Abstract
Introduction:
Endometriosis is a highly prevalent disabling comorbidity of women with migraine. The aim of the current study was to identify menstrual migraine prevalence and features in women with endometriosis.
Methods:
This observational study included data from patients visiting our outpatient clinic from 2015 to 2021. Information was collected from charts and through structured phone interviews. Patients with surgically confirmed endometriosis and migraine were included.
Results:
A total of 92 from 344 endometriosis patients suffered from migraine and a subset of 35 from menstrual migraine (MM). From the subset of women with MM 42.9% reported to have auras. Groups did not differ with regard to rASRM stage. Women with MM reported significantly more pain days/month at present and 5 years ago and more often a MIDAS Grade 4 (p < 0.01). Both groups had less migraine attacks/month now, compared with monthly attacks 5 years ago. All women experienced endometriosis surgery in this interval. There was a trend towards more severe dysmenorrhoea with onset at menarche and towards higher use of analgesics for menstrual pain during the bleeding in the MM group. Less deliveries were noted in the group with MM.
Conclusion:
In women with similar endometriosis stages MM is associated with a higher migraine frequency, more dysmenorrhoea days and a higher burden of disease. Future studies are needed to better understand a potential influence of menstrual migraine on the number of deliveries. Our findings suggest, that close collaboration of neurologists with gynaecologists and shared treatment decisions might be of advantage for patients with the comorbidity.
Introduction
Migraine and endometriosis are disabling conditions with chronic pain affecting women during the reproductive years. Oestrogens play a pivotal role in the pathophysiology of both conditions and symptoms typically dissolve with menopause. 1 Some newer studies indicate in addition, that both conditions are associated with breast cancer.2,3 Today there is high evidence, that there is a shared genetic background, especially related to polymorphisms of sex hormones.4–6 Migraine prevalence was significantly higher in endometriosis patients compared with controls in a population-based study (1.7 fold). 7 Survey-based case-control studies reported migraine in 29%–69% of endometriosis patients, which is far higher than the expected prevalence in the general female population.8–10 Endometriosis is an inflammatory proliferative chronic disease affecting 5%–10% of women of reproductive age.11–13 A combination of immunological, environmental and anatomical factors are discussed as pathomechanisms for the ectopic growth of endometrial tissue. 11 Endometrial tissue in affected patients may release Calcitonin Gene-Related Peptide (CGRP) and shows an increased density of CGRP-positive sensory nerve fibres.14,15 Endometriosis pain is strongly related to the period of menstrual bleeding with dysmenorrhoea and pelvic pain being the most disabling symptoms. 16 Other endometriosis associated symptoms may occur at any type of the cycle. Menstrual migraine (MM) is a special subtype of migraine occurring at days 1 ± 2 in at least two of three cycles and associated with oestrogen withdrawal in the natural cycle or in the pill break in users of combined hormonal contraceptives. 17 MM attacks are more severe, of longer duration and less responsive to acute therapy with, for example, triptans or short-term prevention.18–27 Their high negative impact on quality of life has been described many times. 28 Between 18% and 25% of women with migraine report MM, while the prevalence and features of MM in endometriosis patients with migraine as a comorbidity is unknown.29–31 This is despite the probable high burden caused by the combination of disabling symptoms from endometriosis and MM during the days of the menstrual cycle. It is still unknown, if MM onset in women with endometriosis already occurs with the initiation of dysmenorrhoea during the early years of puberty. Such a coincidence could facilitate earlier diagnosis and treatment potentially contributing to avoiding the development of central pain sensitisation (CPS). CPS leads in patients with chronic pain conditions to pain hypersensitivity. 32 Migraine as well as endometriosis respond to continuous therapy with some progestins in dosages which inhibit ovulation.33–36 With the present study we aim to improve to identify MM features in women with endometriosis, as well as MM prevalence and burden of disease. Considering not only the strong genetic background, but also the high influence of oestrogens on both conditions, we were interested to evaluate, if certain endometriosis features might differ in these patients, and if MM onset and dysmenorrhoea onset are timely connected. Higher awareness of women at risk for the comorbidity might contribute to earlier diagnosis, prevent CPS and decrease burden of disease.
