associated with any organic disease, and secondary dysmenorrhoea, which is associated with an identifiable condition. 4
Common symptoms of dysmenorrhoea in adolescent women include pain or cramping sensations in the lower pelvis accompanied by headaches, dizziness, diarrhoea, bloating, nausea and vomiting, backache and leg pain. 5 Although dysmenorrhoea is a common disorder in adult women, it remains unidentified and undertreated. Dysmenorrhoea disturbs normal daily routine, causing absenteeism from school or work and reduced productivity, thereby affecting the overall quality of life (QoL) of females.2,6 The prevalence of PD in menstruating women is found to be between 45% and 95%. 7 Severe dysmenorrhoea or pain that significantly limits their daily activities is experienced by 2%—28% of women. 7 Most guidelines recommend non-steroidal anti-inflammatory drugs (NSAIDs) as the first-line of treatment.2,8 –10
A Cochrane review of 73 randomised controlled trials (RCTs) established strong evidence to support administration of NSAIDs as first-line treatment for PD9,11 including the common painkillers, aspirin, naproxen, ibuprofen and mefenamic acid. 12 However, a review of 51 clinical trials indicated that 18% of women with dysmenorrhoea showed minimal or no relief using NSAIDs.12,13 Long-term use and higher doses of NSAIDS have been associated with increased frequency of side effects. 14 Administration of OCs lacks robust evidence from high-quality RCTs for menstrual pain management, and thus it is often avoided due to the risk of hormonal imbalance. 9 This lack of evidence underscores the necessity to explore alternative therapies for dysmenorrhoea.
Alternative drug therapies include the use of antispasmodics such as hyoscine butylbromide (HBB) or a combination with analgesics. HBB, first registered in Germany in 1951, is an anticholinergic compound 15 that targets muscarinic receptors in smooth muscles to prevent their contraction. More than 60% of women self-treat dysmenorrhoea4,16,17 with over-the-counter (OTC) drugs, such as paracetamol, NSAIDs and HBB, or their combinations.16 –18 Paracetamol is a simple analgesic used for a multitude of pain conditions. It has a complex mechanism of action that is not fully understood. It is known to inhibit the action of the COX enzyme group with a reduction of prostaglandin synthesis. To this effect, it has a similar mechanism of action to the NSAID group, without many of the adverse side effects. 19
HBB is available as both a prescription and an OTC drug in several countries. 15 It is an anti-muscarinic drug used to treat smooth muscle spasm. Its use reduces the contraction of the uterus. Consequently, the combination of paracetamol and HBB will synergistically reduce uterine contraction and pain in a complementary way/manner.20,21 However, antispasmodics can cause side effects such as nausea, dry mouth, itchy rashes, dizziness, breathing problems, headache and increased pulse rate. 22 Therefore, combination therapy of antispasmodics with NSAIDs or analgesics is recommended for dysmenorrhoea when monotherapy is ineffective. 22 These fixed-dose combinations provide improved therapeutic effects and better patient adherence, while minimising adverse reactions from individual drugs. 22
Although data support effective and safe use of HBB for treating spasmodic genitourinary pain, 23 research on its use for menstrual pain is limited. Moreover, comparative studies on the effectiveness and tolerability of HBB for genitourinary and menstrual pain are insufficient. Owing to the availability of OTC drugs, RCTs may have limited real-world applicability. Moreover, the efficacy reported in various RCTs on OTC medications might not translate to the effectiveness that is observed in a real-world setting. Therefore, the data derived from the real-world evidence (RWE) studies are used to complement results of RCT studies to get insights about different parameters like the effectiveness of the drug, safety of the drug and generalisability beyond controlled environments in order to develop an evidence-based medicine.24,25 Hence, new data are needed to support the use of HBB in dysmenorrhoea.15,26 In recent years, assessment of patients’ experience of disease or treatment through patient-reported outcomes (PROs) has been increasingly utilised in clinical trial settings and for routine care of several health conditions, including pain.27,28 A patient survey was conducted in Germany involving 314 women (mean age: 32.3 years) with dysmenorrhoea who reported pain and cramps with an intensity of 7.45 ± 2.13 on a 0–10 Likert scale; this intensity which was reduced to 2.86 ± 1.81 upon treatment with a combination of HBB and paracetamol, also known as PLUS (HBB Plus). 20 This fixed-dose medication has received approval for the treatment of dysmenorrhoea across 10 countries—Austria, Chile, Germany, Greece, Indonesia, Italy, Korea, Philippines, Portugal and Turkey. 20
In this study, we aimed to describe the effectiveness, utilisation and safety profile of Buscopan® Plus tablet, a fixed-dose combination of HBB and paracetamol (10 mg HBB + 500 mg paracetamol), hereafter referred to as HBB Plus, in self-treating menstrual pain using self-reported survey data. Since 1969, the widely administered dosage for HBB has been 10 mg and for paracetamol, 500 mg. However, as dosage optimisation studies for individual drugs in combination products were not required during that era, there is no available information on how these doses were selected. 29 Therefore, this study considers the current combination to examine patients with dysmenorrhoea in real-world settings to broaden existing research on the real-world self-treatment patterns of dysmenorrhoea with HBB.
Methods
Study design
This non-interventional, prospective cohort study was conducted among adult women with menstrual pain registered with the German online patient platform, Carenity, generated by Else Care SAS. Eligible patients were recruited from Carenity between 15 September 2020 and 15 June 2022 (inclusive dates). The study period for each patient began from the time of enrolment and all the enrolled patients were required to complete a baseline assessment questionnaire prior to treatment with HBB Plus. This was followed by post-treatment assessments at 15 min, 30 min, 45 min, 1 h and 4 h following the first HBB Plus intake to assess the effectiveness of HBB Plus oral tablets over time. Patients were also required to complete additional evening assessments at the first, second and last evenings following HBB Plus treatment. The period of exposure to HBB Plus began at the first HBB Plus intake (occurring within 2 months of first online contact) until 2 days following the last HBB Plus intake. The end of the study period was defined as 2 days following the last HBB Plus intake or date of last completed assessment, whichever was earlier.
All patients provided written informed consent, and the study was conducted in accordance with the guidelines for Good Pharmacoepidemiology Practice defined by the International Epidemiological Association, the ethical principles that have their origins in the Declaration of Helsinki and the French legal and regulatory obligations. All data was handled in strict confidentiality in compliance with the European regulation applicable since 25 May 2018, on data protection and privacy (General Data Protection Regulation no 2016/679). Reporting followed the STrengthening the Reporting of Observational Studies in Epidemiology (STROBE). 30
Study population
Females German residents, aged ⩾18 years, with a history of menstrual pain, who reported the use or intention to use HBB Plus oral tablets for self-treating menstrual pain and who provided written informed consent were included in the study. Patients were allowed to take one to two tablets at a time as required (for a maximum of three times a day, 31 while staying within the dosage limit of 3 g paracetamol per day. Patients who were involved in an interventional clinical trial, were pregnant, or had reached menopause were excluded from the study. All the enrolled patients were grouped in the following cohorts for analysis:
Primary analysis cohort
Patients who completed all the assessments (including the numeric rating scale (NRS) score) for up to 4 h following the first HBB Plus intake were grouped under this cohort. Patients were tagged as completers if they had completed all assessments up to the last evening following HBB Plus intake (including assessments on the first evening after intake, second evening after intake and last evening after 2 days with no HBB Plus intake).
Safety cohort
This cohort included all patients in the study sample who had taken at least one dose of HBB Plus oral tablets. This cohort was used for the analysis of adverse events and other safety-related outcomes.
Measurements
Validated tools
The validated questionnaires included questions to quantify the pain level among patients and its intensity using the NRS score 32 as well as the subjective intensity of pain and the impairment caused by pain using the brief pain inventory-short form (BPI-SF)33 –35 on a scale of 0–10 (0 = no pain; 1–4 = mild; 5–6 = moderate; 7–10 = severe). An overall assessment of the treatment response of the patients was measured using the global perception of improvement (GPI) scale of 1–5 (1 = much better, 2 = better, 3 = about the same, 4 = worse and 5 = much worse). Global satisfaction scores for the effectiveness of HBB Plus were determined at the end of the first intake and at the end of the study (1 = very satisfied, 2 = satisfied, 3 = neither satisfied nor dissatisfied, 4 = dissatisfied and 5 = very dissatisfied). 36
Outcomes
Primary outcome
Pain intensity difference was measured as mean change in NRS score and percentage variation in NRS score between baseline and follow-up points, that is, 15 min, 30 min, 45 min, 1 h and 4 h after the first dose.
The effectiveness (mean change and percentage variation in NRS score between baseline and at each post-treatment assessment) was measured by the decrease in the number of patients who had completed NRS score responses at each assessed time point following the first HBB Plus intake from those who did not take any medication for menstrual pain other than HBB Plus. A decrease of 30% from baseline was considered meaningful.
The percentage of NRS responders was defined as the percentage of patients who reported using only HBB Plus to manage menstrual pain and showed ⩾30% decrease in NRS-measured menstrual pain at each follow-up point from baseline. 37
Time to pain relief
Time to first perceptible relief was defined as the time elapsed between baseline (t = 0) till the time when the patient experienced perceptible pain relief after HBB Plus intake, while the time to meaningful pain relief was defined as the time elapsed from t = 0 till the time when the patient experienced meaningful relief (defined as ⩾30% decrease of NRS compared with baseline) after HBB Plus intake. Both parameters were measured at 15 min, 30 min, 1 h, or 4 h following the first HBB Plus intake. 37
Secondary outcomes
Utilisation of HBB Plus was assessed based on dosage (per assessment, cumulative dose per day and cumulative dose per menstrual period), number of intakes (per day and per menstrual period) and duration of HBB Plus exposure during the menstrual period (number of days and percentage of days during menses with HBB Plus exposure).
Safety
Drug safety was assessed by the number of non-serious and serious treatment-emergent adverse events (TEAEs) occurring during the study period. Serious TEAEs were defined as those resulting in an emergency room (ER) visit and/or hospitalisation.
Perception
Perception was assessed by patients’ (1) GPI scores, (2) patient satisfaction with the effectiveness of HBB Plus and (3) intention to recommend HBB Plus.
Questionnaire data
The questionnaire data assessing study variables and time periods of collection included the following information (Supplemental Table S1):
Demographics/selection criteria: collected via the baseline questionnaire at the time of first online contact.
Menstruation and pain history: collected via baseline questionnaire at the time of first online contact.
HBB Plus treatment history: collected via baseline questionnaire at the time of first online contact.
Current menstruation and treatment utilisation: collected via baseline questionnaire up to 4 h following intake, evening assessments and end-of-study assessment.
Pain symptoms and intensity (NRS score, BPI score): collected via baseline questionnaire up to 4 h following intake, evening assessments and end-of-study assessment.
Perception of HBB Plus treatment (safety, satisfaction, recommendation to others): determined at 4 h following intake, evening assessments and end-of-study assessment.