Materials and methods
This cross-sectional study was conducted at the Clinic for Reproductive Endocrinology in the Department of Gynaecology of the University Hospital of Zurich. Data were collected from patient records and supplemented through telephone interviews. In order to assure correctness of our data, two persons from medical staff were trained in 50 interviews prior to start of the study. The presented data derive from a broader study investigating the characteristics of endometriosis in women with migraine anddepression. 37
Data collection
Patients with the diagnosis of endometriosis and migraine were preselected by searching charts from all endometriosis patients treated in our outpatient clinic from January 2015 to July 2021. Included were premenopausal patients aged >18 years with histopathological examination results confirming endometriosis, who in addition suffered from migraine (Figure 1). Migraine, menstrual migraine and pure menstrual migraine diagnosis was clarified during a telephone interview using the criteria of the International Classification of Headache Disorders (third version, ICHD-3). 17 Postmenopausal women and those with adenomyosis or scar endometriosis were excluded. We furthermore excluded patients, if no histology was available and those in which the stage of endometriosis and localisation were not clearly documented. During a phone contact, we informed patients about the study and asked if they were interested in participating. Those willing to participate received an information sheet and thereafter provided oral consent. Details about the inclusion process can be found in Figure 1. During the interview we collected headache data on age at migraine onset, migraine frequency in the past and at present, aura, menstrual attacks, duration of attacks, migraine triggers, acute and prophylactic medication, family history, history of medication overuse headache, history of status migrainosis and quality of life, using the Migraine Disability Assessment questionnaire (MIDAS). 38 Endometriosis stage according to the American Society for Reproductive Medicine classification (rASRM stage) was identified from the surgery report. 39 Further gynaecological information and information on comorbidities was collected in the interview using a shortened and adapted to our needs version of the ‘Women’s Health Symptom Survey Questionnaire’ recommended from the World Endometriosis Research Foundation. 40 We collected data on medical conditions, operations, family history, pregnancies, deliveries, potential symptoms of endometriosis and medications used at present. Furthermore, we collected detailed information on the menstrual cycle, we focussed on menstrual pain days during adolescence and at present, dysmenorrhoea onset and intensity, number of pregnancies and deliveries, number and timing of endometriosis surgery and number of pain days during the cyclic bleeding as well as use of hormones and analgesics. Pain intensity was rated on a scale from 0 to 3 with 0 meaning no pain and 3 severe pain. The study was approved by the cantonal ethics commission of Zurich (BASEC Nr. 2021-00285) and registered on clinical Trials.gov (NCT04816357).
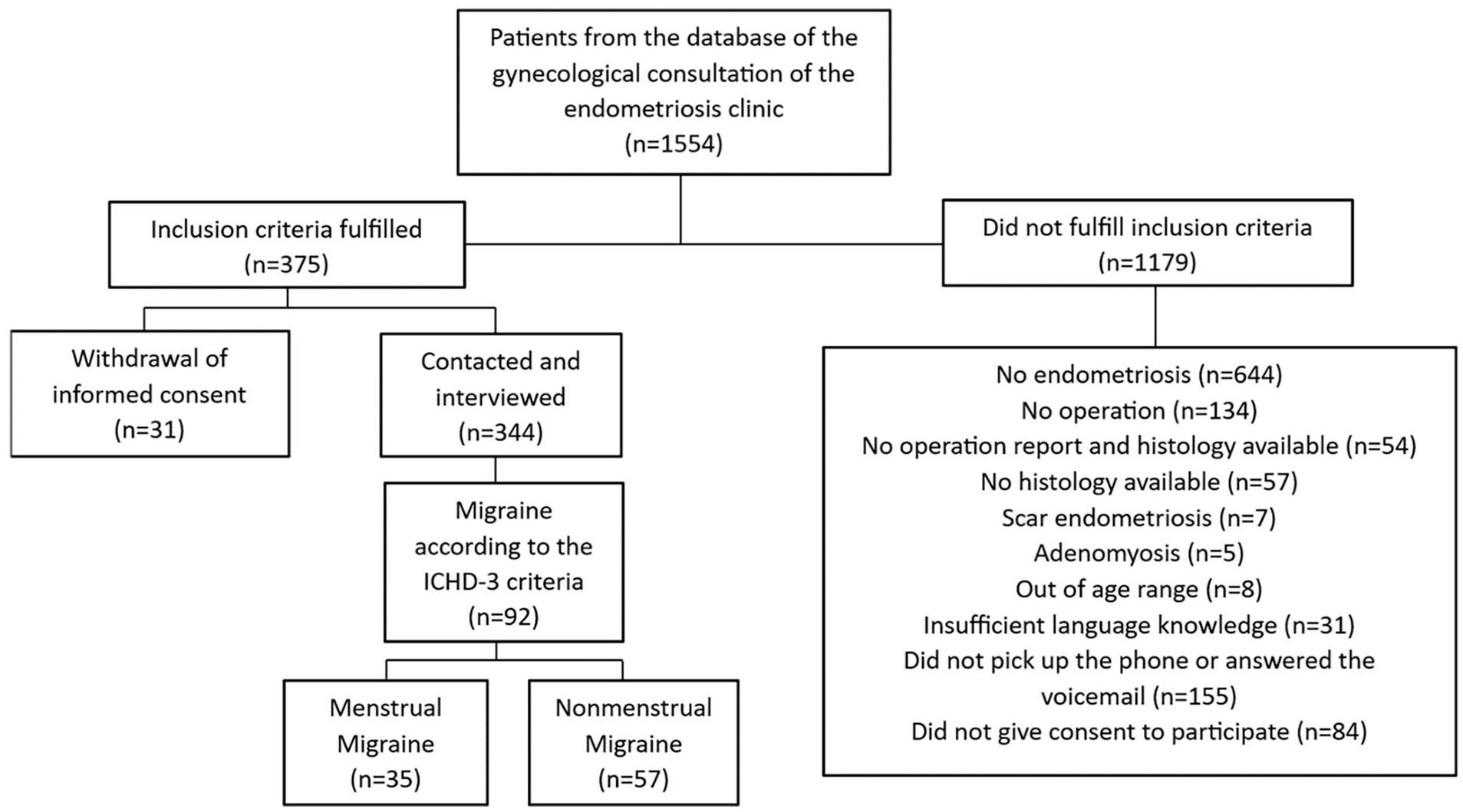
Flow Chart of participants.
Statistical analysis