Statistical analysis
Continuous variables were summarised using descriptive statistics including number of observations (n), mean, standard deviation (SD), median, minimum, Q1, Q3 and maximum value. The 95% confidence intervals (CIs) were presented for all continuous variables. No missing values were imputed for analysis.
Sample size
In a two-sided comparison test using 95% power to detect a minimum difference in NRS scores of 1.0 among patients, a target sample size of 98 patients was determined for primary analysis, considering a population standardised difference of 2.3. Considering that approximately 20% of the patients enrolled would not be eligible for the primary analysis population (assuming that patients would not complete all post-treatment assessments up to 4 h following HBB Plus intake), the recruitment target was set to 123 patients to achieve the power necessary to detect a statistically significant difference in NRS scores. Patients who met all selection criteria and completed all post-treatment assessments up to 4 h were included in the primary analysis cohort until a total of 98 patients were confirmed.
Endpoints
The primary endpoints of mean change in NRS score, percentage variation and BPI scores were presented as continuous variables at baseline and at each post-treatment assessment time point. Variation was observed both overall and by NRS pain intensity level indicated at baseline (mild, moderate and severe). The number and percentage of NRS responders, both overall and by NRS pain intensity level (mild, moderate and severe), as reported at baseline, were presented. The BPI-SF values for pain severity and interference at first intake for NRS responders and non-responders were displayed as continuous variables. To determine the statistically significant difference, a Wilcoxon test with Benjamini-Hochberg type 1 correction was performed; 95% CIs and p-values for mean change between baseline and each post-treatment assessment were presented. Time to first perceptible pain relief and time to meaningful pain relief were presented as continuous variables, stratified by (1) reported use of a pain treatment other than HBB Plus, (2) HBB Plus dose and (3) NRS pain intensity level (mild, moderate and severe) reported at baseline; 95% CIs for means were also presented.
The secondary endpoints measured within the primary study cohort were utilisation-related variables described as both continuous and categorical. Safety variables were described as categorical among the safety cohort. Ordinal logistic regression models were used to determine predictors of GPI scores during the first and last evening assessments. Odds ratios, 95% CIs and p-values were provided for each covariate in the model.
Sensitivity analysis
A mixed model for repeated measures was used to assess the estimation of the mean change in NRS score between baseline and post-treatment assessment. A backward stepwise procedure was used to select covariates significantly and independently associated with NRS score for inclusion in the model. The number of patients included at each covariate level, the least squares mean and 95% CI, model coefficient and p-value were presented for each covariate included in the model.
Results
Baseline characteristics
Patient disposition and characteristics at baseline
A total of 1733 eligible women initiated online contact with the Carenity platform between 15 September 2020 and 15 June 2022. Of these women, 1635/1733 (94%) provided written informed consent and completed the baseline questionnaire, and 972/1635 (56%) met all the inclusion criteria. Among these, 325/972 (33%) patients who had at least one dose of the HBB Plus were included in the safety analysis cohort, 267/972 (27%) patients who had taken at least one dose of HBB Plus oral tablets and having baseline and at least one other assessment available were included in the sensitivity analysis cohort, and 98/972 (10%) who completed all intake assessments (including NRS score at baseline and up to 4 h following first HBB Plus intake) were included in the primary analysis cohort (Supplemental Figure S1).
Among the total enrolled patients, the mean (SD) age at enrolment was 32.7 (7.0) years and in the primary analysis cohort, the mean (SD) age at enrolment was 34.0 (8.0) years. The baseline characteristics of the patients, including age, menstruation, menstrual pain and treatment history are given in Table 1. Additional details on baseline characteristics of patients, overall and cohort wise are given in Supplemental Table S2.
Baseline characteristics of patients in the enrolled, primary analysis, safety and sensitivity analysis cohorts.

N represents the population with available data for a given variable. HBB: hyoscine butylbromide; NSAID: non-steroidal anti-inflammatory drug; SD: standard deviation.
A patient could have multiple answers, in which case the top 3 combinations were described.
In the primary cohort, more than half of the patients (68 (69.4%)) reported experiencing their first menstrual cycle between the ages of 11 and 13 years. The mean (SD) duration of the menstrual cycle reported by patients was 26.7 (3.4) days. Around 56 (57.1%) patients reported experiencing a menstrual cycle lasting between 26 and 30 days. Patients reported a mean (SD) of 3.4 (1.6) days of pain during menstruation and around 45.9% and 49.0% reported pain occurring a few days prior to the occurrence of menstruation and on the first day of menses, respectively. The majority of the patients (88.5%) used only one medication to treat their menstrual pain. Most patients (62.2%) used medications other than HBB Plus in the past to treat their menstrual pain. A quarter of the patients (26.2%) received NSAIDs such as naproxen, while 45.9% received other NSAIDs (Table 1).
Treatment history with HBB Plus
The majority of the patients in the primary analysis cohort (n/N = 76/98, 76%) reported past and continuous use of HBB Plus for self-treating menstrual pain. HBB Plus was never taken for menstrual pain management by 18 (18.4%) patients and was stopped by 4 (4.1%) patients prior to entering the study. Among those who reinitiated use of the drug, 26 (34.2%) did so on the suggestion of a specialist doctor, 22 (28.9%) on the suggestion of a pharmacist, 21 (27.6%) on the suggestion from relatives, while 17 (22.4%) made independent decisions. Most patients (84.2%) reported that HBB Plus was always their first-line treatment for menstrual pain. The most common reasons for use of the drug were quick menstrual pain relief (84.2%), satisfaction with effectiveness (69.7%) and doctor instructions (24.1%). Almost all patients reported taking up to four tablets per day during menstruation, with most patients reporting taking two tablets per day (42.1%; Table 2).
Treatment history with HBB Plus.

N represents the population with available data for a given variable. HBB: hyoscine butylbromide.
A patient could have multiple answers, in which case the top 3 combinations were described.
Dysmenorrhoea symptoms and use of concomitant medications at baseline
In the primary analysis cohort, most patients (95.9%) reported at least one dysmenorrhoea symptom at baseline prior to taking HBB Plus, commonly experienced being the low back pain (70.2%), followed by headache (50.0%), diarrhoea and/or constipation (47.9%) and fatigue/weakness (44.7%). The number of symptoms (mean (SD)) reported at baseline were 3.9 (2.0) and the most frequent combination of symptoms was pain, digestive symptoms and other general symptoms (38.8%; Table 3). Nearly all patients (96.9%) reported that they had not used any treatment other than HBB Plus for pain within the past 24 h.
Pain symptoms and use of concomitant medication at baseline for the primary analysis and sensitivity analysis populations.

N represents the population with available data for a given variable. CI: confidence interval; HBB: hyoscine butylbromide; Max: maximum; Min: minimum; Q1: first quartile; Q3: third quartile; SD: standard deviation.
A patient could have multiple answers, in which case the top 3 combinations are described.
Pain includes lower back pain, headache, pain in the legs and breast pain.
In the primary analysis cohort, patients reported a mean (SD) BPI score of 6.5 (1.9) for pain at its worst corresponding to “severe pain” (indicated by BPI scores of 7–10; Table 4). Patients reported overall mean (SD) BPI scores of 2.2 (2.2) indicating “mild pain” for pain at its least, 5.1 (1.7) indicating “moderate pain” for pain at its average, and 6.4 (1.7) indicating “moderate pain” for pain in the moment (Supplemental Table S3). The mean (SD) composite BPI score (mean score for pain at its worst, least, average and at the moment 38 was 5.0 (1.6), which corresponded to “moderate pain.” At baseline, no patient reported the score of 0 for pain, indicating “no pain” on the pain intensity scale (NRS) prior to treatment with HBB Plus. However, 6.1% patients reported the score of 1–3 indication “mild pain.”
Pain severity at baseline for the primary analysis and sensitivity analysis populations.
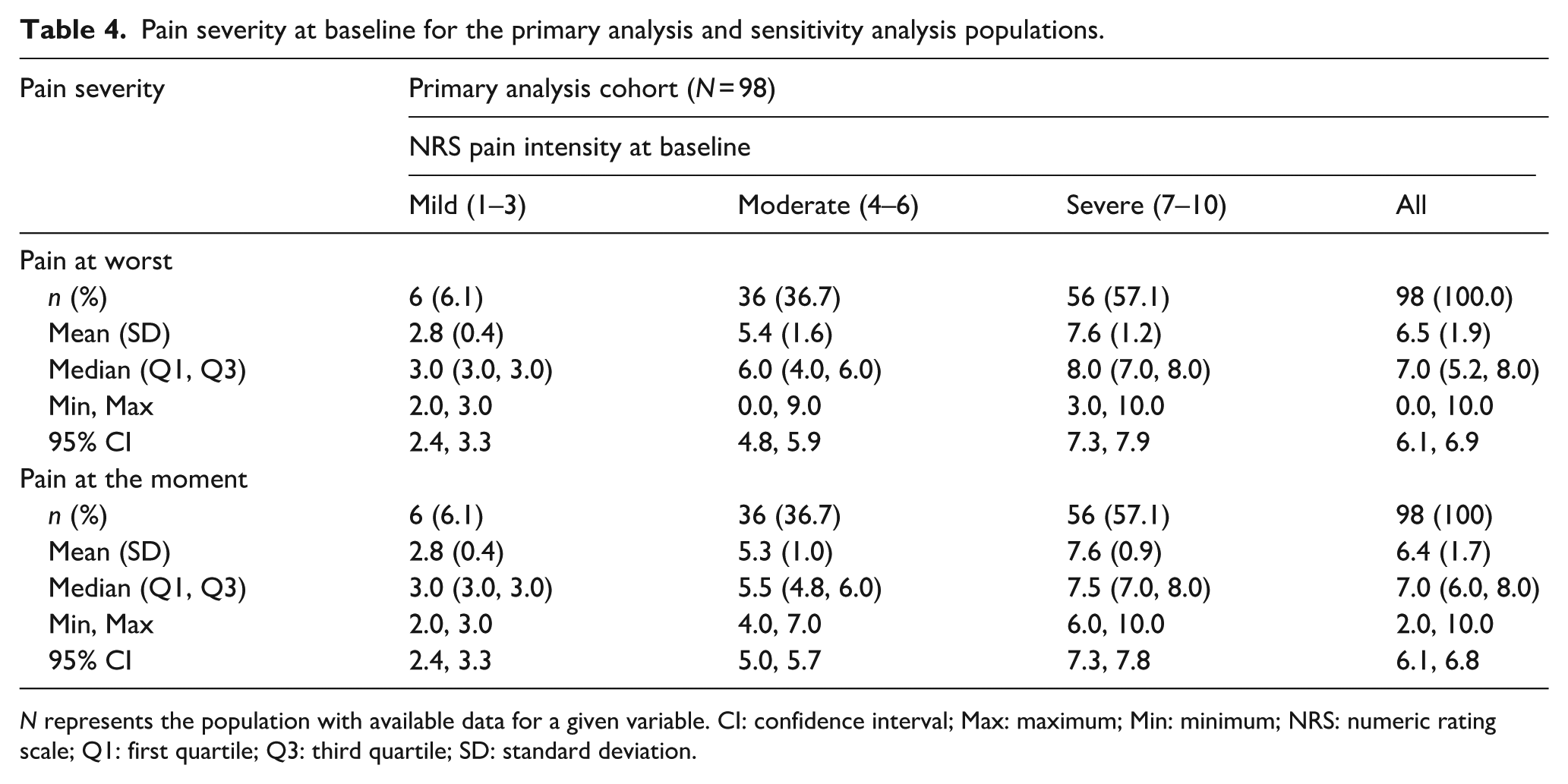
N represents the population with available data for a given variable. CI: confidence interval; Max: maximum; Min: minimum; NRS: numeric rating scale; Q1: first quartile; Q3: third quartile; SD: standard deviation.