Data was analysed using SPSS® statistics (IBM®, Armonk, New York, United States Version 27.0.1.0.). Shares in percent were used for categorical variables and means including standard deviation (SD) were used for numeric variables. To compare categorical variables among groups, we used Chi-Square or Fisher’s exact test (expected frequencies <5), depending on expected frequencies. To compare numeric variables between two groups, we used the independent sample t-test for normally distributed variables and Wilcoxon-Mann-Whitney test for not normally distributed variables. In addition, binary logistic regression was used to evaluate a potential association between age and menarche and migraine onset in both subgroups. A two-tailed p-value ⩽0.05 was considered statistically significant.
Results
In this study involving 375 premenopausal patients with endometriosis and 92 patients suffered from both, endometriosis and migraine (Figure 1). About 38% (n = 35) of the migraineurs experienced menstrual migraine and 61% (n = 57) reported non-menstrual migraine (nMM; Table 1). Only two women in the menstrual migraine group suffered from pure menstrual migraine indicating, that they only experienced migraine attacks at cycle days 1 ± 2 and never on any other day. The groups did not differ with regard to age, but BMI was slightly lower in the MM group. Age at menarche and age at first severe dysmenorrhoea did not differ (p = 0.66 and 0.56). A higher percentage of women in the MM group experienced severe dysmenorrhoea onset with menarche 48.6% versus 36.8% (p < 0.1). A similar trend was seen for the number of monthly pain days during the bleeding and use of analgesics for bleeding-related pain (p ⩽ 0.1). There were no differences between the two groups with regard to the ASRM stage or the number of surgeries (p = 0.28 and 0.22; Table 1). Nevertheless, women with MM had less pregnancies and significantly less deliveries (p = 0.09 and 0.04). The groups did not differ with regard to a history of depression or treatment for a psychic condition (p = 0.35 and 0.66). Table 2 demonstrates migraine characteristic for both groups. There were no differences with regard to age at migraine onset (p = 0.8). Women with menstrual migraine suffered from a higher migraine frequency, now and in the past, but not during adolescence (p = 0.001, 0.002 and 0.36). In both groups the rate of women having migraine with aura (MA) was high with (41.2%–42.9%; p = 0.9). Frequency of medication overuse headache, or history of status migrainosus did not differ between groups (p = 0.44 and 0.3). The only migraine trigger, which was significantly higher in the MM group was menstrual bleeding (p > 0.001). In more than 50% of the women migraine was not present before starting use of combined hormonal contraceptives (CHC). This was more pronounced in the group of women without menstrual migraine (p = 0.057; Table 2). Hormone use at the time of the interview did not differ between groups (CHC 6.8 % nMM, 14.3% MM; progestin-only methods 15.3% nMM 2.9% MM, no hormones 77.9% nMM; 82.8%). A correlation between age at menarche and migraine onset (p = 0.7 and 0.5) or between age at menarche and onset of severe dysmenorrhoea (p = 0.6 and 0.68) could not be observed in the whole group, nor in the two subgroups. Most of MIDAS associated quality of life items was lower in the women with MM. Both average MIDAS Grade and MIDAS Score were higher in women with MM (0.01; Table 3).