Study outcomes
Pain intensity difference
After treatment with HBB Plus, the NRS score decreased from the baseline at 15 min (5.7 (1.8)), at 30 min (4.5 (2.0)), at 45 min (3.6 (2.1)), at 1 h (2.9 (2.0)) and at 4 h (2.2 (2.0)). A significant mean (SD) change in pain NRS score from the baseline observed were at 15 min (−0.8 (1.2); p < 0.001), at 30 min (−2.0 (1.8); p < 0.001), at 45 min (−2.9 (2.3); p < 0.001), at 1 h (−3.6 (2.3); p < 0.001) and at 4 h (−4.3 (2.3); p < 0.001; Figure 1 and Supplemental Table S3). The respective percentage mean (SD) change from baseline is given in Supplemental Table S3. Sensitivity analysis showed similar result indicating significant decrease in pain NRS score from baseline with increasing time from treatment intake—at 15 min (least squares [LS] mean (standard error): −1.2 (0.5); p < 0.001), 30 min (−2.4 (0.5); p < 0.001), 45 min (−3.3 (0.5); p < 0.001), 1 h (−4.0 (0.5); p < 0.001) and 4 h (−4.7 (0.5); p < 0.001).

Mean change variation in the NRS score between baseline and each post-treatment assessment.
Based on pain severity at baseline (mild, moderate and severe), the mean percentage variation in NRS-measured for menstrual pain was observed to have increased over time from 15 min to 4 h post-treatment. At 15 min following treatment, the mean (SD) percentage variation in pain between baseline and post-treatment assessment were similar across pain severity levels (mild: −13.9% (22.2%); moderate: −11.5% (16.2%) and severe: −13.4% (17.6%)). From 30 min to 4 h post-treatment, the highest mean (SD) percentage change in variation was observed among patients who reported severe pain at baseline compared with other baseline severity levels at each respective assessment point (30 min: −31.6% (24.6); 45 min: −46.8% (29.0); 1 h: −57.0% (27.7) and 4 h: −65.8% (27.9); Figure 2 and Supplemental Table S3).

Percent variation in NRS score for menstrual pain from baseline to each post-treatment assessment by pain severity at baseline among the primary analysis cohort.
NRS responders
The percentage of NRS responders increased with time since the first HBB Plus intake with an overall 90.8% of patients as responders after up to 4 h following HBB Plus treatment; the percentage of responders was 13.3% at 15 min, 42.9% at 30 min, 59.2% at 45 min, 75.5% at 1 h and 86.7% after 4 h (Figure 3). Compared with non-responders, responders demonstrated lower mean (SD) BPI scores at every pain level (worst: 6.7 (1.6) versus 7.2 (1.2); least: 1.5 (1.8) versus 3.7 (2.4); average: 4.7 (1.7) versus 5.7 (1.9); and at the moment: 2.7 (2.1) versus 5.3 (3.0)) assessed on the first evening following HBB Plus intake (Supplemental Table S4).

NRS responders at each post-treatment assessment period, overall and by baseline severity.
Dysmenorrhoea symptoms
Across all post-treatment assessment time points, majority of patients who reported at least one symptom at baseline (95.9%, prior to treatment) also reported at least one symptom at each assessment following treatment. However, this proportion decreased with time following treatment (15 min: 97.9%; 30 min: 91.5%; 45 min: 83.0%; 1 h: 76.6%; 4 h: 70.2%).
Difference in BPI score
Patients with menstrual pain at its worst reported a slight increase in mean (SD) BPI scores on the first evening following treatment compared with baseline (6.7 (1.6) versus 6.5 (1.9); p = 0.03). By the third evening, the BPI score decreased to 6.1 (2.1; p = 0.21 compared with baseline) and continued to drop, reaching 2.4 (1.8) by the seventh evening (p = 0.04 compared with baseline). Mean BPI scores for average pain were significantly lower in all evening assessments compared to baseline (all p < 0.02). Scores for pain at the moment also showed significant reductions compared to baseline (all p < 0.001). In terms of the composite score of pain severity, the mean BPI scores for all evening assessments were significantly lower than baseline, decreasing from 5.0 (1.6) at baseline to 1.6 (1.3) by the seventh evening (all p < 0.02; Supplemental Table S5).
Time to first perceptible relief and meaningful relief
Following HBB Plus intake, the mean (SD) time to first perceptible relief was 39.6 (41.8) min. When stratified by pain severity at baseline, the mean (SD) time to first perceptible relief was shortest among those with severe pain (32.1 (24.0) min) and moderate pain (39.7 (43.6) min) at baseline and longest among those with mild pain (110.0 (87.7) min). Mean (SD) time to first perceptible relief was shorter among patients who took 10 mg of HBB Plus (one tablet; 29.4 (20.6) min) than among patients who took 20 mg of HBB Plus (two tablets; 84.7 (72.6) min).
Following HBB Plus intake, the mean (SD) time to first meaningful relief was 65.8 (73.2) min. When stratified by pain severity at baseline, the mean (SD) time to first meaningful relief was shortest among those with severe pain (53.8 (56.6) min) and moderate pain (65.5 (76.2) min) at baseline and longest among those with mild pain (170.0 (108.4) min). Mean (SD) time to first meaningful relief was shorter among patients who took 10 mg of HBB Plus (one tablet; 47.5 (49.1) min) than among patients who took 20 mg of HBB Plus (two tablets; 139.2 (104.4) min).
Utilisation
In the primary analysis cohort of 98 patients, the median (Q1, Q3) cumulative dose of HBB Plus taken by patients over their menstrual pain period was 50.0 (30.0, 70.0) mg. The mean (SD) duration of menstrual pain was reported as 3.4 (1.6) days, ranging from 1 to 9 days. During the menstrual pain period, the minimum cumulative dose taken was one tablet of 10 mg, with a maximum of 23 tablets, amounting to 230 mg. During this period, the number of intakes ranged from 1 to 14, with a mean (min–max) number of intakes being 1.3 (1.0–3.0) per day per respondent. Patients were exposed to HBB Plus for a median (Q1, Q3) of 3.0 (2.0, 4.0) days during the menstrual period with the median (Q1, Q3) percentage of 66.7% (50.0%, 100.0%). On the day of the first intake assessment, 46.9% reported taking only one HBB Plus tablet, and 36.7% reported taking two tablets with a median (Q1, Q3) first intake of 2 (1, 2) tablets. A total of 32 patients had at least one HBB Plus intake with a corresponding pain assessment during the first three other intake periods with majority of the patients taking the lowest dose of 10 mg (68.8%; Figure 4).

HBB Plus dose taken at three other intakes for patients with one post-baseline assessment.
Safety
Among patients from the safety analysis cohort (N = 267), 5 (1.9%) TEAEs were reported. Among these TEAEs, 2 (40.0%) were categorised as mild, 1 (20.0%) as moderate and 2 (40.0%) as severe. These TEAEs included severe pain, circular loop problems, palpitations, iron deficiency and very severe abdominal pain with dark blood. All the TEAEs reported were deemed non-serious and did not lead to hospitalisation or an ER visit (Table 5).
Treatment-emergent adverse events for the safety analysis cohort.

n represents the population with available data for a given variable. AEs: adverse events; ER: emergency room; TEAE: treatment-emergent adverse event.
Perception
Global perception of improvement score
After HBB Plus intake, the majority of the patients reported a GPI score corresponding to “better” or “much better” at the time of first assessment (91.8%), at first evening (87.7%), and last evening assessment (98.2%). Only 2.0% and 7.9% of patients reported feeling “worse” or “much worse” at the time of first assessment following intake and first evening following intake, respectively. However, by the last evening assessment, no patients reported feeling “worse” or “much worse” following treatment, and only one patient reported feeling “about the same” at this time point (Supplemental Table S6).
Patient satisfaction
In the first intake assessment, 46.9% patients were “satisfied” and 42.9% were “very satisfied” with their use of the HBB Plus. In the last evening assessment, 47.4% patients were “satisfied” and 47.4% were “very satisfied” while no patient reported of being “dissatisfied” or “very dissatisfied” (Supplemental Table S7).
Discussion
The HBB containing medications are widely used to treat abdominal cramps and HBB is considered as an indispensable drug by the World Health Organization. 39 Though HBB has been used for over 70 years to treat abdominal cramps, there is a paucity of both interventional and non-interventional research on its efficacy and safety in gastro-urinary tract spasm including dysmenorrhoea. 40 Self-reported data are recommended for understanding first-hand patterns of utilisation and perception of HBB in a real-world setting. Such studies provide insights into patient preferences for OTC drugs for self-treatment. Moreover, PROs, capturing patients’ perspectives are increasingly considered as gold standard for clinical research outcome metrics. 22 Thus, this study aimed to describe the effectiveness of the HBB Plus tablet in women suffering from dysmenorrhoea using data from self-reported questionnaires. The study also discussed about the other objectives, which included describing utilisation patterns, safety and perception of pain (perceived reductions and satisfaction) among women to self-treat menstrual pain using HBB Plus.
A previous study in 2022 highlighted that dysmenorrhoea, a common gynaecologic problem, significantly affects routine life with symptoms like diarrhoea, sweating, breast tenderness, headache, nausea, tachycardia, vomiting and mood changes. 41 Such symptoms can interfere with normal daily activities, such as mood, sleep, walking, ability to work and social relationships. 42 This is consistent with our study that reported low back pain, headache, diarrhoea and/or constipation and fatigue/weakness as the most commonly reported symptoms at baseline. However, a decrease in symptoms was observed with treatment over time. Such evidence underscores the benefits of HBB for pain management and improving QoL in self-treated patients, supporting the integral role of OTC drugs in safeguarding public health.