Baseline characteristics and endometriosis-related gynaecologic features.

n.r.:no response
Migraine characteristics. .

MIDAS Parameters in women with Endometriosis and Menstrual or Non-menstrual Migraine.

Questions in this table are given in a shortened form. The observation time for MIDAS includes a 90 day interval. Data were compared using nonparametric Wilcoxon test.
Currently, 59% of the patients did not use prophylactic agents, 37% used magnesium or riboflavin on a regular basis, while 3.1% used onabotulinumtoxin A or beta-blockers. Pain medication was prescribed from neurologists in 18.5% and GPs in 35.1%. Another 35.1% of the participants bought the medication in the pharmacy without prescription or received it from other sources (11.3%).
Discussion
Endometriosis and migraine are frequent, highly disabling, oestrogen-dependent comorbidities with a shared genetic background.4–6 It is possible that women with endometriosis might differ from those without endometriosis with regard to MM course and features. Oestrogen interacts with the local inflammatory process involving sensory nerve fibres and macrophages produced in the endometriosis lesions. 41 In the present study we found, that 40% of migraine patients with endometriosis suffered from MM. Within this subgroup 42% reported migraine with aura. In comparison with the nMM group the MM group experienced a higher frequency of monthly migraine attacks and had a significantly lower quality of life with more days characterised by more days with reduced productivity at work, missed leisure activities and without the ability to perform household work. Also, the MIDAS Grade was significantly higher (Table 3). This was observed despite no significant differences in the ASRM stage between women with MM and nMM. Regarding endometriosis complaints the patients with MM reported more often severe dysmenorrhoea onset with menarche already (48.6% vs 35.8% p = 0.1) and more endometriosis-related pain days during the menstrual bleeding (p = 0.1). The delivery rate was significantly lower in the MM group (p = 0.04).
Menstrual migraine frequency, onset and aura
Considering both, the high genetic background of the comorbidity based amongst others on oestrogen-receptor polymorphisms, and the relevant role of oestrogens in the pathophysiology of endometriosis we expected, that the majority of women might suffer from MM and that migraine onset might have a relation to age at menarche and onset of dysmenorrhoea. Insofar we were surprised that migraine onset also in endometriosis patients is quite late (age around 20 years) in relation to age at menarche (12.7–12.8 years) and also age at first severe dysmenorrhoea (age 17–18 years). This is around 5–6 years after menarche. Interesting is, that migraine onset and severe dysmenorrhoea onset are timely related in both groups. Further studies are needed to understand if initiation of severe dysmenorrhoea in endometriosis patients has an impact on the course of migraine. It is also noteworthy, that around 50% of our MM patients suffered from severe dysmenorrhoea already at the time of menarche.
The comorbidity does not seem to be associated with onset of symptoms already during early adolescence, but the in general high rate of severe dysmenorrhoea with menarche could be an indication for the clinician to check for a history or family history of migraine. Even more relevant might be the choice of a contraceptive or hormone for endometriosis treatment, if there is an indication for a bidirectional comorbidity.
In a recent high-quality diary-based study with migraine patients recruited in a headache centre, the prevalence of MM was around 15% higher than in our cross-sectional study, while the, prevalence of migraine with aura did not differ from ours with 40%. 42 The study did check report if participants suffered from endometriosis. Another recently published gene-based study found substantial genetic correlation between migraine with aura and migraine without aura, suggesting that both conditions might be more alike than dissimilar. 43 Menstrual migraine has been described to be typically without aura, however migraines with aura could occur during the non-menstrual attacks of these women.18,24,29 Newer diary-based data confirm the notion, that typically auras reported from patients with MM do not occur during the perimenstrual time frame of cycle days 1 ± 2. 42 Prevalence of menstrual migraine did in the latter study not differ between women with migraine with or without aura what is in line with our findings.
Still, it is noteworthy, that in our study neither age at migraine onset was earlier, nor was family history more frequently positive in women suffering from MM. 44
The percentage of women, who have ever been treated for a psychological condition (51%–57%) was higher in our trial including endometriosis patients, than in studies with migraine patients, but did not differ so much from the percentages in the Verhagen trial, which reported the percentage of lifetime depressions in migraineurs with 38%–43%.42,45,46 Major depression rate in our trial was 25% and did not differ between groups, but there was a high rate of non-responders.