This study observed that around 99% of the patients experienced pain relief within 4 h following the first intake of HBB Plus. The difference in pain between baseline (prior to HBB Plus treatment) and during post-treatment assessments significantly increased over time. This outcome was consistent with the findings from the sensitivity analysis, thereby assuring the robustness of the primary result. Patients with severe pain experienced the most pain relief 30 min after HBB Plus intake. These findings are similar to an RCT by Lacy et al. in self-reported functional abdominal pain, which showed a significant mean NRS score reduction from baseline at 15 min after taking the first dose of HBB (−0.4 points (95% CI: −0.7, −0.1), p = 0.0137). Treatment with HBB recorded a clinically relevant reduction of at least two points on NRS (~30% pain relief) in 45 min in patients with gastrointestinal-related abdominal cramping. 43
The current study reported an increase in the percentage of patients responding to HBB Plus treatment as the time from intake of the study drug increased. The study observed 13.3% responders after 15 min, 42.9% after 30 min, 59.2% after 45 min, 75.5% after 1 h and 86.7% after 4 h. Overall, 90.8% patients were categorised as responders after 4 h post drug intake based on commonly accepted criteria of the number of patients with at least a 30% decrease from baseline. As compared with non-responders, responders demonstrated lower mean (SD) BPI scores at every level of pain, that is, worst pain, least pain, average pain and pain at the moment. Also, the mean BPI scores for average pain (p < 0.02) and pain at the moment (p < 0.001) were significantly lower in all evening assessments compared with baseline. A previous trial involving ambulatory patients with irritable colon studied the efficacy and tolerability of combination therapy of HBB Plus, monotherapies of HBB and paracetamol against placebo. The study reported 81%, 76%, 72% and 64% of patients as responders, defined as slight or remarkable improvement in the symptoms from baseline, belonging to HBB Plus group, monotherapy of HBB and paracetamol groups and placebo group, respectively. 44 More than 90% of patients in our study were “satisfied” or “very satisfied” with the use of HBB to self-treat menstrual pain and reported that they would recommend it to others. These results were consistent with a survey study in Germany by Storr et al., which evaluated HBB Plus for abdominal cramps and discomfort among 1686 patients. 45 At least 95% of the patients rated HBB or HBB Plus as good or very good in relieving abdominal pain compared with peppermint oil (combination of four different extracts). 45 Additionally, Stewen et al. found that 45.5% and 51.6% of patients rated the global efficacy of HBB Plus as very good or good, respectively. 20 In our study, only 3% women in the primary analysis cohort reported using a concomitant medication in addition to HBB Plus, suggesting a high reliance on HBB for pain relief. This strengthens the assessment of therapeutic effectiveness and reliability based on data on satisfaction. Though patient satisfaction is subjective to the patient perspective, it supports drawing an affirmative correlation between effectiveness and preference of drug with patient satisfaction.
Approximately 30% of women in our study reported using hormonal OCs at the time of study enrolment, which is consistent with existing prevalence estimates of hormonal OC use in European countries. 46
The summary of product characteristics 31 mentions that 10 mg dose of HBB may cause symptoms like fever, nausea, vomiting, changes in bowel movements, abdominal tenderness, decreased blood pressure, fainting, or blood in stool as commonly observed AEs. Painful or red eye with loss of vision, anaphylactic shock, anaphylactic reactions, dyspnoea, other hypersensitivity, tachycardia, dry mouth, skin reactions (e.g., urticaria, pruritus), abnormal sweating and urinary retention are either rare or uncommon AEs. 31 A recent meta-analysis on antispasmodics reported HBB with the lowest frequency of treatment-related adverse events. 47 In addition to a recent review article that confirmed the benefits of analgesic/antispasmodic combination products, 22 previous studies have reported that 4 g per day dose of HBB and paracetamol is considered to be safe.40,48 Overall, these results are in line with our study, which reported only 5 TEAEs, all non-serious and not requiring hospitalisation or ER visit.
Previous review articles highlight that fixed-dose combination products with synergistic or additive properties achieve significant therapeutic effects, faster onset and improved responses with smaller doses than individual drugs. 22 Previous placebo-controlled RCT has shown that HBB, paracetamol and their combination (HBB Plus) are effective and well-tolerant for recurring abdominal cramps when used three times daily continuously for 3 weeks. 49 Similarly, another 4-week trial involving 712 ambulatory patients with irritable colon demonstrated a statistically significant improvement in abdominal pain with HBB Plus compared to the placebo group and HBB Plus compared to the paracetamol group. 44 de los Santos et al. reported that combination therapies, including HBB Plus were more effective than placebo in controlling menstrual pain and other symptoms of PD. 50 Early double-blind cross-over study by Kemp 51 and Odeblad 52 showed also positive effects of HBB in dysmenorrhoea. A non-interventional, prospective study based on pharmacy-based patient survey reported that in 314 women with dysmenorrhoea, pain and cramp intensity of 7.45 ± 2.13 on a 0–10 Likert scale decreased to 2.86 ± 1.81 after treatment with HBB Plus, with 64.6%—70.4% improvement in daily functions. Symptom relief within 60 min was observed in 84.7% patients; 97.2% rated tolerability as good. 20
Though the current study had achieved a small sample size of around 100 patients in the primary analysis cohort, the results produced were similar to those found in a previously conducted clinical trial and RWE.43,45 It should be noted that the target sample size for this study was reached, providing adequate power to detect a change in the NRS score. Because we conducted this study in real-world setting, the results are likely generalisable to other individuals with dysmenorrhoea, as the patients in our study were homogeneous with regards to their baseline symptom complaints that led to HBB Plus intake.