The monthly migraine frequency (MMF) did not differ between groups during adolescence. In the later course of life MMF was significantly higher in women with MM (Table 2). For the latter group we found in addition a trend to more days with endometriosis pain days during the menstrual bleeding, what may contribute to the probably higher burden of disease. We can only speculate, why for both groups the MMF was 40%–50% lower at the time of the interview in comparison to the frequency 5 years earlier (Table 2). As only 3.1% of the patients used prophylactic agents apart from magnesium or riboflavin, the observed decrease in MMF is rather not related to the new-start of prophylactic agents. All participants in our study underwent endometriosis surgery within the recent years. There are two mechanisms how endometriosis surgery could impact the course of migraine. On one hand the CGRP release, an important trigger for migraine attacks, from endometriosis lesions is diminished. On the other hand, reduction in pelvic pain might contribute to reverse central pain sensitisation. Therefore, we hypothesize that endometriosis surgery might not only reduce dysmenorrhoea and pelvic pain, but also might have had a positive impact on the course of both types of migraine.
Burden of disease, surgery and pregnancies
In accordance with findings in migraine patients without the comorbidity we found for endometriosis patients with MM that reduction in quality of life was much more pronounced than in those with nMM (Table 3). The high negative impact of MM on the private life is reflected in the number of days when family/social activities are missed. If this burden is the reason, why the pregnancy and delivery rate in this subgroup is lower requires further investigation. It is well known, that endometriosis is associated with infertility. 47 Infertility rates in migraine patients have not yet been investigated. As neither ASRM stages nor the age at first endometriosis operation or the number of endometriosis operations differ, we suggest, that the higher burden of disease in our MM-subpopulation is a main cause for the lower delivery rate. This hypothesis is in accordance with a recently published narrative review postulating that migraine may influence fertility related to the obstacles patients face stemming the impact of migraine on their social relationships. 48 Looking at the balanced age distribution of both groups (women >35 years; MM = 52%; nMM = 54%) we regard it as unlikely that a difference in the distribution of age between the groups could bias this result. Staying without children might in addition diminish psychological well-being. 49 As both migraine and endometriosis are associated with depression we checked in the interview, if women had ever experienced treatment for psychic problems or a depression. In spite of the higher burden of disease reported for our MM patient we did not find a difference in psychiatric comorbidity between groups, however the percentage of such a treatment was strikingly high (>50%) for both groups (Table 1). The majority of data related to migraine and depression do not discriminate between migraine subtypes and do not consider fertility or endometriosis, but this topic requires more attention.50–53
Finally, we found a trend towards more monthly days with endometriosis pain during the menstrual bleeding in MM patients, what could be the cause for the higher number of days using analgesics in this period (p < 0.1; Table 1). The progestin desogestrel exerts a positive impact on menstrual and non-menstrual migraine and might insofar be considered as an important option for the treatment of both conditions.35,54,55 It is still not clear today, if the oestrogen-induced disabling migraine attacks during the perimenstrual phase would also improve with treatment options like physiotherapy or stress relaxation.
Limitations
Strength of this study are the inclusion of only women with histologic confirmed endometriosis and a clear staging, and the collection of migraine data by skilled experts in a phone interview. However, potentially data related to pain and migraine frequency in the past might be affected to recall bias. Insofar our data have to be confirmed in prospective studies. Women were recruited from a specialised endometriosis clinic and not a headache clinic. This might explain, why the overall frequency of migraine/month was rather low. However, we do not expect that the general outcomes related to high burden, migraine onset in relation to menarche, low delivery rate and course of MM are much affected from this.
Conclusions
In women with similar ASRM endometriosis stages MM is associated with a higher migraine frequency, a lower delivery rate, more dysmenorrhoea days and a much lower quality of life in comparison to nMM. In neither group age of migraine onset correlated with onset of severe dysmenorrhoea or age at menarche. Future studies are needed to better understand a potential influence of menstrual migraine or its burden on the number of deliveries. Oestrogen-free hormonal treatment options can be useful for both conditions and may contribute to reduce the burden of disease. Our findings suggest that collaboration between neurologists and gynaecologists and shared treatment decisions might be of advantage in endometriosis/migraine patients.
Footnotes
Acknowledgements
The authors thank all patients who agreed to participate in this study.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analysed during the current study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