The self-reported study outcomes align with clinical outcomes that measure the effectiveness of HBB Plus. The results also demonstrated that the majority of patients experienced improvement in symptoms within 15 min. This study assessed outcomes every 15 min till 1 h following HBB Plus intake to ensure timely reporting of the outcomes and likely minimised recall bias that may have occurred if assessed less frequently or at later times post-treatment. The robustness of the results was also confirmed by sensitivity analyses.
Limitations
The current study is associated with a few limitations. In the trials conducted in real-world settings, which are primarily conducted in a digital space, 53 there is a considerable delay observed between recruitment and active participation for pain-related dropout rates. Thus, there is a significant delay in reaching the desired sample size of patients due to the difficulty in recruiting eligible patients to form a representative sample size. 24 Additionally, the study participants may not completely represent the general population as the digital survey app may have introduced selection bias not evident in the summarised demographic data ─ selection of participants with a greater affinity for technology. However, the inherent sampling limitations 54 did not significantly affect the conclusion of the study owing to the enhanced suitability and user-friendliness of digital apps for evaluating pain impact and related lifestyle factors. 55 No attempts were made to externally validate the patient-reported data and the study completely relied on self-reported data by participants, considering only that the patients could accurately capture the perceived effects of OTC treatment on their pain. Also, there was a variation in compliance related to patients taking HBB Plus before the pain baseline evaluation, which resulted in the exclusion of such patients. Not all the patients completed all the post-treatment assessments, as the data was collected via self-report during the 4 h following HBB Plus intake, which could have led to the potential selection bias. In other cases, enrolled women did not experience any pain during their menstrual cycle occurring during the study period. Generally, women reported that it was difficult to anticipate their menstrual pain for study enrolment in case of an irregular cycle. Therefore, HBB Plus combination, being an OTC drug and associated with the benefits of availability and convenience presents as a potential self-treatment option for pain management in such a population with irregular periods. Although the use of self-reported data is beneficial for understanding first-hand accounts of patient symptoms, treatment utilisation and perception, PROs are also subject to risk of misclassification and recall bias.
However, results of our sensitivity analysis suggested that patients in the primary analysis cohort (with completed assessments up to 4 h) and the sensitivity analysis cohort (at least one completed assessment in 4 h following treatment) were similar in terms of patient characteristics and observed mean change in NRS score between baseline and post-treatment assessment. Therefore, this selection criterion for the primary analysis cohort appeared to impact the study results only slightly, if at all.
Conclusion
The objective of this study was to assess the effectiveness, utilisation patterns, perception and safety of HBB Plus for self-treatment of menstrual pain in women with dysmenorrhoea. Based on real-world data from patient-reported questionnaires, the findings indicate that HBB Plus significantly reduces pain within the first 4 h after treatment. Women reported substantial symptom improvement, satisfaction with HBB Plus and a willingness to recommend it to others for menstrual pain relief. Overall, this real-world study demonstrated that treatment with HBB Plus is effective and well-tolerated, as there was a decrease in the proportion of symptoms with treatment over time. The outcomes of this study show similarities to a recently published post hoc analysis of another real-world study. 20 Taking the results of both studies together, there is considerable support from real-world data for the use of HBB Plus as an additional treatment option for the self-treatment of dysmenorrhoea.
Supplemental Material
sj-docx-1-whe-10.1177_17455057251406956 – Supplemental material for A real-world study to describe the effectiveness and usage patterns of hyoscine butylbromide plus tablets in patients with dysmenorrhoea
Supplemental material, sj-docx-1-whe-10.1177_17455057251406956 for A real-world study to describe the effectiveness and usage patterns of hyoscine butylbromide plus tablets in patients with dysmenorrhoea by Robert Lange, Caroline Amand and Lorraine Harrington in Women's Health
Supplemental Material
sj-docx-2-whe-10.1177_17455057251406956 – Supplemental material for A real-world study to describe the effectiveness and usage patterns of hyoscine butylbromide plus tablets in patients with dysmenorrhoea
Supplemental material, sj-docx-2-whe-10.1177_17455057251406956 for A real-world study to describe the effectiveness and usage patterns of hyoscine butylbromide plus tablets in patients with dysmenorrhoea by Robert Lange, Caroline Amand and Lorraine Harrington in Women's Health
Footnotes
Acknowledgements
The authors thank Didem Sen (Opella) for coordinating the development of the manuscript and facilitating author discussions. The authors acknowledge Carenity, part of the EvidentIQ group, a German online patient platform; and Vijay Rayasam, an employee of Opella, who provided writing and editorial support.
Ethical considerations
The study was conducted in accordance with the guidelines for Good Pharmacoepidemiology Practice defined by the International Epidemiological Association, the ethical principles that have their origins in the Declaration of Helsinki and the French legal and regulatory obligations. All data was handled in strict confidentiality in compliance with the European regulation applicable since 25 May 2018, on data protection and privacy (General Data Protection Regulation no 2016/679).
Consent to participate
All participants provided written informed consent form.
Consent for publication
Not applicable.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was funded by Opella (grant number 2019-PA-001).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RL and CA are employees of Opella and may hold shares and/or stock options in the company. LH declared no potential conflicts of interest with respect to the research authorship and or publication of this article.
Data availability statement
Qualified researchers may request access to patient-level data and related study documents including the clinical study report, study protocol with any amendments, blank case report form, statistical analysis plan and dataset specifications. Patient-level data will be anonymised and study documents will be redacted to protect the privacy of participants. Further details on Opella’s data sharing criteria, eligible studies, and process for requesting access can be found at: ![]() .
.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


